Chapter 9 Chemical Names and Formulas Ms Wang

















- Slides: 21

Chapter 9 Chemical Names and Formulas Ms. Wang Lawndale High School

Chapter 9. 1 – Naming Ions Monatomic Ions • Consist of a single atom with a positive or negative charge –Example: Li+, Na+, K+, Cl-, Ca 2+, Mg 2+, F-, S 2–Which ones are cations? Which ones are anions? Polyatomic Ions • Composed of more than one atom • The entire unit is comprised of one charge • Example: SO 42 -, NO 2 -, Cl. O 2 -

Chapter 9. 2 – Naming Ionic Compounds • Binary Compound – composed of 2 elements

Rules for Naming Chemical Formulas Write the cation first and then the anion l Cation keeps its name l Anion changes its ending The correct formula contains the fewest positive and negative ions needed to make the electrical charge ZERO.

Anion changes its ending… –ine, From -ygen, To -fur, -ogen Fluorine becomes fluoride Oxygen becomes oxide -ide Example: Write the chemical formula for Calcium Chloride Name Cation Anion Calcium chloride Ca 2+ Cl- Total number of ions 3 Chemical Formula Ca. Cl 2

Practice 1. 2. 3. 4. 5. 6. 7. 8. Cs 2 O Sr. F 2 Sn. S KBr Cu. O Na. I K 2 S Ca. O Cesium Oxide Strontium Fluoride Tin (II) Sulfide Potassium Bromide Copper (II) Oxide Sodium Iodide Potassium Sulfide Calcium Oxide

How to Write Formulas for Ionic Compounds Question: What is the formula for aluminum oxide? Step 1: Write the symbol and charges for the cation and anion Aluminum =Al 3+ Oxygen = O 2 Step 2: Write the symbol for the ions side by side, beginning with the cation Al 3+O 2 Step 3: Find the least common multiple of the ions’ charges. The least common multiple of 2 and 3 is 6. To be neutral you would need 6 + and 6 To get 6+, you need two Al 3+ ions To get 6 -, you need three O 2 - ions Al 2 O 3

How to Write Formulas for Ionic Compounds Example: what is the formula for Magnesium Chloride? 1. Magnesium =Mg 2+ Chloride =Cl- 2. Mg 2+Cl 3. Least common multiple of 2 and 1 = 2 To get 2+, need one Mg To get 2 -, need two Cl Mg. Cl 2 CHEATER METHOD: JUST CRIS-CROSS!!!!

Transition Metals For metals with more than one charge, the charge on the metal ion must be stated. These are typically transition metals. Ex. Iron (II) Chloride Fe 2+ Cl- Fe. Cl 2 Ex. Iron (III) Chloride Fe 3+ Cl- Fe. Cl 3 Example Write the name of Ti. O? 1. Find out the charge on the ion that only has one charge. O 22. Then evaluate what charge the other ion must have in order to fulfill the formula. Ti must be 2+ Titanium (II) Oxide

Practice Write the formulas for the following compounds: Copper (II) Chloride 1. Cu. Cl 2 2. Cu. Cl Copper (I) Chloride 3. Cr 2 O 3 Chromium (III) Oxide 4. Mn. O Manganese (II) Oxide 5. Be. Cl 2 Beryllium Chloride 6. Na. F Sodium Fluoride

Polyatomic Ions: ions made from more than one element. They stay together when bonding. Example: Lithium Phosphate Li + PO 43 Li 3 PO 4 Example: Magnesium Hydroxide Mg 2+ OHMg(OH)2

Write the Formula for the following 1. 2. 3. 4. 5. 6. 7. 8. Calcium Nitrate Iron (III) Sulfate Copper (II) Sulfite Potassium Permanganate Chromium (III) Nitrate Sodium Perchlorate Calcium Acetate Magnesium Dichromate Ca(NO 3)2 Fe 2(SO 4)3 Cu. SO 3 KMn. O 4 Cr(NO 3)3 Na. Cl. O 4 Ca(C 2 H 3 O 2)2 Mg. Cr 2 O 7

Name the following Compounds 1. 2. 3. 4. 5. 6. 7. 8. Mg(OH)2 Ti 2(Cr. O 4)3 Co. PO 4 Ag. NO 3 Cu. SO 4 Be. C 2 O 4 Fe. Cr. O 4 Cr. PO 4 Magnesium Hydroxide Titanium (III) Chromate Cobalt (III) Phosphate Silver Nitrate Copper (II) Sulfate Beryllium Oxalate Iron (II) Chromate Chromium (III) Phosphate

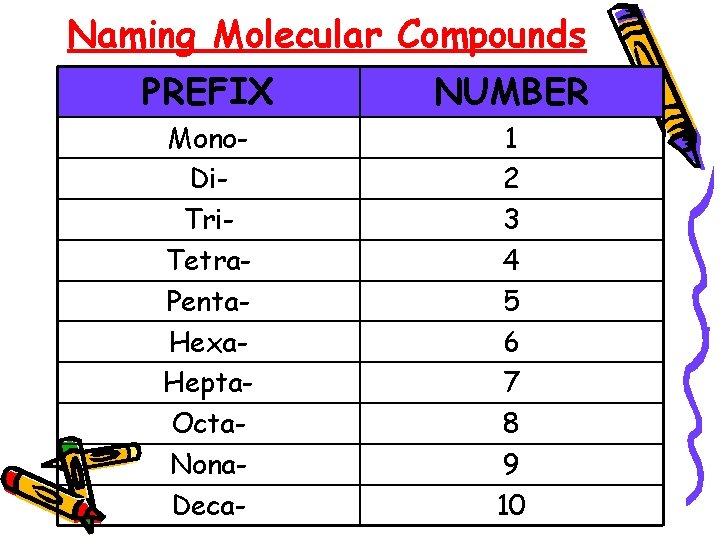
Chapter 9. 3 – Naming Molecular Compounds • Remember that molecules are composed of two nonmetals • the prefix in the name of a binary molecule tells how many atoms of each element are present in each molecule of the compound

Naming Molecular Compounds PREFIX NUMBER Mono. Di. Tri. Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca- 1 2 3 4 5 6 7 8 9 10

Writing Formulas for Binary Molecular Compounds 1 st Step: Write the cation 2 nd Step: Write the anion with the appropriate prefix Example #1: NCl 3 Nitrogen Trichloride Example #2: BCl 3 Boron Trichloride Example #3: N 2 O 3 Dinitrogen Trioxide Example #4: N 2 H 4 Dinitrogen Tetrahydride

Practice (1. ) SO 3 (2. ) CS 2 (3. ) Cl 2 O 7 (4. ) Carbon Tetrabromide (5. ) Diphosphorus Trioxide (6. ) Phosphorus Pentachloride (7. ) Iodine Heptafluoride (8. ) Chlorine Trifluoride (9. ) Iodine Dioxide Sulfur Trioxide Carbon Disulfide Dichlorine Heptaoxide CBr 4 P 2 O 3 PCl 5 IF 7 Cl. F 3 IO 2

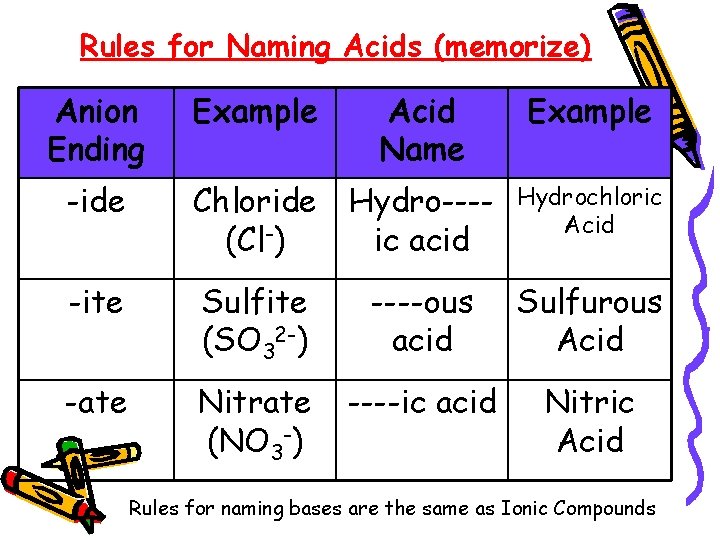
Chapter 9. 4 – Naming Acids and Bases Acids – compound that contains one or more hydrogen atoms and produces hydrogen ions (H+) Bases – compound that produces hydroxide ions when dissolved in water (OH-)

Rules for Naming Acids (memorize) Anion Ending Example Acid Name Example -ide Chloride (Cl-) Hydro---ic acid Hydrochloric Acid -ite Sulfite (SO 32 -) ----ous acid Sulfurous Acid -ate Nitrate (NO 3 -) ----ic acid Nitric Acid Rules for naming bases are the same as Ionic Compounds

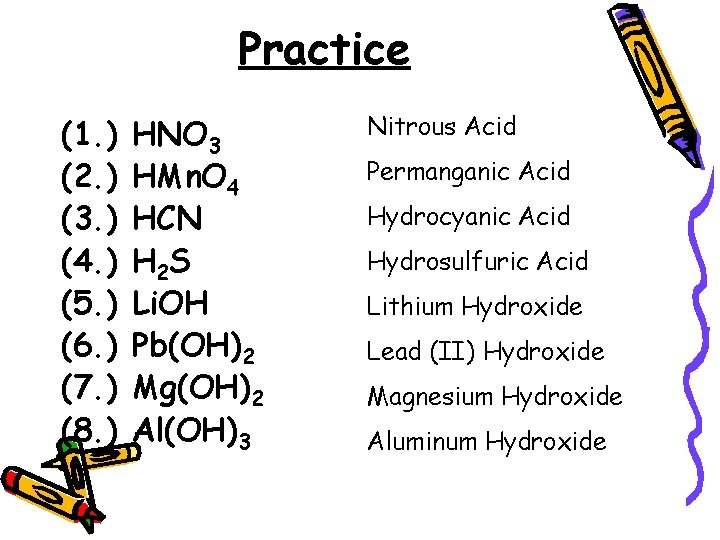
Practice (1. ) (2. ) (3. ) (4. ) (5. ) (6. ) (7. ) (8. ) HNO 3 HMn. O 4 HCN H 2 S Li. OH Pb(OH)2 Mg(OH)2 Al(OH)3 Nitrous Acid Permanganic Acid Hydrocyanic Acid Hydrosulfuric Acid Lithium Hydroxide Lead (II) Hydroxide Magnesium Hydroxide Aluminum Hydroxide

Homework Section 9. 1 – Page 258 #’s 1 -9 Section 9. 2 – Page 266 #’s 14 -19 Section 9. 3 – Page 270 #’s 22 -25 Section 9. 4 – Page 273 #’s 29 -33 Chapter 9 Assessment Page 281 #’s 42 -61, 65 -70, 82, 83