Chapter 9 Chemical Bonding I Lewis Theory Chapter

Chapter 9 Chemical Bonding I: Lewis Theory Chapter 9

Chapter 9 and 10 Bonding Theories • Lewis Model (Theory) – Ch 9 – Treats electrons as dots and bonds as dashes • Valence Bond theory – Ch 10 – Examines Molecular Geometry – Look at molecules in Three dimensions – More quantum mechanical view of bonding • Molecular Orbital theory – Ch 10 – Fully quantum mechanical view of bonding 2

Chapter 9 and 10 Bonding Theories • Which theory is correct? • Why do we have more than one? • Different theories of bonding are useful to examine different aspect of bonding and chemical behavior. • We will come back to this idea as we go along. 3

Chapter 9 Chemical Bonding I: Lewis Theory 9. 1 9. 2 9. 3 9. 4 9. 5 9. 6 9. 7 9. 8 9. 9 Bonding Models and Aids Drugs Types of Chemical Bonds Representing Valence Electrons with Dots Ionic Bonding: Lewis Structures and Lattice Energies Covalent Bonding: Lewis Structures Electronegativity and Bond Polarity Lewis Structures of Molecular Compounds and Polyatomic Ions Resonance and Formal Charge Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets 9. 10 Bond Energies and Bond Lengths 9. 11 Bonding in Metals: The Electron Sea Model 4

Section 9. 1 Bonding Models and Aids Drugs HIV Protease and Bonding Theory • HIV Protease – Protein that is essential to the ability of the Human Immunodeficiency virus (HIV) to survive and replicate – The structure was solved using X Ray crystallography • To construct a molecule to disable the protease researchers used bonding theory 5

Section 9. 2 Types of Chemical Bonds • Chemical Bonds form because they lower the potential energy between the charged particles that compose atoms • We will discuss metallic bonding in Section 9. 11 6

Section 9. 3 Representing Valence Electrons with Dots Lewis symbols • To write a Lewis Symbol for an element – Abbreviation for the element – Represent the valence electrons with dots – Number of valence electrons = group number for representative elements 7

Section 9. 3 Representing Valence Electrons with Dots Chemical Bond and The Octet Rule • In the Lewis Model a Chemical Bond is the transfer or sharing of electrons to attain the most stable electron configuration for the bonded atoms. – Ionic bonds – transfer electrons – Covalent bonds – share electrons • Octet (8 valence electrons) is the most stable electron configuration – Period 1 elements have a duet 8

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Ionic Bonding and Electron Transfer • We can use Lewis Symbols to represent the electron transfer that takes place in the formation of an ionic bond. • Both the K and Cl attain an octet in the transfer • K 1 s 22 p 63 s 23 p 64 s 1 K+ 1 s 22 p 63 s 23 p 6 = Ar – K loses the 4 s electron • Cl 1 s 22 p 63 s 23 p 5 Cl– 1 s 22 p 63 s 23 p 6 = Ar 9

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Lattice energy • The formation of ionic compounds is quite exothermic • Na (s) + ½ Cl 2 (g) Na. Cl (s) DH f = – 411 k. J/mol • Where does all this energy come from? 10

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Lattice energy The energy associated with the formation of a crystalline lattice of cations and anions from the gaseous ions. Fig 9 -3 11

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Lattice energy • When the Na + (g) and Cl – (g) come together into a crystal lattice the potential energy decreases according to Coulombs Law • The decrease in potential energy corresponds to the energy released. 12

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies The Born – Haber Cycle • A Method to determine the value of the lattice energy. • The Heat of formation is calculated from solid sodium and molecular chlorine – Na (s) + ½ Cl 2 (g) Na. Cl (s) DH f = – 411 k. J/mol • The Lattice energy is determined from gaseous ionic sodium and gaseous ionic chlorine – Na+ (g) + Cl – (g) Na. Cl (s) DH° = lattice energy 13
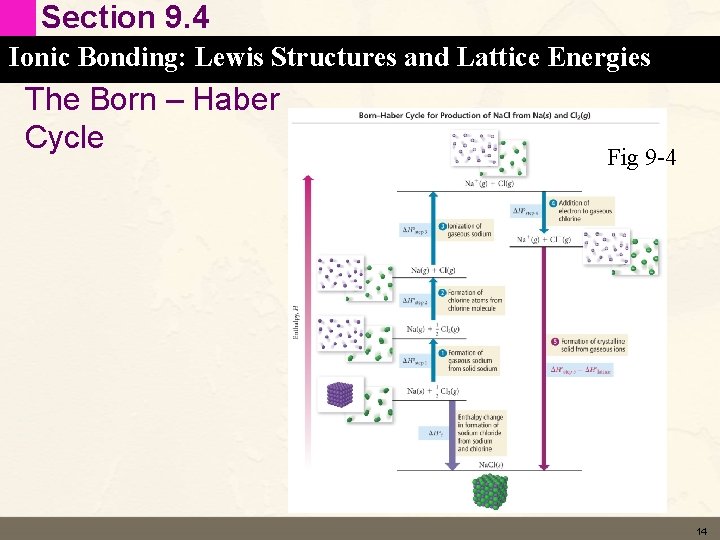
Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies The Born – Haber Cycle Fig 9 -4 14
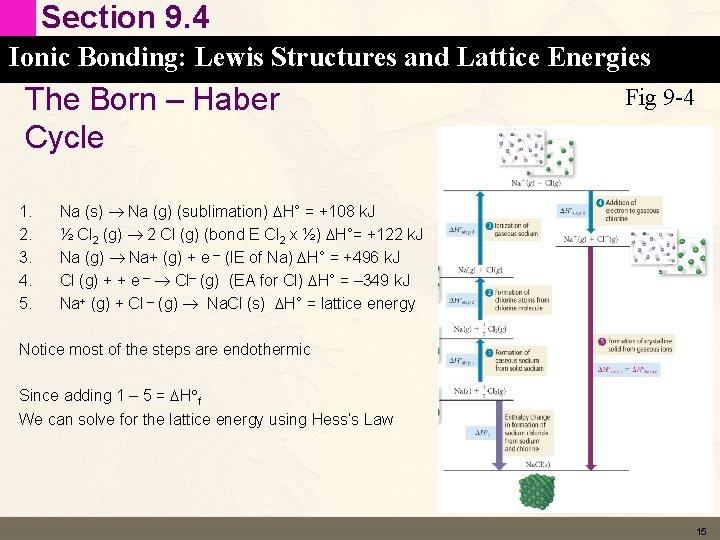
Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies The Born – Haber Cycle 1. 2. 3. 4. 5. Fig 9 -4 Na (s) Na (g) (sublimation) DH° = +108 k. J ½ Cl 2 (g) 2 Cl (g) (bond E Cl 2 x ½) DH°= +122 k. J Na (g) Na+ (g) + e – (IE of Na) DH° = +496 k. J Cl (g) + + e – Cl– (g) (EA for Cl) DH° = – 349 k. J Na+ (g) + Cl – (g) Na. Cl (s) DH° = lattice energy Notice most of the steps are endothermic Since adding 1 – 5 = DH f We can solve for the lattice energy using Hess’s Law 15

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies The Born – Haber Cycle • DH f = DH 1 + DH 2 + DH 3 + DH 4 + lattice energy • Lattice energy = DH f – (DH 1 + DH 2 + DH 3 + DH 4) 16
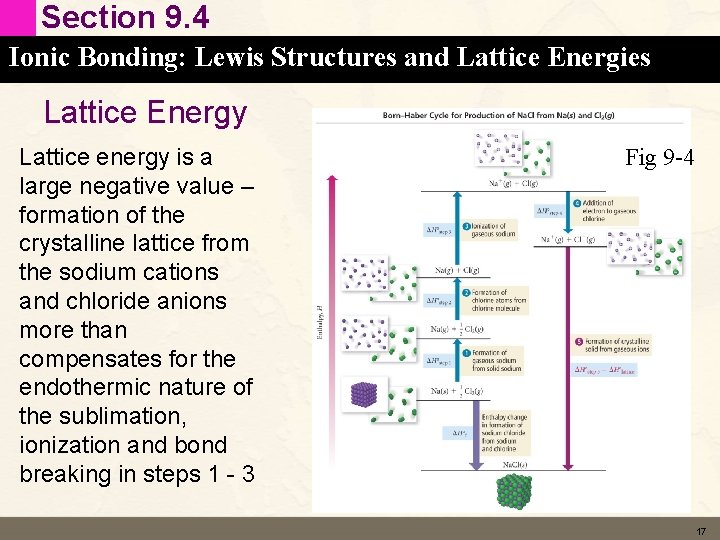
Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Lattice Energy Lattice energy is a large negative value – formation of the crystalline lattice from the sodium cations and chloride anions more than compensates for the endothermic nature of the sublimation, ionization and bond breaking in steps 1 - 3 Fig 9 -4 17

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Think – pair - share • Use Coulombs law to explain why the lattice energy for Ca. O is so much larger than the value for Na. F. 18

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Solution • Ca 2+ x O 2– gives a value of E roughly 4 x as large as Na+ x F– 19

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Think – pair - share • Use Coulombs law to explain why lattice energies decrease for metal chlorides as we go down a group in the periodic table. 20

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Solution • • • As we move down a group in the periodic table the size of the cation increases According to Coulomb’s Law the magnitude of the potential energy depends on the inverse of the radius between the interacting particles. As the radius increases the value of E (lattice energy) decreases. 21

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Ionic Bonding: Models and Reality • • How well does the Lewis Model for Ionic Bonding predict the behavior of ionic compounds? High melting and boiling point − − − Ions are charged spheres that have a uniform field of force around them and hence attract oppositely charged ions from all directions. Hence, ionic bond is non -directional. Bonding is non directional and involves all the particles in the lattice. Breaking all these interactions leads to high MP and BP. 22

Section 9. 4 Ionic Bonding: Lewis Structures and Lattice Energies Ionic Bonding: Models and Reality • • How well does the Lewis Model for Ionic Bonding predict the behavior of ionic compounds? Do not conduct electricity as solids − • No free ions moving around in the solid state Conduct electricity when dissolved in water − Free ions floating around 23

Section 9. 5 Covalent Bonding: Lewis Structures Molecular Compounds as Lewis Structures • Lewis Structures for Molecular Compounds – Depicts neighboring atoms as sharing some (or all) of their valence electrons – In order to attain an octet (or duet for Hydrogen) 24
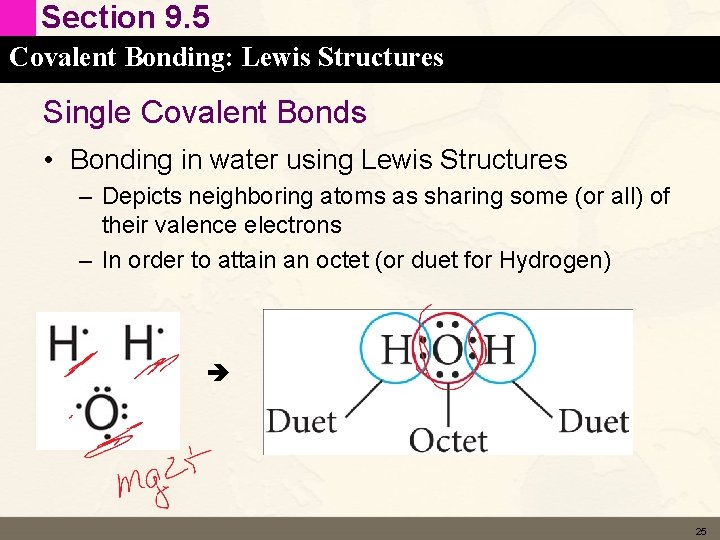
Section 9. 5 Covalent Bonding: Lewis Structures Single Covalent Bonds • Bonding in water using Lewis Structures – Depicts neighboring atoms as sharing some (or all) of their valence electrons – In order to attain an octet (or duet for Hydrogen) 25
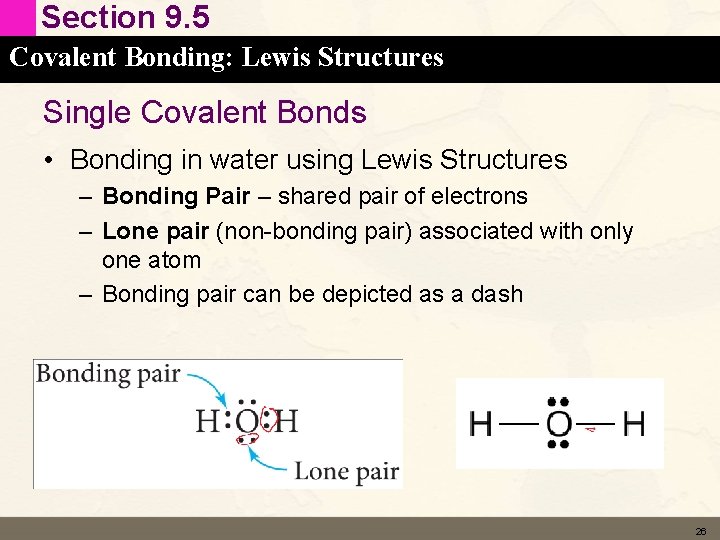
Section 9. 5 Covalent Bonding: Lewis Structures Single Covalent Bonds • Bonding in water using Lewis Structures – Bonding Pair – shared pair of electrons – Lone pair (non-bonding pair) associated with only one atom – Bonding pair can be depicted as a dash 26

Section 9. 5 Covalent Bonding: Lewis Structures Single Covalent Bonds • Bonding in chlorine using Lewis Structures • Notice the mistake in the book on page 392 in the Lewis Symbol for Chlorine. 27

Section 9. 5 Covalent Bonding: Lewis Structures Double and triple bonds • Atoms can share more than one electron pair to attain the octet. • Oxygen shares two electron pairs – Double bond • Nitrogen shares three – Triple bond (strong) – : N: : : N: 28
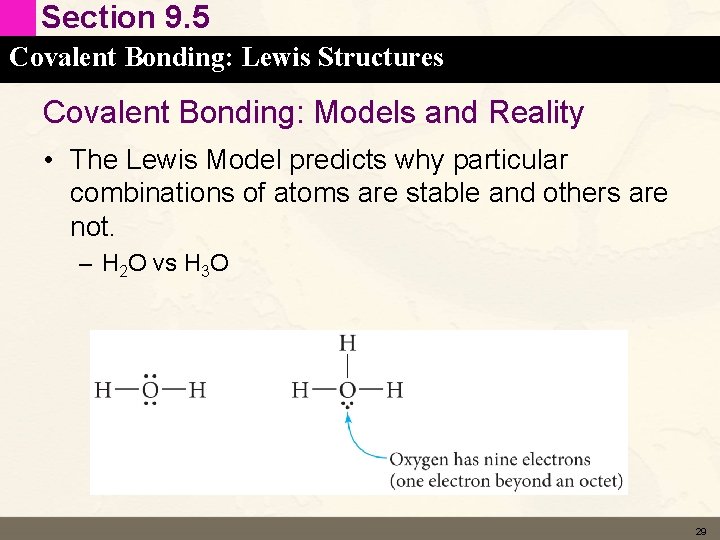
Section 9. 5 Covalent Bonding: Lewis Structures Covalent Bonding: Models and Reality • The Lewis Model predicts why particular combinations of atoms are stable and others are not. – H 2 O vs H 3 O 29
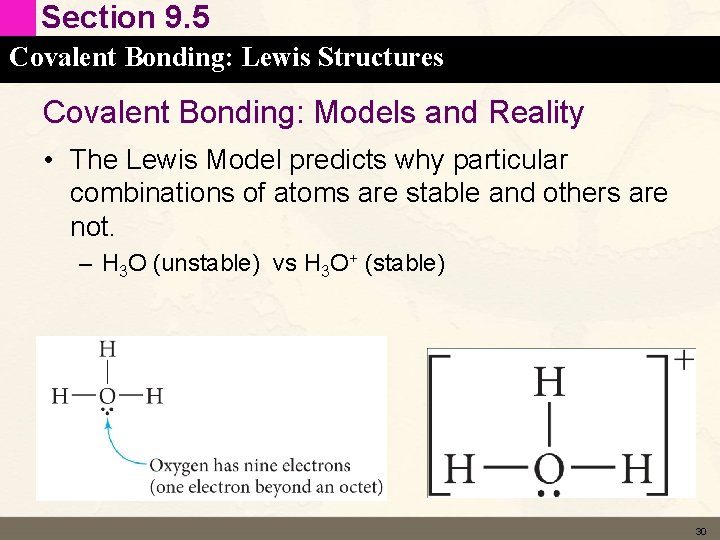
Section 9. 5 Covalent Bonding: Lewis Structures Covalent Bonding: Models and Reality • The Lewis Model predicts why particular combinations of atoms are stable and others are not. – H 3 O (unstable) vs H 3 O+ (stable) 30

Section 9. 5 Covalent Bonding: Lewis Structures Covalent Bonding: Models and Reality • In the section on ionic bonding we discussed how ionic bonds were non directional and hold together an entire array or atoms • In contrast covalent bonds are highly directional and each bond links just two atoms. • Instead of having to break all the strong interactions in the entire array to melt or boil we are instead breaking up relatively weak interactions between the molecules 31
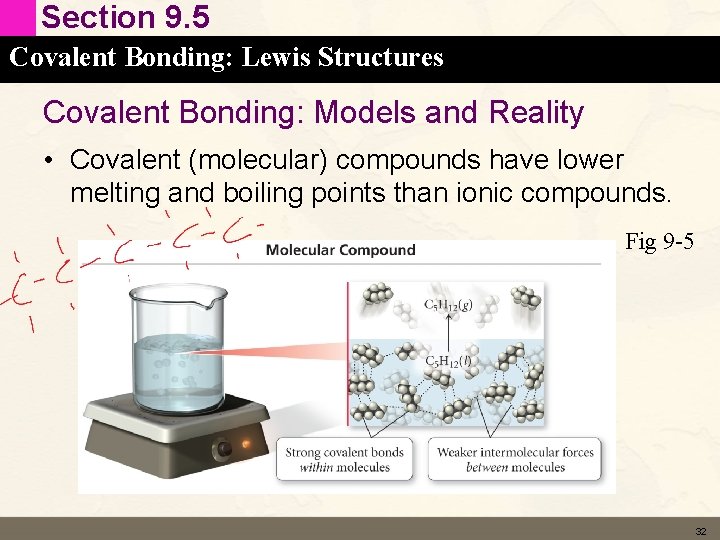
Section 9. 5 Covalent Bonding: Lewis Structures Covalent Bonding: Models and Reality • Covalent (molecular) compounds have lower melting and boiling points than ionic compounds. Fig 9 -5 32

Section 9. 6 Electronegativity and Bond Polarity Polar Covalent Bonds • The Lewis structure for HF implies that the 2 electrons that form the bond between H and F are shared equally. • This is not what we observe in the lab. 33
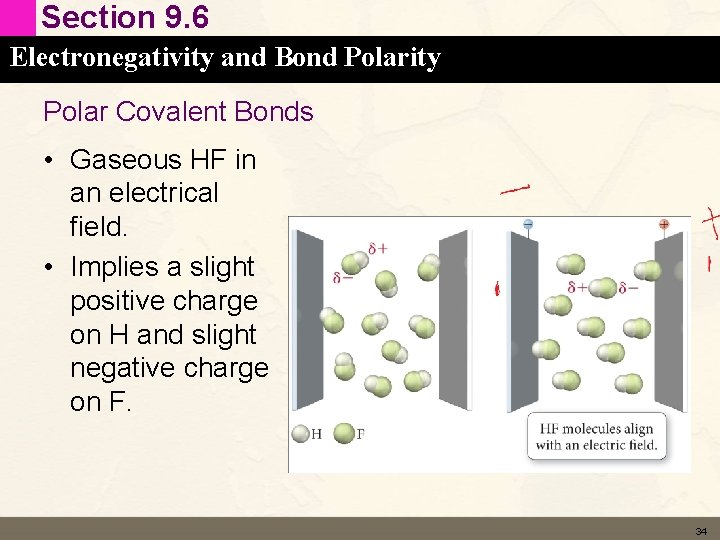
Section 9. 6 Electronegativity and Bond Polarity Polar Covalent Bonds • Gaseous HF in an electrical field. • Implies a slight positive charge on H and slight negative charge on F. 34

Section 9. 6 Electronegativity and Bond Polarity Polar Covalent Bonds • HF is a polar covalent bond • Most covalent bonds between dissimilar atoms have some degree of polarity. 35
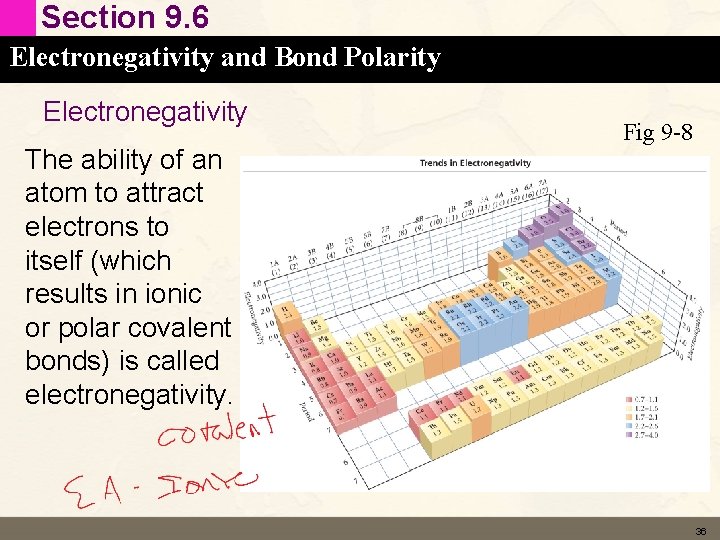
Section 9. 6 Electronegativity and Bond Polarity Electronegativity The ability of an atom to attract electrons to itself (which results in ionic or polar covalent bonds) is called electronegativity. Fig 9 -8 36

Section 9. 6 Electronegativity and Bond Polarity Electronegativity • Where did the values for electronegativity come from? • Compare H-H and F-F bond strength to H-F bond strength. • If H-F is purely covalent then H-F should be the average of H-H and H-F. • Not the case • H-F bond is a little stronger than the average of H-H and H-F. 37

Section 9. 6 Electronegativity and Bond Polarity Electronegativity • The extra energy of the H-F bond is due to the fact that when F is bonded to H it can hold the electrons a little more tightly – thus the bond is harder to break. • Essentially what we are measuring here is the amount of Fluorine’s electronegativity. 38
Section 9. 6 Electronegativity and Bond Polarity Electronegativity • Lots of measurements like this led to the values in the table. • Fluorine (being the most electronegative) was assigned a value of 4. • All the other elements are assigned a number relative to fluorine. 39
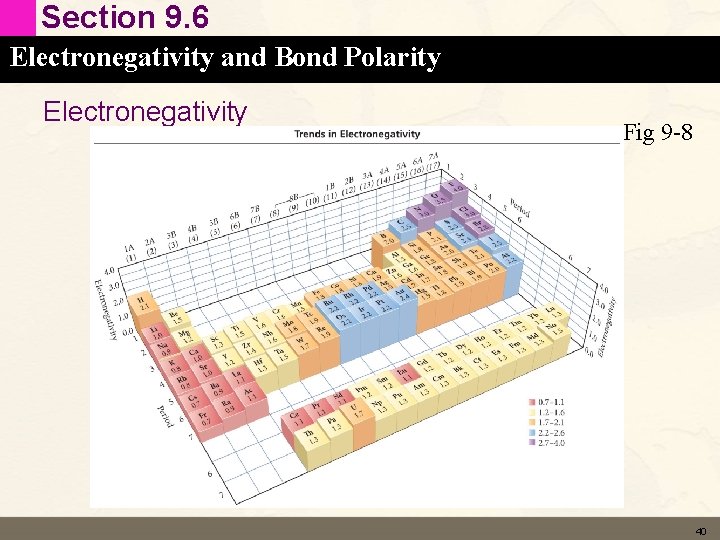
Section 9. 6 Electronegativity and Bond Polarity Electronegativity Fig 9 -8 40

Section 9. 6 Electronegativity and Bond Polarity Concept Check - Trends in Electronegativity • Explain the following Trends in Electronegativity in terms of the effective nuclear charge • Increases across a period. • Decreases down a column (group). 41

Section 9. 6 Electronegativity and Bond Polarity Solution - Trends in Electronegativity • Explain the following Trends in Electronegativity in terms of the effective nuclear charge • Increases across a period. – More protons added to the nucleus in the same principle energy level (higher Zeff) attract electrons more strongly. • Decreases down a column (group). – Atoms get larger – more principle energy levels shield the nucleus reducing Zeff. 42

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • Bonds can be (arbitrarily) classified as either non polar (pure covalent), polar or ionic based on the electronegativity difference of the bonded atoms. • Cl 2 non-polar • HCl polar • Na. Cl ionic 43
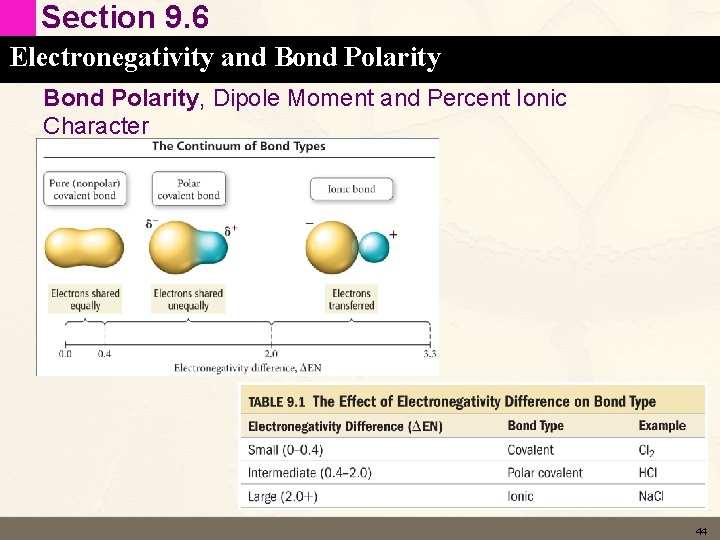
Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character 44

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • The table on the last slide characterizes bonds on the difference in electronegativity between the atoms. • We can also calculate how polar a bond is. • Dipole moment (m) occurs whenever there is a separation of positive and negative charge. • m = qr 45

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • • • For a completely ionic bond m = qr m = (1. 6 x 10 – 19 C) (130 x 10 – 12 m) m = 2. 1 x 10 – 12 C m m = 6. 2 D • Where D = debye (1 D = 3. 34 x x 10 – 30 C m 46

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • So for a completely ionic bond • Dipole moment ~ 6 • Smaller the magnitude of the charges or the smaller the distance between the charges the value of the dipole moment would decrease. 47

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • If we compare the actual (measured) dipole moment of a bond to what the dipole moment would be if the electron were completely transferred to the other atom we can determine the percent ionic character of the bond. 48

Section 9. 6 Electronegativity and Bond Polarity, Dipole Moment and Percent Ionic Character • As the difference in electronegativity increases the % ionic character increases. – No bond actually reaches 100% ionic. – In general bonds with greater than 50% ionic character are considered ionic bonds. 49

Section 9. 6 Electronegativity and Bond Polarity Concept Check • The HCl (g) molecule has a dipole moment of 1. 08 D. What is the percent ionic character. 50

Section 9. 6 Electronegativity and Bond Polarity Solution • The HCl (g) molecule has a dipole moment of 1. 08 D. What is the percent ionic character. 51

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions How to draw Lewis Structures • 1. Write a skeletal structure – Hydrogens are always terminal – Most electronegative terminal • 2. Sum the valence electrons for all the atoms • 3. Distribute the electrons so all the atoms have an octet – Start with terminal atoms – End with central atom • 4. If any atoms lack an octet, form double or triple bonds as needed. 52

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Lewis Structure for Phosphorus Trichloride • 1. Write a skeletal structure – Hydrogens are always terminal – Most electronegative terminal • 2. Sum the valence electrons for all the atoms • 3. Distribute the electrons so all the atoms have an octet – Start with terminal atoms – End with central atom • 4. If any atoms lack an octet, form double or triple bonds as needed. P 5, Cl; 3 x 7 = 21. Total = 26 53

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Lewis Structure for Sulfur Dioxide • 1. Write a skeletal structure – Hydrogens are always terminal – Most electronegative terminal • 2. Sum the valence electrons for all the atoms • 3. Distribute the electrons so all the atoms have an octet – Start with terminal atoms – End with central atom • 4. If any atoms lack an octet, form double or triple bonds as needed. O–S– O S; 6, O; 2 x 6 = 12. Total = 18 : O – S – O: : O – S = O 54

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Lewis Structure for Polyatomic Ions • • Uses the same rules Add electrons for negative charge Subtract electrons for positive charge Place ion inside brackets 55

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Concept Check Draw a Lewis structure for each of the following molecules or ions: NH 3 CO 2 CCl 4 CN – 56
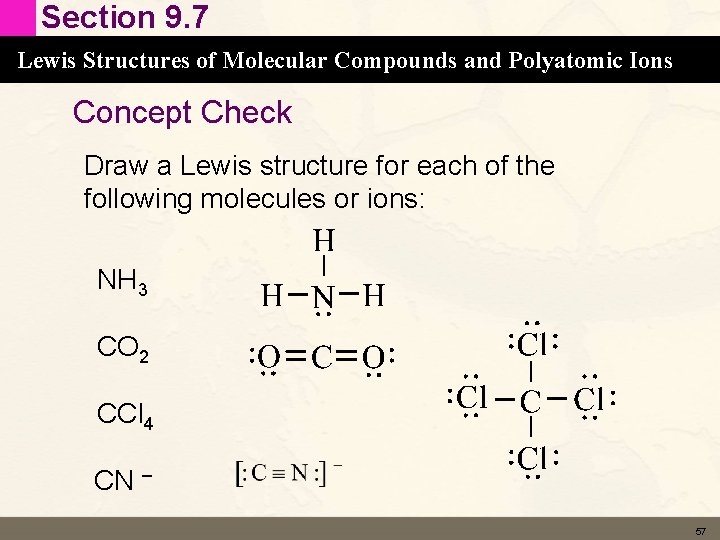
Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Concept Check Draw a Lewis structure for each of the following molecules or ions: NH 3 CO 2 CCl 4 CN – 57
Section 9. 8 Resonance and Formal Charge What if there is more than one Lewis Structure? • Resonance – More than one valid Lewis structure for a compound. • Formal Charge – An electron “book keeping” system that allow us to pick the “best” Lewis structure. 58

Section 9. 8 Resonance and Formal Charge Resonance • Two Lewis structures for Ozone (O 3) • Both satisfy the octet rule • The actual structure is an intermediate between the two • Resonance hybrid. 59

Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Concept Check Draw the resonance structures for the nitrate ion. 60
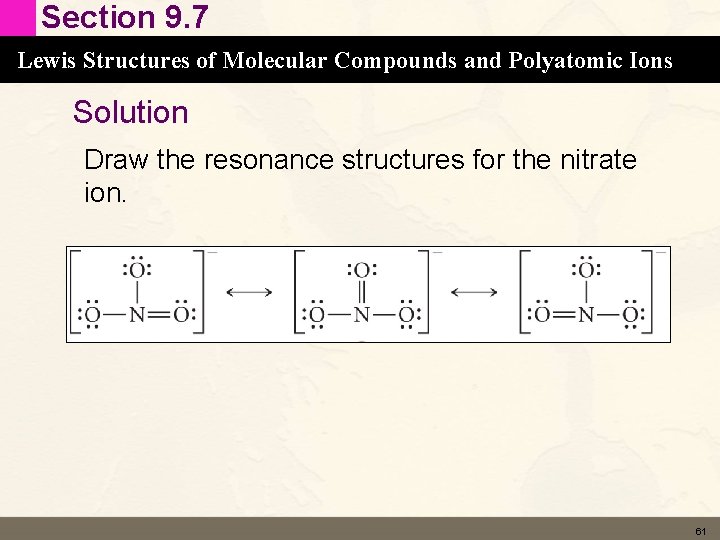
Section 9. 7 Lewis Structures of Molecular Compounds and Polyatomic Ions Solution Draw the resonance structures for the nitrate ion. 61

Section 9. 8 Resonance and Formal Charge Resonance • The concept of resonance is an adaptation of the Lewis model that helps account for the fact that even though we draw the electrons in a Lewis structure as localized to an individual atom, in reality electrons are delocalized over several atoms or bonds. – Delocalization contributes to the stability of the molecule. – The stabilization due to the formation of resonance structures is called resonance stabilization. 62

Section 9. 8 Resonance and Formal Charge • In the previous example both resonance structures were equivalent. • What if the resonance structures were not equivalent to each other? • If one was more stable than another. • How would we choose between them? 63
Section 9. 8 Resonance and Formal Charge • To decide between two seemingly equivalent Lewis structures we assign each atom a formal charge and use a set of rules to determine which is the better structure 64

Section 9. 8 Resonance and Formal Charge • Formal Charge = # valence electrons – # electrons “owned” in the Lewis structure • Formal charge = # valence electrons – (# of non bonding electrons + ½ # of bonding electrons) 65

Section 9. 8 Resonance and Formal Charge • Formal Charges for H and F in HF • Formal charge ignores the effects of electronegativity. • Assumes the electrons are shared equally 66

Section 9. 8 Resonance and Formal Charge • How do we use FC to decide between resonance structures? 1. Sum of FC in an molecule = 0. 2. Sum of FC in an ion = charge of ion. 3. Smaller FC is better. 4. Negative FC should reside on more electronegative atom. 67

Section 9. 8 Resonance and Formal Charge • Resonance structures for the cyanate ion. 68

Section 9. 8 Resonance and Formal Charge Learning Check • Assign a formal charge to each atom in the resonance forms of N 2 O. Which resonance form is likely to contribute most to the correct structure of N 2 O. 69

Section 9. 8 Resonance and Formal Charge Solution • Assign a FC to each atom in the resonance forms of N 2 O. Which resonance form is likely to contribute most to the correct structure of N 2 O. 70

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets The exceptions make the rule • The Lewis Model is a model • It is not perfect • It can’t perfectly account for and explain everything • Odd Electron species • Incomplete octets • Expanded octets 71

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Odd electron species • Odd Electron species • Molecules or ions with odd numbers of electrons in their Lewis structures – Free radicals (radicals) • Unstable - reactive 72

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Incomplete octets • Incomplete octets – B tends to only be surrounded by 6 electrons • Why doesn’t B form a double bond with F? 73

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Incomplete octets • Boron can complete its octet via reaction with a compound that donates a pair of electrons. 74
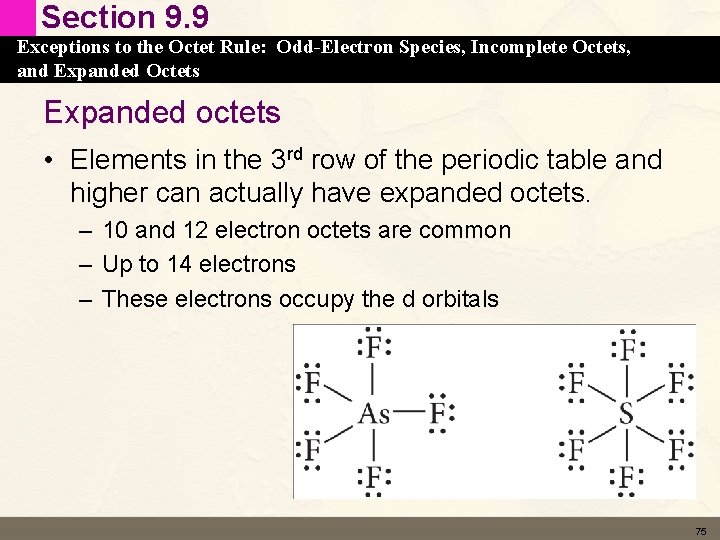
Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Expanded octets • Elements in the 3 rd row of the periodic table and higher can actually have expanded octets. – 10 and 12 electron octets are common – Up to 14 electrons – These electrons occupy the d orbitals 75

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Expanded octets • Can only expand the octet in the 3 rd row and above. – Requires the d orbitals • How can you decide whether to draw a Lewis structure with an expanded octet? 76

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Learning Check • Write Lewis structures for the following compounds where the central atom has an expanded octet. • Br. F 4 • PF 5 77

Section 9. 9 Exceptions to the Octet Rule: Odd-Electron Species, Incomplete Octets, and Expanded Octets Solution • Write Lewis structures for the following compounds where the central atom has an expanded octet. • Br. F 4 • PF 5 78

Section 9. 10 Bond Energies and Bond Lengths DH°rxn from Bond Energy • In Chapter 6 (Thermochemistry) we learned three different ways to determine the DHrxn – Calorimetry – Hess’s Law – Standard enthalpies of formation DH°rxn • Turns out we can also determine DHrxn from bond energies. 79

Section 9. 10 Bond Energies and Bond Lengths Bond Energy • The bond energy of a chemical bond is the energy required to break 1 mole of the bond in the gas phase. – Cl 2 (g) 2 Cl (g) – HCl (g) H(g) + Cl (g) DH = 243 k. J DH = 431 k. J • Bond energies are always positive – takes energy to break bonds – The more positive the bond energy the stronger the bond and the more stable (less reactive) the compound. 80

Section 9. 10 Bond Energies and Bond Lengths Bond Energy • Same kind of bond has different energy in different compounds – – H 3 C–H (g) H 3 C (g) + H(g) F 3 C–H (g) F 3 C (g) + H(g) Br 3 C–H (g) Br 3 C (g) + H(g) Cl 3 C–H (g) Cl 3 C (g) + H(g) DH = 438 k. J DH = 446 k. J DH = 402 k. J DH = 401 k. J • We can determine an average C–H bond energy. 81
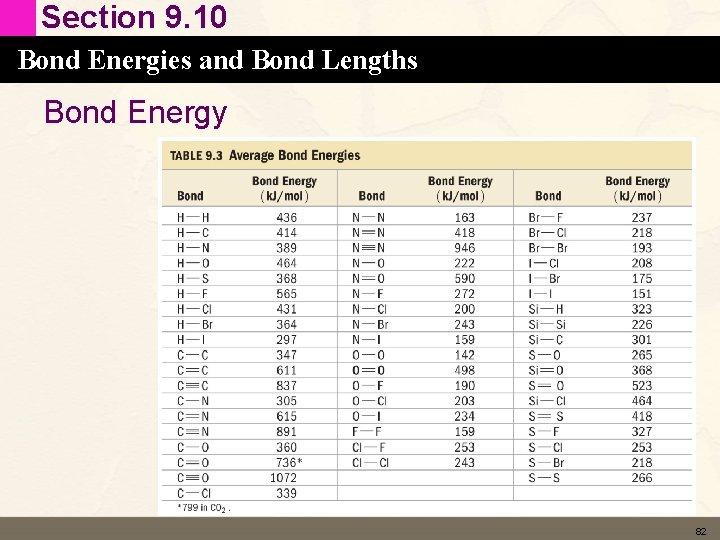
Section 9. 10 Bond Energies and Bond Lengths Bond Energy 82

Section 9. 10 Bond Energies and Bond Lengths Using Bond Energies to Estimate Enthalpy Changes for Reactions • Energy for bonds broken is positive (energy in) • Energy for bonds formed is negative (energy released) 83

Section 9. 10 Bond Energies and Bond Lengths Using Bond Energies to Estimate Enthalpy Changes for Reactions • Energy for bonds broken (reactants) • Energy for bonds formed (products) DHrxn = SDH reactants – SDH products 84

Section 9. 10 Bond Energies and Bond Lengths Using Bond Energies to Estimate Enthalpy Changes for Reactions • Exothermic – weak bonds break – strong bonds form • Endothermic – strong bonds break – weak bonds form 85

Section 9. 10 Bond Energies and Bond Lengths Sample problem with bond energy • C (s) + 2 H 2 O (g) 2 H 2 (g) + CO 2 (g) DHrxn = SDH reactants – SDH products • (4 O – H) – (2 H – H + 2 C=O) • [4 x 464] – [(2 x 436) + (2 x 799)] • 1856 – (872 + 1598) = – 614 k. J /mol 86
Section 9. 10 Bond Energies and Bond Lengths Learning Check • Use bond energies to calculate DHrxn for the reaction N 2 (g) + 3 H 2 (g) 2 NH 3 (g) 87

Section 9. 10 Bond Energies and Bond Lengths Solution • Use bond energies to calculate DHrxn for the reaction N 2 (g) + 3 H 2 (g) 2 NH 3 (g) 1 N 2 = 946 + 3 H 2 = 3(436) – 2 NH 3 = 6 N-H (389) [946 + 3(436)] – [6(389)] [946 + 1308] – 2334 2254 – 2334 = – 80 k. J/mol 88

Section 9. 10 Bond Energies and Bond Lengths • Longer bonds are weaker • Shorter bonds are stronger 89
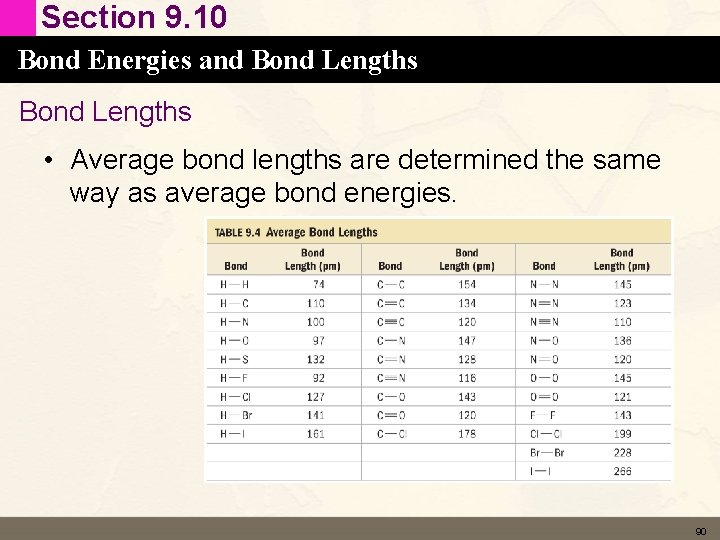
Section 9. 10 Bond Energies and Bond Lengths • Average bond lengths are determined the same way as average bond energies. 90

Section 9. 11 Bonding in Metals: The Electron Sea Model Bonding in Metals • We have looked at bonding between – Metal and non metal – ionic bonding – Non metal and non metal – covalent bonding • What about bonding in metals? • When metals bond together each atom donates its electrons to an “electron sea”. 91

Section 9. 11 Bonding in Metals: The Electron Sea Model • Sodium metal is an array of positively charged Na+ ions floating in a sea of negatively charged electrons (e –) 92

Section 9. 11 Bonding in Metals: The Electron Sea Model • The electron sea model accounts for the properties of metals: – Metals conduct electricity because electrons are not localized – they are free to move – Metals are good conductors because the electrons are free to move – Metals are malleable and ductile because there are no directional bonds – the metal ions slide freely past each other. 93
- Slides: 93