Chapter 9 Elements Compounds and Mixtures Section 3






























- Slides: 32

Chapter 9 Elements, Compounds, and Mixtures Section 3 Mixtures

0807. 9. 4 Differentiate between a mixture and a compound. I can describe properties of mixtures. I can describe methods for separating mixtures.

Mixtures • Imagine that you roll out some dough, add tomato sauce and sprinkle some cheese on top. • Then, you add green peppers, mushrooms, olives, and pepperoni. • WHAT HAVE YOU JUST MADE? ? ?

Properties of Mixtures • A mixture is a combination of two or more substances that are not chemically combined. • When two or more materials are put together, they form a mixture if they do not react to form a compound. • For example, cheese and tomato sauce do not react when they are used to make a pizza. • So what does that make a pizza?

No Chemical Changes in a Mixture • NO chemical change • In some mixtures, happens when a such as the pizza, you mixture is made. can see each component. • So, each substance in However, a mixture has the such as salt water, same chemical you cannot see all the makeup it had before components. the mixture formed. • So in a sense, they keep their identity.

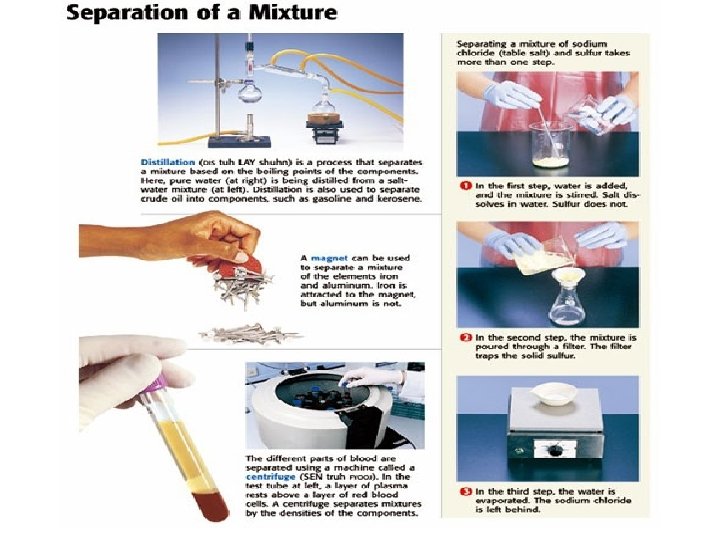
Separating Mixtures Through Physical Methods • If you don’t like peppers on your pizza, you simply just pick them off. • This would be what type of change? • PHYSICAL • The identities of the substance do not change. • However, not all mixtures are that easy to separate. • You cannot just pick salt out of a salt water mixture. • You would need to heat the mixture until the water evaporates so that all that is left is the salt.


The Ratio Component in a Mixture • Components in a • Feldspar is pink in mixture do not need to color. be mixed in a definite ratio. • Mica is black in color. • For example, granite is a mixture made of • Quartz is colorless. three minerals: feldspar, mica, and quartz.

The Ratio Of Components in a Mixture • Depending on how much of each mineral is mixed in granite, the color will vary. • For instance, if granite has more feldspar in it, it will look like a pinkish color. • If granite has more mica in it, it will have a black color. • If granite has more quartz in it, it will have more of a gray color. • Although they are different colors, the mixture still makes granite.


Solutions • A solution is a mixture that appears to be a single substance. • A solution is composed of particles of two or more substance that are distributed evenly among each others. • The process in which particles separate and spread evenly throughout a mixture is known as dissolving.

Solutions • In solutions, the solute • A solute must be is the substance that soluble, meaning able is dissolved. to dissolve. • The solvent is the • A substance that is substance in which insoluble, or unable to the solute is dissolved. dissolve, forms a mixture that is not a solution.


• A homogeneous mixture has two substances where particles the size of atoms and molecules are blended completely. (most mixed) • To the eye, the mixture appears to be pure substance. When the particles stay intermixed and don’t settle into layers we call the homogeneous mixture a solution. • Homogeneous mixture results from the process of dissolving. The sugar particles are completely separated from the clumps of other sugar particles. The attractive forces between the sugar particles and the water particles is strong enough to pull the sugar particles away from the main clump.


Examples of Solutions • You may think that solutions are only liquids. • However, solutions may also be gases, such as air. • Solutions may also be solids, such as steel. • Alloys are solid solutions of metals or nonmetals dissolve in metals.


Particles in a Solution • The particles in solutions are so small that they never settle out. • They also cannot be removed by filtering. • They are even so small they do not scatter light.

Concentration of Solutions • A measure of the amount of solute dissolved in a solvent is concentration. • Concentration can be expressed in grams of solute per milliliter of solvent. ? ?

Concentrated or Dilute? • Solutions can be • A concentrated item described as being has more solute. concentrated or dilute. • A diluted solution has less solute.

Solubility • If you add too much sugar to a glass of lemonade, not all of the sugar can dissolve. • Some of it sinks to the bottom. • To find the maximum amount of sugar that can dissolve, you would need to know the solubility of sugar. • The solubility of a solute is the ability of the solute to dissolve in a solvent at a certain temperature.


Dissolving Gases in Liquids • Most solids are more soluble in liquids at higher temperatures. • But, gases become less soluble in liquids as the temperature is raised. • A soft drink goes flat faster when warm • The gas that is dissolved in the soft drink cannot stay dissolved when the temperature increases. So the gas escapes, and the soft drink becomes flat.

Dissolving Solids Faster in Liquids • Several things affect how fast a solid will dissolve. – Stirring or Shaking – Heating – Crushing

Suspensions • Think about a snow globe. What happens when you shake the snow globe? • When you stop shaking the globe, the snow settles to the bottom. This mixture is called a suspension. • A suspension is a mixture in which particles of a material are dispersed throughout a liquid or gas but are large enough that they settle out.


Suspensions • The particles in a suspension are large enough to scatter or block light. • The particles are also too large to stay mixed without being stirred or shaken. • Also, if allowed to sit, the particles will settle out to the bottom. • A suspension can be separated by passing it through a filter.

• A heterogeneous mixture has large clumps of particles that don’t fully separate and doesn’t get intermixed with the other substance. • The difference in particles can be seen in the texture, color, shape and size. • A suspension is a heterogeneous mixture that has clumps that stay floating. . Example: fine sand or silt in water or tomato juice.

Colloids • A colloid is a mixture in which the particles are dispersed throughout but are not heavy enough to settle out. • The particles in a colloid are relatively small and are fairly well mixed. You might be surprised at the number of colloids you see each day. • Milk, mayonnaise, and deodorant. • Particles in colloids can scatter light but cannot be filtered out.


0807. 9. 4 Differentiate between a mixture and a compound. I can describe properties of mixtures. I can describe methods for separating mixtures.

Label each figure below with the type of substance it BEST models: colloid, compound, element, solution, or suspension.