Chapter 9 Acids Bases Salts 9 9 The

Chapter 9 Acids, Bases, & Salts 9. 9 The Strengths of Acids and Bases 1

Strengths of Acids • A strong acid completely ionizes (100%) in aqueous solutions. HCl(g) + H 2 O(l) H 3 O+ (aq) + Cl− (aq) • A weak acid dissociates only slightly in water to form a few ions in aqueous solutions. H 2 CO 3(aq) + H 2 O(l) H 3 O+(aq) + HCO 3− (aq) 2

Strong Acids In water, the dissolved molecules of a strong acid • dissociate into ions. • give large concentrations of H 3 O+ and the anion (A-). Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 3

Weak Acids In weak acids, • only a few molecules dissociate. • most of the weak acid remains as the undissociated (molecular) form of the acid. • the concentrations of the H 3 O+ and the anion (A-) are small. HA(aq) + H 2 O(l) H 3 O(aq) + A−(aq) Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 4
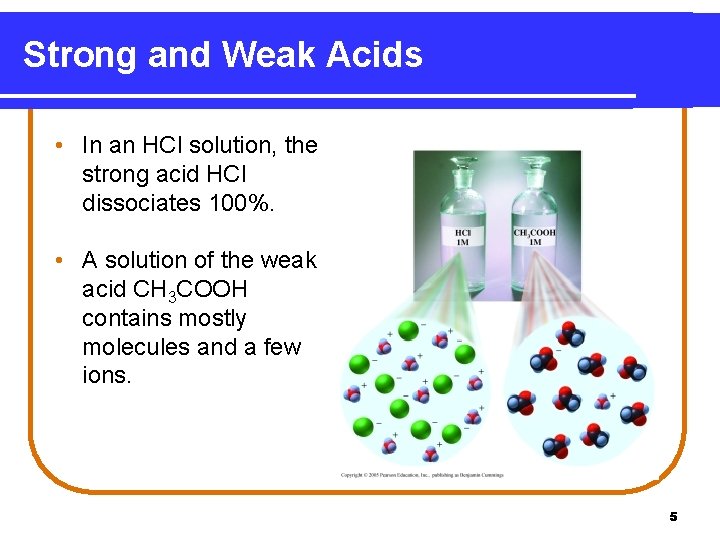
Strong and Weak Acids • In an HCl solution, the strong acid HCl dissociates 100%. • A solution of the weak acid CH 3 COOH contains mostly molecules and a few ions. 5
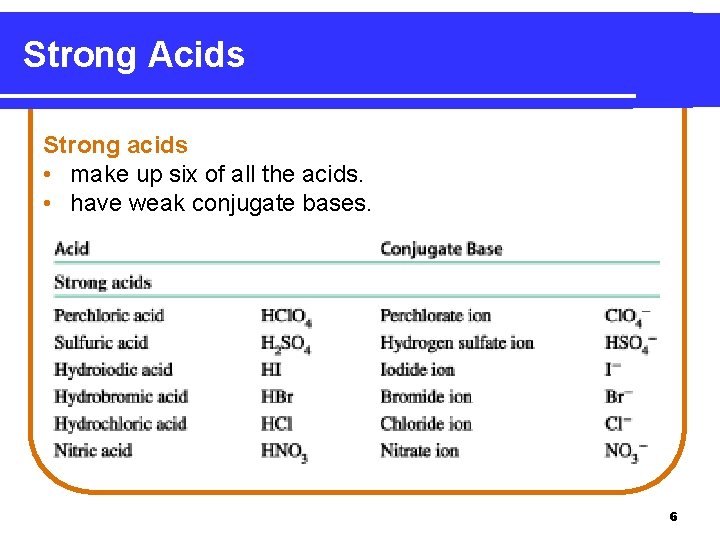
Strong Acids Strong acids • make up six of all the acids. • have weak conjugate bases. 6
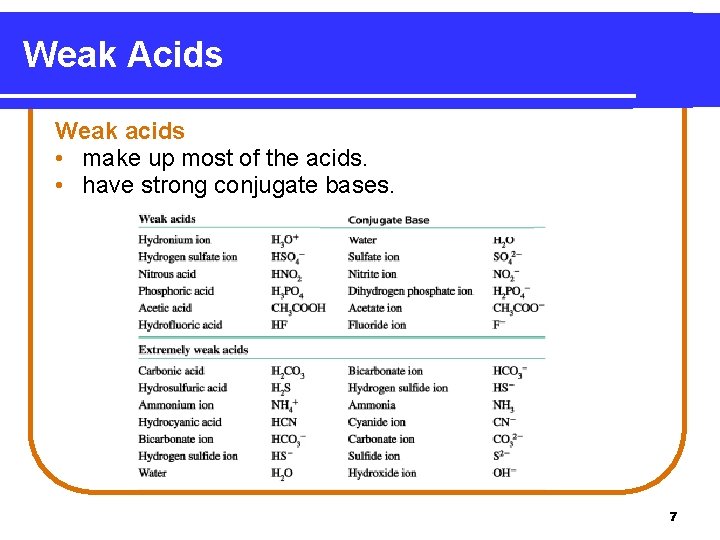
Weak Acids Weak acids • make up most of the acids. • have strong conjugate bases. 7
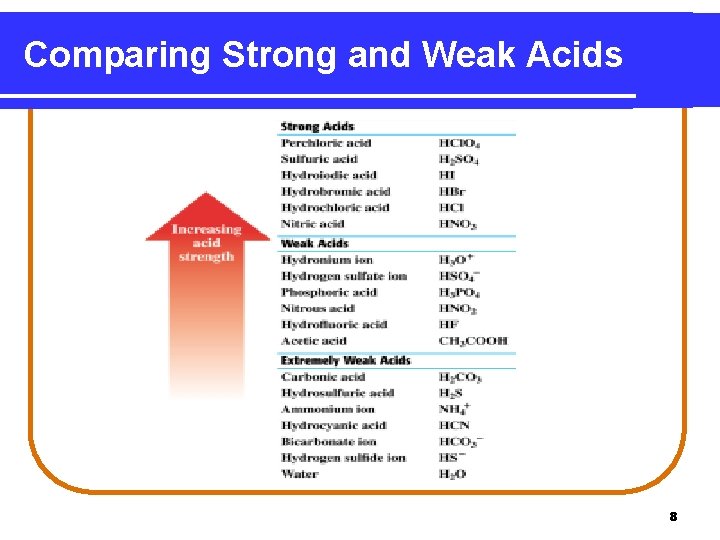
Comparing Strong and Weak Acids 8

Strong Bases Strong bases • are formed from metals of Groups 1 A (1) and 2 A (2). • include Li. OH, Na. OH, KOH, and Ca(OH)2. • dissociate completely in water. KOH(s) K+(aq) + OH−(aq) 9

Weak Bases Weak bases • are most other bases. • dissociate only slightly in water. • form only a few ions in water. NH 3(g) + H 2 O(l) NH 4+(aq) + OH−(aq) 10

Learning Check Identify each of the following as a strong or weak acid or base. A. B. C. D. E. HBr HNO 2 Na. OH H 2 SO 4 Cu(OH)2 11

Solution Identify each of the following as a strong or weak acid or base. A. B. C. D. E. HBr HNO 2 Na. OH H 2 SO 4 Cu(OH)2 strong acid weak acid strong base strong acid weak base 12

Learning Check Identify the stronger acid in each pair. 1. HNO 2 or H 2 S 2. HCO 3− or HBr 3. H 3 PO 4 or H 3 O+ 13

Solution Identify the stronger acid in each pair. 1. HNO 2 2. HBr 3. H 3 O+ 14

Acid Dissociation Constant l This is the equilibrium constant for the dissociation of an acid HA(aq) + H 2 O(l) H 3 O+(aq) + A-(aq) The equilibrium expression for this reaction is [H 3 O+][A-] K= [HA][H 2 O] The acid dissociation constant, Ka, is simply [H 3 O+][A-] K[H 2 O] = Ka = [HA] 15

Base Dissociation Constant l This is the equilibrium constant for the dissociation of a base B(aq) + H 2 O(l) OH-(aq) + HB+(aq) The equilibrium expression for this reaction is [OH-][HB+] K= [B][H 2 O] The base dissociation constant, Kb, is simply [OH-][HB+] K[H 2 O] = Kb = [B] 16

Learning Check Write dissociation reactions and expressions for Ka or Kb for each of the following weak acids and bases: 1. 2. 3. 4. Hydrocyanic acid (HCN) Phosphoric acid (H 3 PO 4) (1 st H only) Dihydrogen phosphate ion (H 2 PO 4 -) (1 st H only) Ammonia (NH 3) 17
![Solution +][CN-] [H 1. HCN H+ + CN-; Ka = [HCN] +][H PO -] Solution +][CN-] [H 1. HCN H+ + CN-; Ka = [HCN] +][H PO -]](http://slidetodoc.com/presentation_image_h/c9d0d92797791e882f914434c8b8c813/image-18.jpg)
Solution +][CN-] [H 1. HCN H+ + CN-; Ka = [HCN] +][H PO -] [H 2 4 2. H 3 PO 4 H+ + H 2 PO 4 -; Ka = [H PO ] 3 4 [H+][HPO 42 -] 3. H 2 PO 4 - H+ + HPO 42 -; Ka = [H PO -] 2 4 [OH-][NH 4+] 4. NH 3 OH- + NH 4+; Kb = [NH ] 3 Note that [H 3 O+] = [H+] 18
- Slides: 18