CHAPTER 8 Writing Chemical Equations 2013 Marshall Cavendish



















- Slides: 24

CHAPTER 8 Writing Chemical Equations © 2013 Marshall Cavendish International (Singapore) Private Limited

Chapter 8 Writing Chemical Equations 8. 1 Chemical Equations 8. 2 Ionic Equations 2

8. 1 Chemical Equations Learning Outcomes At the end of this section, you should be able to: • interpret chemical equations with state symbols; • write balanced chemical equations with state symbols. 3

8. 1 Chemical Equations What is a Chemical Equation? A chemical equation shows what happens in a chemical reaction. It tells us • which reactants and products are involved in the reaction; • the relative amounts of reactants and products; • the physical states of the reactants and products of the reaction. Example: sodium + chlorine → sodium chloride (word equation) 2 Na + Cl 2 → 2 Na. Cl (chemical equation) 4

8. 1 Chemical Equations Balancing a Chemical Equation A balanced chemical equation must contain equal numbers of atoms of each element on both sides of the equation. The reactant(s) are written on the left-hand side of the equation. The product(s) are written on the right-hand side of the equation. 2 H 2(g) + O 2(g) → 2 H 2 O(l) The chemical equation is balanced by adding a number in front of the chemical formula. This is the same as multiplying the formula by that number. The state symbols (s) – solid (g) – gas (l) – liquid (aq) – aqueous solution 5

8. 1 Chemical Equations Writing a Balanced Chemical Equation Example: Reacting hydrogen and oxygen to get water Step 1: Write down the chemical formulae of the reactants and products to get the chemical equation. H 2 + O 2 H 2 O 6

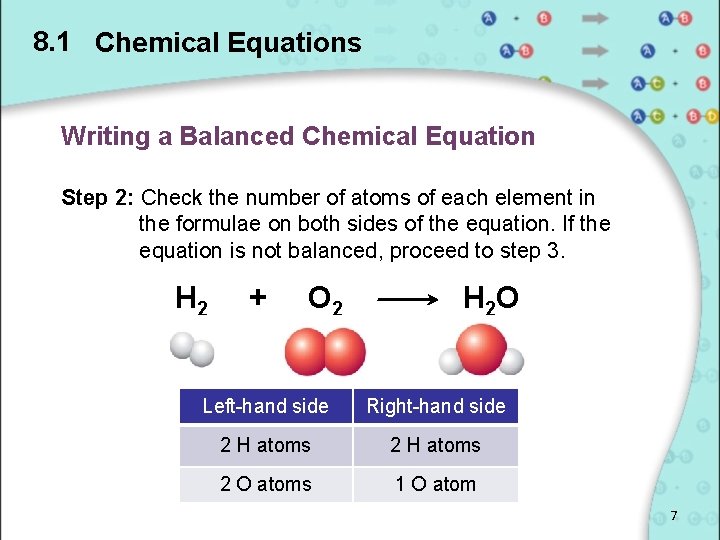
8. 1 Chemical Equations Writing a Balanced Chemical Equation Step 2: Check the number of atoms of each element in the formulae on both sides of the equation. If the equation is not balanced, proceed to step 3. H 2 + O 2 H 2 O Left-hand side Right-hand side 2 H atoms 2 O atoms 1 O atom 7

8. 1 Chemical Equations Writing a Balanced Chemical Equation Step 3 a: To balance the number of oxygen atoms, put a ‘ 2’ in front of H 2 O. This means that two molecules of water are formed. H 2 + O 2 2 H 2 O The equation is still not balanced because there are four hydrogen atoms on the right-hand side and only two on the left-hand side. 8

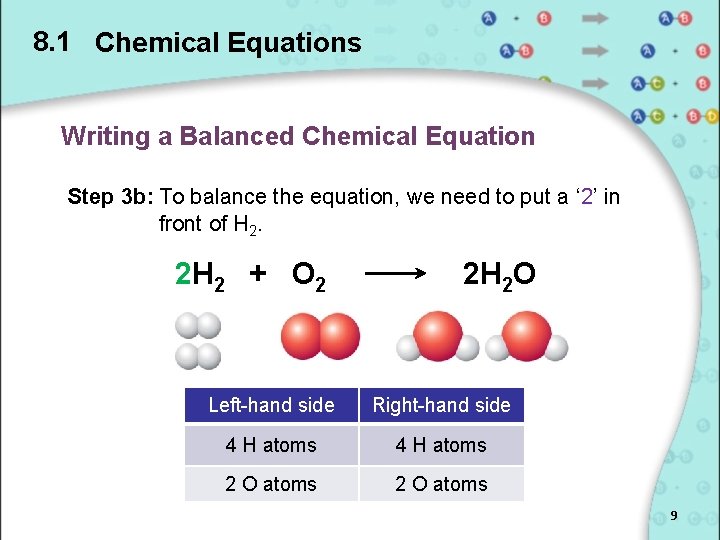
8. 1 Chemical Equations Writing a Balanced Chemical Equation Step 3 b: To balance the equation, we need to put a ‘ 2’ in front of H 2. 2 H 2 + O 2 2 H 2 O Left-hand side Right-hand side 4 H atoms 2 O atoms 9

8. 1 Chemical Equations Writing a Balanced Chemical Equation Step 4: Add the state symbols to indicate the physical state of each reactant and product. State Symbol Solid (s) Liquid (l) Gas (g) Aqueous (aq) 2 H 2(g) + O 2(g) 2 H 2 O(l) Aqueous means dissolved in water 10

Chapter 8 Writing Chemical Equations 8. 1 Chemical Equations 8. 2 Ionic Equations 11

8. 2 Ionic Equations Learning Outcome At the end of this section, you should be able to: • write ionic equations with state symbols. 12

8. 2 Ionic Equations What is an Ionic Equation? An ionic equation is a simplified chemical equation that shows the reactions involving ions in aqueous solution. Spectator Ions In chemical reactions, there are ions that do not take part in the chemical reaction. Such ions are called spectator ions. 13

8. 2 Ionic Equations Writing an Ionic Equation Let’s look at the reaction between hydrochloric acid and sodium hydroxide. hydrochloric acid + sodium hydroxide HCl(aq) + H+(aq) + Cl–(aq) Na. OH(aq) → sodium chloride + water Na. Cl(aq) + H 2 O(l) Na+(aq) + OH–(aq) Na+(aq) + Cl–(aq) Na+(aq) and Cl–(aq) are spectator ions. Removing them from the equation, we are left with: H+(aq) + OH–(aq) → H 2 O(l) 14

8. 2 Ionic Equations How to Write an Ionic Equation Example: Reaction of hydrochloric acid with sodium hydroxide Step 1: Write a balanced chemical equation including state symbols. HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O(l) 15

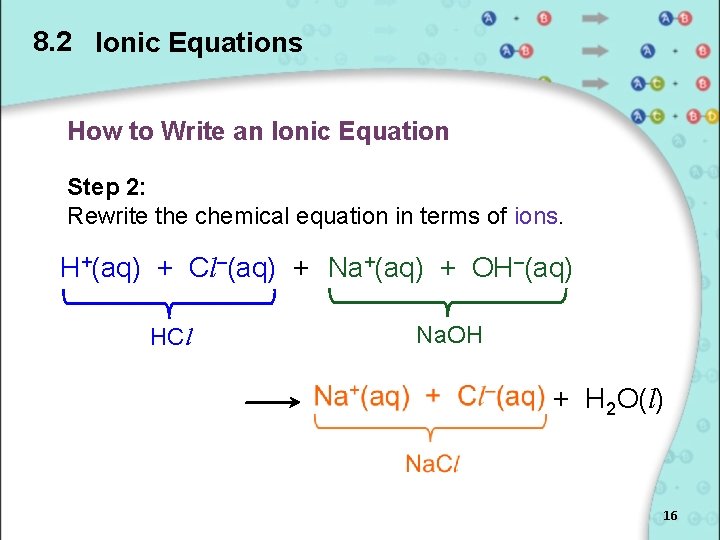
8. 2 Ionic Equations How to Write an Ionic Equation Step 2: Rewrite the chemical equation in terms of ions. H+(aq) + Cl–(aq) + Na+(aq) + OH–(aq) HCl Na. OH + H 2 O(l) 16

8. 2 Ionic Equations Step 3: Cancel out the spectator ions. H+(aq) + Cl–(aq) + Na+(aq) + OH–(aq) → Na+(aq) + Cl–(aq) + H 2 O(l) Step 4: Write the ionic equation. H+(aq) + OH–(aq) H 2 O(l) 17

8. 2 Ionic Equations Substances that do not ionise in solution: • Insoluble solids (e. g metals, insoluble salts) • Covalent liquids (such as water) • Gases Do not break them into ions when writing an ionic equation. These substances should be written in full, for example, H 2 O(l) and Ag. Cl (s). 18

8. 2 Ionic Equations Example: Reaction of sodium chloride with silver nitrate solution Step 1: Write a balanced chemical equation including state symbols. Na. Cl(aq) + Ag. NO 3(aq) → Ag. Cl(s) + Na. NO 3(aq) 19

8. 2 Ionic Equations Step 2: Rewrite the chemical equation in terms of ions. Na+(aq) + Cl–(aq) + Ag+(aq) + NO 3–(aq) → Ag. Cl(s) + Na+(aq) + NO 3–(aq) 20

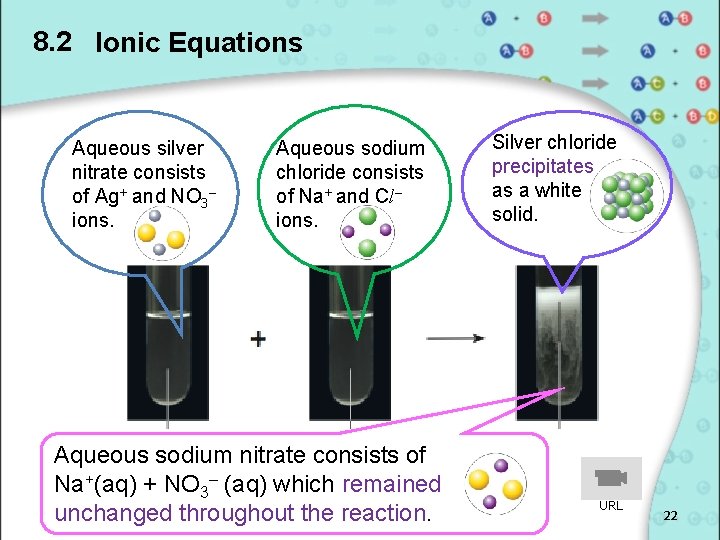
8. 2 Ionic Equations Step 3: Cancel out the spectator ions. Na+(aq) + Cl–(aq) + Ag+(aq) + NO 3–(aq) → Ag. Cl(s) + Na+(aq) + NO 3– (aq) Step 4: Write the ionic equation. Cl–(aq) + Ag+(aq) → Ag. Cl(s) Can you give another example of ionic precipitation? 21

8. 2 Ionic Equations Aqueous silver nitrate consists of Ag+ and NO 3– ions. Aqueous sodium chloride consists of Na+ and Cl– ions. Aqueous sodium nitrate consists of Na+(aq) + NO 3– (aq) which remained unchanged throughout the reaction. Silver chloride precipitates as a white solid. URL 22

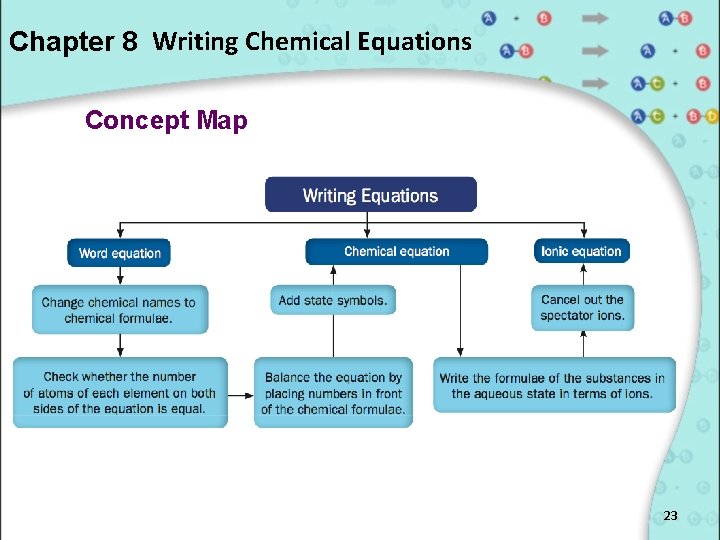
Chapter 8 Writing Chemical Equations Concept Map 23

Chapter 8 Writing Chemical Equations The URLs are valid as at 15 October 2012. Acknowledgements (slide 1) chemical reactions © Aushulz | Wikimedia Commons | CC BY 3. 0 (http: //creativecommons. org/licenses/by/3. 0/deed. en) (slide 22) © Marshall Cavendish International (Singapore) 24