Chapter 9 Fluids Three common phases of matter













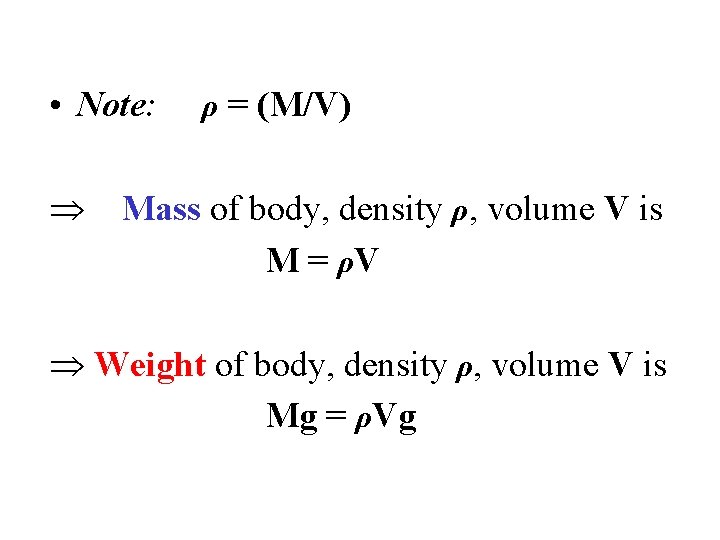























- Slides: 43


Chapter 9: Fluids • Three (common) phases of matter: 1. Solid: Maintains shape & size (approx. ), even under large forces. ( crystalline or amorphous) 2. Liquid: No fixed shape. Takes shape of container. 3. Gas: Neither fixed shape, nor fixed volume. Expands to fill container. 4. Plasma: collection of free electrically charged ions. Lump 2. & 3. into category of FLUIDS. Fluids: Have the ability to flow.

Chapter 9. 2: Deformation of Solids • All objects are deformable with the application of external forces. • When the forces ar 3 e removed the object tends to go back to its normal shape and size. ( Elastic Behavior)

Elastic Properties of Solids • Stress and Strain • Stress is related to the force causing deformation • Strain is the degree of deformation • Elastic modulus= Stress • Strain

Young's Modulus - Modulus of Elasticity (or Tensile Modulus) Hooke's Law • Most metals have deformations that are proportional with the imposed loads over a range of loads. Stress is proportional to load and strain is proportional to deformation expressed by the Hooke's law like • E = stress / strain = (Fn / A) (dl / lo) • Where E = Young's modulus (N/m 2) (lb/in 2, psi) – ε = dl / lo = σ / E (3) – where – dl = change of length (m, in) – lo = initial length (m, in) – ε = unitless measure of engineering strain – E = Young's modulus (Modulus of Elasticity) (Pa, psi)

• Modulus of Elasticity or Young's Modulus are commonly used for metals and metal alloys and expressed in terms 106 lbf/in 2, N/m 2 or Pa. • Tensile modulus are often used for plastics and expressed in terms 105 lbf/in 2 or GPa.

Shear Modulus • S = stress / strain = (Fp / A) (s / d) • Where • • • S = shear modulus (N/m 2) (lb/in 2, psi) Fp = force parallel to the faces which they act A = area (m 2, in 2) s = displacement of the faces (m, in) d = distance between the faces displaced (m, in)

Bed of Nails • A "volunteer" removes glasses and jewelry and lies on a bed of 4000 nails. A male volunteer can take off his shirt so that his bare skin is against the nails. A plywood sheet is placed over him and several concrete blocks piled on top. The demonstrator then smashes the blocks with a sledge hammer.

Its All For Science

Physics Principles • Several physics principles are involved here. The force from any one nail is reduced by spreading the weight over many nails. The inertia of the blocks partially protects the person below from the force of impact. The smashing of the blocks absorbs much of the energy of the blow.

TOO FEW NAILS • Using too few nails in this demo was a form of torture. In the illustration below Roman consul Marcus Atilius Regulus is tortured to death by Carthaginians in about 255 BC. The illustration was painted in about 1415 in Paris.

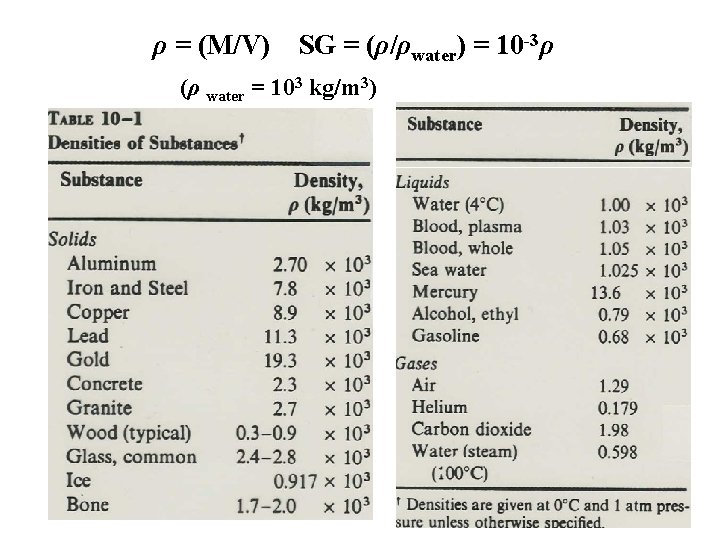
Sect. 9 -3: Density & Specific Gravity • Density, ρ (lower case Greek rho, NOT p!) of object, mass M & volume V: (mass per unit volume) ρ (M/V) (kg/m 3 = 10 -3 g/cm 3) • Specific Gravity (SG): Ratio of density of a substance to density of water at 4 degree Celsius. ρwater = 1 g/cm 3 = 1000 kg/m 3 See table!!

ρ = (M/V) SG = (ρ/ρwater) = 10 -3ρ (ρ water = 103 kg/m 3)

NOTE: 1. The density for a substance varies slightly with temperature, since volume is temperature dependent 2. The values of densities for various substances are an indication of the average molecular spacing in the substance. They show that this spacing is much greater than it is in a solid or liquid
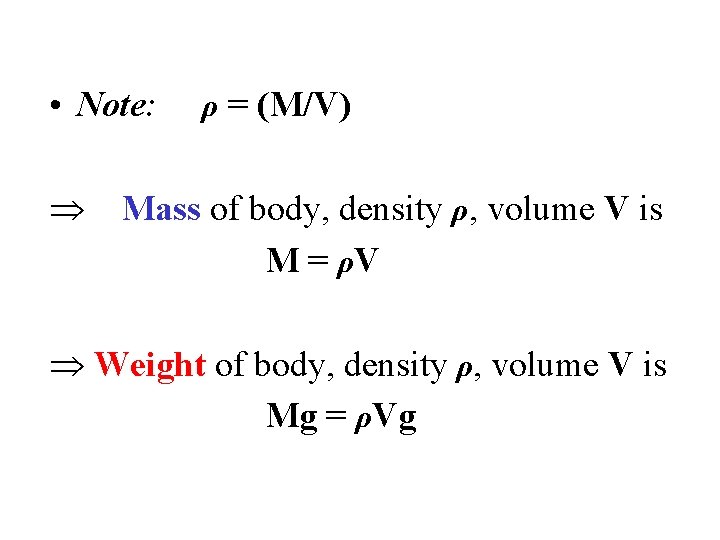
• Note: ρ = (M/V) Mass of body, density ρ, volume V is M = ρV Weight of body, density ρ, volume V is Mg = ρVg

FLUID DYNAMICS • To do fluid dynamics using Newton’s Laws, we obviously need to talk about forces in fluids. • Unlike solids: – Static Fluids do not sustain shearing forces (stresses). Shearing forces are exerted parallel to fluid surfaces. – Static Fluids do not sustain tensile forces (stresses). Tensile forces are exerted perpendicular to the fluid surface. • The only force that can be exerted on an object submerged in a Static Fluid is one that tends to compress the object from all sides • The force exerted by a Static Fluid on an object is always perpendicular to the surfaces of the object

Section 9 -3: Pressure in Fluids • Definition: Pressure = Force/Area P (F/A) F applied perpendicular to A SI units: N/m 2 1 N/m 2 = 1 Pa (Pascal)


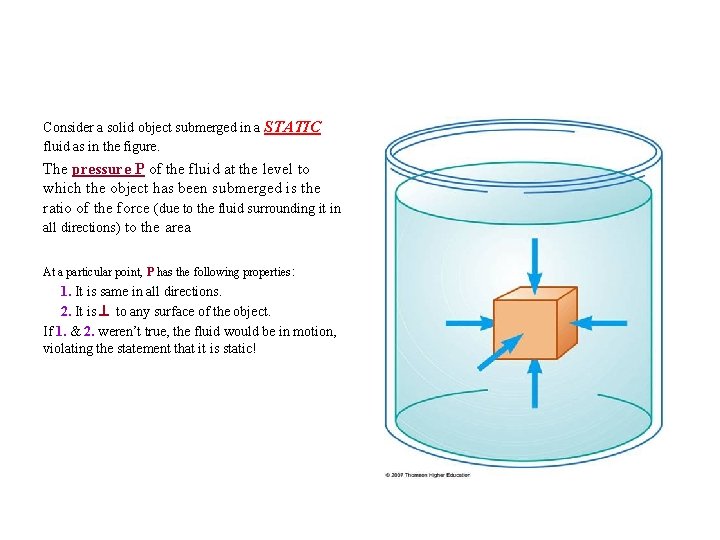
Consider a solid object submerged in a STATIC fluid as in the figure. The pressure P of the fluid at the level to which the object has been submerged is the ratio of the force (due to the fluid surrounding it in all directions) to the area At a particular point, P has the following properties : 1. It is same in all directions. 2. It is to any surface of the object. If 1. & 2. weren’t true, the fluid would be in motion, violating the statement that it is static!

• P is any fluid surface: P = (F /A)

• Note that pressure is a scalar, in contrast with force, which is a vector. It is proportional to the magnitude of the force • Suppose the pressure varies over an area. Consider a differential area d. A. That area has a force d. F on it and d. F = P d. A • The direction of the force producing a pressure is perpendicular to the area of interest.

Pressure Measurements • A possible means of measuring the pressure in a fluid is to submerge a measuring device in the fluid. • A common device is shown in the lower figure. It is an evacuated cylinder with a piston connected to an ideal spring. It is first calibrated with a known force. • After it is submerged, the force due to the fluid presses on the top of the piston & compresses the spring. • The force the fluid exerts on the piston is then measured. Knowing the area A, the pressure can then be found.

Sect. 9. 3: Variation of Pressure with Depth • • • Experimental Fact: Pressure depends on depth. See figure. If a static fluid is in a container, all portions of the fluid must be in static equilibrium. All points at the same depth must be at the same pressure – Otherwise, the fluid would not be static. Consider the darker region, which is a sample of liquid with a cylindrical shape – It has a cross-sectional area A – Extends from depth d to d + h below the surface The liquid has a density r – Assume the density is the same throughout the fluid – This means it is an incompressible liquid


Chapter 9. 4 Pressure with depth • There are three external forces acting on the darker region. These are: – The downward force on the top, P 0 A – Upward force on the bottom, PA – Gravity acting downward, Mg • The mass M can be found from the density: • The net force on the dark region must be zero: ∑Fy = PA – P 0 A – Mg = 0 • Solving for the pressure gives P = P 0 + rgh • So, the pressure P at a depth h below a point in the liquid at which the pressure is P 0 is greater by an amount rgh

• Experimental Fact: Pressure depends on depth. Consider a fluid at rest. Depth h below surface: At rest ∑Fy = 0 or, F - mg = 0 F = mg = ρVg, V =Ah F = ρAhg P F/A = ρgh Pressure at depth h (fluid at rest) P = ρgh

• Depth h below surface of liquid: P = ρgh Change in pressure with change in depth: P = ρg h (for a fluid at rest only!) P = P 2 -P 1 = ρg(h 2 -h 1) ρg h

Example 10 -2

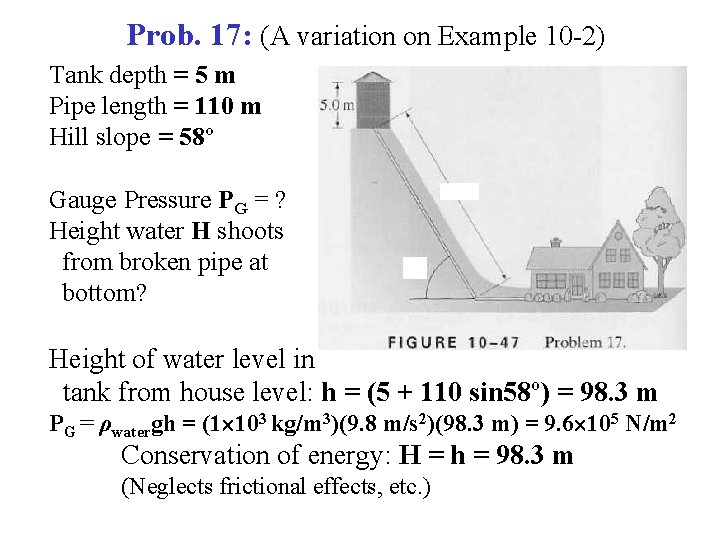
Example: (A variation on the previous example) • • • Tank depth = 5 m Pipe length = 110 m Hill slope = 58º • • Gauge Pressure PG = ? Height water H shoots from broken pipe at bottom? • • • Height of water level in tank from house level: h = (5 + 110 sin 58º) = 98. 3 m PG = ρwatergh = (1 103 kg/m 3)(9. 8 m/s 2)(98. 3 m) = 9. 6 105 N/m 2 Conservation of energy: H = h = 98. 3 m (Neglects frictional effects, etc. )

Section 10 -4: Atmospheric Pressure • Earth’s atmosphere: A fluid. – But doesn’t have a fixed top “surface”! • Change in height h above Earth’s surface: Change in pressure: P = ρg h • Sea level: PA 1. 013 105 N/m 2 = 101. 3 k. Pa 1 atm – Old units: 1 bar = 1. 00 105 N/m 2 • Physics: Cause of pressure at any height: Weight of air above that height!

Gauge Pressure • Pressure gauges (like tire gauges, etc. ) measure difference between atmospheric pressure PA & internal pressure (of tire, for example). • Gauge pressure: PG = P - PA

Conceptual Example 10 -4 P=? Pressure on A: Pdown = P + Pmg Pup = PA At rest ∑Fy = 0 Pup = Pdown or PA = P + Pmg P = PA - Pmg < PA So, air pressure holds fluid in straw!

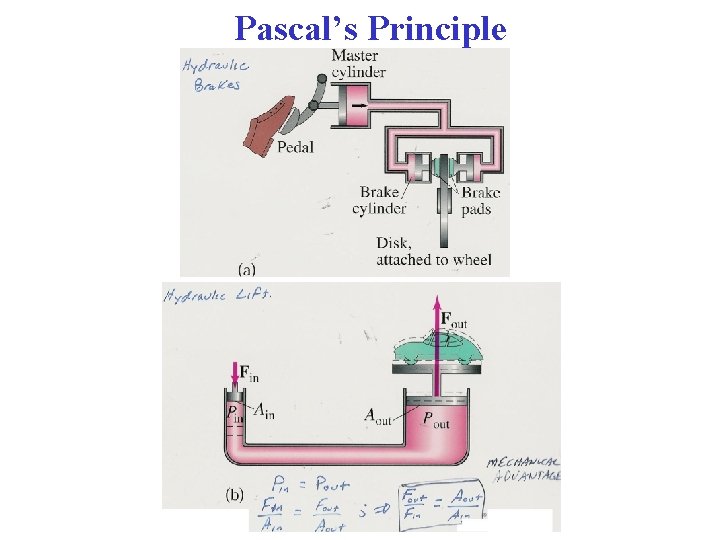
Section 10 -5: Pascal’s Principle • Experimental fact: An external pressure P applied to confined fluid increases the pressure throughout by P Pascal’s Principle • Simple example: Water in a lake (at rest). At depth h below surface, pressure is P = PA + ρgh (PA = atmospheric pressure)

Pascal’s Law • Named for French scientist Blaise Pascal • A change in the pressure applied to a fluid is transmitted undiminished to every point of the fluid and to the walls of the container

Pascal’s Principle

Pascal’s Law Example • • • Car lift in a service station. See figure. A large output force can be applied by means of a small input force. Volume of liquid pushed down on left must equal volume pushed up on right. Circular cross section system. On left r 1 = 5 cm = 0. 05 m. On right r 2 = 15 cm = 0. 15 m. Car’s weight mg = 13, 300 N. N’s 2 nd Law on right: ∑Fy = 0 = F 2 – mg. Or F 2 = 13, 300 N Calculate minimum F 1 to lift the car & pressure P in the system. Gives Pascal’s Law F 1 = (A 1/A 2)F 2 = 14, 800 N P = (F 1/A 1) = 1. 88 105 Pa

• • • The volume of liquid pushed down on left must equal volume pushed up on right. So, Combining this with Gives This means that Work 1 = Work 2 So, Pascal’s Law is consequence of Conservation of Mechanical Energy

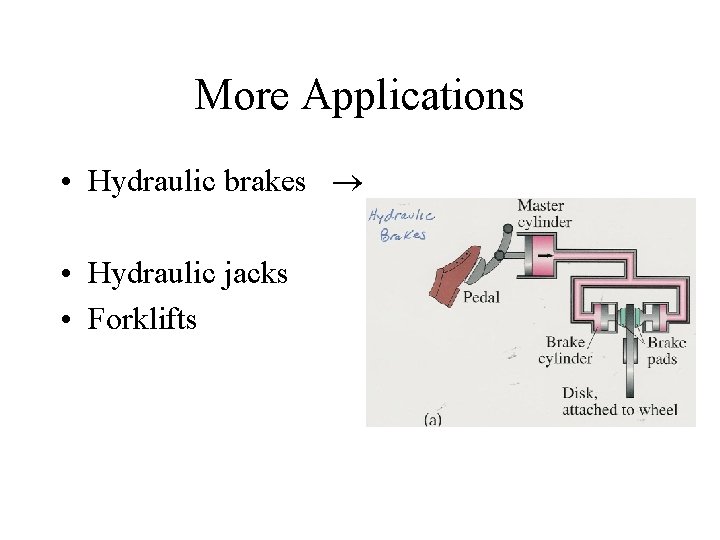
More Applications • Hydraulic brakes • Hydraulic jacks • Forklifts

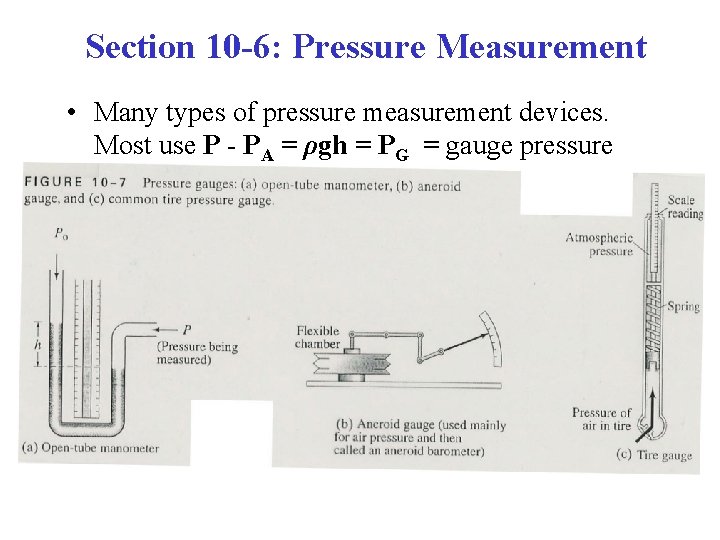
Section 10 -6: Pressure Measurement • Many types of pressure measurement devices. Most use P - PA = ρgh = PG = gauge pressure

Various Pressure Units • Gauge Pressure: PA = ρgh Alternate unit of pressure: Instead of calculating ρgh, common to use standard liquid (mercury, Hg or alcohol, where ρ is standard) & measure h Quote pressure in length units! For example: “millimeters of mercury” mm Hg For h = 1 mm Hg = 10 -3 m Hg ρmercury gh = (1. 36 104 kg/m 3) (9. 8 m/s 2)(10 -3 m) = 133 N/m 2 = 133 Pa 1 Torr (another pressure unit!) mm Hg & Torr are not proper SI pressure units!

• About as many pressure units as there are measurement devices!! • Preferred (SI) unit: 1 Pa (Pascal) = 1 N/m 2

Mercury Barometer • Weather reports: Barometric pressure (atmospheric pressure): 28 -32 inches Hg 76 cm = 760 mm = 29. 29 inches When h = 760 mm, P = ρmercury gh = 1. 013 105 N/m 2 = 1 atm • If use water P = 1 atm = ρwater gh h 10 m 30 feet!

Prob. 17: (A variation on Example 10 -2) Tank depth = 5 m Pipe length = 110 m Hill slope = 58º Gauge Pressure PG = ? Height water H shoots from broken pipe at bottom? Height of water level in tank from house level: h = (5 + 110 sin 58º) = 98. 3 m PG = ρwatergh = (1 103 kg/m 3)(9. 8 m/s 2)(98. 3 m) = 9. 6 105 N/m 2 Conservation of energy: H = h = 98. 3 m (Neglects frictional effects, etc. )