Chapter 9 Molecular Geometry Bonding Theories Overview u

Chapter 9 Molecular Geometry & Bonding Theories

Overview u u Molecular Shapes VSEPR Model o. Predicting Shapes o. Effect of Nonbonding Electrons u u Polarity of Molecules Covalent Bonding

u Hybrid Orbitals osp, sp 2, sp 3 ohybrids containing d orbitals u Multiple Bonds osigma (s) & pi (p) olocalized & delocalized u Molecular Orbitals oelectron configurations & bond order odiamagnetism & paramagnetism

Molecular Shapes & VSEPR u Shapes defined by bond angles olinear, 180° angles otrigonal planar, 120° angles otetrahedral, 109. 5° angles u VSEPR o Valence Shell Electron Pair Repulsion theory oelectron pairs are arranged symmetrically with maximum separation
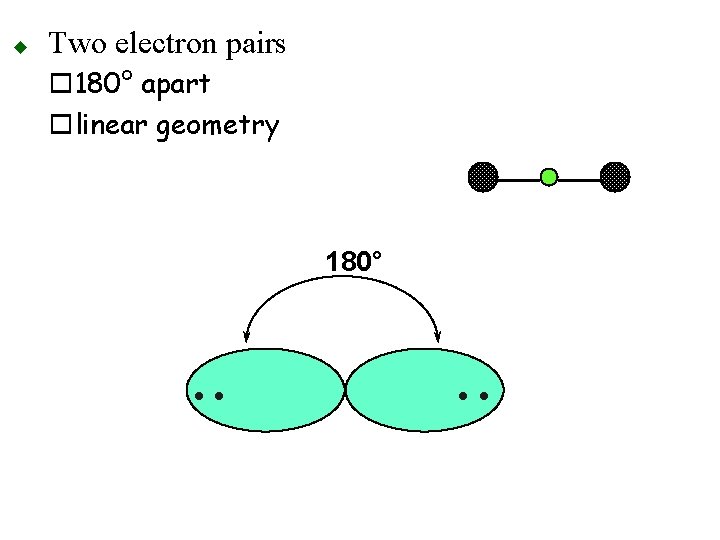
u Two electron pairs o 180° apart olinear geometry 180° • •
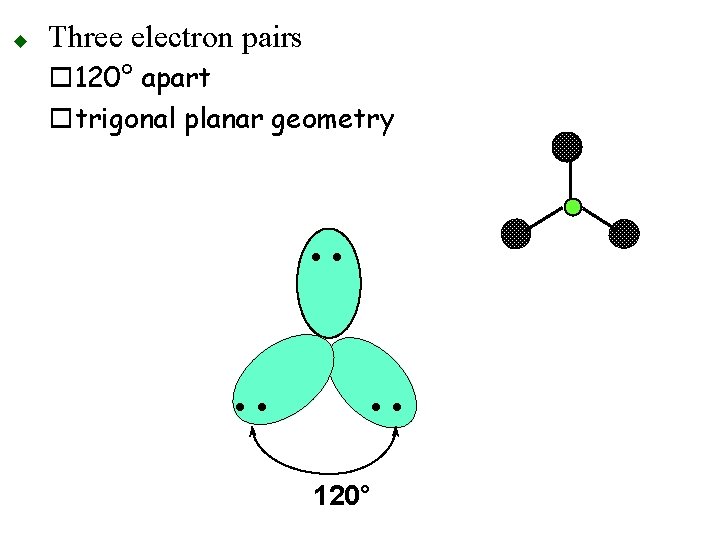
u Three electron pairs o 120° apart otrigonal planar geometry • • 120°
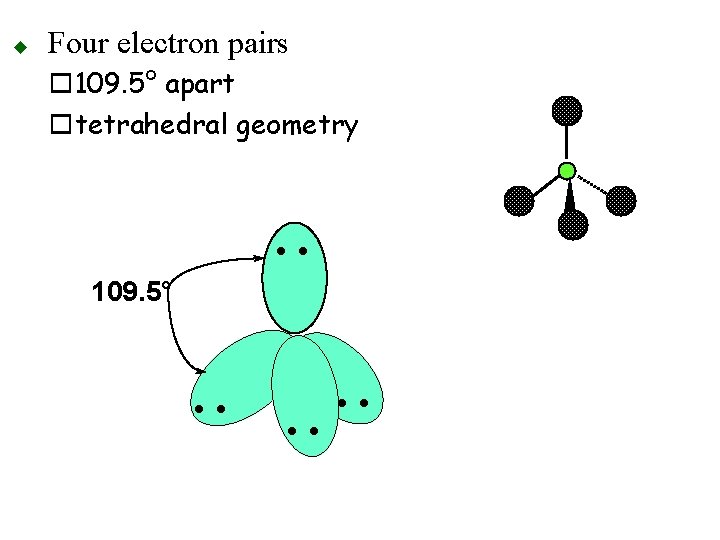
u Four electron pairs o 109. 5° apart otetrahedral geometry • • 109. 5° • •
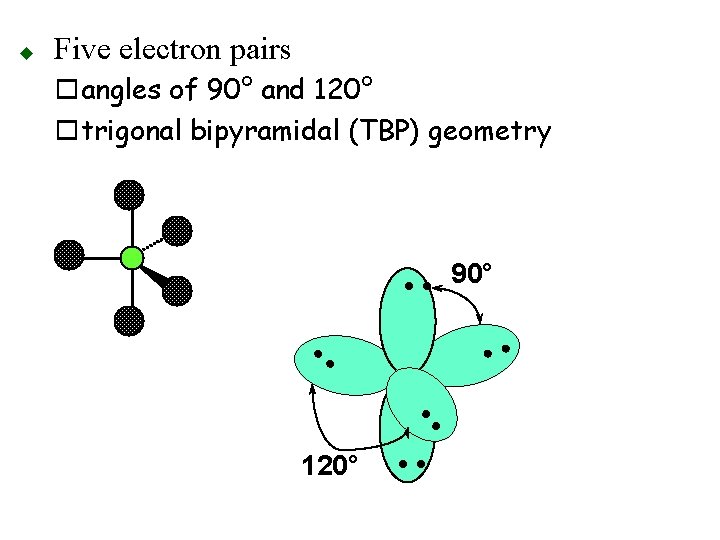
Five electron pairs oangles of 90° and 120° otrigonal bipyramidal (TBP) geometry • • 90° • • 120° • • u
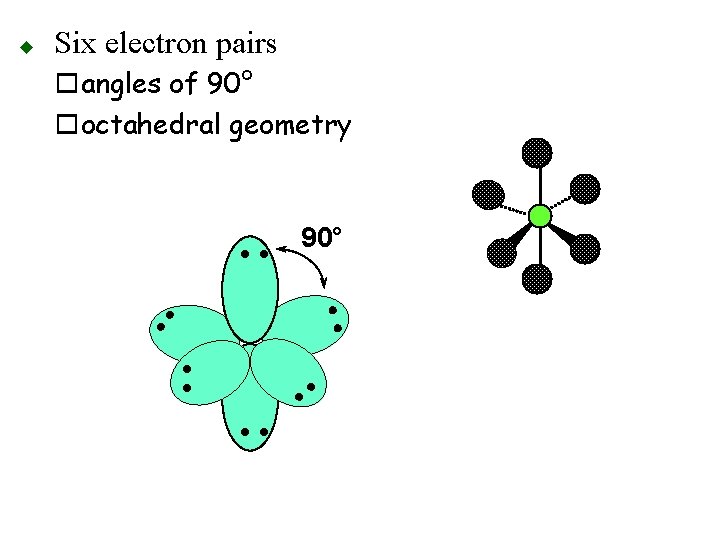
Six electron pairs oangles of 90° ooctahedral geometry 90° • • • • u • •

Geometries u Electron pair geometry oarrangement of electron pairs around a central atom u Molecular Geometry oarrangement of atoms around a central atom u When all electron pairs are bonding pairs oelectron pair geometry = molecular geometry u When there are unshared electron pairs oelectron pair geometry ¹ molecular geometry

u To determine electron pair geometry odraw Lewis dot structure ocount shared & unshared electron pairs around central atom • a multiple bond is counted as only one bonding pair when predicting geometry odetermine electron pair geometry based on the number of electron pairs • • • 2 pair = 3 pair = 4 pair = 5 pair = 6 pair = linear trigonal planar tetrahedral trigonal bipyramidal octahedral

Molecular Geometries with One or More Unshared Pairs u Two Pairs oelectron pair geometry linear obonding pairs 2 onon-bonding pairs 0 omolecular geometry linear
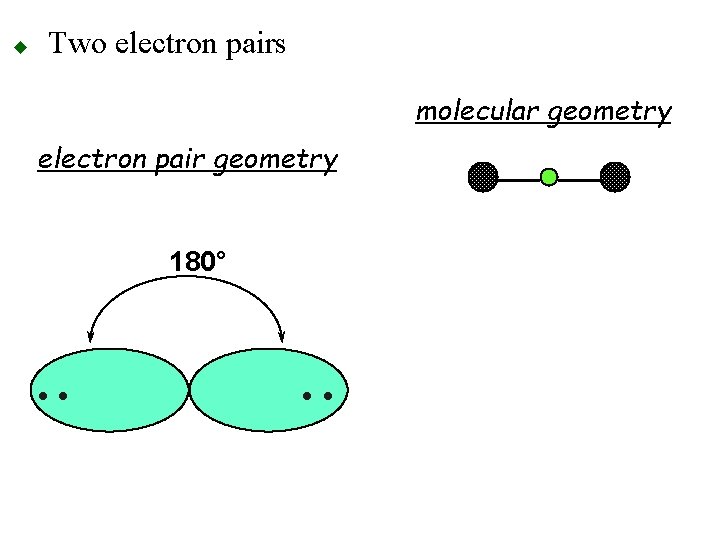
u Two electron pairs molecular geometry electron pair geometry 180° • •

u Three Pairs oelectron pair geometry planar trigonal obonding pairs 3 2 onon-bonding pairs 0 1 omolecular geometry bent trig. pl.
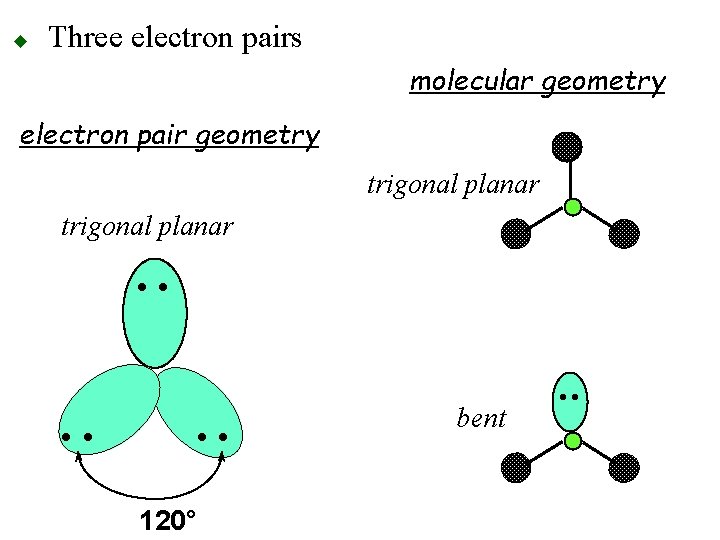
u Three electron pairs molecular geometry electron pair geometry trigonal planar • • 120° bent • •
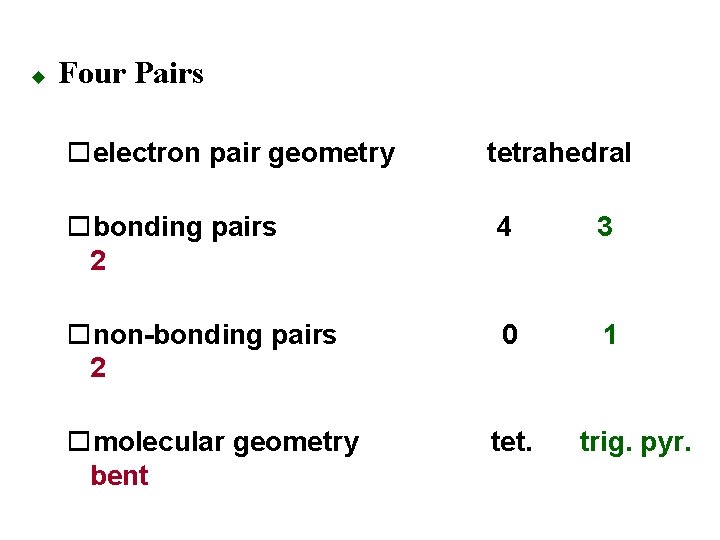
u Four Pairs oelectron pair geometry tetrahedral obonding pairs 2 4 3 onon-bonding pairs 2 0 1 omolecular geometry bent tet. trig. pyr.
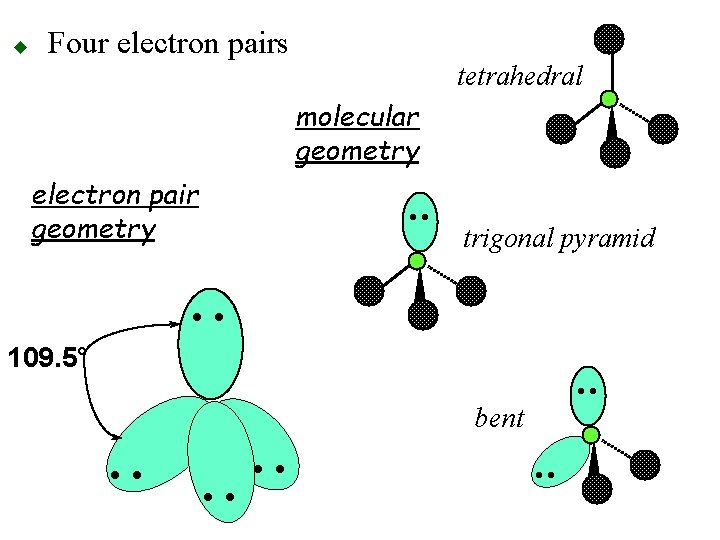
u Four electron pairs tetrahedral molecular geometry electron pair geometry • • trigonal pyramid • • 109. 5° • • bent • •

u Five Pairs oelectron pair geometry trigonal bipyramid obonding pairs 2 5 4 3 onon-bonding pairs 3 0 1 2 seesaw T- omolecular geometry shp. Lin. tbp
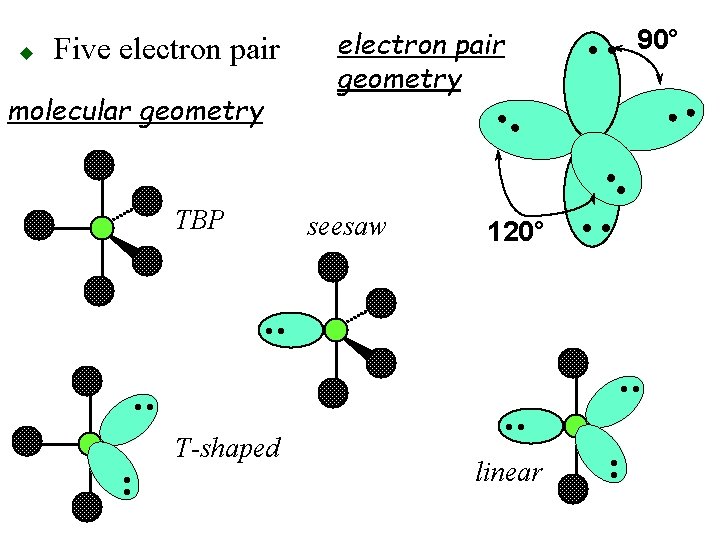
Five electron pair molecular geometry electron pair geometry 90° • • TBP seesaw 120° • • • • T-shaped • • linear • • u

u Six Pairs oelectron pair geometry octahedral obonding pairs 4 2 6 5 onon-bonding pairs 2 4 0 1 omolecular geometry sq. pl. lin. oct sq. pyr.
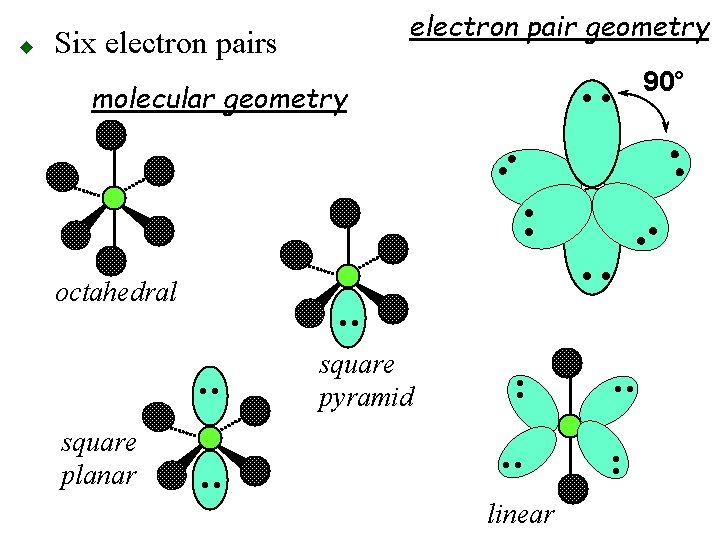
molecular geometry 90° • • Six electron pairs • • octahedral • • linear • • square planar • • square pyramid • • u electron pair geometry

Molecular Polarity u Molecules are always non-polar if all covalent bonds are non-polar o. N 2, P 4, Cl 2 u Molecules with polar bonds can be polar or nonpolar o. H - Cl polar bond, polar molecular o. O=C=O two polar bonds but total molecule is non-polar
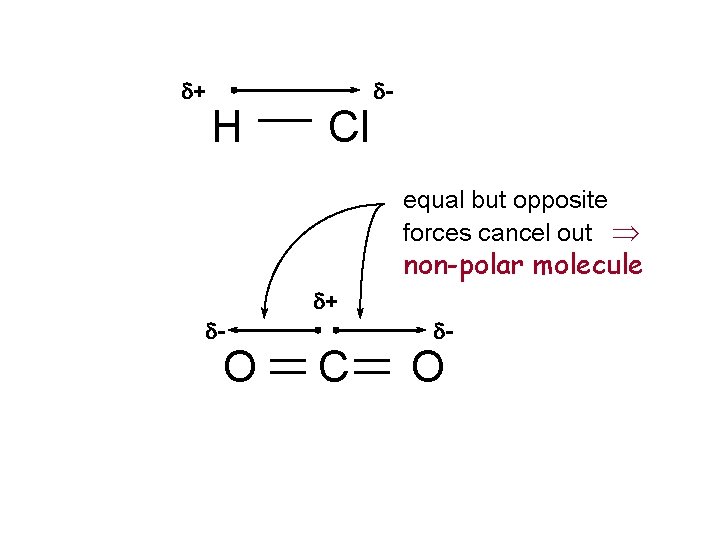
d+ H Cl d- equal but opposite forces cancel out Þ non-polar molecule d+ d- O C d- O
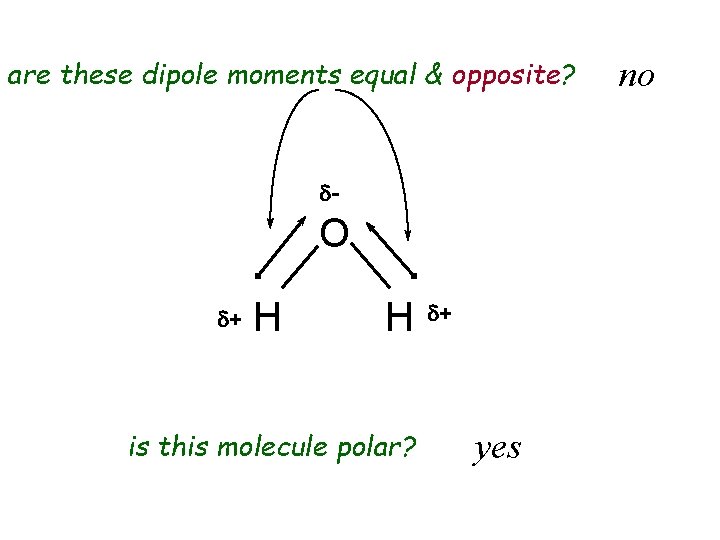
are these dipole moments equal & opposite? d- O d+ H H is this molecule polar? d+ yes no
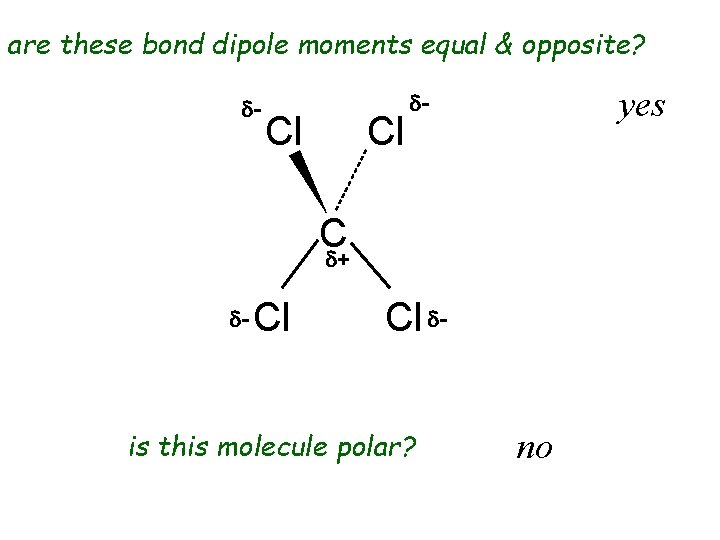
are these bond dipole moments equal & opposite? d- Cl Cl yes d- C d+ d- Cl Cl d- is this molecule polar? no
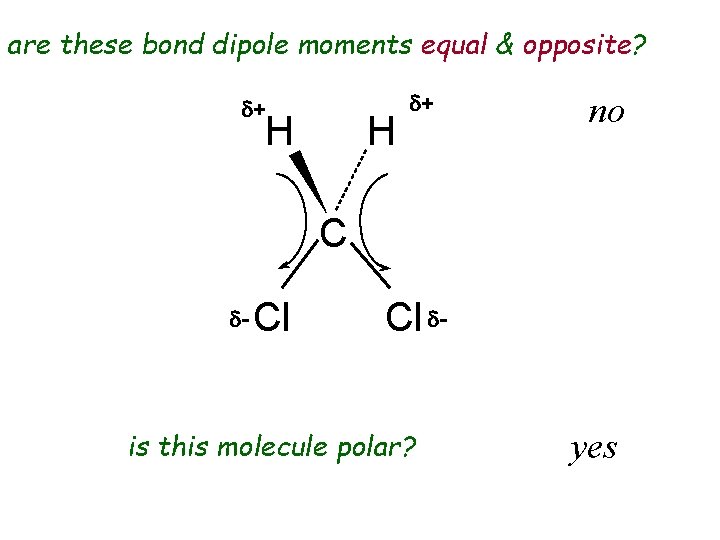
are these bond dipole moments equal & opposite? d+ H H d+ no C d- Cl Cl d- is this molecule polar? yes
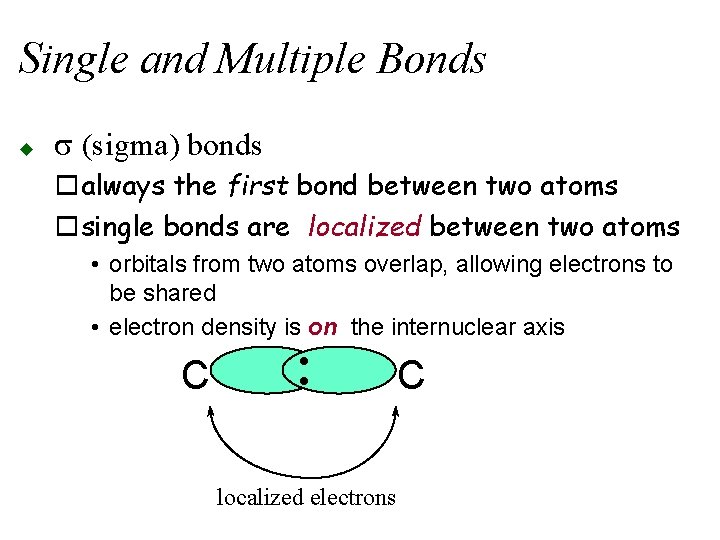
Single and Multiple Bonds u s (sigma) bonds oalways the first bond between two atoms osingle bonds are localized between two atoms • orbitals from two atoms overlap, allowing electrons to be shared • electron density is on the internuclear axis • • C localized electrons C
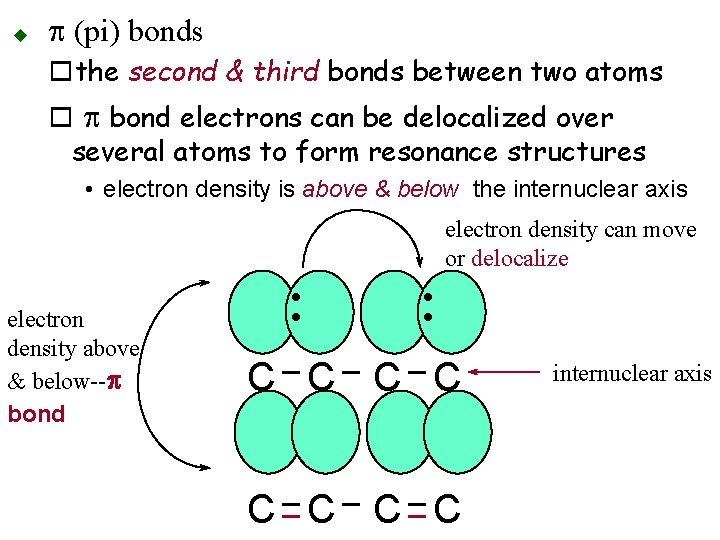
u p (pi) bonds othe second & third bonds between two atoms o p bond electrons can be delocalized over several atoms to form resonance structures • electron density is above & below the internuclear axis electron density can move or delocalize • • electron density above & below--p bond C C C C internuclear axis

Hybridization u u allows for greater number of bonds types of hybridization osp mixing of one s orbital & one p orbital • ¯ 2 s 2 p sp p osp 2 mixing of one s orbital & two p orbitals • ¯ 2 s 2 p sp 2 p osp 3 mixing of one s orbital & three p orbitals • ¯ 2 s 2 p sp 3

u in sp hybridization othe two sp hybrid orbitals form two s bonds with linear geometry oremaining two p orbitals form p bonds u in sp 2 hybridization othe three hybrid orbitals form three s bonds with trigonal planar geometry othe remaining one p orbital forms a p bond u in sp 3 hybridization othe four hybrid orbitals form four s bonds with tetrahedral geometry osp 3 hybrid atoms can form no p bonds as they have no unhybridized p orbitals

Molecular Orbitals u mathematical combinations of atomic orbitals odelocalized over whole molecule on atomic orbitals produce n molecular orbitals • ½ are bonding orbitals and ½ are antibonding orbitals u bond order o# bonding electrons - # antibonding electron 2
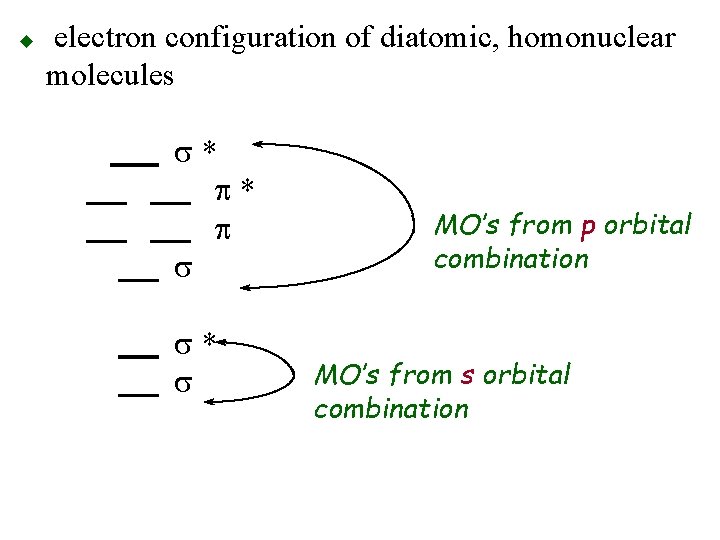
u electron configuration of diatomic, homonuclear molecules s* p* p s s* s MO’s from p orbital combination MO’s from s orbital combination
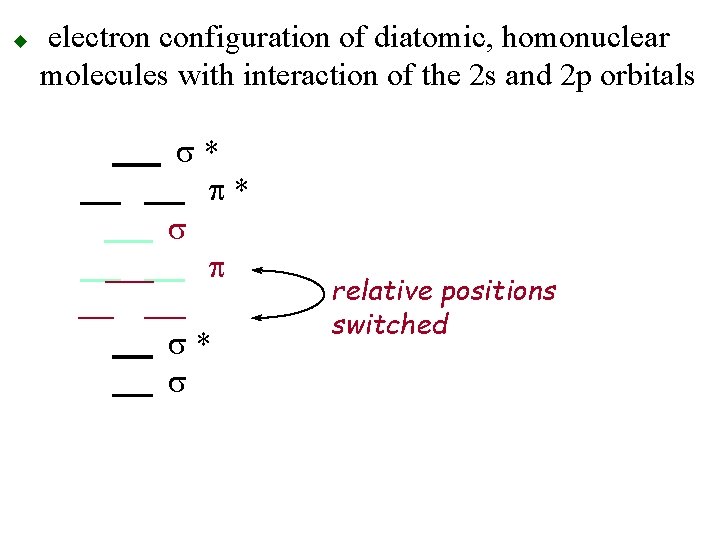
u electron configuration of diatomic, homonuclear molecules with interaction of the 2 s and 2 p orbitals s* p* s p s* s relative positions switched
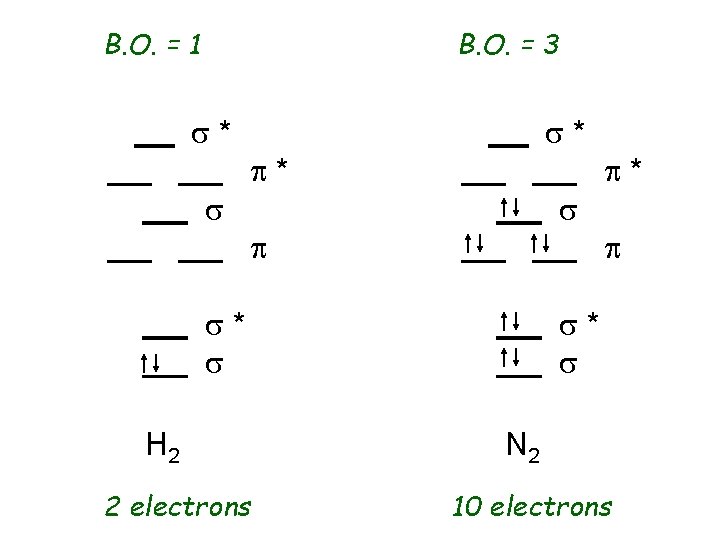
B. O. = 1 B. O. = 3 s* s s* p* s p s* s H 2 2 electrons p* p s* s N 2 10 electrons
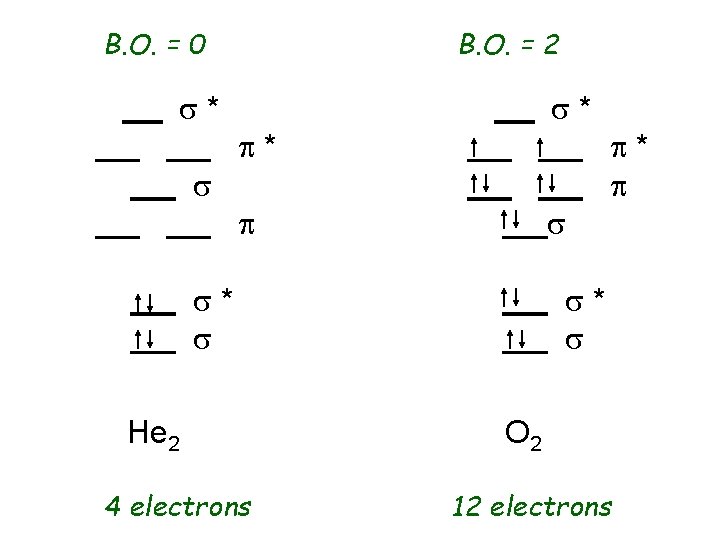
B. O. = 0 s* s B. O. = 2 s* p* p s s* s He 2 4 electrons p* p s* s O 2 12 electrons
- Slides: 35