Chapter 9 Chemical Bonds Types of Bonds Ionic

Chapter 9: Chemical Bonds Types of Bonds • Ionic – Metal and nonmetal – Electron transfer – Infinite lattice • Covalent – Nonmetal and nonmetal – Shared electrons – Individual molecules • Metallic – Metal and metal – “electron sea” – Infinite lattice

Ionic Bonds: Octet Rule In forming ionic compounds, atoms tend to gain or lose electrons in order to achieve a stable valence shell electron configuration of 8 electrons. Group I metals --> Group II metals --> Al (group III) --> Group VII (17) --> Group VI (16) --> Group V (15) --> +1 cations (Li+, Na+, etc) +2 cations (Mg 2+, Ca 2+, etc) Al 3+ -1 anions (F–, Cl–, Br–, etc) -2 anions (O 2–, S 2–, etc) -3 anions (N 3–, P 3–) e. g. Na 2 s 22 p 63 s 1 --> Na+ 2 s 22 p 6 {~Ne} Cl 3 s 23 p 5 --> Cl– 3 s 23 p 6 {~Ar} Remember Ch. 2 Figure 2. 14 (p 62)

Ionic Bonds: Lewis Symbols simple notation for showing number of valence electrons Cl group VII (7 valence e–) O group VI (6 valence e–) 3 s 23 p 5 2 s 22 p 4 • • Cl • • O • • • e. g. use Lewis symbols to illustrate the formation of a compound of sodium and sulfur Na 2 S Na+ combined with S 2–

Lattice Energy Ionic Bonding; electrostatic attraction of positive (cation) and negative (anion) ions e– transfer Neutral atoms cation + anion (IE + EA) Ionic compound Lattice energy (lattice energy) energy released when gaseous ions combine to form crystalline solid (an ionic compound) e. g. lattice energy of Na. Cl is 787 k. J/mol: Na+(g) + Cl–(g) --> Na. Cl(s) DH = – 787 k. J Bigger ions smaller lattice energy Higher charge larger lattice energy
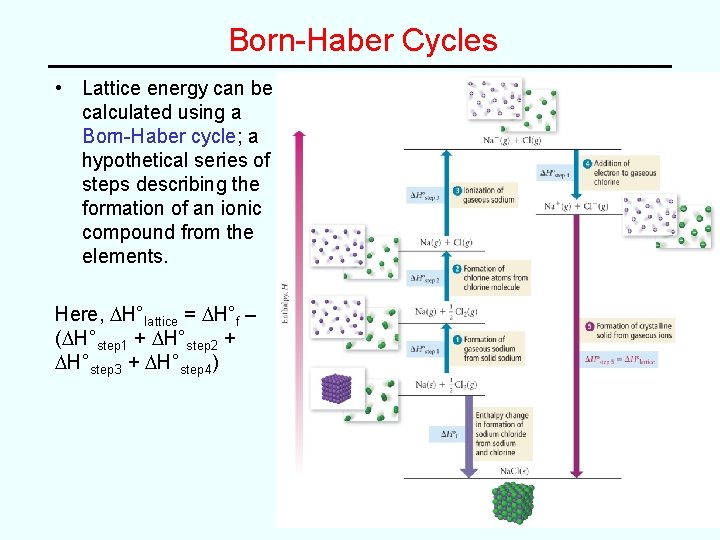
Born-Haber Cycles • Lattice energy can be calculated using a Born-Haber cycle; a hypothetical series of steps describing the formation of an ionic compound from the elements. Here, DH°lattice = DH°f – (DH°step 1 + DH°step 2 + DH°step 3 + DH°step 4)
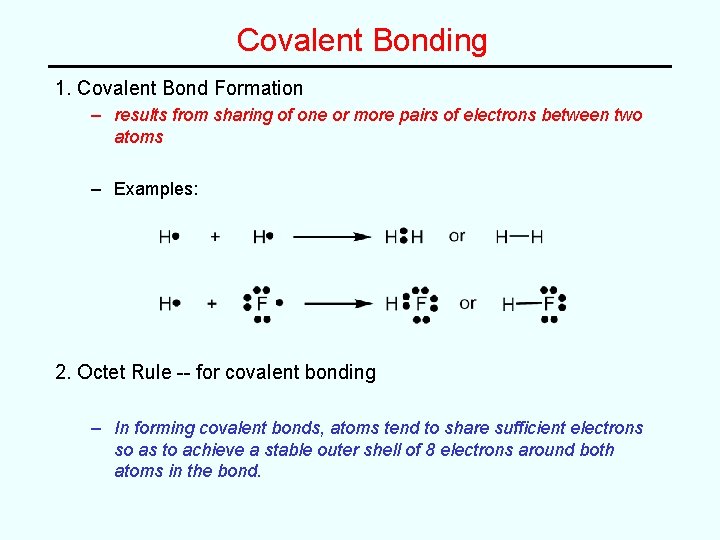
Covalent Bonding 1. Covalent Bond Formation – results from sharing of one or more pairs of electrons between two atoms – Examples: 2. Octet Rule -- for covalent bonding – In forming covalent bonds, atoms tend to share sufficient electrons so as to achieve a stable outer shell of 8 electrons around both atoms in the bond.
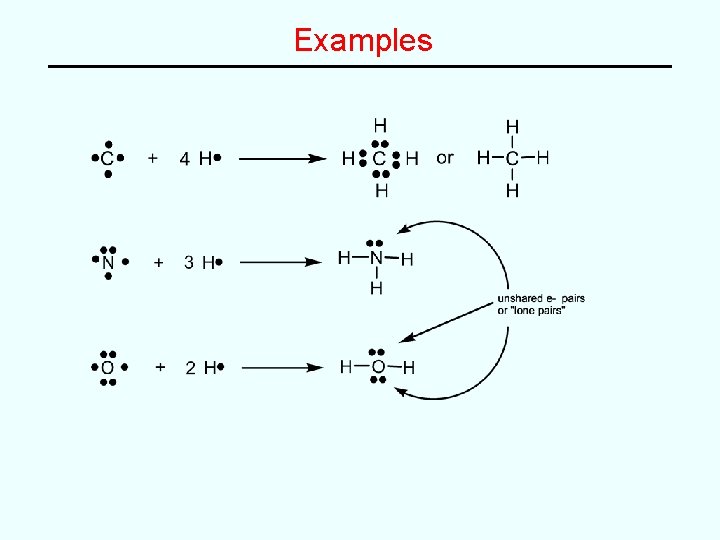
Examples
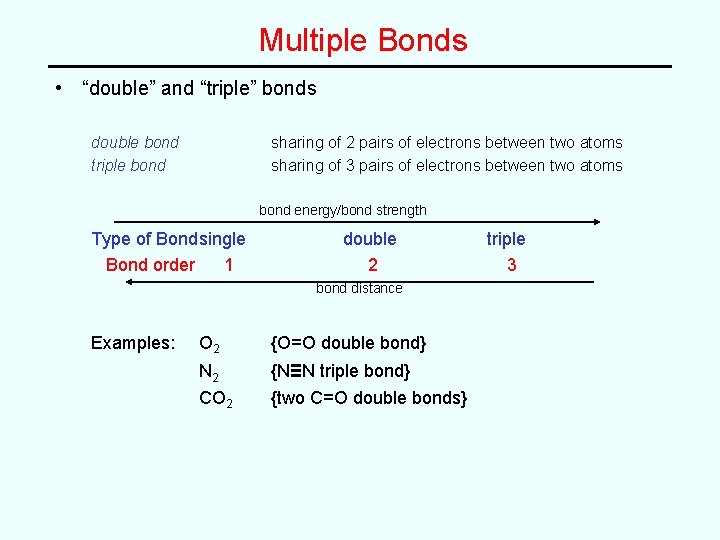
Multiple Bonds • “double” and “triple” bonds double bond triple bond sharing of 2 pairs of electrons between two atoms sharing of 3 pairs of electrons between two atoms bond energy/bond strength Type of Bondsingle Bond order 1 double 2 bond distance Examples: O 2 {O=O double bond} N 2 CO 2 {N≡N triple bond} {two C=O double bonds} triple 3
Electronegativity and Bond Polarity electronegativity tendency of an atom in a molecule to attract electrons to itself Electronegativity Increases Periodic Table e. g. Cl is more electronegative than H, so there is partial charge separation in the H-Cl bond: • • • The H-Cl bond is described as “polar” and is said to have a “dipole” The entire HCl molecule is also polar as a result More complex molecules can be polar or nopolar, depending on their 3 -D shape (Later)

Lewis Electron Dot Structures General Procedure -- stepwise process --Write the skeletal structure (which atoms are bonded? ) --H atoms terminal --More electronegative atoms terminal (e. g. halogens) --Count all valence electrons (in pairs) and charges --Place 2 electrons (1 pair) in each bond --Complete the octets of the terminal atoms --Use multiple bonds if needed to complete the octet of the central atom, or --Put any remaining electron pairs on the central atom --Show formal charges Apply the OCTET RULE as follows: --H never has more than 2 electrons (I. e. one bond) --2 nd row elements (e. g. C, N, O) almost always have an octet and never have more than 8 electrons (sometimes boron has only 6) --3 rd row and higher elements can have more than 8 electrons but only after the octets of any 2 nd row elements are completed

Formal Charge • The “apparent” charge on an atom in a covalent bond • = (# of valence e– in the isolated atom) - (# of bonds to the atom) - (# of unshared electrons on the atom) minimize formal charges whenever possible (but the octet rule takes priority!) NH 4+ CO Put a circle around the formal charge. All nonzero formal charges must be shown in a Lewis structure.

Lewis Electron Dot Structures General Procedure -- stepwise process --Write the skeletal structure (which atoms are bonded? ) --H atoms terminal --More electronegative atoms terminal (e. g. halogens) --Count all valence electrons (in pairs) and charges --Place 2 electrons (1 pair) in each bond --Complete the octets of the terminal atoms --Use multiple bonds if needed to complete the octet of the central atom, or --Put any remaining electron pairs on the central atom --Show formal charges and resonance forms as needed Apply the OCTET RULE as follows: --H never has more than 2 electrons (I. e. one bond) --2 nd row elements (e. g. C, N, O) almost always have an octet and never have more than 8 electrons (sometimes boron has only 6) --3 rd row and higher elements can have more than 8 electrons but only after the octets of any 2 nd row elements are completed

Resonance • When multiple bonds are present, a single Lewis structure may not adequately describe the compound or ion -- occurs whenever there is a “choice” of where to put a multiple bond. e. g. the HCO 2– ion is a “resonance hybrid” of two “contributing resonance structures” The C-O bond order is about 1. 5 (average of single and double bonds) All reasonable resonance structures must be shown in a Lewis structure.

Lewis Dot Structure Checklist • • • Correct total # of valence electrons Correct connectivity NO atoms with > octet (or duet) in group 2 or below All atoms have octets (or duets), if possible Lone electrons clearly shown Nonzero formal charges included (and circled) – Note it should be “ 2+” not “+2” – If the charge is 1+ or 1 - do not write the “ 1”! • Important resonance structures included • Ions have brackets and overall charge (not circled)

Exceptions to the Octet Rule • Odd-Electron Species – “radicals” or “free radicals” • Incomplete Octets – Group 3 elements often have 6 electrons total, e. g. Al. Cl 3 – Often form bonds to complete the octets • Expanded octets – Only 3 rd row or higher

Sample Problems • Write Lewis Electron Dot Structures (including formal charges and/or resonance as needed) for the following compounds and ions. PF 3 HCN SF 5– NO 2– SOCl 2 O 3 HNO 3 H 2 CO N 3 –

Sample Problems • Write Lewis Electron Dot Structures (including formal charges and/or resonance as needed) for the following compounds and ions. PF 3 HCN SF 5– NO 2– SOCl 2 O 3 HNO 3 H 2 CO N 3 –

Bond Energies and Estimating DH • Bond Energy—energy required to break 1 mole of the bond in the gas phase, e. g. • HCl(g) H(g) + Cl(g) DH = 431 k. J/mol • CH 4(g) CH 3(g) + H(g) DH = 438 k. J/mol • Endothermic! • Estimating DHrxn using bond energies; DHrxn = ∑(DH bonds broken) – ∑(DH bonds formed)

Sample Problem Ethanol is a possible fuel. Use average bond energies to calculate DHrxn for the combustion of gaseous ethanol.

Sample Problem Ethanol is a possible fuel. Use average bond energies to calculate DHrxn for the combustion of gaseous ethanol. CH 3 CH 2 OH(g) + 3 O 2(g) 2 CO 2(g) + 3 H 2 O(g) DHrxn = -1245 k. J/mol
- Slides: 20