Chapter 9 Dissolution and Precipitation Equilibria 9 1

Chapter 9 Dissolution and Precipitation Equilibria 9 -1 The Nature of Solubility Equilibria 9 -2 The Solubility of Ionic Solids 9 -3 Precipitation and the Solubility Product 9 -4 The Effects of p. H on Solubility 9 -5 Complex Ions and Solubility 9 -6 Controlling Solubility in Qualitative Analysis 11/5/2020 OFB Chapter 9 1

Saturated Solution: a solution in equilibrium with excess solute e. g. , Na. Cl solubility in grams per 100 grams water is approximately 36. 0 grams = saturated solution Unsaturated Solution: contains less than the equilibrium concentration of the solute Supersaturated Solution: a solution that temporarily contains more of a solute than the equilibrium quantity 11/5/2020 OFB Chapter 9 2
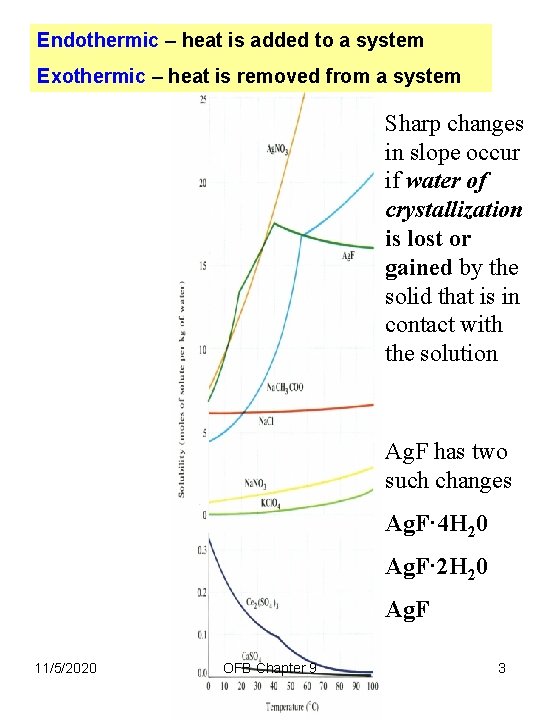
Endothermic – heat is added to a system Exothermic – heat is removed from a system Sharp changes in slope occur if water of crystallization is lost or gained by the solid that is in contact with the solution Ag. F has two such changes Ag. F· 4 H 20 Ag. F· 2 H 20 Ag. F 11/5/2020 OFB Chapter 9 3

9 -2 Solubility of Salts This chapter considers only salts which are sparingly soluble or insoluble for which concentrations of saturated salts are [salt] = 0. 1 Mol L-1 or less 11/5/2020 OFB Chapter 9 4

Solubility Product Ksp Describes a chemical equilibrium in which an excess solid salt is in equilibrium with a saturated aqueous solution of its separated ions. General equation AB (s) ↔ A+ (aq) + B- (aq) 11/5/2020 OFB Chapter 9 5

The Solubility of Ionic Solids The Solubility Product Ag. Cl(s) ↔Ag+ (aq) + Cl-(aq) Ksp = = The solid Ag. Cl, which is in excess, is understood to have a concentration of 1 mole per liter. Ksp= 1. 6 10 -10 at 25 o. C 11/5/2020 OFB Chapter 9 6

The Solubility of Ionic Solids The Solubility Product Ag 2 SO 4(s) ↔ 2 Ag+(aq) + SO 42 -(aq) Ksp = Fe(OH)3(s) ↔Fe+3(aq) + 3 OH-1(aq) Ksp = 11/5/2020 OFB Chapter 9 7

The Solubility of Ionic Solids The Solubility Product Exercise 9 -1 Write the Ksp equation for the dissolution of aluminum hydroxide (Al(OH)3) in water. Al(OH)3(s) ↔Al 3+(aq) + 3 OH-(aq) 11/5/2020 OFB Chapter 9 8

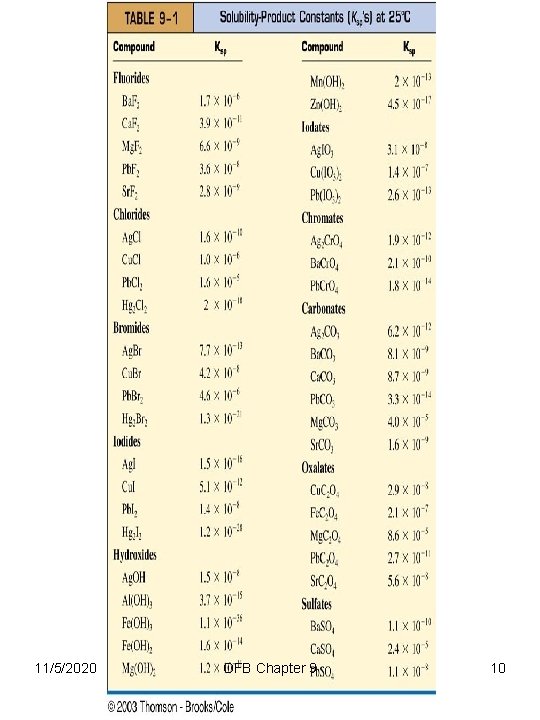
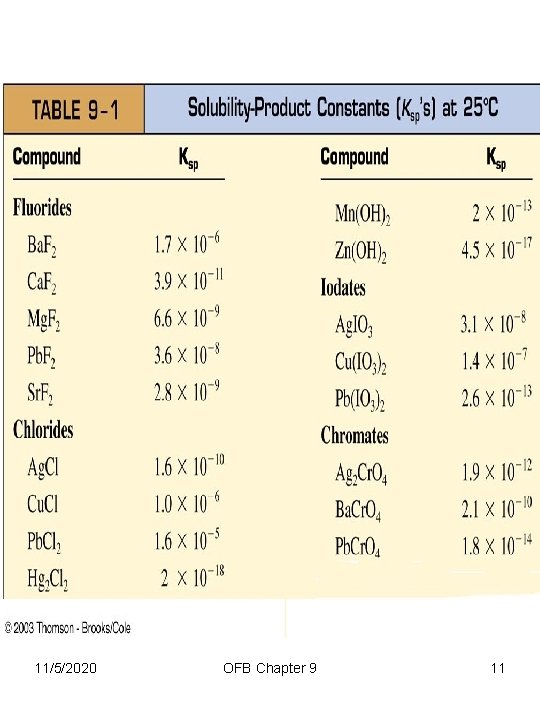
The Solubility of Ionic Solids The Solubility Product TABLE 9 -1 contains Ksp values at 25 C 11/5/2020 OFB Chapter 9 9
11/5/2020 OFB Chapter 9 10
11/5/2020 OFB Chapter 9 11

11/5/2020 OFB Chapter 9 12

The Solubility of Salts Solubility and Ksp Exercise 9 -2 Determine the mass of lead(II) iodate dissolved in 2. 50 L of a saturated aqueous solution of Pb(IO 3)2 at 25 o. C. The Ksp of Pb(IO 3)2 is 2. 6 10 -13. Pb(IO 3)2(s) ↔Pb 2+(aq) + 2 IO 3 -(aq) [y] [Pb 2+][IO 3 -]2 = Ksp [y] [2 y] y = 4. 0 10 -5 [Pb(IO 3)2] = [Pb 2+] = y = 4. 0 10 -5 mol L-1 [IO 3 -] = 2 y = 8. 0 10 -5 mol L-1 Gram solubility of = (4. 0 10 -5 mol L-1) (557 g mol-1) Lead (II) iodate = 0. 0223 g L-1 2. 50 L Pb=207. 2 11/5/2020 O=16 I=126. 9 OFB Chapter 9 Pb(IO 3)2 = 557 g per mole 13

The Solubility of Salts Solubility and Ksp Exercise 9 -3 Compute the Ksp of silver sulfate (Ag 2 SO 4) at 25 o. C if its mass solubility is 8. 3 g L-1. 1 Ag 2 SO 4(s) ↔ 2 Ag+(aq) + 1 SO 42 -(aq) [y] 11/5/2020 [2 y] OFB Chapter 9 [y] 14

The Solubility of Salts Solubility and Ksp Exercise 9 -3 Compute the Ksp of silver sulfate (Ag 2 SO 4) at 25 o. C if its mass solubility is 8. 3 g L-1. 1 Ag 2 SO 4(s) ↔ 2 Ag+(aq) + 1 SO 42 -(aq) [y] [2 y] [y] = (8. 3 g Ag 2 SO 4 L-1) (1 mol Ag 2 SO 4/311. 8 g) [y] = [2. 66 10 -2 ] mol Ag 2 SO 4 L-1 [Ag+]2[SO 42 -] = Ksp 11/5/2020 OFB Chapter 9 15

Review The Nature of Solubility Equilibria Dissolution and precipitation are reverse of each other. Dissolution (Solubility) General reaction X 3 Y 2 (s) ↔ 3 X+2 (aq) + 2 Y-3 (aq) [s] [3 s] Ksp = [X+2]3 [Y-3]2 s= [2 s] = (3 s)3 (2 s)2 molar solubility expressed in moles per liter 11/5/2020 OFB Chapter 9 16

Review The Nature of Solubility Equilibria Dissolution and precipitation are reverse of each other. Precipation General reaction X 3 Y 2 (s) ↔ 3 X+2 (aq) + 2 Y-3 (aq) Mix [X+2] and [Y-3] [X+2] [Y-3] Does a ppt of X 3 Y 2 form? Reaction quotient before mixing occurs: Q(init) = [X+2]3(init)[Y-3]2(init) 2 9 +2]3 [Y 11/5/2020 OFB-3 Chapter K = [X ] sp Q>K? 17
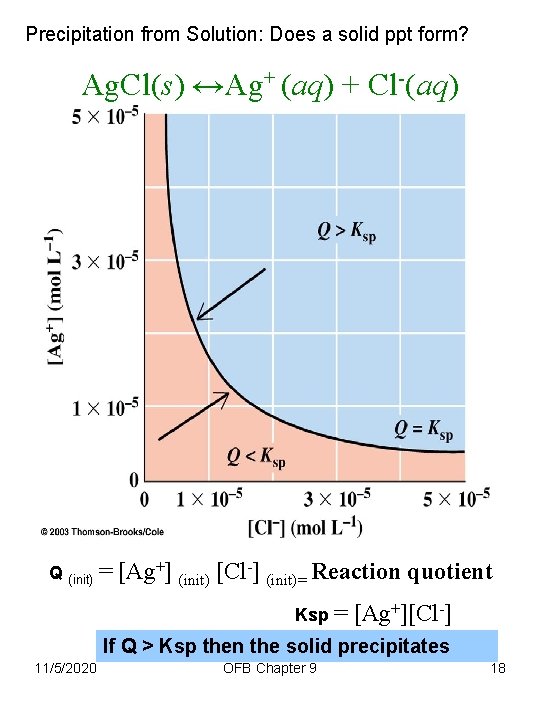
Precipitation from Solution: Does a solid ppt form? Ag. Cl(s) ↔Ag+ (aq) + Cl-(aq) Q (init) = [Ag+] (init) [Cl-] (init)= Reaction quotient Ksp = [Ag+][Cl-] If Q > Ksp then the solid precipitates 11/5/2020 OFB Chapter 9 18

Precipitation and the Solubility Product Precipitation from Solution Exercise 9 -4: The Ksp of thallium (I) iodate is 3. 1 10 -6 at 25 o. C. Suppose that 555 m. L of a 0. 0022 M solution of Tl. NO 3 is mixed with 445 m. L of a 0. 0022 M solution of Na. IO 3. Does Tl. IO 3 precipitate at equilibrium? Evaluate : Reaction quotient before mixing occurs: Q(init) = [Tl+](init)[IO 3 -](init) If Q(init) > Ksp, solid Tl. IO 3 precipitates until Q = Ksp If Q(init) < Ksp, no solid Tl. IO 3 can appear. [Tl+] Q > Ksp Solid ppt Q < Ksp No ppt 11/5/2020 OFB Chapter 9 19 [IO 3 -]

Exercise 9 -4 The Ksp of thallium(I) iodate is 3. 1 10 -6 at 25 o. C. Suppose that 555 m. L of a 0. 0022 M solution of Tl. NO 3 is mixed with 445 m. L of a 0. 0022 M solution of Na. IO 3. Does Tl. IO 3 precipitate at equilibrium? Tl(IO 3) (s) ↔Tl+(aq) + IO 3 -(aq) [Tl+](init) = (0. 0022 mol L-1)(555 m. L/1000 m. L) = 0. 0012 mol L-1 [IO 3 -](init) = (0. 0022 mol L-1)(445 m. L/1000 m. L) = 0. 00098 mol L-1 Q(init) = [Tl+](init)[IO 3 -](init) = (0. 0012)(0. 00098) = 1. 17 10 -6 Q(init) ? Ksp = 1. 17 10 -6 < 3. 1 10 -6 Because Q(init) < Ksp, 11/5/2020 solid Tl. IO 3 does NOT precipitate! OFB Chapter 9 20

Precipitation and the Solubility Product The Common Ion Effect If a solution and a solid salt to be dissolved in it have an ion in common, then the solubility of the salt is depressed. 11/5/2020 OFB Chapter 9 21

The Common Ion Effect Exercise 9 -6 The Ksp of thallium(I) iodate (Tl. O 3) is 3. 1 10 -6 at 25 o. C. Determine the molar solubility of Tl. IO 3 in 0. 050 mol L-1 KIO 3 at 25 o. C. Tl. IO 3(s) ↔ Tl+(aq) + [Tl+] (mol L-1) IO 3 -(aq) [IO 3 -] (mol L-1) Initial concentration Change in concentration Equilibrium concentration [Tl+][IO 3 -] = Ksp Assume s is small s = [Tl. IO 3]= 6. 2 × 10 -5 mol L-1 which is -3 11/5/2020 Chapter 9 to the 1. 76 x 1022 depressed 28 times OFB relative conc. without the common ion

The Effects of p. H on Solubility of Hydroxides Many solids dissolve more readily in more acidic solutions Zn(OH)2(s) ↔Zn 2+(aq) + 2 OH-(aq) [Zn 2+][OH-]2 = Ksp = 4. 5 10 -17 If p. H decreases (or made more acidic), the [OH-] decreases. In order to maintain Ksp the [Zn 2+] must increase and consequently more solid Zn(OH)2 dissolves. 11/5/2020 OFB Chapter 9 23

The Effects of p. H on Solubility of Hydroxides Exercise 9 -7 Estimate the molar solubility of Fe(OH)3 in a solution that is buffered to a p. H of 2. 9. [OH-] = 3 y In pure water: [Fe 3+] = y y(3 y)3 = 27 y 4 = Ksp = 1. 1 10 -36 y = 4. 5 10 -10 mol L-1 = [Fe 3+] = [Fe(OH)3]= [OH-] = 3 y = 1. 3 10 -9 mol L-1 p. OH = 8. 87 (and p. H = 5. 13) In pure water, Fe(OH)3 is 5 x 10 6 less soluble = 2. 9 11/5/2020 than at p. HOFB Chapter 9 24

9 -7 The Effects of p. H on Solubility • • Solubilities of Hydroxides Solubility of Salts and Weak Bases Selective Precipitation of Ions Metal Sulfides Somewhat more complicated due to other competing reactions. E. g. , MS + H 2 O ↔ M 2+ + OH- + HS(Metal Sulfide) But as before solubility of Metal Sulfides increase as p. H decreases Ksp = [M 2+][OH-][HS-] As p. H decreases (or made more acidic), the [OH-] decreases. In order to maintain Ksp the [M 2+] must increase and consequently more solid Metal Sulfide 11/5/2020 OFB Chapter 9 25 dissolves.

• Examples / Exercises – 9 -1, – 9 -2, – 9 -3, – 9 -4, – 9 -5, – 9 -6, – 9 -7 11/5/2020 Ksp calculations ppt Q ? Ksp Equilibrium concentrations Common Ion effect Effect of p. H of on solubility OFB Chapter 9 26
- Slides: 26