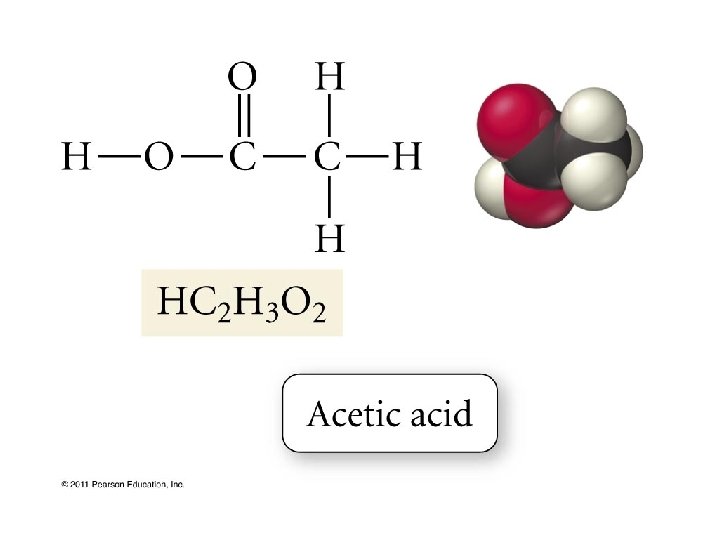
Chapter 9 Acids Bases Acids Acid a substance

Chapter 9: Acids & Bases
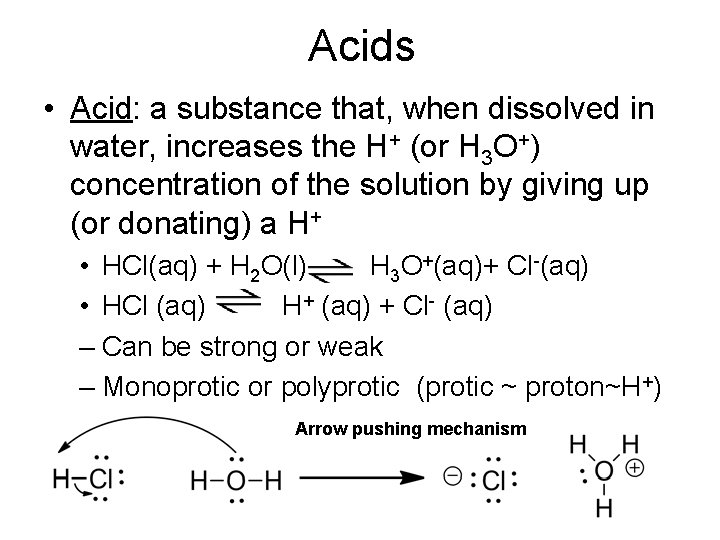
Acids • Acid: a substance that, when dissolved in water, increases the H+ (or H 3 O+) concentration of the solution by giving up (or donating) a H+ • HCl(aq) + H 2 O(l) H 3 O+(aq)+ Cl-(aq) • HCl (aq) H+ (aq) + Cl- (aq) – Can be strong or weak – Monoprotic or polyprotic (protic ~ proton~H+) Arrow pushing mechanism

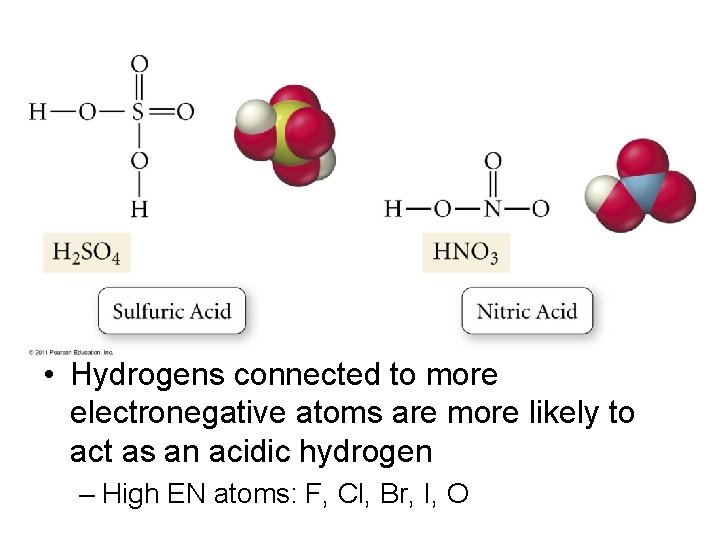
• Hydrogens connected to more electronegative atoms are more likely to act as an acidic hydrogen – High EN atoms: F, Cl, Br, I, O
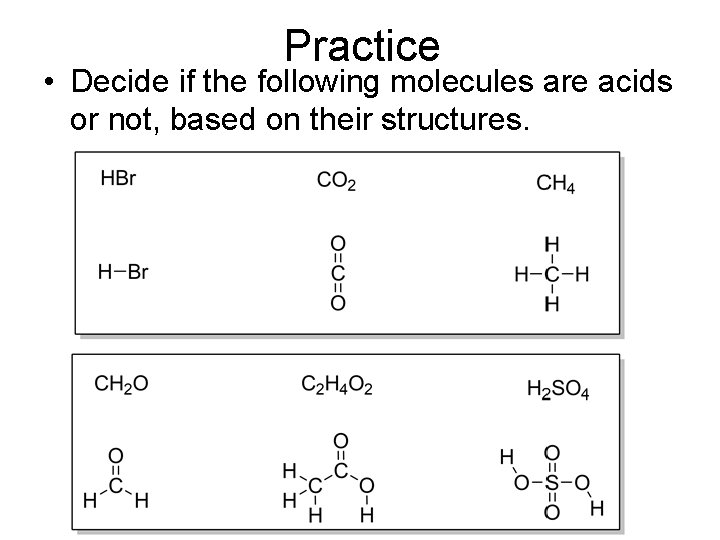
Practice • Decide if the following molecules are acids or not, based on their structures.

• Strong acid: an acid that completely ionizes/dissociates in water • HNO 3(aq) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq)
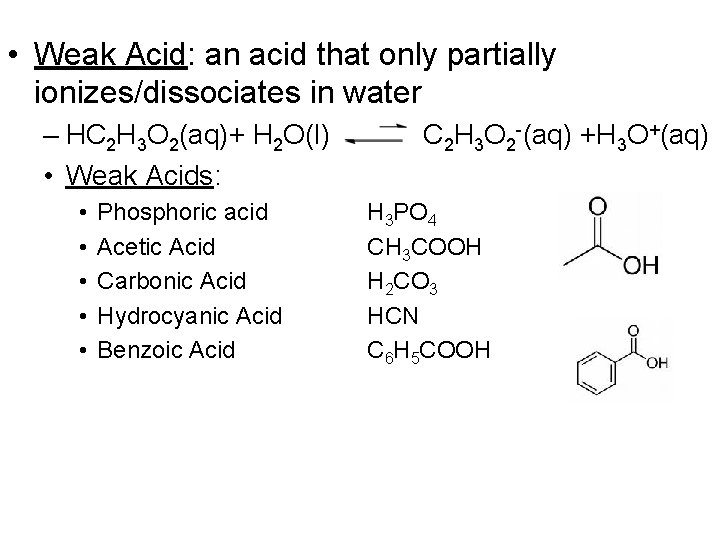
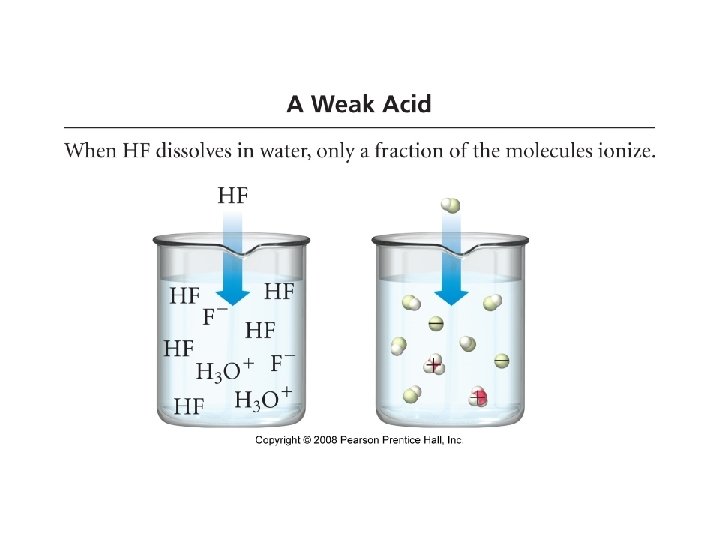
• Weak Acid: an acid that only partially ionizes/dissociates in water – HC 2 H 3 O 2(aq)+ H 2 O(l) • Weak Acids: • • • Phosphoric acid Acetic Acid Carbonic Acid Hydrocyanic Acid Benzoic Acid C 2 H 3 O 2 -(aq) +H 3 O+(aq) H 3 PO 4 CH 3 COOH H 2 CO 3 HCN C 6 H 5 COOH

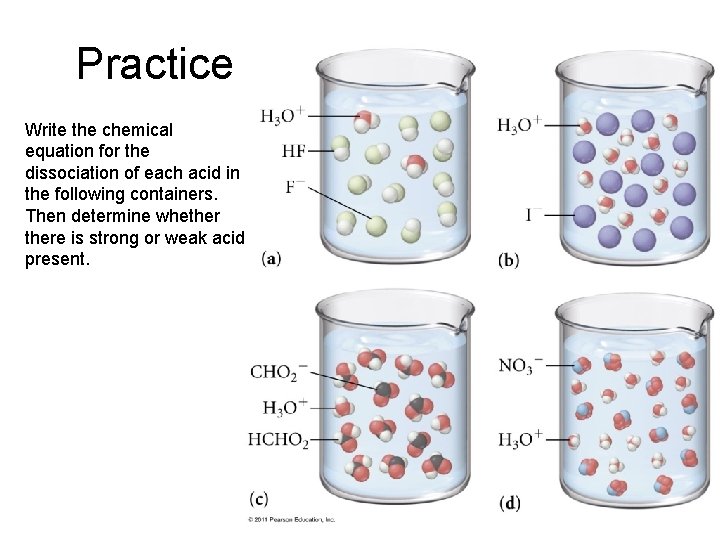
Practice Write the chemical equation for the dissociation of each acid in the following containers. Then determine whethere is strong or weak acid present.
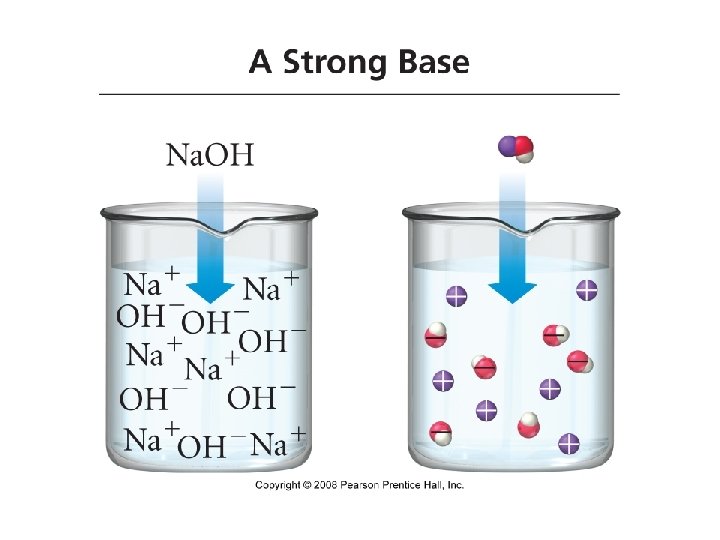
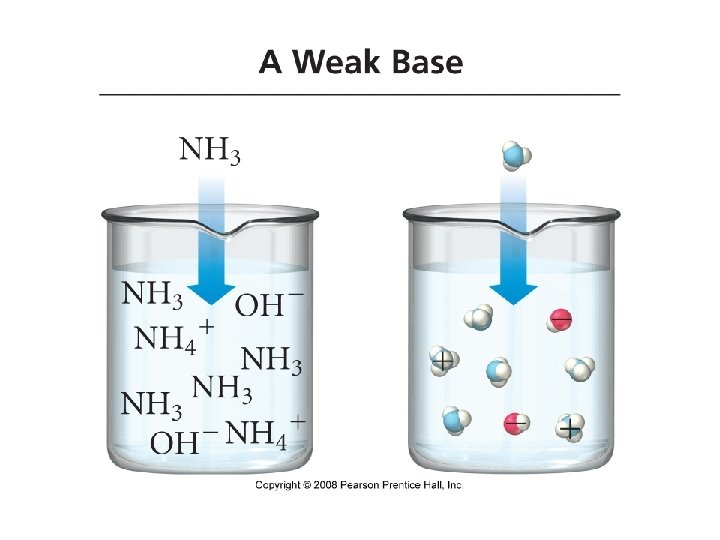
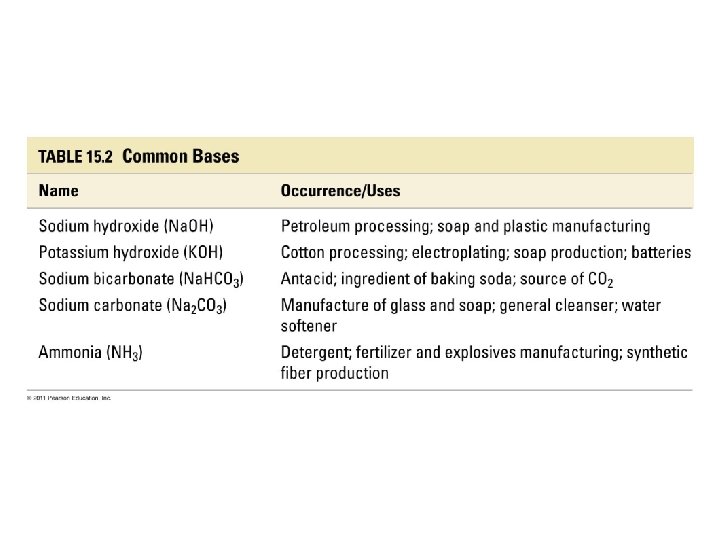
Bases • Base: a substance that, when put in water, increases the concentration of OH- ions or a substance that accepts H+ ions • Na. OH(aq) + H 2 O(l) Na+(aq) + OH-(aq) +H 2 O • Na. OH(aq) Na+(aq) + OH-(aq) • Strong Bases: bases that completely ionize in water Arrow pushing mechanism

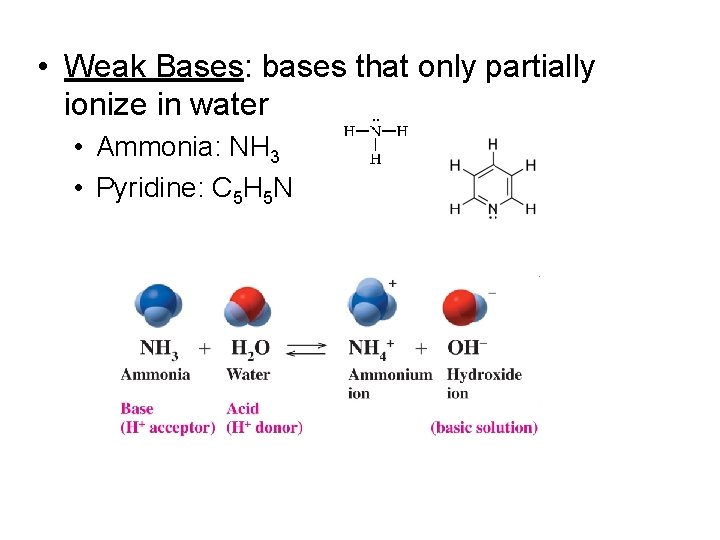
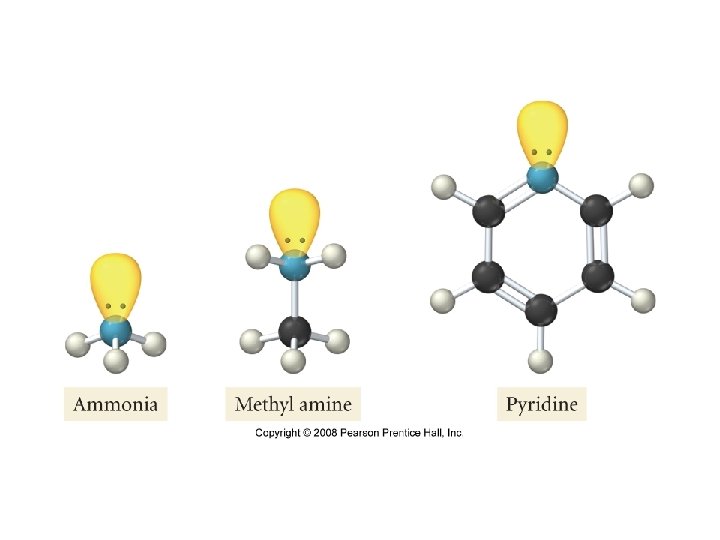
• Weak Bases: bases that only partially ionize in water • Ammonia: NH 3 • Pyridine: C 5 H 5 N

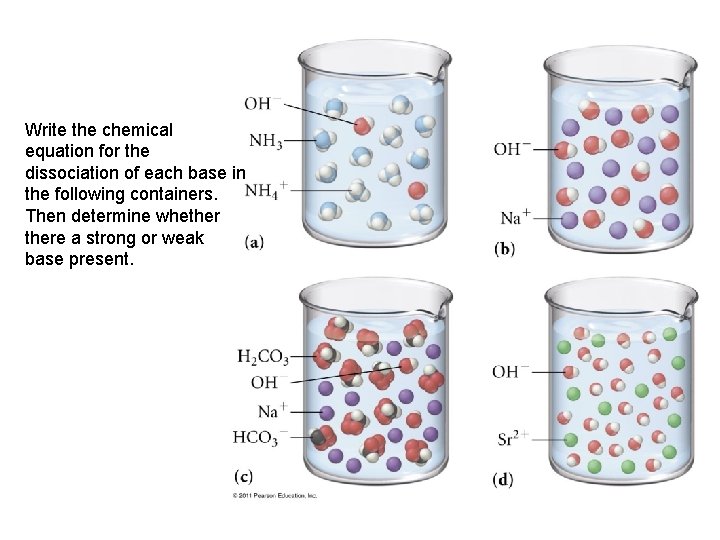
Write the chemical equation for the dissociation of each base in the following containers. Then determine whethere a strong or weak base present.

Polyprotic Acids • Polyprotic acids: acids that can release more than one H+ • HCl(aq) +H 2 O(l) Cl-(aq) + H 3 O+(aq) HCl = monoprotic acid • Complete ionization of H 2 SO 4 (sulfuric acid) H 2 SO 4(aq) + H 2 O(l) HSO 4 -(aq) + H 3 O+(aq) HSO 4 -(aq) + H 2 O(l) SO 42 -(aq) + H 3 O+(aq) H 2 SO 4 = polyprotic acid
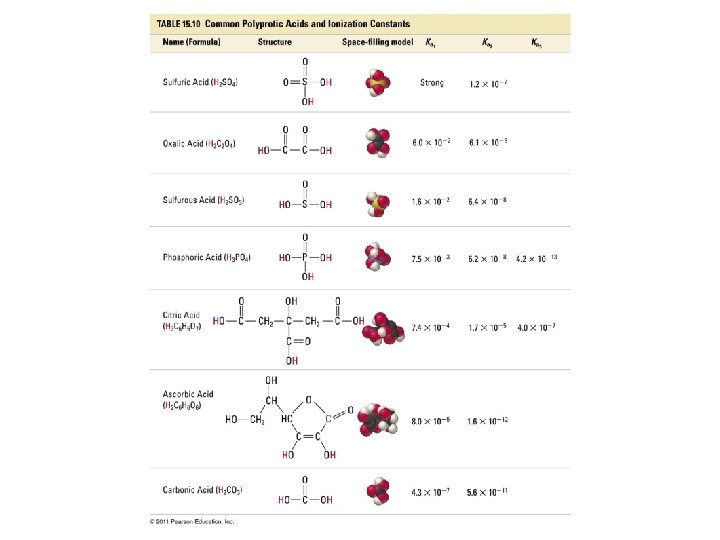

Problem • Write out the equation(s) for the complete ionization of phosphoric acid, H 3 PO 4, in water.

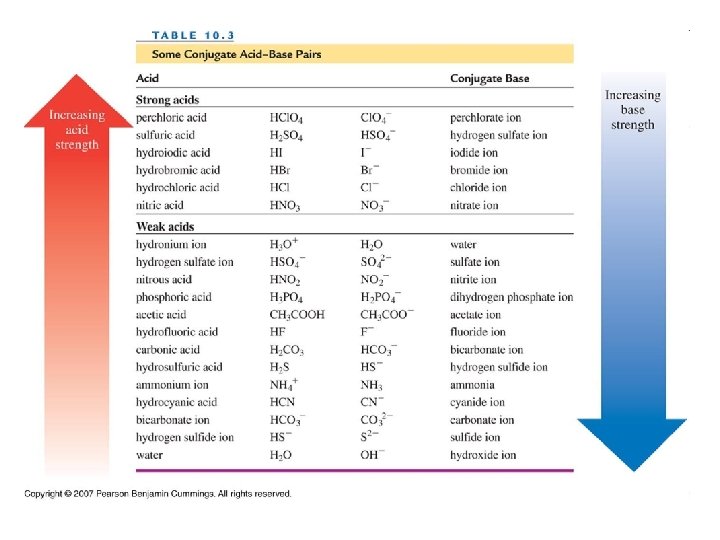
Congugate Acid-Base Pairs Each acid has a conjugate base and every base has a conjugate acid-base pair 1 HA +B A− + BH+ conjugate acid-base pair 2
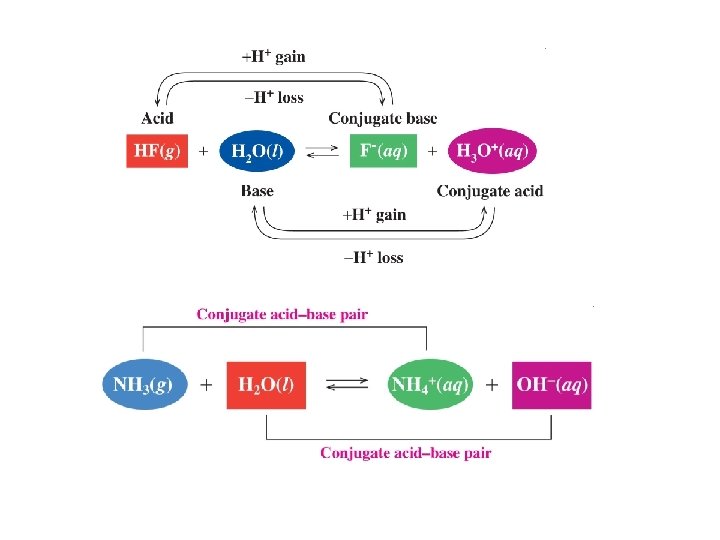

Problems Identify the conjugate bases for the following: 1) HBr 2) H 2 S 3) H 2 CO 3 Identify the conjugate acids for the following 1) NO 22) NH 3 3) OH-

In the following equations, identify the conjugate acid and base pairs: 1) HCl + H 2 O H 3 O+ + Cl- 2) C 5 H 5 N + H 2 O C 5 H 6 N+ + OH-

Acid Ionization Constant • Acid Ionization Constant (Ka): the equilibrium constant for the ionization reaction of an acid with water • HA + H 2 O A - + H 3 O + • Large Ka = Strong acid • Small Ka = Weak acid
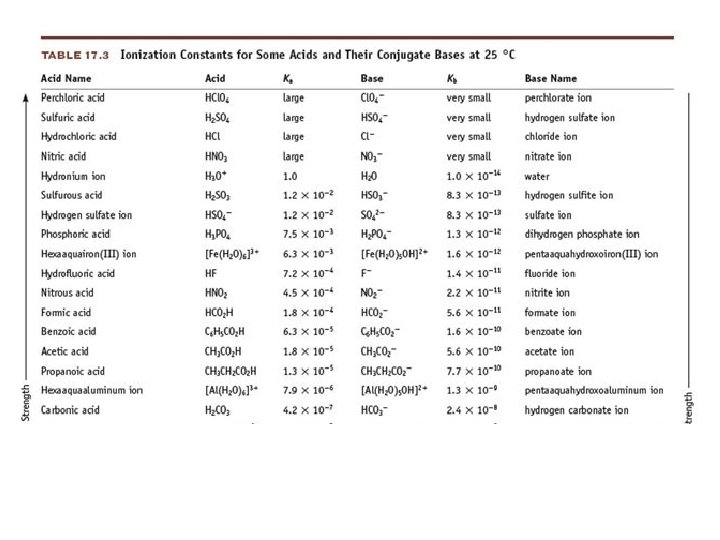

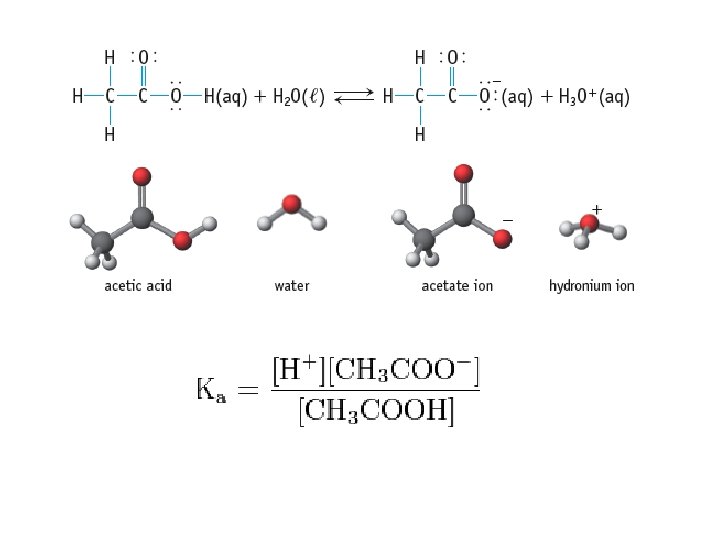

Questions 1) Write the equilibrium constant expression for the dissociation of HCN (hydrocyanic acid) in water 2) Write the equilibrium expression for the dissociation of HF in water 3) If the Ka for HCN is 4. 9 x 10 -10 and the Ka for HF is 7. 2 x 10 -4, which acid is stronger?

4) Predict whether the equilibrium for each of the following reactions favors the reactants or products. a) H 3 PO 4(aq) + H 2 O(l) H 3 O+(aq) + H 2 PO 4(aq) b) NH 4+(aq) + H 2 O(l) H 3 O+(aq) + NH 3(aq)
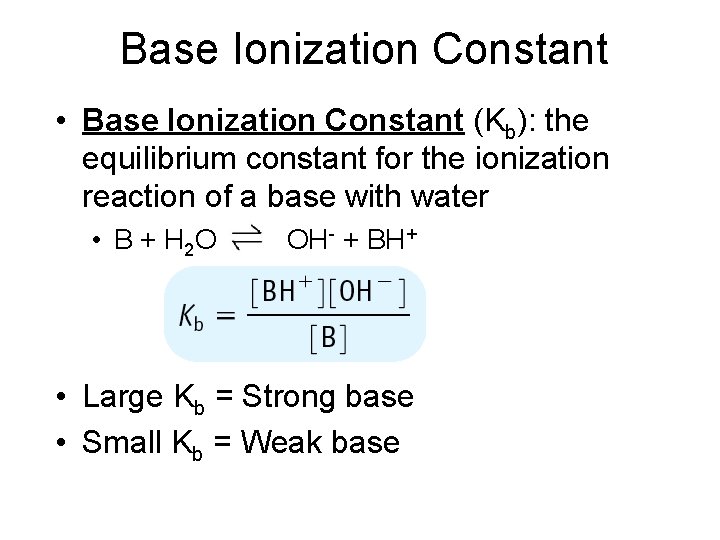
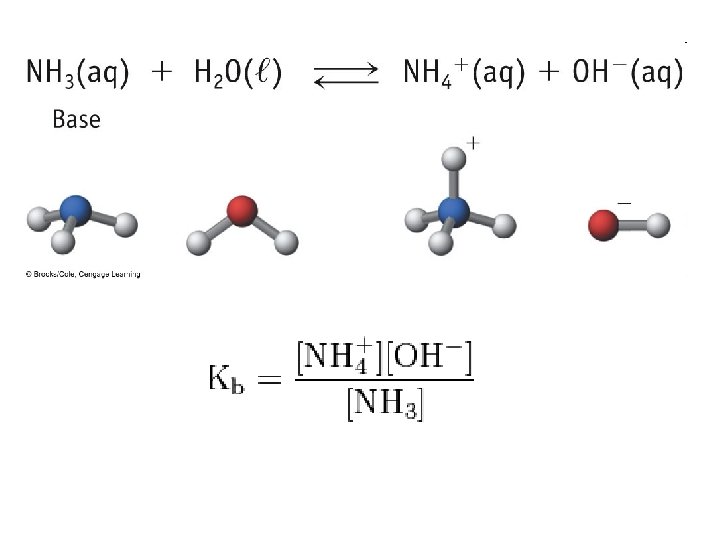
Base Ionization Constant • Base Ionization Constant (Kb): the equilibrium constant for the ionization reaction of a base with water • B + H 2 O OH- + BH+ • Large Kb = Strong base • Small Kb = Weak base
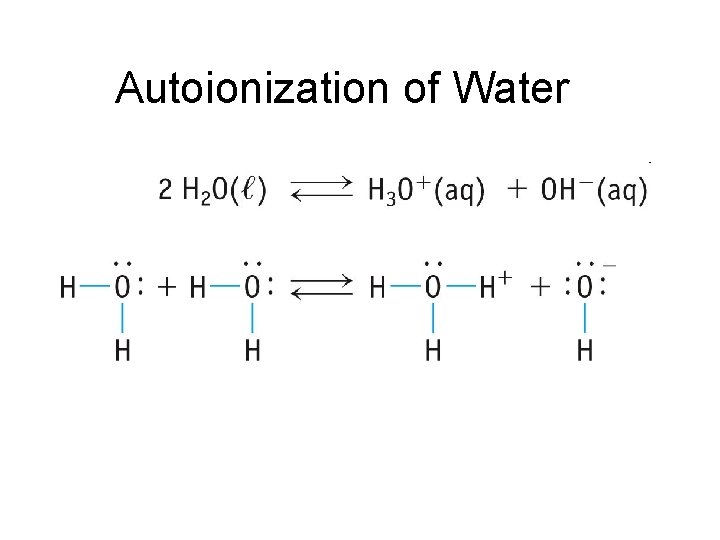
Autoionization of Water
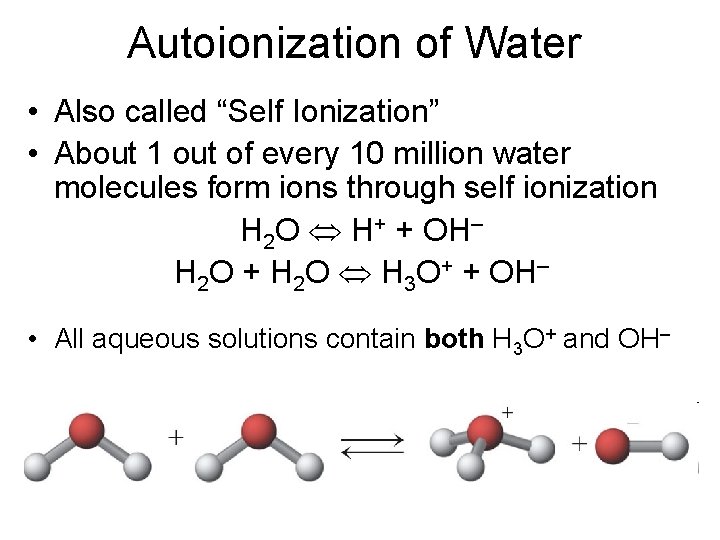
Autoionization of Water • Also called “Self Ionization” • About 1 out of every 10 million water molecules form ions through self ionization H 2 O H+ + OH– H 2 O + H 2 O H 3 O+ + OH– • All aqueous solutions contain both H 3 O+ and OH–

Ion Product Constant for Water • Ion Product Constant for Water (Kw): the numerical value obtained by multiplying the molar concentrations for hydronium and hydroxide ions present in pure water at 25°C • Kw = [H 3 O+][OH-] = 1. 00 x 10 -14 at 25 o. C • the concentration of H 3 O+ and OH– are equal in pure water • [H 3 O+] = [OH–] = 10 -7 M @ 25°C

Ion Product of Water • the product of the H 3 O+ and OH– concentrations is always the same number • Kw =[H 3 O+][OH–] = 1. 00 x 10 -14 @ 25°C – if you measure one of the concentrations, you can calculate the other • as [H 3 O+] increases the [OH–] must decrease so the product stays constant – inversely proportional
![[H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10 [H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10](http://slidetodoc.com/presentation_image_h2/70d28b1cdfa326e390e7f930e0abf8ca/image-45.jpg)
[H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10 -5 + H OH- [OH-]10 -14 10 -13 10 -11 10 -9 10 -7 10 -9 Base 10 -11 H+ + H OH OH 10 -7 10 -5 10 -3 10 -14 H+ OH 10 -1 100 Even though it may look like it, neither H+ nor OH- will ever be 0
![Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–] Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–]](http://slidetodoc.com/presentation_image_h2/70d28b1cdfa326e390e7f930e0abf8ca/image-46.jpg)
Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–] • [H 3 O+] = [OH–] = 1 x 10 -7 • acidic solutions have a larger [H 3 O+] than [OH–] • [H 3 O+] > 1 x 10 -7; [OH–] < 1 x 10 -7 • basic solutions have a larger [OH–] than [H 3 O+] • [H 3 O+] < [OH–] • [H 3 O+] < 1 x 10 -7; [OH–] > 1 x 10 -7
![Questions 1) Calculate the [OH ] at 25°C when the [H 3 O+] = Questions 1) Calculate the [OH ] at 25°C when the [H 3 O+] =](http://slidetodoc.com/presentation_image_h2/70d28b1cdfa326e390e7f930e0abf8ca/image-47.jpg)
Questions 1) Calculate the [OH ] at 25°C when the [H 3 O+] = 1. 5 x 10 -9 M, and determine if the solution is acidic, basic, or neutral 2) Calculate the [H 3 O+] at 25°C in a solution that has a [OH ] 1. 3 x 10 -10 M and determine if the solution is acidic, basic, or neutral 3) Calculate the [H 3 O+] at 25°C in a solution that has a [OH ] 1. 0 x 10 -7 M and determine if the solution is acidic, basic, or neutral
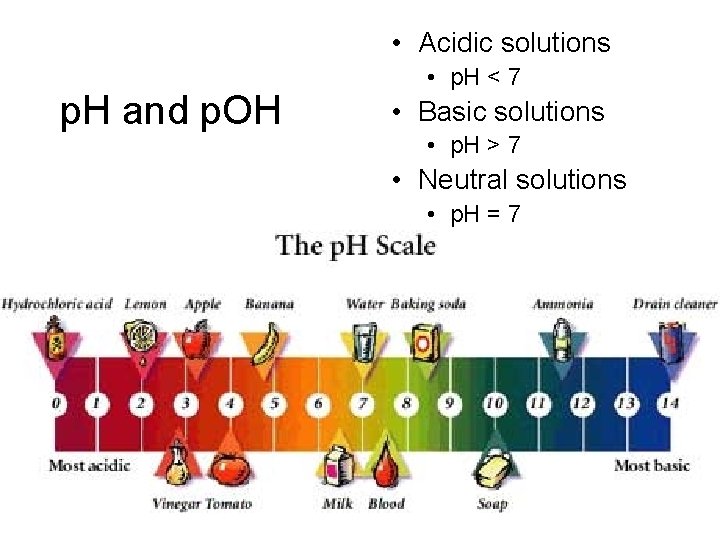
• Acidic solutions p. H and p. OH • p. H < 7 • Basic solutions • p. H > 7 • Neutral solutions • p. H = 7
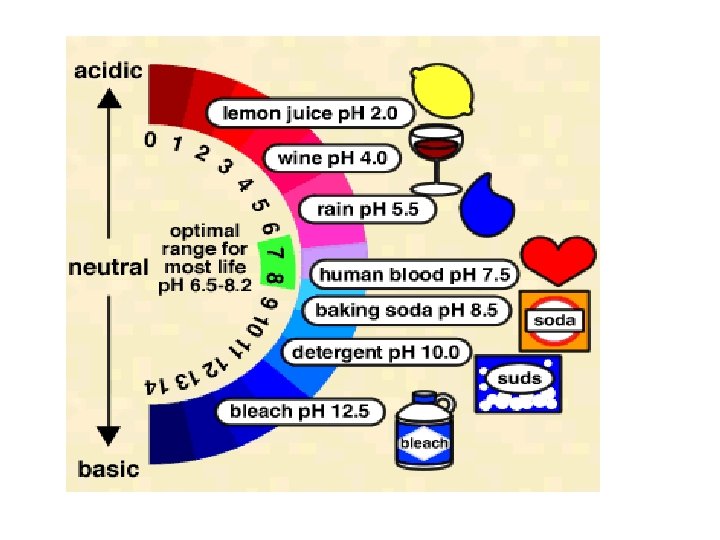

• p. H is a measure of the concentration of H+ or H 3 O+ in solution • p. H = -log [H+] OR p. H = -log [H 3 O+] • [H 3 O+] = 10 -p. H • p. OH is a measure of the concentration of OH- in solution • p. OH = -log [OH-] • [OH-] = 10 -p. OH • p. H + p. OH = 14

Problems 1) What is the p. H for an HCl solution with an [H 3 O+] = 2. 3 x 10 -2? 2) What is the p. H of an Na. OH solution with an [H 3 O+] = 3. 39 x 10 -10? 3) What is the p. H of a KOH solution with a [OH-] = 1. 1 x 10 -2? 4) What is the [H 3 O+] for a solution with a p. H of 8. 45. What is the [OH-]?

Classification of Water Soluble Substances • Electrolytes: solutes that separate into ions when dissolved in water (they’re soluble) – Have the ability to conduct electricity – 2 types • Strong electrolytes • Weak electrolytes
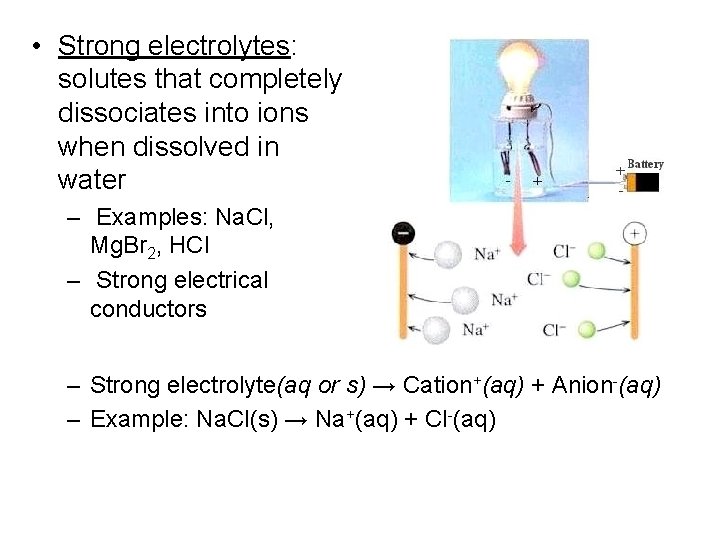
• Strong electrolytes: solutes that completely dissociates into ions when dissolved in water – Examples: Na. Cl, Mg. Br 2, HCl – Strong electrical conductors – Strong electrolyte(aq or s) → Cation+(aq) + Anion-(aq) – Example: Na. Cl(s) → Na+(aq) + Cl-(aq)
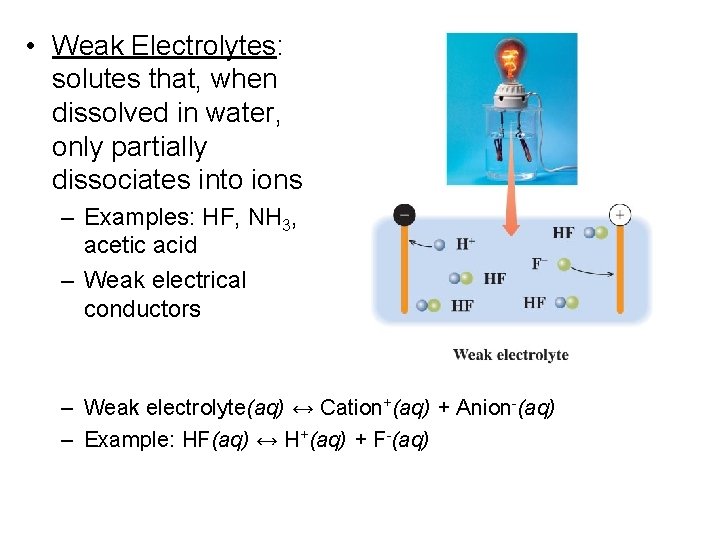
• Weak Electrolytes: solutes that, when dissolved in water, only partially dissociates into ions – Examples: HF, NH 3, acetic acid – Weak electrical conductors – Weak electrolyte(aq) ↔ Cation+(aq) + Anion-(aq) – Example: HF(aq) ↔ H+(aq) + F-(aq)
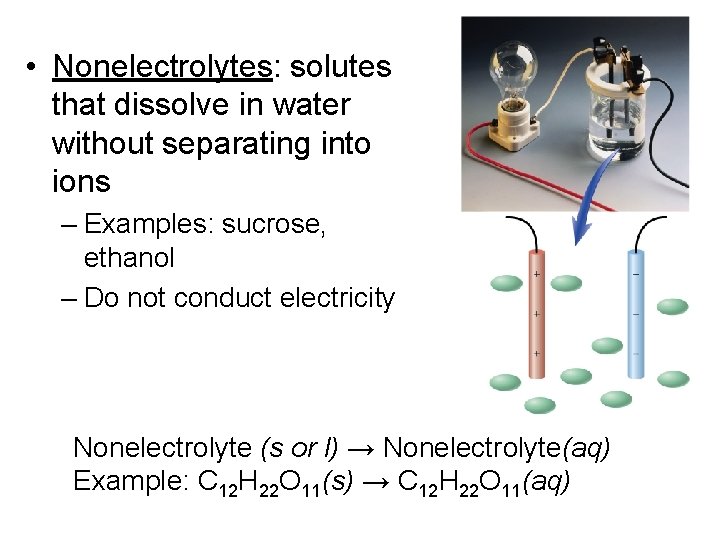
• Nonelectrolytes: solutes that dissolve in water without separating into ions – Examples: sucrose, ethanol – Do not conduct electricity Nonelectrolyte (s or l) → Nonelectrolyte(aq) Example: C 12 H 22 O 11(s) → C 12 H 22 O 11(aq)

Problems 1) The following salts are strong electrolytes. Write a balanced equation for their dissociation in water a) Li. Br b) Fe. Cl 3 2) HCN is a weak acid. Write a balanced equation for its dissociation in water 3) Do you expect Fructose (C 6 H 12 O 6) to be a strong electrolyte, weak electrolyte, or nonelectrolyte? Write a balanced equation for its solvation in water

Neutralization Reactions • When strong acids and bases in aqueous solution react with each other, they form water and a salt • HX(aq) + MOH(aq) HOH(l) + MX(aq) Water Salt • HCl(aq) + Na. OH(aq) H 2 O(l) + Na. Cl(aq)

Problems Write out the chemical equations for the following reactions 1) HBr and KOH 2) HCl and Mg(OH)2 3) H 2 SO 3 and Na. OH
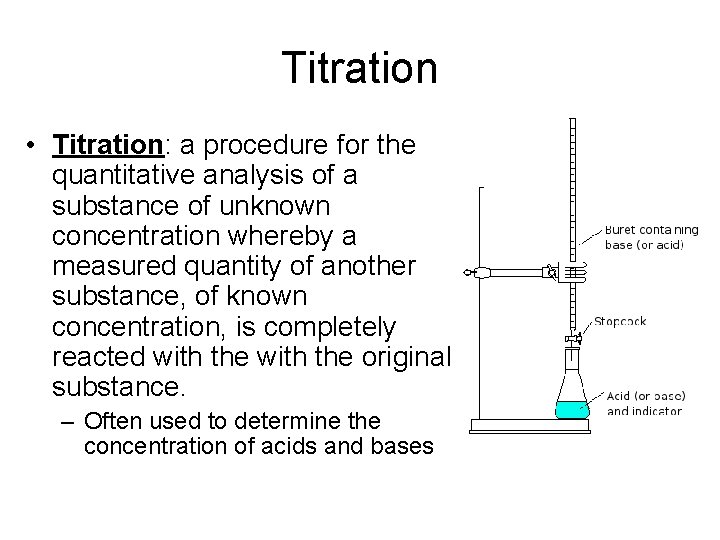
Titration • Titration: a procedure for the quantitative analysis of a substance of unknown concentration whereby a measured quantity of another substance, of known concentration, is completely reacted with the original substance. – Often used to determine the concentration of acids and bases
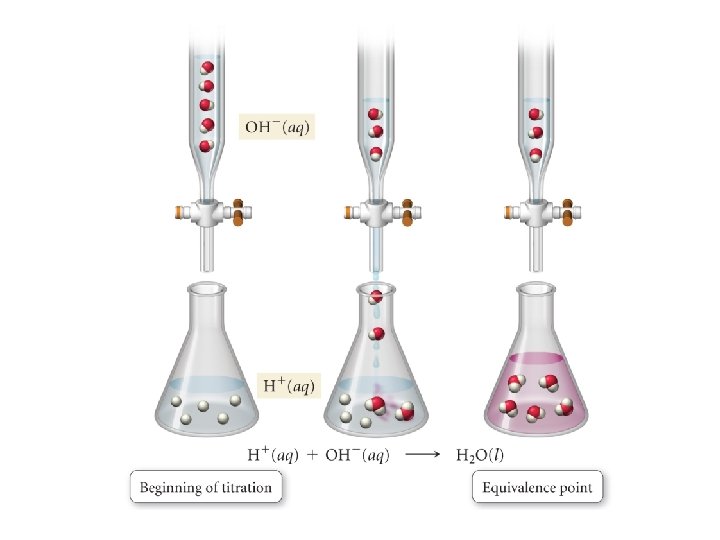
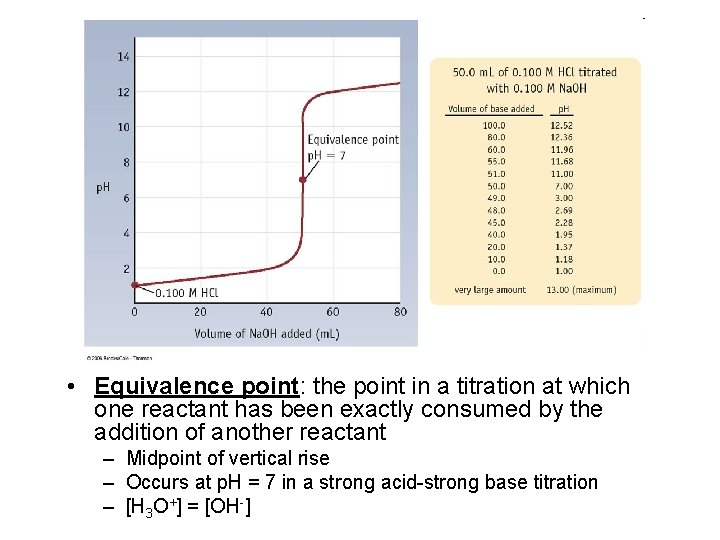
• Equivalence point: the point in a titration at which one reactant has been exactly consumed by the addition of another reactant – Midpoint of vertical rise – Occurs at p. H = 7 in a strong acid-strong base titration – [H 3 O+] = [OH-]

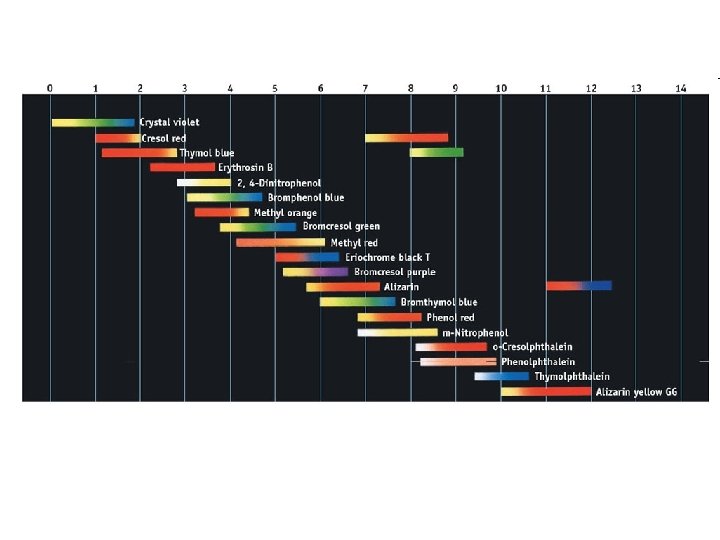
Indicators • Acid-Base Indicator: a chemical that changes color with a change in p. H – Added to solutions in small amounts in order to determine to solution’s p. H visually – Usually organic compounds – Weak acid or base – establishes an equilibrium with the H 2 O and H 3 O+ in the solution
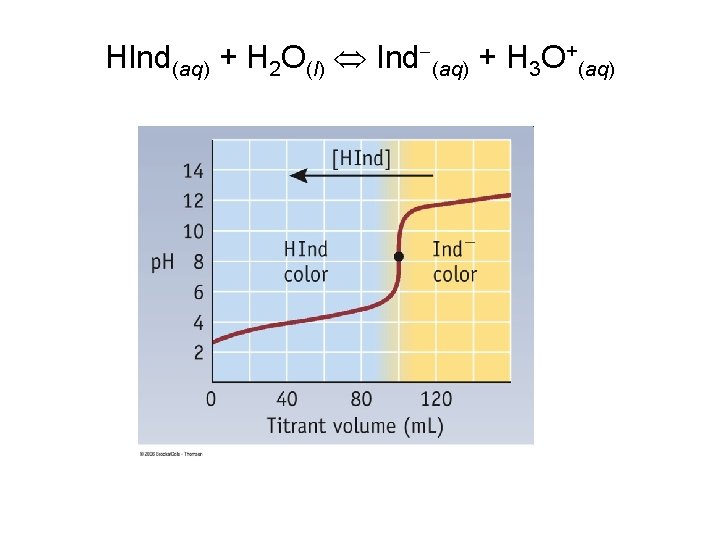
HInd(aq) + H 2 O(l) Ind (aq) + H 3 O+(aq)
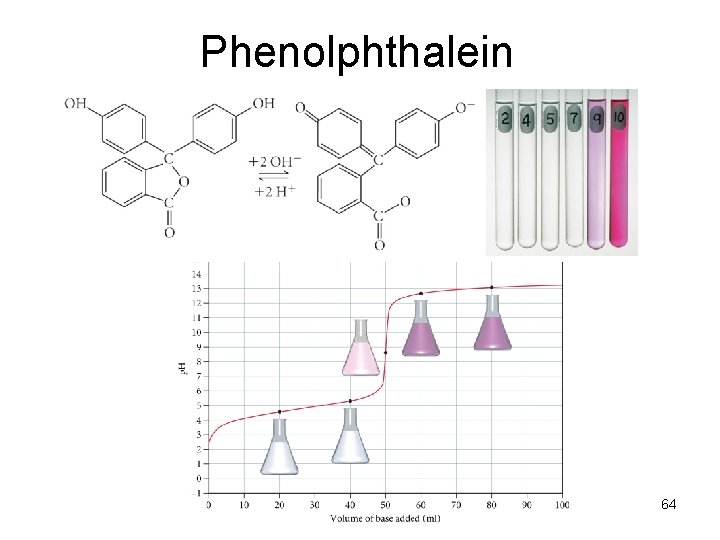
Phenolphthalein 64
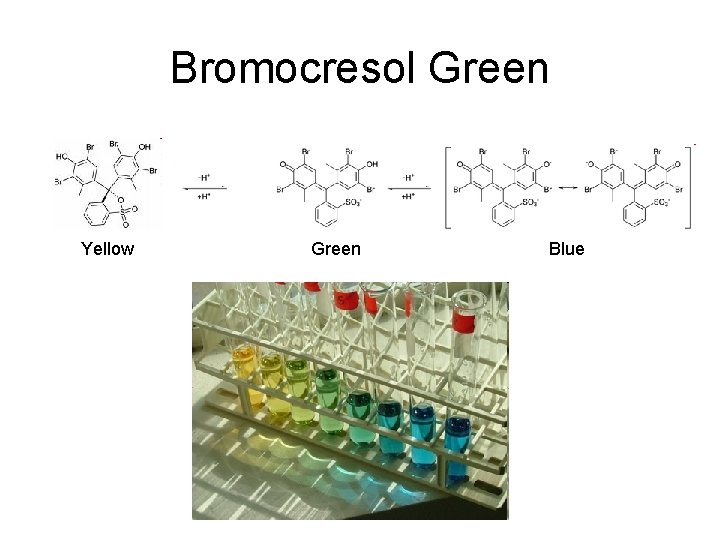
Bromocresol Green Yellow Green Blue
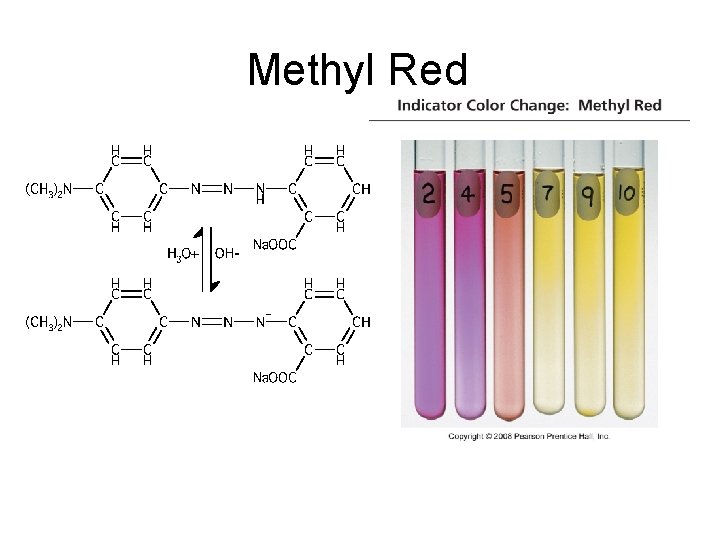
Methyl Red

3 steps to solving a titration problem (unknown acid molarity) • 1. How many moles of base were added to titrate the unknown acid? – Use concentration and volume of base added – M =mol/L • 2. What is the molar ratio that the base reacts with the acid, this will give you moles of acid? – Use the overall/net chemical equation, just like stoichiometry • 3. With the moles of acid, what is the initial concentration of the acid? – Use the initial volume of acid – M = mol/L

Questions 1) You have 25. 0 m. L of a HCl solution of unknown concentration. If you titrate your acid with 17. 3 m. L of a 0. 877 M Na. OH solution, what is the original concentration of your acid? Na. OH +HCl H 2 O + Na. Cl M = mol/L M x L = mol MNa. OH = 0. 877 MNa. OH x LNa. OH = mol. Na. OH LNa. OH = 0. 0173 LNa. OH mol. Na. OH x (1 mol. HCl / 1 mol. Na. OH) = mol. HCl LHCl = 0. 0250 LHCl mol. HCl / LHCl = MHCl 0. 877 MNa. OH x 0. 0173 LNa. OH = 0. 01517 mol. Na. OH x (1 mol. HCl / 1 mol. Na. OH) = 0. 01571 mol. HCl / 0. 0250 LHCl = 0. 607 MHCl

2) You also have 25. 0 m. L of a sulfuric acid solution. If you titrate this solution with 32. 1 m. L of a 1. 50 M KOH solution, what is the concentration of your acid?

Chapter 9 review • Acid, strong acid, weak acid, Base, strong base, weak base • Polyprotic acids • Acid-conjugate base, Base-conjugate acid • Neutralization reaction • Acid ionization constant, Base ionization constant • Auto ionization of water • Strong and weak electrolytes • p. H and p. OH • Titration, acid-base indicators, equivalence point,

• • • • • Definitions 8 and 9 Chapter 8 Solution, solvent, solute Solubility, Henry’s Law Unsaturated, and super saturated solutions Concentration, Molarity Osmosis, osmotic pressure, turgor, Hypotonic, hypertonic, and isotonic solutions Mixture, colloids, suspension Chapter 9 Acid, strong acid, weak acid, Base, strong base, weak base Polyprotic acids Acid-conjugate base, Base-conjugate acid Neutralization reaction Acid ionization constant, Base ionization constant Auto ionization of water Strong and weak electrolyte p. H and p. OH Titration, acid-base indicators, equivalence point,
- Slides: 72