Chapter 9 9 1 Carbon Compounds An compound
Chapter 9

9 -1 Carbon Compounds • An_____ compound contains ____ and_____, often combined with a few other elements such as oxygen and nitrogen. • There are millions of organic compounds- more than _______ percent of all known compounds.

Forms of Carbon • _____, ______ and_____are forms of carbon. • In each form, there is a different _____ of bonded_____ atoms.
Diamond • Cutting, grinding, and drilling tools are often coated with diamond because no substance is _______ than ____. • Diamond is an example of a ________. • In a network solid, all the atoms are linked by _____ bonds.

Graphite • It is extremely_______ and______. • It is a good lubricant for moving______ parts in machinery. • Pencil “_______” is a mixture of graphite and clay. • Carbon atoms are arranged______ spaced layers.

Fullerenes • ______ are large hollow spheres or cages of carbon.

Saturated Hydrocarbons • A _____ is an organic compound that contains only the elements____ and_______. • _____is a saturated hydrocarbon. • In a _____hydrocarbon, all the bonds are single bonds. • A saturated hydrocarbon contains the ______ possible number of hydrogen atoms for each carbon atom. • Another name for a hydrocarbon is an_____

Saturated Hydrocarbons • Factors that determine the properties of a hydrocarbon are the ______ of carbon atoms and how the atoms are _____. • The carbon atoms can be arranged in a _____ chain, _____chain, or a _______

Straight Chains • Look at figure 4 on page 264. • ______ formula shows the type and the number of atoms in a molecule of the compound. • A _______ formula show those atoms are arranged. • The number of carbon atoms in a straightchain _____ affects the state of alkane at room temperature.

Branched Chains • Compounds with the same molecular formula but different structural formulas are _____.

Rings • The carbon atoms in cyclobutane are linked in a four-carbon ____. • Most ring alkanes, or cyclic hydrocarbons, have rings with ______ or______. • Look at figure 5 on page 265.

9 -1 Continued Unsaturated Hydrocarbons • A hydrocarbon that contains one or more_____ or_____ bonds is an_____ hydrocarbon. • There are three types of unsaturated hydrocarbons-_____, and ______ hydrocarbons • See figure 6 on page 266

Alkenes/Alkynes/ Aromatic Hydrocarbons • _______ – hydrocarbons that have one or more carbon-carbon____ bond. Example – ethene • _______- straight- or branched-chain hydrocarbons that have one or more_____ bond • _____ Hydrocarbons contain similar _____ structures

Fossil Fuels • _______fuels are mixtures of hydrocarbons that formed from the remains of____ or animals. • Three types of fossil fuels are_______, and_____.

Coal • ____ is a solid fossil fuel that began to form about 300 million years ago in ancient swamp. • Burning coal produces more_______ than burning other fossil fuel does

Natural Gas • The second main fossil fuel formed from the remains of____ organisms. • Used for_____, _______, and to generate some _____.

Petroleum • The _______ main fossil fuel, also formed from the remains of marine organisms. • Often known as _____oil, is pumped from deep beneath Earth’s ____. • It must be separated into simpler mixtures, or fractions, such as _______ and heating ______.

Combustion of Fossil Fuels • The primary products of the complete combustion of fossil fuels are _____ and ____. • Some _____ oxides and _____dioxide are produced during the combustion of fossil fuels. • C 3 H 8 +5 O 2 3 CO 2 + 4 H 2 O

Incomplete Combustion • In stoves and furnaces, there may not be enough____ available for complete combustion of all the fuel. • A deadly gas, carbon____ is produced. • 2 C 3 H 8 +7 O 2 6 CO + 8 H 2 O

Acid Rain • The combustion of fossil fuels causes the _______ of rain to increase. • Sulfur dioxide and nitrogen oxides released into the atmosphere also dissolve in water, forming sulfuric acid, _______, and nitric acid, _____ • Acid rain damages _____ structures, ______ and ______.

Complete the following questions • Name three categories of unsaturated hydrocarbons • Name three main fossil fuels. • What are the two primary products of the complete combustion of fossil fuels? • What are three ways that carbon atoms can be arranged in hydrocarbon molecules?

9 -2 Substituted Hydrocarbons • A hydrocarbon in which one or more hydrogen atoms have been replaced by an atom or group of atoms is a _______ hydrocarbon. • The substituted atom or group of atoms is called a_____ group because it determines the ______ of the compound. • ____, organic______, organic_____, and esters are substituted hydrocarbons.

Alcohols • Methanol, ______ is used as a fuel in some motorcycles. • Ethanol, _____, is often mixed with gasoline to help the gasoline burn more completely. • Ethanol and methanol are_____ • The functional group in an alcohol is a hydroxyl group, ____ Drawing:

Organic Acids • The functional group in organic acids is a _____group, -_______ • Organic acids tend to have ______ and______. Drawing:
Organic Bases • _____ are organic bases. • The functional group in an amine is an ______ group -_____ • Aminos are found in _______, and_____.
Esters • _____ form when organic______ react with ____. • The second product of the reaction is _____. • Easter are used in many processed foods to produce_____ such as strawberry, banana, and grape. Drawing

Complete the following Questions 1. What functional groups are found in alcohols, organic acids, and organic bases? 2. Which types of compounds can react to produce esters? 3. What are two properties of organic acids?

9 -3 Polymers • A _____ is a large molecule that forms when many smaller molecules are linked together by covalent bonds. • The smaller molecules that join together to form a polymer are _____. • Poly- means “______” • Mono – means “______”

Polymers • Polymers can be classified as ____ polymers or _____ polymers. • Many important types of ______ molecules are natural polymers. • ______ polymers are developed by chemists in research laboratories and manufactured in factories

Synthetic Polymers • ____, _______, and_____ are three examples of compounds that can be______.

Rubber • The supply of ______rubber is limited. • Chemists produce synthetic rubber using hydrocarbons from_____ • ____will resist wear and be less likely to leak if they are made of synthetic rubber.

Nylon • _______fibers are very strong, durable, and shiny. • Nylon is used in ________, _________, and ____.

Polyethylene • Plastic milk bottles and plastic wrap are made of ______. • The number of carbon atoms in a polyethylene chain affects the______of the polymer. • The more carbon atoms in the chain, the _____ the polymer is.

Review Questions 1. Describe the two ways that polymers can be classified. 2. Name three synthetic polymers

9 -3 Continued Natural Polymers • 1. 2. 3. 4. Four types of polymers produced in plant and animal cells are : _______________

Starches • Typically, a _____ contains hundreds of glucose monomers. • Plants store starches for _______ and to build stems, _____ and roots. • Simple sugars, slightly more complex sugars such as _______, and polymers built from sugar monomers are all classified as ______.

Cellulose • The _______ cellulose is the main component of cotton and wood. • _______gives strength to plant stems and tree trunks

Nucleic Acids • _______ are large nitrogen -containing polymers found mainly in the nuclei of cells. • There are two types of nucleic acids, ____ and ____ • The monomers in a nucleic acid are _______.
Protein • A ______ is a polymer in which at least 100 _______ monomers are linked through bonds between an amino group and a carboxyl group. • An _______ is a compound that contains both ______and_____functional groups in the same molecule. • Proteins make up the fibers of your_____, your hair and _______, and the hemoglobin in your blood.

Review Question • What are four types of polymers that can be found in the cells of organisms. • What two functional groups are found in amino acids?
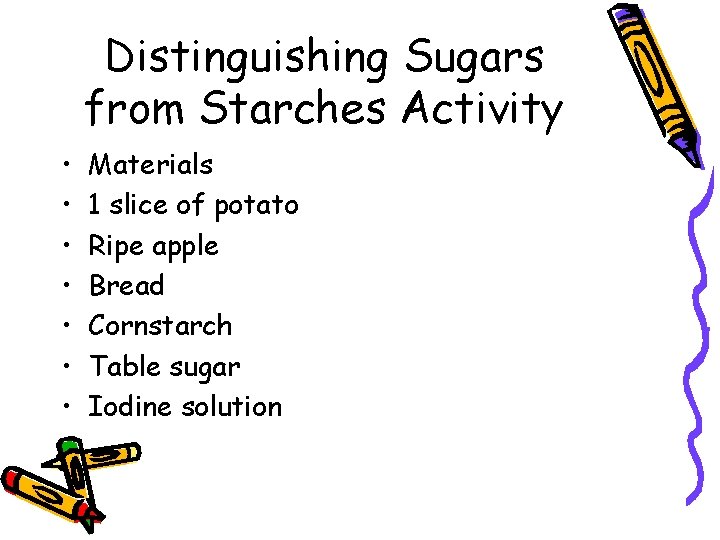
Distinguishing Sugars from Starches Activity • • Materials 1 slice of potato Ripe apple Bread Cornstarch Table sugar Iodine solution

Observations

Analyze and Conclude 1. How can an iodine solution be used to distinguish a sugar from a starch? 2. Which of the food samples contain starch? 3. What other foods would turn dark when tested with iodine solution?
- Slides: 43