Chapter 9 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 9 Lecture Basic Chemistry Fourth Edition Chapter 9 Chemical Quantities in Reactions 9. 1 Mole Relationships in Chemical Equations Learning Goal Given a quantity in moles of a reactant or product, use the mole– mole factor from the balanced equation to calculate the number of moles of another substance in the reaction. © 2014 Pearson Education, Inc.

Chapter 9 Readiness Key Math Skills • Calculating a Percentage (1. 4 C) Core Chemistry Skills • Counting Significant Figures (2. 3) • Using Significant Figures in Calculations (2. 4) • Writing Conversion Factors from Equalities (2. 6) • Using Conversion Factors (2. 7) © 2014 Pearson Education, Inc.

Chapter 9 Readiness Core Chemistry Skills (continued) • Using Energy Units (3. 4) • Calculating Molar Mass (7. 2) • Using Molar Mass as a Conversion Factor (7. 3) • Balancing a Chemical Equation (8. 2) • Balancing Combustion Reactions (8. 3) © 2014 Pearson Education, Inc.

Law of Conservation of Mass The law of conservation of mass indicates that in an ordinary chemical reaction • matter cannot be created or destroyed • no change in total mass occurs in a reaction • mass of products is equal to mass of reactants © 2014 Pearson Education, Inc.

Law of Conservation of Mass Tarnish (Ag 2 S) forms when silver reacts with sulfur in the air to form silver sulfide. The law of conservation of mass states: 2 Ag(s) + S(s) Ag 2 S(s) 2 mol Ag + 1 mol S = 1 mol Ag 2 S 2(107. 9 g) + 1(32. 07 g) = 1(247. 9 g) In the chemical reaction of Ag and S, the mass of the reactants is the same as the mass of the product, Ag 2 S. © 2014 Pearson Education, Inc.
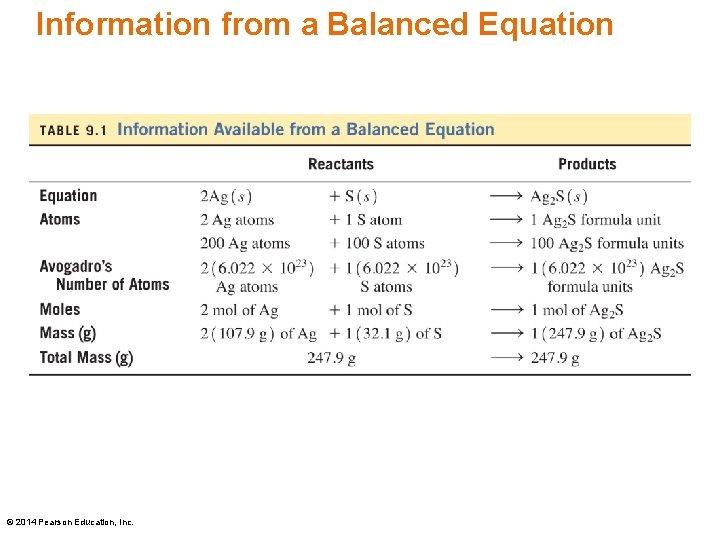
Information from a Balanced Equation © 2014 Pearson Education, Inc.

Mole−Mole Factors from a Balanced Equation A mole−mole factor is a ratio of the moles for two substances in an equation. 2 Fe(s) + Fe and S 3 S(s) Fe and Fe 2 S 3 S and Fe 2 S 3 © 2014 Pearson Education, Inc. Fe 2 S 3(s)

Learning Check Consider the following equation: 3 H 2(g) + N 2(g) 2 NH 3(g) A. A mole factor for H 2 and N 2 is B. A mole factor for NH 3 and H 2 is © 2014 Pearson Education, Inc.

Solution Consider the following equation: 3 H 2(g) + N 2(g) 2 NH 3(g) A. A mole factor for H 2 and N 2 is B. A mole factor for NH 3 and H 2 is © 2014 Pearson Education, Inc.

Guide to Calculating Quantities of Reactants and Products © 2014 Pearson Education, Inc.

Calculating Moles of a Reactant How many moles of Fe are needed for the reaction of 12. 0 moles of O 2? 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) Step 1 State the given and needed quantities (moles). Given: 12. 0 moles of O 2 Need: moles of Fe © 2014 Pearson Education, Inc.

Calculating Moles of a Reactant How many moles of Fe are needed for the reaction of 12. 0 moles of O 2? 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) ? mol Step 2 12. 0 mol Write a plan to convert the given to the needed quantity of moles of O 2 © 2014 Pearson Education, Inc. Mole−Mole factor moles of Fe

Calculating Moles of a Reactant How many moles of Fe are needed for the reaction of 12. 0 moles of O 2? 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) ? mol 12. 0 mol Step 3 Use coefficients to write mole−mole factors. 3 moles of O 2 = 4 moles of Fe © 2014 Pearson Education, Inc.

Calculating Moles of a Reactant How many moles of Fe are needed for the reaction of 12. 0 moles of O 2? 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) ? mol 12. 0 mol Step 4 Set up the problem to give the needed quantity (moles). Three SF’s © 2014 Pearson Education, Inc. Exact Three SF’s

Learning Check How many moles of NO 2 can be produced when 3. 0 moles of O 2 react? N 2(g) + 2 O 2(g) 2 NO 2(g) © 2014 Pearson Education, Inc.

Solution How many moles of NO 2 can be produced when 3. 0 moles of O 2 react? N 2(g) + 2 O 2(g) 2 NO 2(g) Step 1 State the given and needed quantities (moles). Given: 3. 0 moles of O 2 Need: moles of NO 2 © 2014 Pearson Education, Inc.

Solution How many moles of NO 2 can be produced when 3. 0 moles of O 2 react? N 2(g) + 2 O 2(g) 2 NO 2(g) 3. 0 mol ? Mol Step 2 Write a plan to convert the given to the needed quantity of moles of O 2 © 2014 Pearson Education, Inc. Mole−Mole factor moles of NO 2

Solution How many moles of NO 2 can be produced when 3. 0 moles of O 2 react? N 2(g) + 2 O 2(g) 2 NO 2(g) 3. 0 mol ? Mol Step 3 Use coefficients to write mole−mole factors. 2 moles of O 2 = 2 moles of NO 2 © 2014 Pearson Education, Inc.

Solution How many moles of NO 2 can be produced when 3. 0 moles of O 2 react? N 2(g) + 2 O 2(g) 2 NO 2(g) 3. 0 mol ? Mol Step 4 Set up the problem to give the needed quantity (moles). Two SF’s © 2014 Pearson Education, Inc. Exact Two SF’s
- Slides: 19