Chapter 9 Molecular Geometries and Bonding Theories 1

Chapter 9 Molecular Geometries and Bonding Theories 1
Molecular Shapes • The shape of a molecule plays an important role in its reactivity. • By noting the number of bonding and nonbonding electron pairs we can easily predict the shape of 2 the molecule.
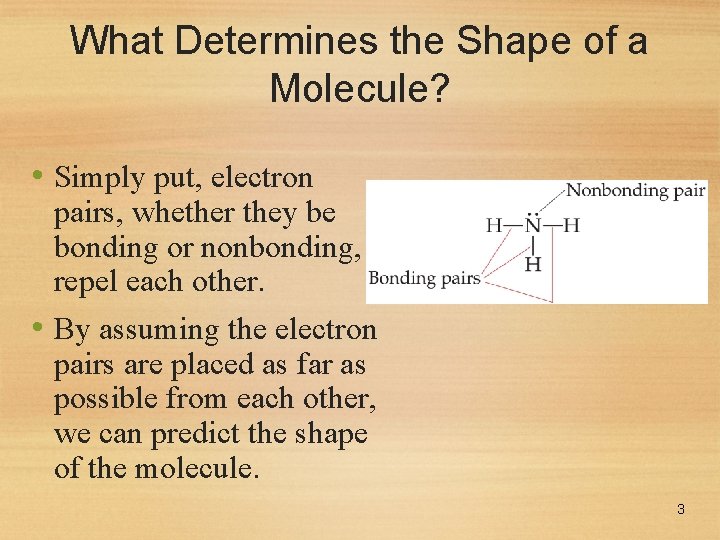
What Determines the Shape of a Molecule? • Simply put, electron pairs, whether they be bonding or nonbonding, repel each other. • By assuming the electron pairs are placed as far as possible from each other, we can predict the shape of the molecule. 3
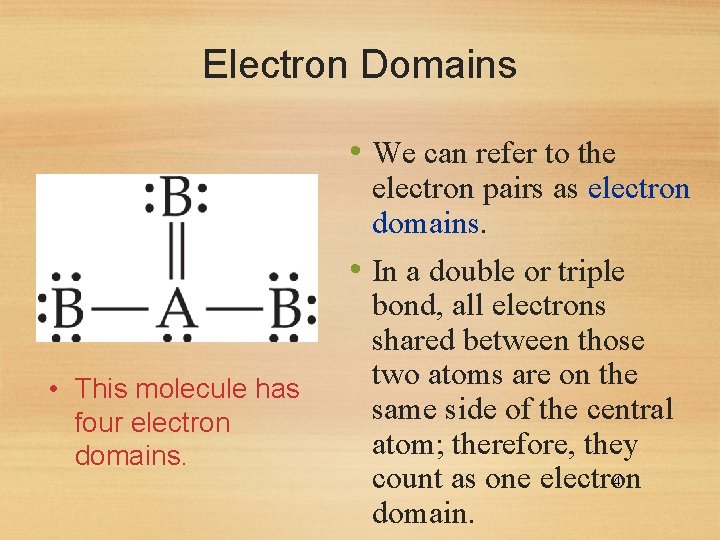
Electron Domains • We can refer to the electron pairs as electron domains. • In a double or triple • This molecule has four electron domains. bond, all electrons shared between those two atoms are on the same side of the central atom; therefore, they 4 count as one electron domain.

Valence Shell Electron Pair Repulsion Theory (VSEPR) The best arrangement of a given number of electron domains is the one that minimizes the repulsions among them. 5
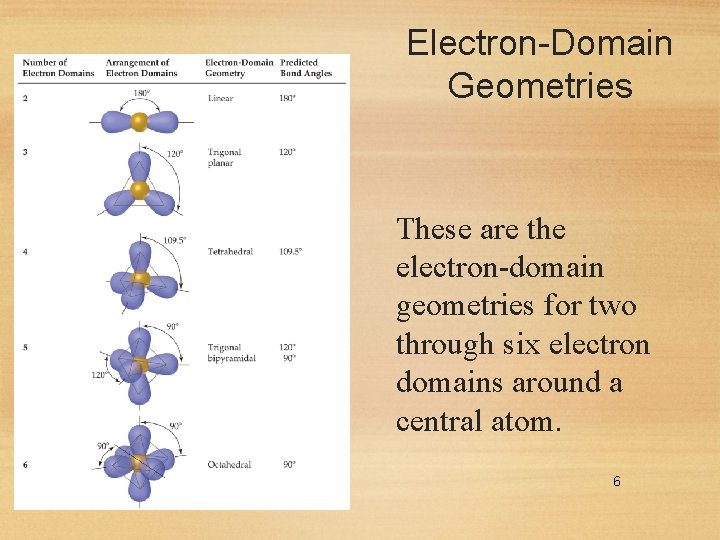
Electron-Domain Geometries These are the electron-domain geometries for two through six electron domains around a central atom. 6

Electron-Domain Geometries • count the number of electron domains in the Lewis structure. • The geometry will be that which corresponds to that number of electron domains. 7
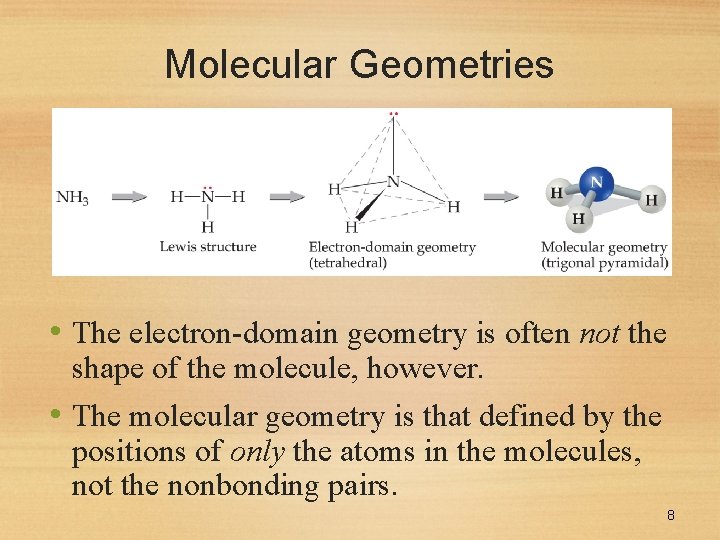
Molecular Geometries • The electron-domain geometry is often not the shape of the molecule, however. • The molecular geometry is that defined by the positions of only the atoms in the molecules, not the nonbonding pairs. 8
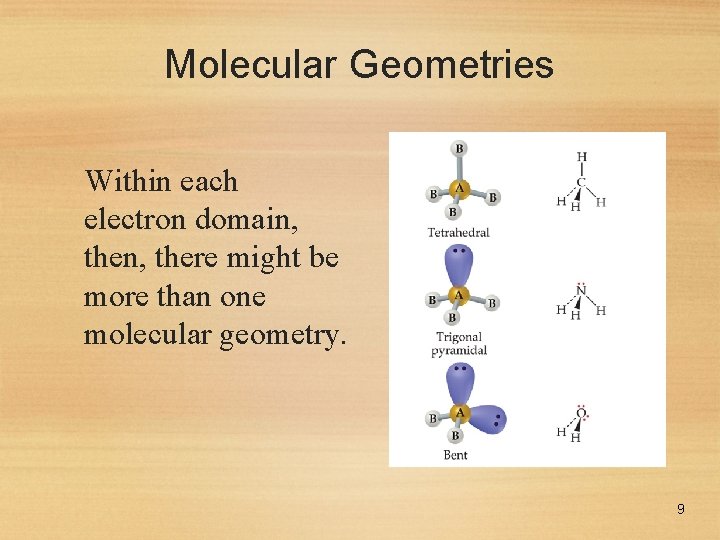
Molecular Geometries Within each electron domain, then, there might be more than one molecular geometry. 9

Linear Electron Domain • In this domain, there is only one molecular geometry: linear. • NOTE: If there are only two atoms in the molecule, the molecule will be linear no matter what the electron domain is. 10
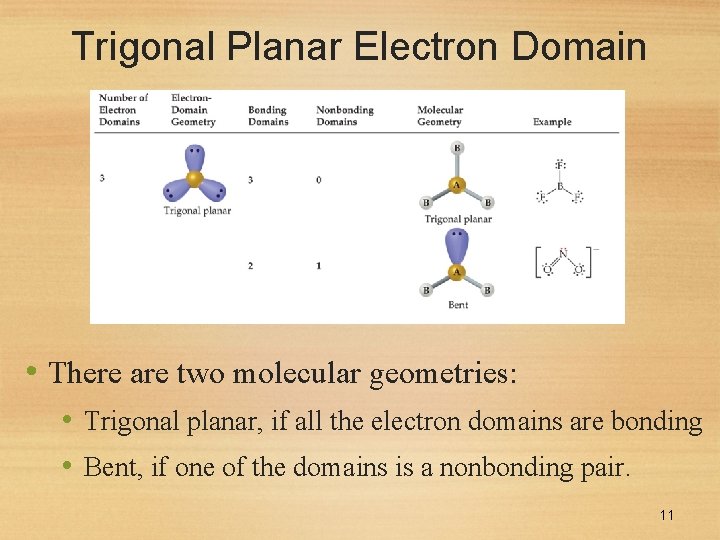
Trigonal Planar Electron Domain • There are two molecular geometries: • Trigonal planar, if all the electron domains are bonding • Bent, if one of the domains is a nonbonding pair. 11
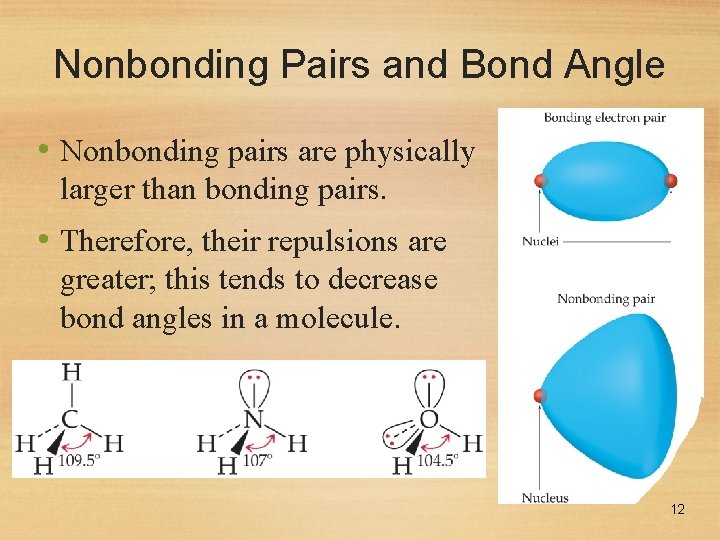
Nonbonding Pairs and Bond Angle • Nonbonding pairs are physically larger than bonding pairs. • Therefore, their repulsions are greater; this tends to decrease bond angles in a molecule. 12
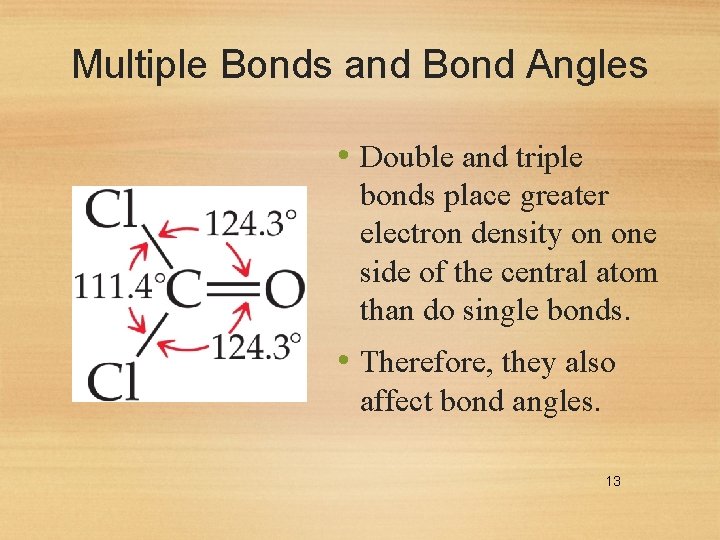
Multiple Bonds and Bond Angles • Double and triple bonds place greater electron density on one side of the central atom than do single bonds. • Therefore, they also affect bond angles. 13

Tetrahedral Electron Domain • There are three molecular geometries: • Tetrahedral, if all are bonding pairs • Trigonal pyramidal if one is a nonbonding pair • Bent if there are two nonbonding pairs 14

Trigonal Bipyramidal Electron Domain • There are two distinct positions in this geometry: • Axial • Equatorial 15
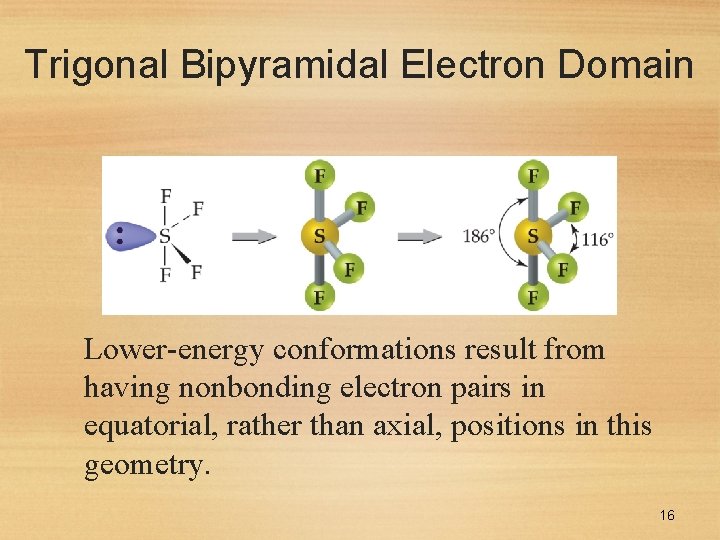
Trigonal Bipyramidal Electron Domain Lower-energy conformations result from having nonbonding electron pairs in equatorial, rather than axial, positions in this geometry. 16

Trigonal Bipyramidal Electron Domain • There are four distinct molecular geometries in this domain: • Trigonal bipyramidal • Seesaw • T-shaped • Linear 17

Octahedral Electron Domain • All positions are equivalent in the octahedral domain. • There are three molecular geometries: • Octahedral • Square pyramidal • Square planar 18
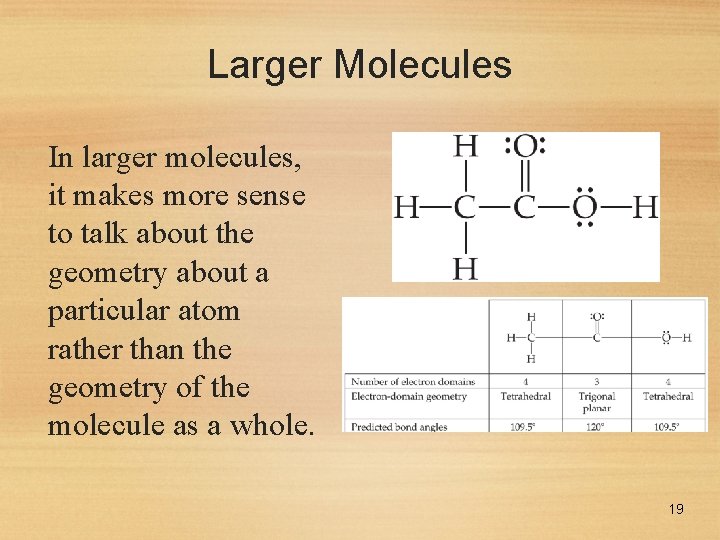
Larger Molecules In larger molecules, it makes more sense to talk about the geometry about a particular atom rather than the geometry of the molecule as a whole. 19

Larger Molecules This approach makes sense, especially because larger molecules tend to react at a particular site in the molecule. 20
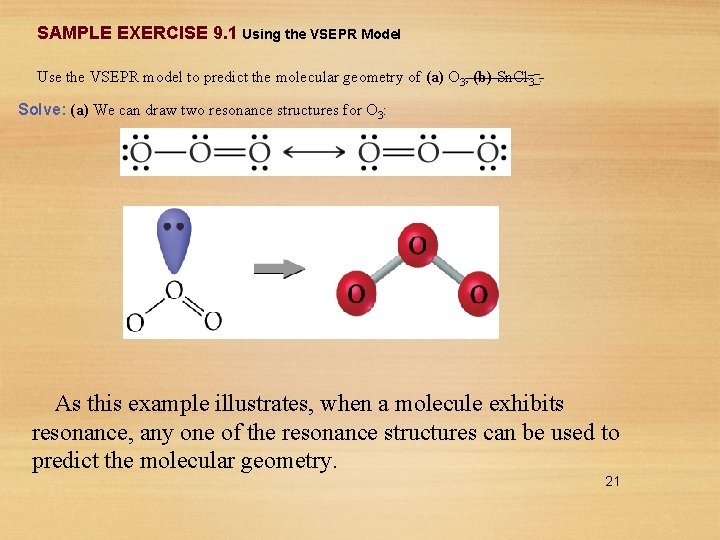
SAMPLE EXERCISE 9. 1 Using the VSEPR Model Use the VSEPR model to predict the molecular geometry of (a) O 3, (b) Sn. Cl 3–. Solve: (a) We can draw two resonance structures for O 3: As this example illustrates, when a molecule exhibits resonance, any one of the resonance structures can be used to predict the molecular geometry. 21
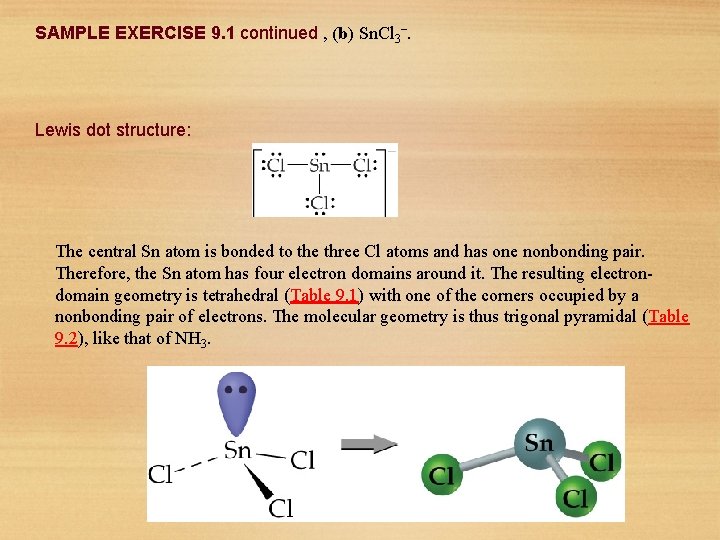
SAMPLE EXERCISE 9. 1 continued , (b) Sn. Cl 3–. Lewis dot structure: The central Sn atom is bonded to the three Cl atoms and has one nonbonding pair. Therefore, the Sn atom has four electron domains around it. The resulting electrondomain geometry is tetrahedral (Table 9. 1) with one of the corners occupied by a nonbonding pair of electrons. The molecular geometry is thus trigonal pyramidal (Table 9. 2), like that of NH 3. 22

PRACTICE EXERCISE Predict the electron-domain geometry and the molecular geometry for (a) Se. Cl 2, (b) CO 32–. 23

SAMPLE EXERCISE 9. 2 Molecular Geometries of Molecules with Expanded Valence Shells Use the VSEPR model to predict the molecular geometry of (a) SF 4, (b) IF 5. 24

PRACTICE EXERCISE Predict the electron-domain geometry and molecular geometry of (a) Cl. F 3, (b) ICl 4–. 25

SAMPLE EXERCISE 9. 3 Predicting Bond Angles Eyedrops for dry eyes usually contain a water-soluble polymer called polyvinyl alcohol, which is based on the unstable organic molecule called vinyl alcohol: Predict the approximate values for the H—O—C and O —C—C bond angles in vinyl alcohol. 26

SAMPLE EXERCISE 9. 3 continued PRACTICE EXERCISE Predict the H—C—H and C—C—C bond angles in the following molecule, called propyne: 27

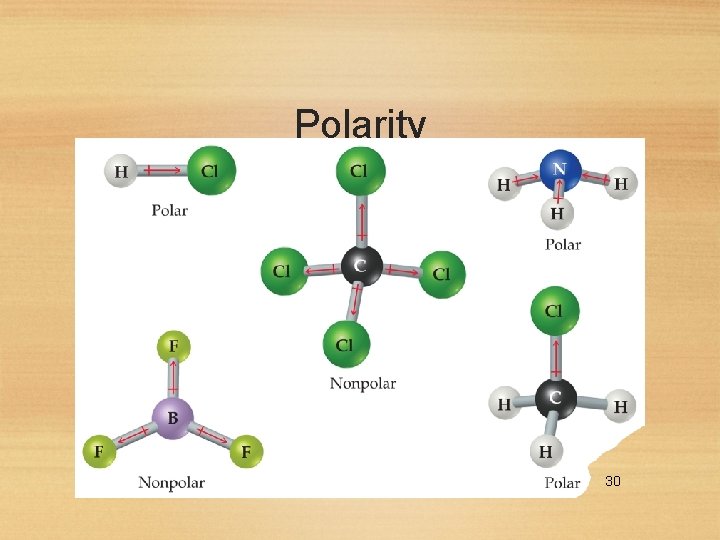
Polarity • In Chapter 8 we discussed bond dipoles. • But just because a molecule possesses polar bonds does not mean the molecule as a whole will be polar. 28

Polarity By adding the individual bond dipoles, one can determine the overall dipole moment for the molecule. 29
Polarity 30

SAMPLE EXERCISE 9. 4 Polarity of Molecules Predict whether the following molecules are polar or nonpolar: (a) Br. Cl, (b) SO 2, (c) SF 6. 31

PRACTICE EXERCISE Determine whether the following molecules are polar or nonpolar: (a) NF 3, (b) BCl 3. 32
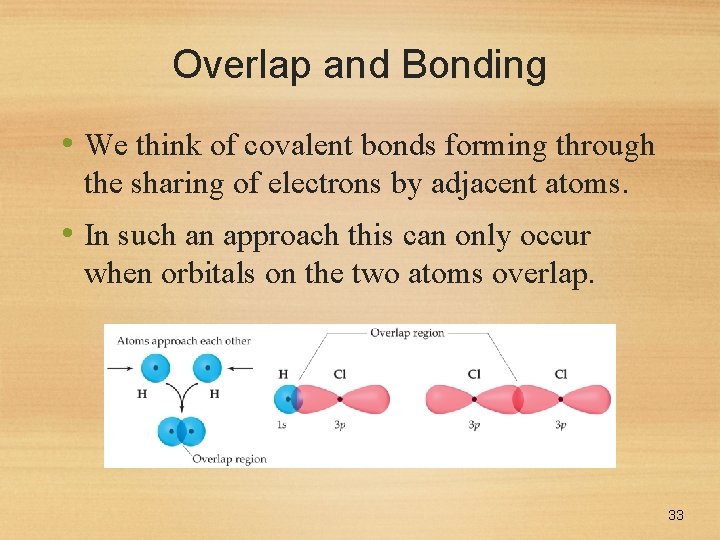
Overlap and Bonding • We think of covalent bonds forming through the sharing of electrons by adjacent atoms. • In such an approach this can only occur when orbitals on the two atoms overlap. 33
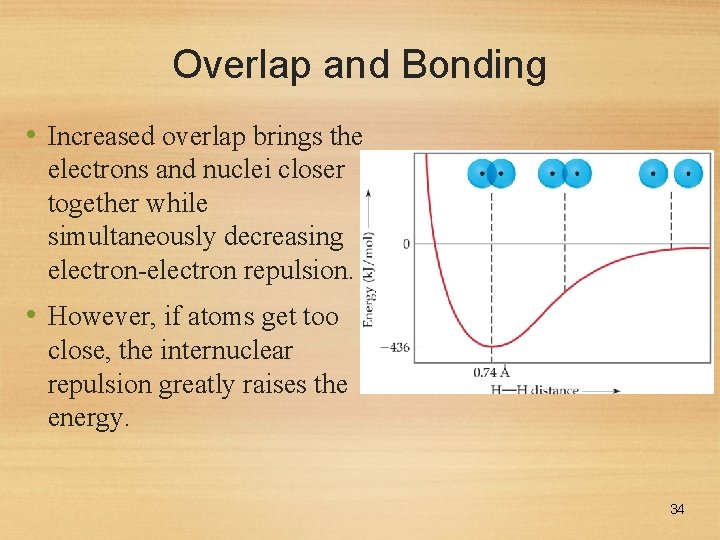
Overlap and Bonding • Increased overlap brings the electrons and nuclei closer together while simultaneously decreasing electron-electron repulsion. • However, if atoms get too close, the internuclear repulsion greatly raises the energy. 34
Hybrid Orbitals But it’s hard to imagine tetrahedral, trigonal bipyramidal, and other geometries arising from the atomic orbitals we recognize. 35

Hybrid Orbitals • Consider beryllium: • In its ground electronic state, it would not be able to form bonds because it has no singly-occupied orbitals. 36

Hybrid Orbitals But if it absorbs the small amount of energy needed to promote an electron from the 2 s to the 2 p orbital, it can form two bonds. 37

Hybrid Orbitals • Mixing the s and p orbitals yields two degenerate orbitals that are hybrids of the two orbitals. • These sp hybrid orbitals have two lobes like a p orbital. • One of the lobes is larger and more rounded as is the s orbital. 38
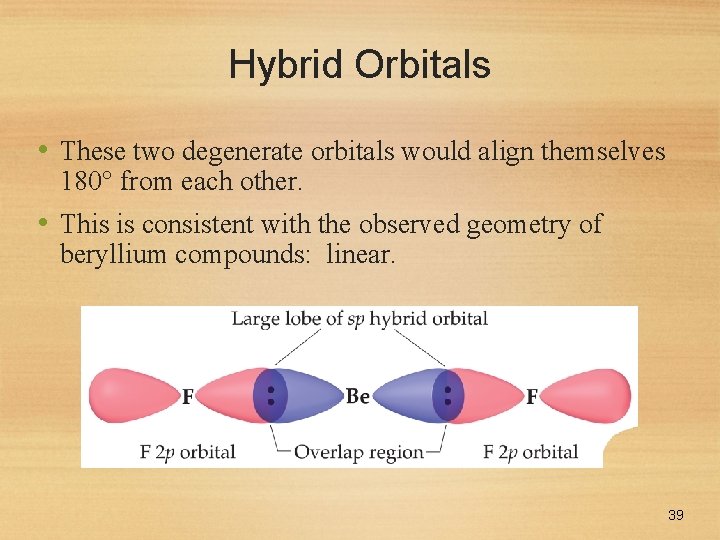
Hybrid Orbitals • These two degenerate orbitals would align themselves 180 from each other. • This is consistent with the observed geometry of beryllium compounds: linear. 39
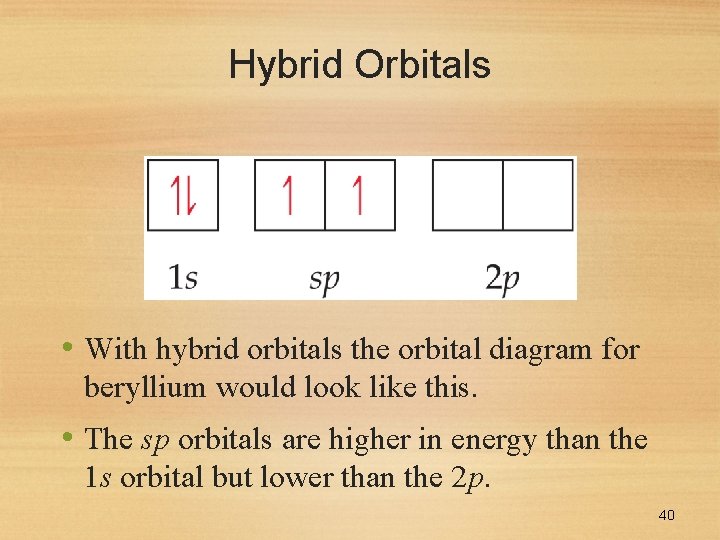
Hybrid Orbitals • With hybrid orbitals the orbital diagram for beryllium would look like this. • The sp orbitals are higher in energy than the 1 s orbital but lower than the 2 p. 40
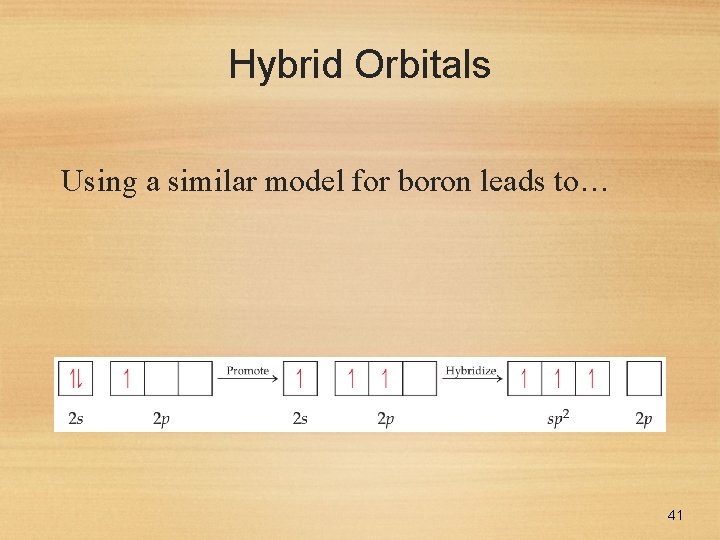
Hybrid Orbitals Using a similar model for boron leads to… 41
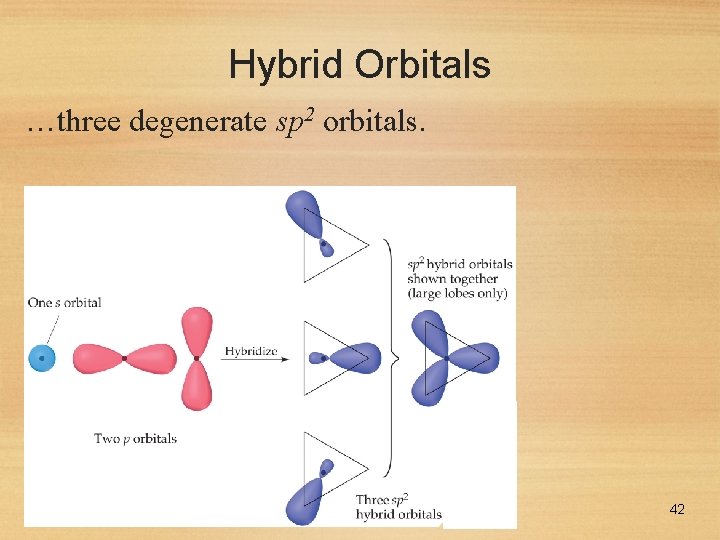
Hybrid Orbitals …three degenerate sp 2 orbitals. 42
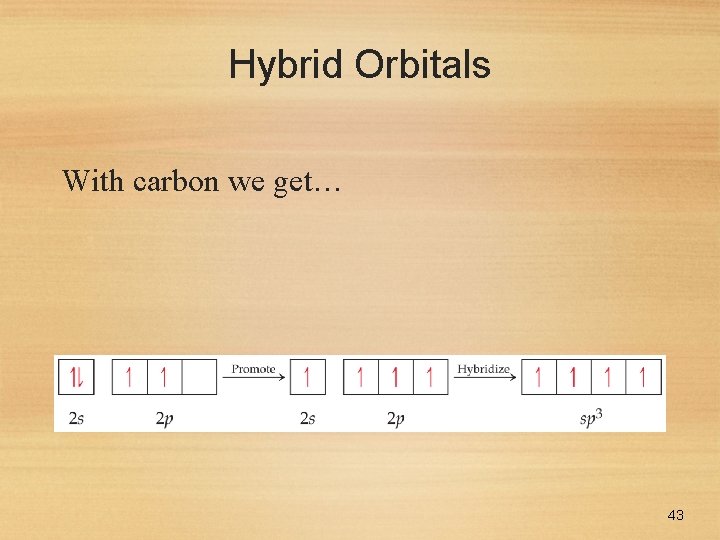
Hybrid Orbitals With carbon we get… 43
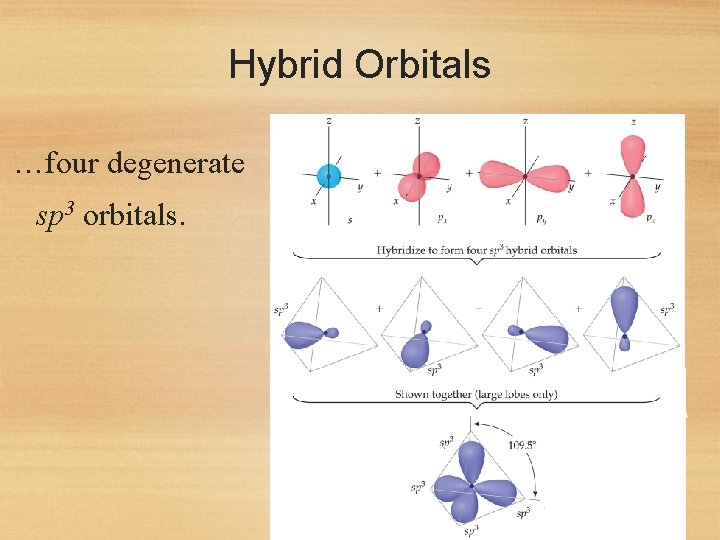
Hybrid Orbitals …four degenerate sp 3 orbitals. 44
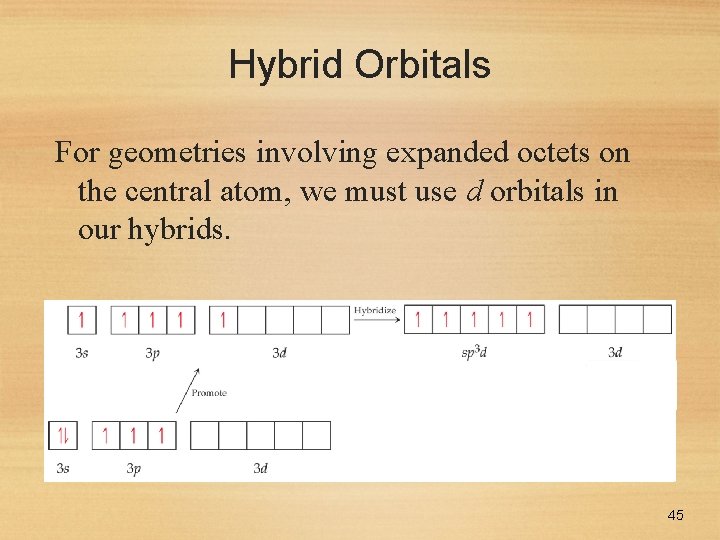
Hybrid Orbitals For geometries involving expanded octets on the central atom, we must use d orbitals in our hybrids. 45
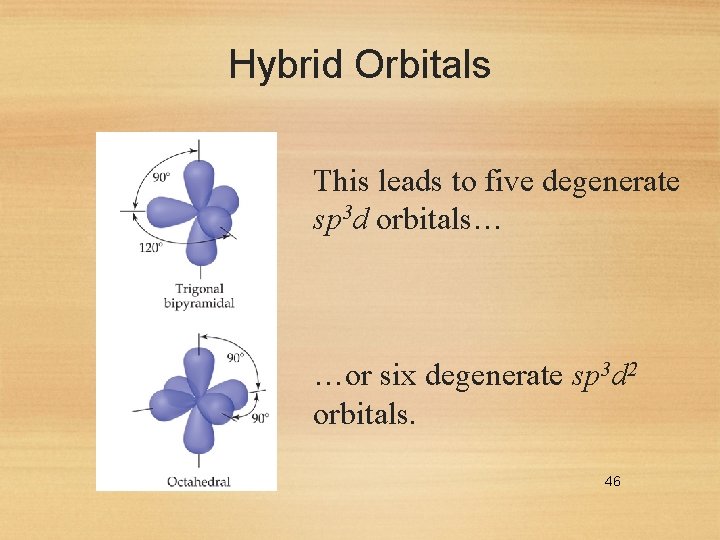
Hybrid Orbitals This leads to five degenerate sp 3 d orbitals… …or six degenerate sp 3 d 2 orbitals. 46

Hybrid Orbitals Once you know the electron-domain geometry, you know the hybridization state of the atom. 47

SAMPLE EXERCISE 9. 5 Hybridization Indicate the hybridization of orbitals employed by the central atom in (a) NH 2–, (b) SF 4 (see Sample Exercise 9. 2). PRACTICE EXERCISE Predict the electron-domain geometry and the hybridization of the central atom in (a) SO 32–, (b) SF 6. 48

Valence Bond Theory • Hybridization is a major player in this approach to bonding. • There are two ways orbitals can overlap to form bonds between atoms. 49
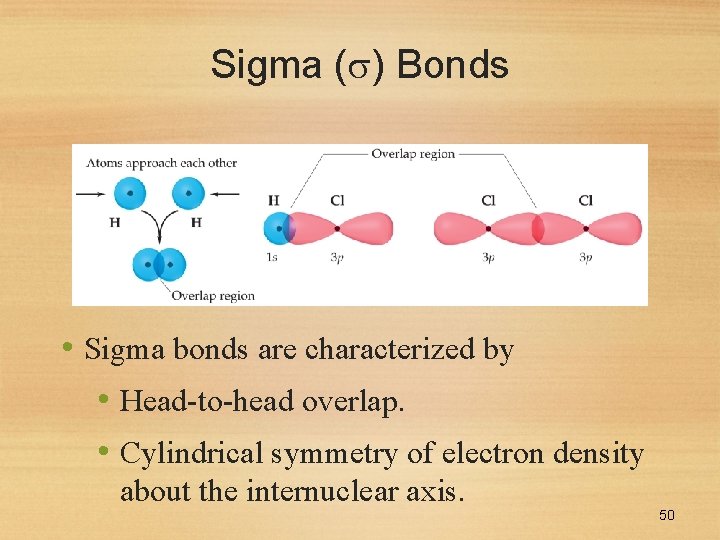
Sigma ( ) Bonds • Sigma bonds are characterized by • Head-to-head overlap. • Cylindrical symmetry of electron density about the internuclear axis. 50
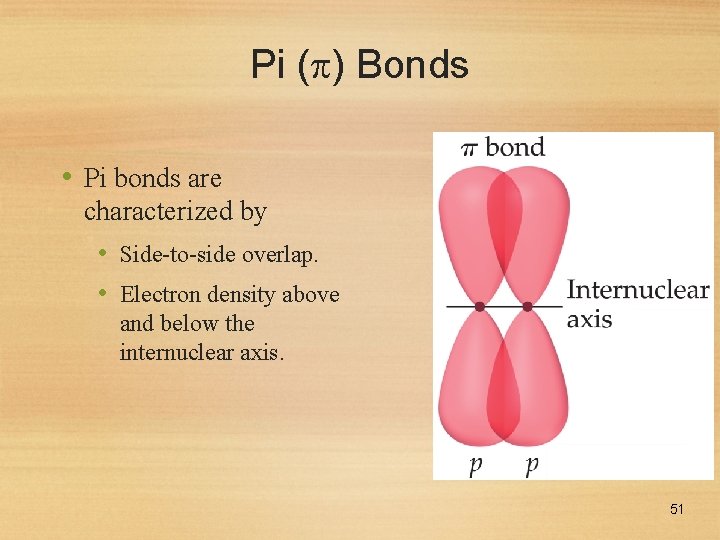
Pi ( ) Bonds • Pi bonds are characterized by • Side-to-side overlap. • Electron density above and below the internuclear axis. 51

Single Bonds Single bonds are always bonds, because overlap is greater, resulting in a stronger bond and more energy lowering. 52
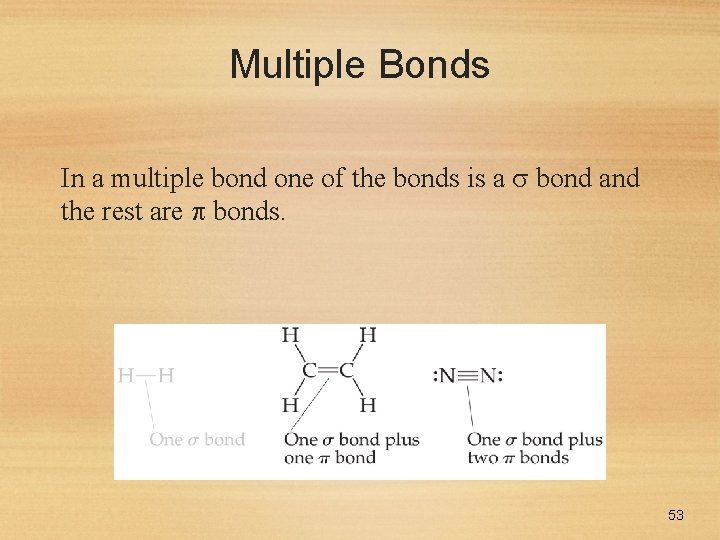
Multiple Bonds In a multiple bond one of the bonds is a bond and the rest are bonds. 53
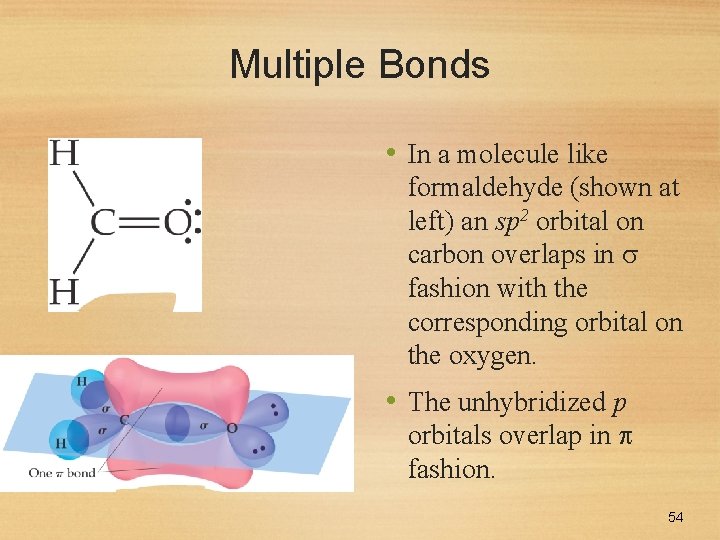
Multiple Bonds • In a molecule like formaldehyde (shown at left) an sp 2 orbital on carbon overlaps in fashion with the corresponding orbital on the oxygen. • The unhybridized p orbitals overlap in fashion. 54
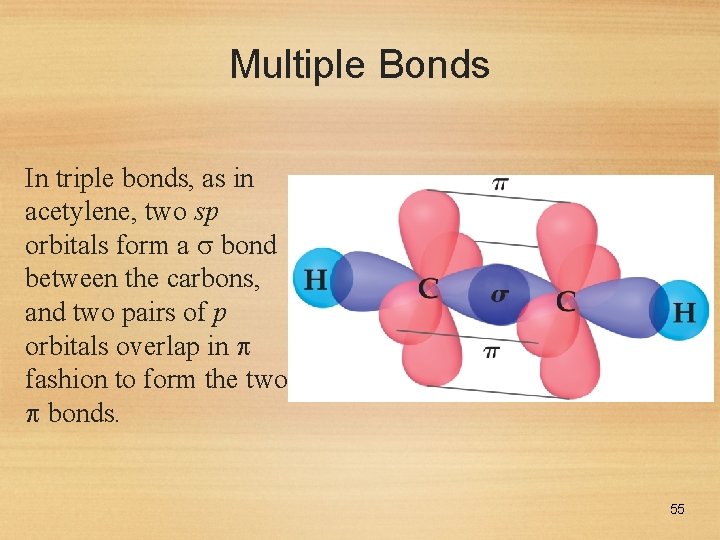
Multiple Bonds In triple bonds, as in acetylene, two sp orbitals form a bond between the carbons, and two pairs of p orbitals overlap in fashion to form the two bonds. 55

Delocalized Electrons: Resonance When writing Lewis structures for species like the nitrate ion, we draw resonance structures to more accurately reflect the structure of the molecule or ion. 56
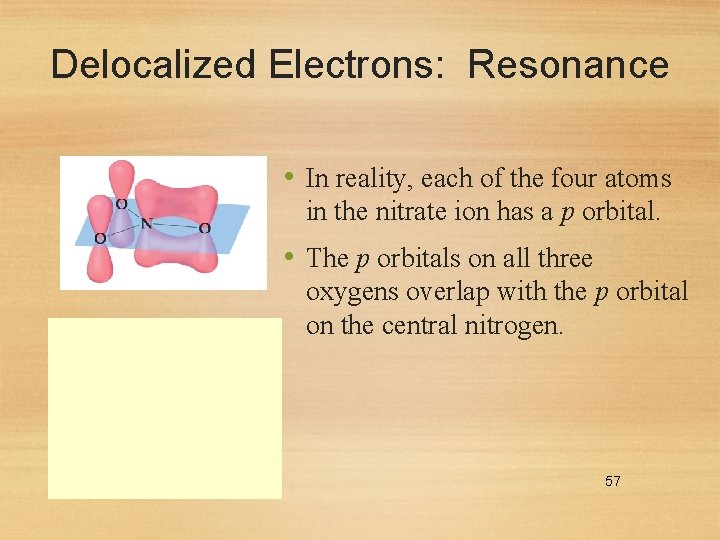
Delocalized Electrons: Resonance • In reality, each of the four atoms in the nitrate ion has a p orbital. • The p orbitals on all three oxygens overlap with the p orbital on the central nitrogen. 57
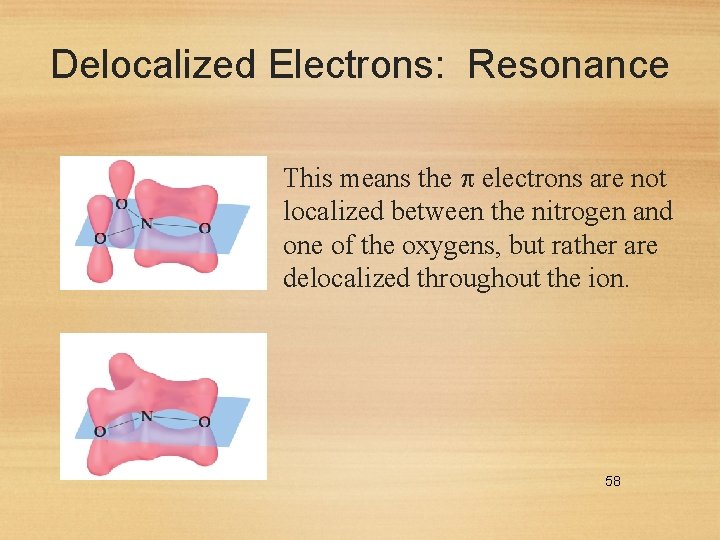
Delocalized Electrons: Resonance This means the electrons are not localized between the nitrogen and one of the oxygens, but rather are delocalized throughout the ion. 58
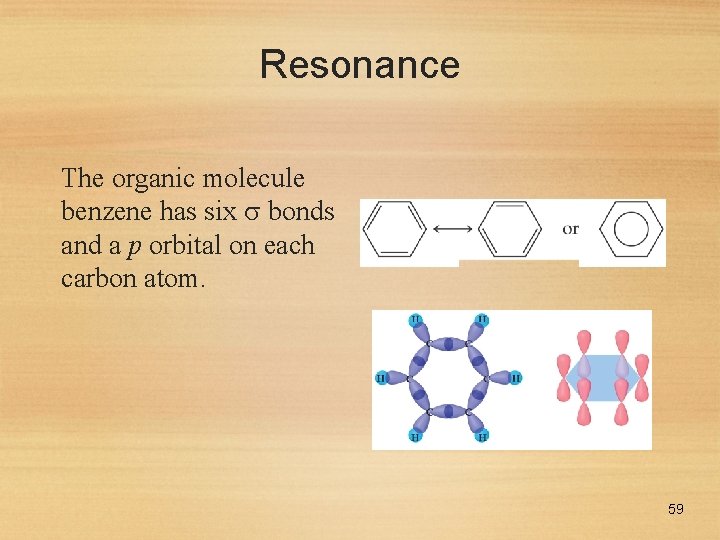
Resonance The organic molecule benzene has six bonds and a p orbital on each carbon atom. 59

Resonance • In reality the electrons in benzene are not localized, but delocalized. • The even distribution of the electrons in benzene makes the molecule unusually stable. 60

SAMPLE EXERCISE 9. 6 Describing and Bonds in a Molecule Formaldehyde has the Lewis structure Describe how the bonds in formaldehyde are formed in terms of overlaps of appropriate hybridized and unhybridized orbitals. 61
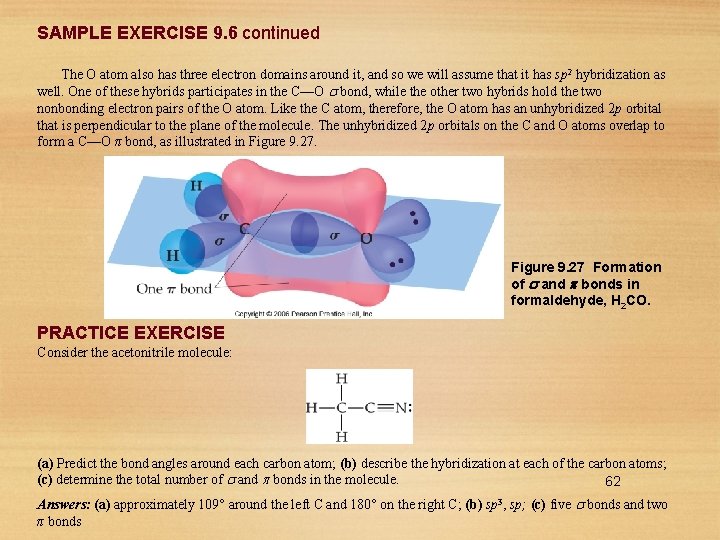
SAMPLE EXERCISE 9. 6 continued The O atom also has three electron domains around it, and so we will assume that it has sp 2 hybridization as well. One of these hybrids participates in the C—O bond, while the other two hybrids hold the two nonbonding electron pairs of the O atom. Like the C atom, therefore, the O atom has an unhybridized 2 p orbital that is perpendicular to the plane of the molecule. The unhybridized 2 p orbitals on the C and O atoms overlap to form a C—O π bond, as illustrated in Figure 9. 27 Formation of and bonds in formaldehyde, H 2 CO. PRACTICE EXERCISE Consider the acetonitrile molecule: (a) Predict the bond angles around each carbon atom; (b) describe the hybridization at each of the carbon atoms; (c) determine the total number of and bonds in the molecule. 62 Answers: (a) approximately 109° around the left C and 180° on the right C; (b) sp 3, sp; (c) five bonds and two π bonds

SAMPLE EXERCISE 9. 7 Delocalized Bonding Describe the bonding in the nitrate ion, NO 3–. Does this ion have delocalized bonds? Solution Analyze: Given the chemical formula for a polyatomic anion, we are asked to describe the bonding and determine whether the ion has delocalized bonds. Plan: Our first step in describing the bonding in NO 3– is to construct appropriate Lewis structures. If there are multiple resonance structures that involve the placement of the double bonds in different locations, that suggests that the component of the double bonds is delocalized. Solve: In Section 8. 6 we saw that NO 3– has three resonance structures: In each of these structures the electron-domain geometry at nitrogen is trigonal planar, which implies sp 2 hybridization of the N atom. The sp 2 hybrid orbitals are used to construct the three N—O bonds that are present in each of the resonance structures. 63
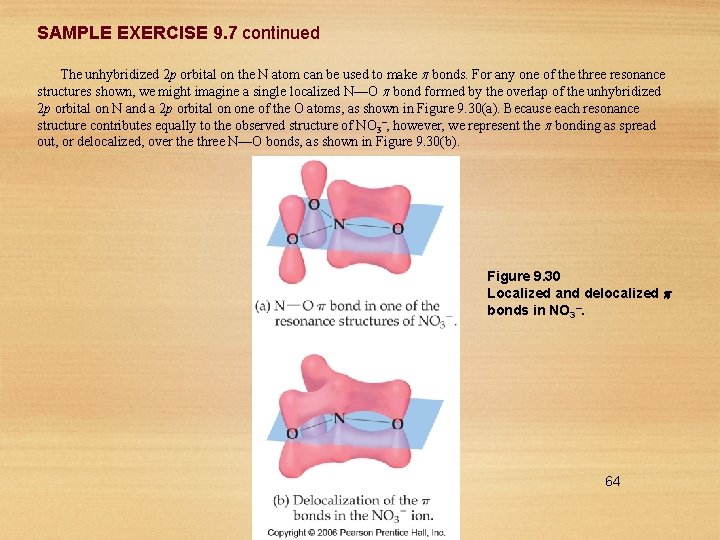
SAMPLE EXERCISE 9. 7 continued The unhybridized 2 p orbital on the N atom can be used to make bonds. For any one of the three resonance structures shown, we might imagine a single localized N––O bond formed by the overlap of the unhybridized 2 p orbital on N and a 2 p orbital on one of the O atoms, as shown in Figure 9. 30(a). Because each resonance structure contributes equally to the observed structure of NO 3–, however, we represent the bonding as spread out, or delocalized, over the three N—O bonds, as shown in Figure 9. 30(b). Figure 9. 30 Localized and delocalized bonds in NO 3–. 64

SAMPLE EXERCISE 9. 7 continued PRACTICE EXERCISE Which of the following molecules or ions will exhibit delocalized bonding: SO 3, SO 32–, H 2 CO, O 3, NH 4+? 65
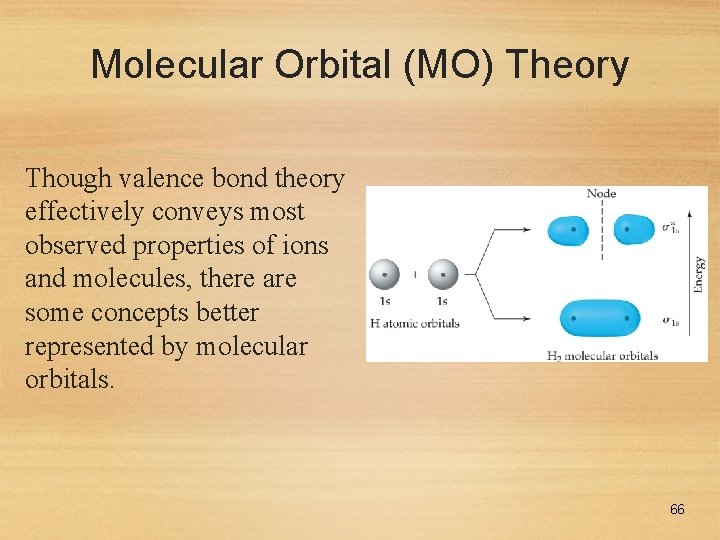
Molecular Orbital (MO) Theory Though valence bond theory effectively conveys most observed properties of ions and molecules, there are some concepts better represented by molecular orbitals. 66
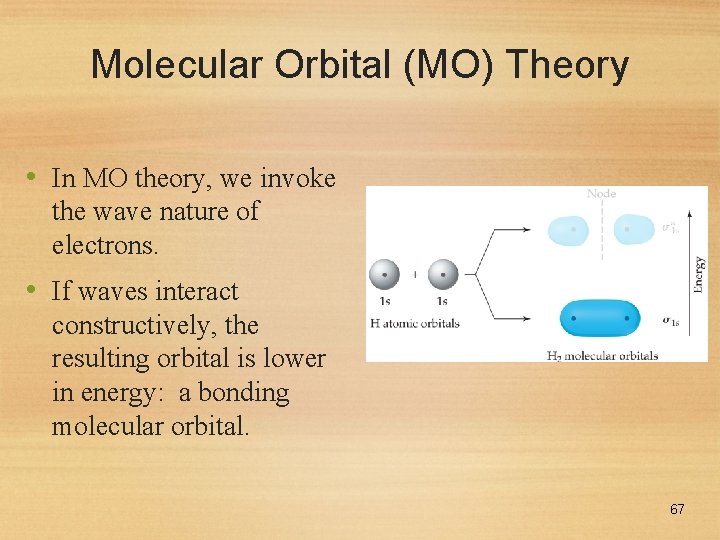
Molecular Orbital (MO) Theory • In MO theory, we invoke the wave nature of electrons. • If waves interact constructively, the resulting orbital is lower in energy: a bonding molecular orbital. 67
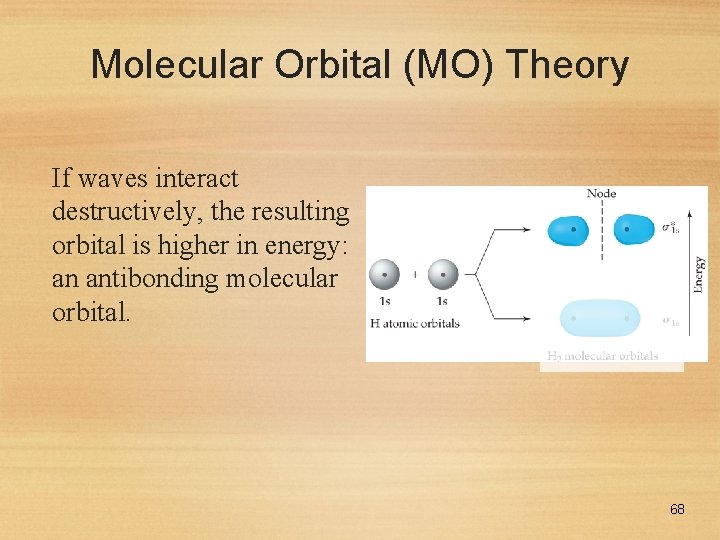
Molecular Orbital (MO) Theory If waves interact destructively, the resulting orbital is higher in energy: an antibonding molecular orbital. 68
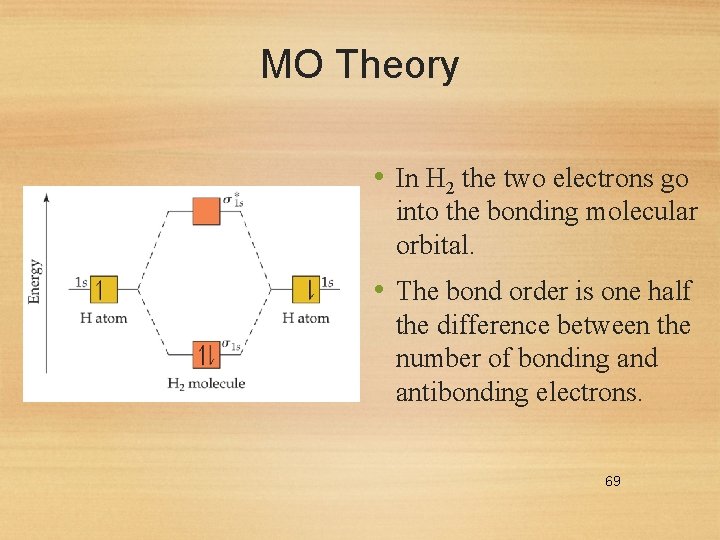
MO Theory • In H 2 the two electrons go into the bonding molecular orbital. • The bond order is one half the difference between the number of bonding and antibonding electrons. 69
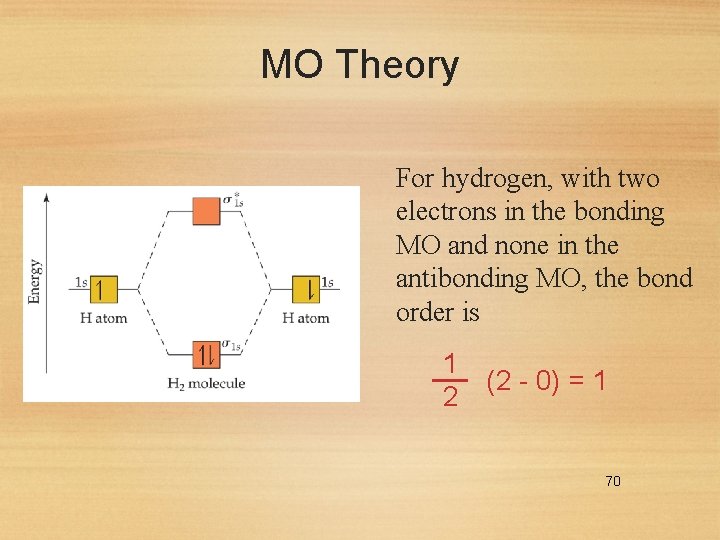
MO Theory For hydrogen, with two electrons in the bonding MO and none in the antibonding MO, the bond order is 1 (2 - 0) = 1 2 70
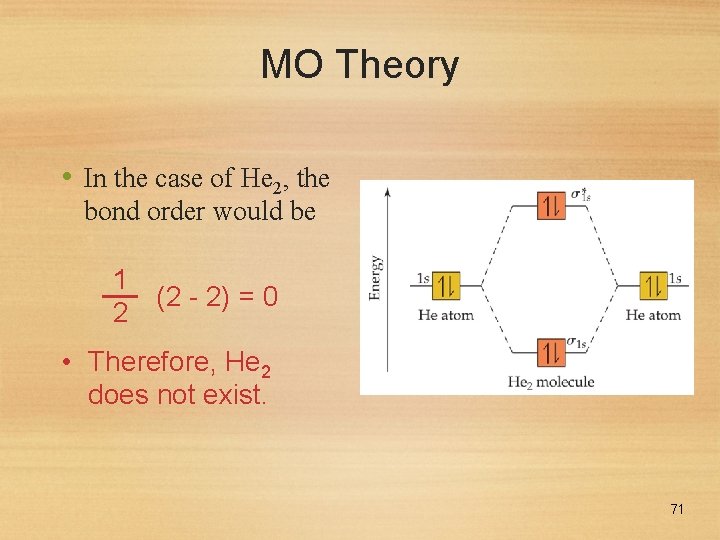
MO Theory • In the case of He 2, the bond order would be 1 (2 - 2) = 0 2 • Therefore, He 2 does not exist. 71
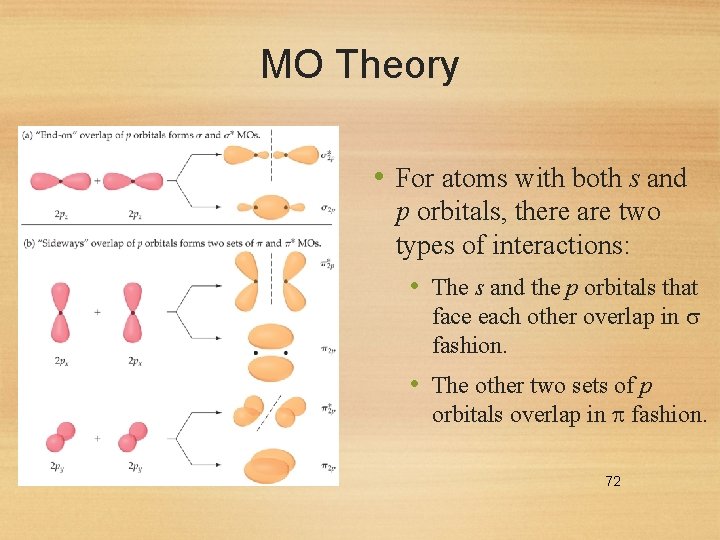
MO Theory • For atoms with both s and p orbitals, there are two types of interactions: • The s and the p orbitals that face each other overlap in fashion. • The other two sets of p orbitals overlap in fashion. 72
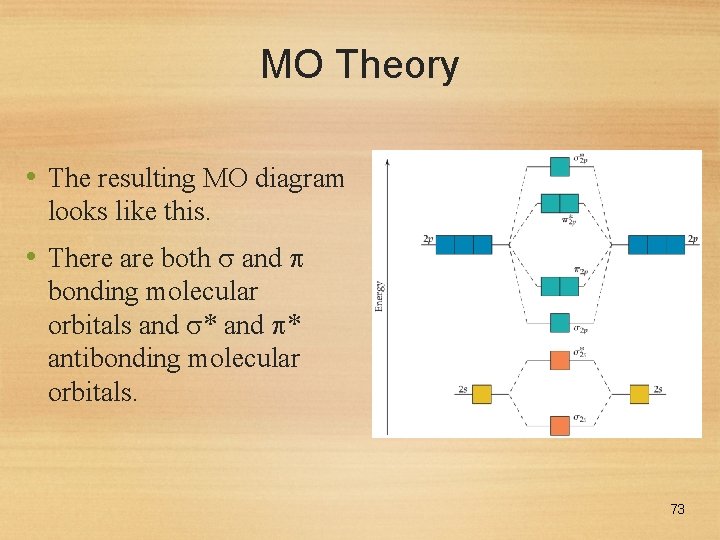
MO Theory • The resulting MO diagram looks like this. • There are both and bonding molecular orbitals and * antibonding molecular orbitals. 73
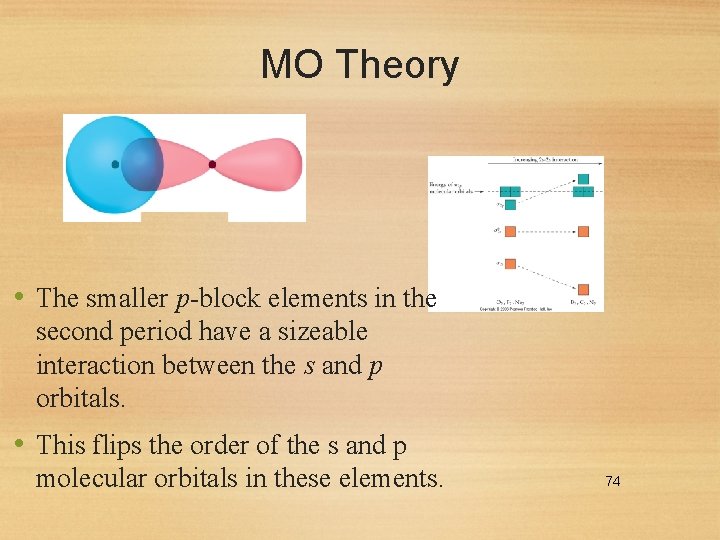
MO Theory • The smaller p-block elements in the second period have a sizeable interaction between the s and p orbitals. • This flips the order of the s and p molecular orbitals in these elements. 74
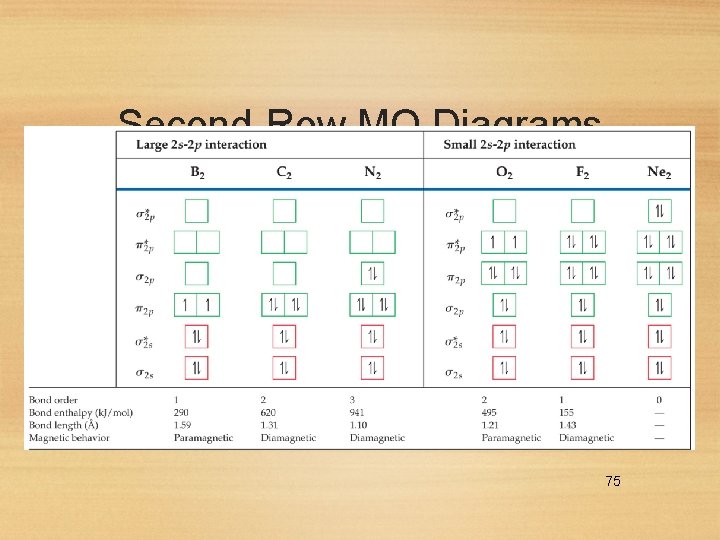
Second-Row MO Diagrams 75

SAMPLE EXERCISE 9. 8 Bond Order What is the bond order of the He 2+ ion? Would you expect this ion to be stable relative to the separated He atom and He+ ion? Solution Analyze: We will determine the bond order for the He 2+ ion and use it to predict whether the ion is stable. Plan: To determine the bond order, we must determine the number of electrons in the molecule and how these electrons populate the available MOs. The valence electrons of He are in the 1 s orbital, and the 1 s orbitals combine to give an MO diagram like that for H 2 or He 2 (Figure 9. 35). If the bond order is greater than 0, we expect a bond to exist and the ion is stable. Answer: The energy-level diagram for the He 2+ ion is shown in Figure 9. 36. This ion has three electrons. Two are placed in the bonding orbital, the third in the antibonding orbital. Thus, the bond order is 76
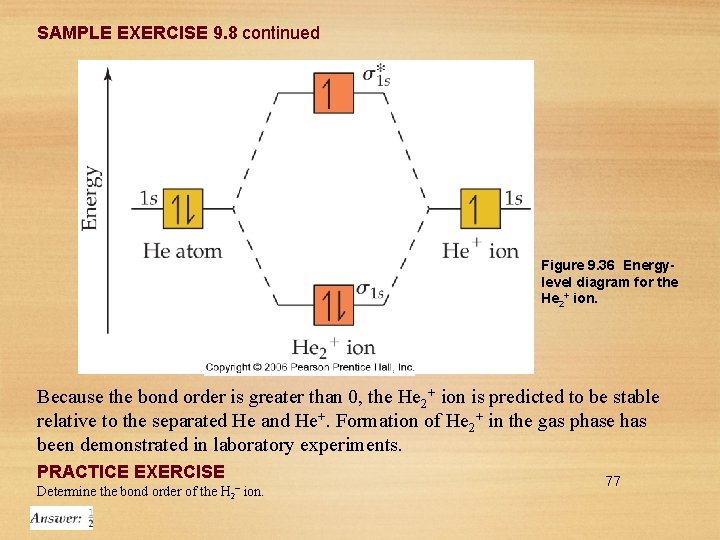
SAMPLE EXERCISE 9. 8 continued Figure 9. 36 Energylevel diagram for the He 2+ ion. Because the bond order is greater than 0, the He 2+ ion is predicted to be stable relative to the separated He and He+. Formation of He 2+ in the gas phase has been demonstrated in laboratory experiments. PRACTICE EXERCISE Determine the bond order of the H 2– ion. 77

SAMPLE EXERCISE 9. 9 Molecular Orbitals of a Second-Row Diatomic Ion Predict the following properties of O 2+: (a) number of unpaired electrons, (b) bond order, (c) bond enthalpy and bond length. PRACTICE EXERCISE Predict the magnetic properties and bond orders of (a) the peroxide ion, O 22–; (b) the acetylide ion, C 22–. 78

SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together Elemental sulfur is a yellow solid that consists of S 8 molecules. The structure of the S 8 molecule is a puckered eight-membered ring (Figure 7. 30). Heating elemental sulfur to high temperatures produces gaseous S 2 molecules: Solution (a) Sulfur is a group 16 element with an [Ne]3 s 23 p 4 electron configuration. It is expected to be most similar electronically to oxygen (electron configuration, [He]2 s 22 p 4), which is immediately above it in the periodic table. • (Chapter 7, Introduction) (b) The Lewis structure of S 8 is 79

SAMPLE INTEGRATIVE EXERCISE continued There is a single bond between each pair of S atoms and two nonbonding electron pairs on each S atom. Thus, we see four electron domains around each S atom, and we would expect a tetrahedral electron-domain geometry corresponding to sp 3 hybridization. • (Sections 9. 2, 9. 5) Because of the nonbonding pairs, we would expect the S—S—S angles to be somewhat less than 109°, the tetrahedral angle. Experimentally, the S—S—S angle in S 8 is 108°, in good agreement with this prediction. Interestingly, if S 8 were a planar ring (like a stop sign), it would have S—S—S angles of 135°. Instead, the S 8 ring puckers to accommodate the smaller angles dictated by sp 3 hybridization. (c) The MOs of S 2 are entirely analogous to those of O 2, although the MOs for S 2 are constructed from the 3 s and 3 p atomic orbitals of sulfur. Further, S 2 has the same number of valence electrons as O 2. Thus, by analogy to our discussion of O 2, we would expect S 2 to have a bond order of 2 (a double bond) and to be paramagnetic with two unpaired electrons in the molecular orbitals of S 2. • (Section 9. 8) (d) We are considering the reaction in which an S 8 molecule falls apart into four S 2 molecules. From parts (b) and (c), we see that S 8 has S—S single bonds and S 2 has double bonds. During the course of the reaction, therefore, we are breaking eight S––S single bonds and forming four double bonds. We can estimate the enthalpy of the reaction by using Equation 8. 12 and the average bond enthalpies in Table 8. 4: Because Hrxn > 0, the reaction is endothermic. • (Section 5. 4) The very positive value of Hrxn suggests that high temperatures are required to cause the reaction to occur. 80
- Slides: 80