Chapter 9 Covalent Bonding Covalent bond Sharing of

Chapter 9 Covalent Bonding
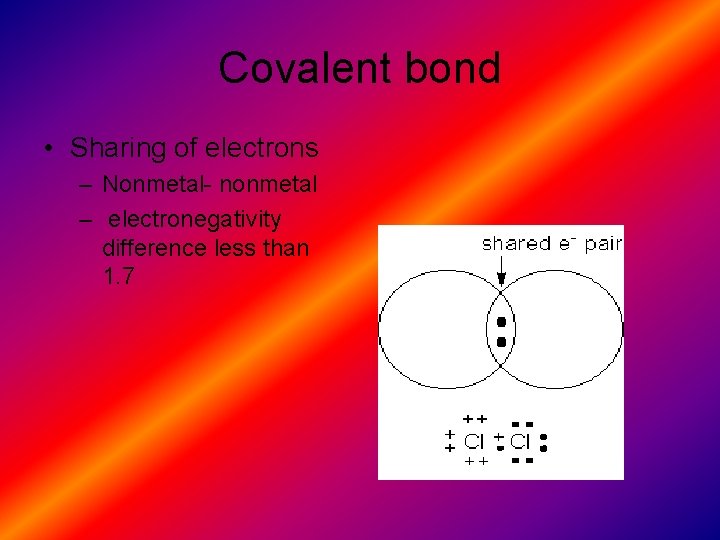
Covalent bond • Sharing of electrons – Nonmetal- nonmetal – electronegativity difference less than 1. 7

Molecule • Base unit of a covalent bond

Diatomic molecule • Naturally formed 2 atom groups – H 2, N 2, 02, F 2, Cl 2, Br 2, I 2

2 types of covalent bonds • Polar covalent- bonding electrons are shared unequally • Nonpolar covalent- bonding electrons are shared equally – Balanced distribution of charge

How do you tell the difference in polar and nonpolar? • If the 2 atoms have a difference in electronegativity the bond is polar, if no difference then the bond is nonpolar • If polar bonds draw the electron to one side of the molecule and the other side has none the molecule is polar

Bond length • Distance between two bonded atoms at their minimum potential energy – Average distance between bonded atoms

Bond energy • Energy required to break a bond

Coordinate covalent bond • One atom donates both electrons to the bond

Nomenclature for covalent bonds • (nonmetal- nonmetal) • 2 systems: – Stock- name(+), (+) oxidation # in roman numerals, name(-) – Classical- use prefixes except for a single (+) ion • *Never use mono first*

Prefixes • • • Mono Di Tri Tetra Penta Hexa Hepto Octa Nano Deca

Naming acids • Binary: hydro + root + ic acid • Ternary: polyatomic, drop –ate, add –ic acid

Oxyacids • +1 0 per- ate per - ic • Memory -ate - ic • -1 0 -ite -ous • -2 0 - hypo-ite hypo-ous
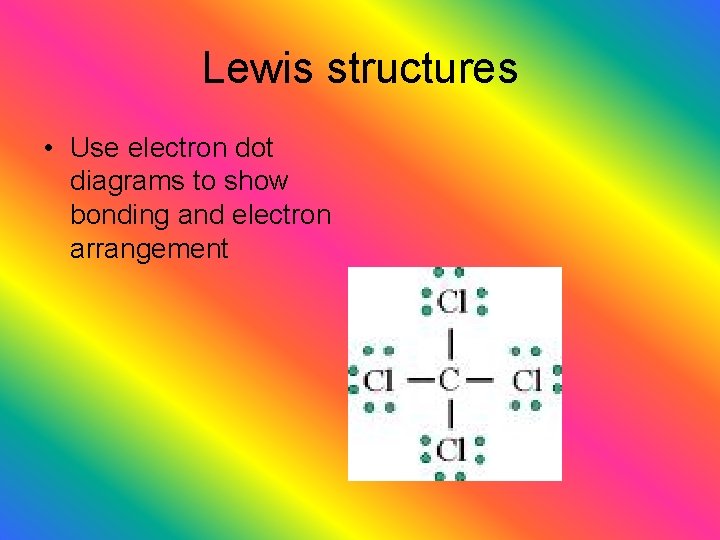
Lewis structures • Use electron dot diagrams to show bonding and electron arrangement

Symbols uses in molecular structural formulas unshared pair- (lone pair) pair of electrons that is not involved in bondingbelongs to one atoms single bond- 2 e- shared double bond- 4 e- shared triple bond- 6 e- shared
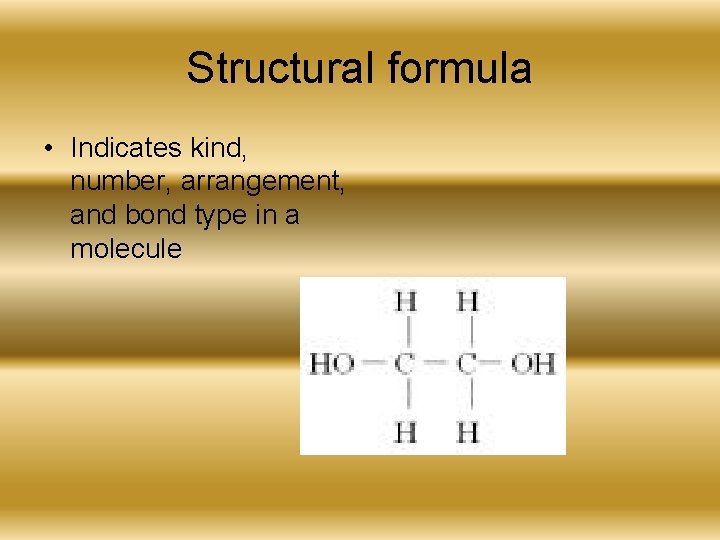
Structural formula • Indicates kind, number, arrangement, and bond type in a molecule

Sigma bond • s - electrons are shared along the bond axis

Pi bond p – electrons are shared above and below axis

Resonance • More than one valid Lewis structure can be drawn

VSEPR theory • Valance shell electron pair repulsion – Model for molecular geometry – Bond angles – Arrangement minimizes repulsion of e- around the central atom – Molecules adjust their shape, so that valence e- are as far apart as possible

3 types of repulsion 1. Unshared-unshared 2. Unshared- shared 3. Shared-shared

hybridization • A process in which atomic orbitals are mixed to form new identical hybrid orbitals

Hybrid orbitals • Orbital of equal energy produced by the combination of two or more atomic orbitals • (sp, sp 2, sp 3)

Intramolecular forces • Forces within a molecule that hold atoms together

Intermolecular forces • Forces of attraction between molecules (Van der Waals)

Dipole • Molecule that has two poles (polar)

Dipole moment • Measure of the strength of the dipole and is a property that results from the asymmetrical charge distribution in a polar molecule- depends on size and distance m= Qd

Van der Waals forces • Groups of intermolecular forces

Dipole- dipole force • Between polar molecules

Induce dipole • A normally nonpolar molecule is transformed into a dipole
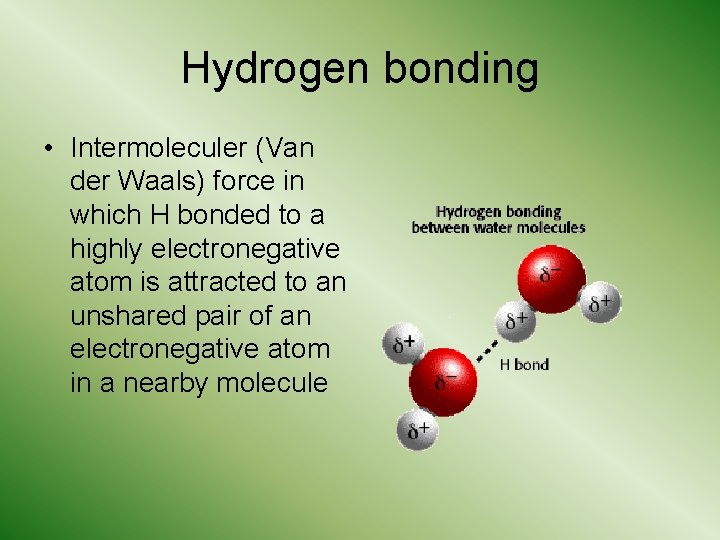
Hydrogen bonding • Intermoleculer (Van der Waals) force in which H bonded to a highly electronegative atom is attracted to an unshared pair of an electronegative atom in a nearby molecule

London Dispersion forces • (dispersion force) result from the constant motion of e- ’s and the creation of instanteous dipoles – Force generated in a temporary dipole interaction – Most important Van der Waals force – Proposed by Fritz London in 1930 – Strength increases with the number of e- in the interacting atoms

- Slides: 33