Chapter 8 Understanding Drug Labels Copyright 2016 Cengage

Chapter 8 Understanding Drug Labels Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Understanding Drug Labels • Recognize pertinent information on drug labels, including: § § § Drug form Dosage strength Supply dosage or concentration Total volume of drug container Administration route Expiration date Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Understanding Drug Labels • You must also: § Differentiate between brand generic names of drugs. § Find the directions for mixing or preparing the supply dosage of drugs, as needed. § Recognize and follow drug alerts. § Locate the lot or control number, National Drug Code, barcode symbols, and controlled substance classifications. § Determine if containers are for single-dose or multidose use. § Identify combination drugs. § Describe supply dosage expressed as a ratio or percent. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Drug Names • Brand, trade, and proprietary – Manufacturer’s name for drug • Generic – Established, nonproprietary name Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Brand Generic Names • Brand name usually most prominent • Generic name must be on all drug labels (by law) • Nurses must cross-check medications to prevent errors especially when only the generic name is on label Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
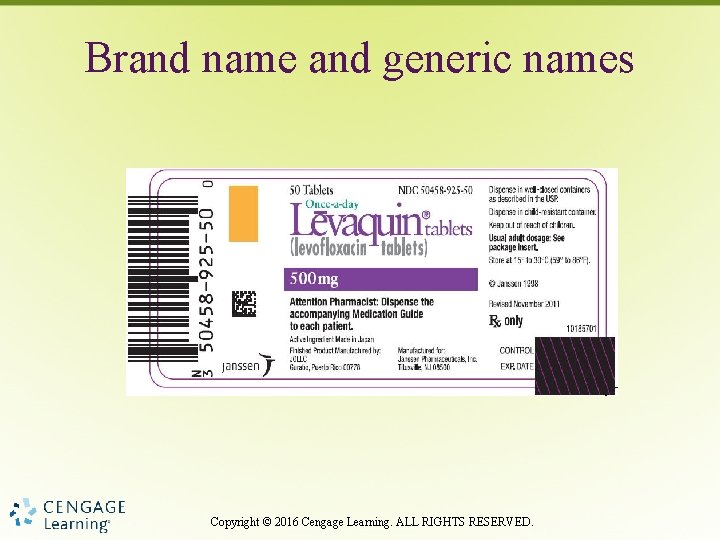
Brand name and generic names Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Brand Generic Names Used with permission from Aventis Pharmaceuticals. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Generic Name Used with permission from Abbott Laboratories. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Brand Generic Name Copyright Pfizer Inc. Reproduced with permission. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

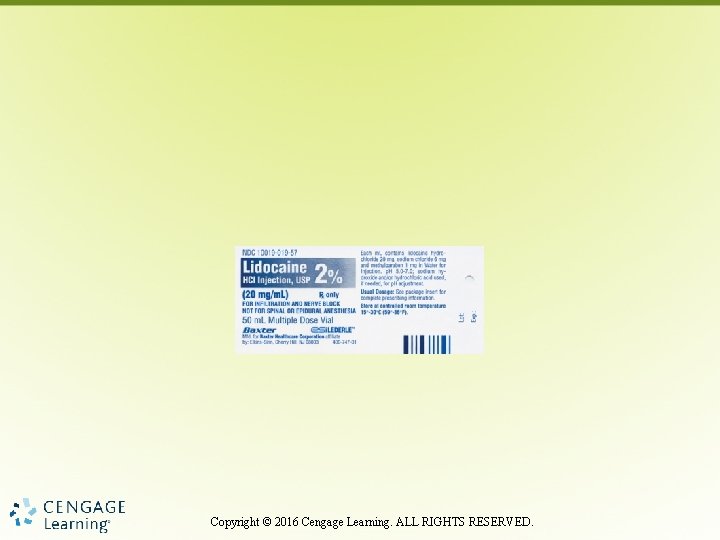
Dosage Strength • Dosage weight or amount of drug provided in specific unit of measurement Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Dosage Strength Image courtesy of Baxter Healthcare Corporation. All rights reserved. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Form • Structure and composition of drug – – – Solid forms for oral use Liquid, powder, suspension forms for oral use Injectable Suppositories Creams Patches Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Form Copyright Pfizer Inc. Reproduced with permission. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Supply Dosage • Both dosage strength and form – Dosage per tablet – Dosage per milliliter (liquid medication) Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
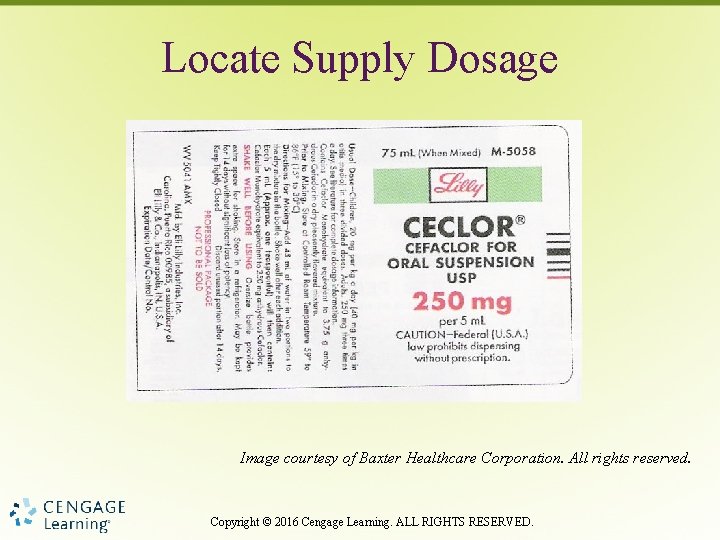
Locate Supply Dosage Image courtesy of Baxter Healthcare Corporation. All rights reserved. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
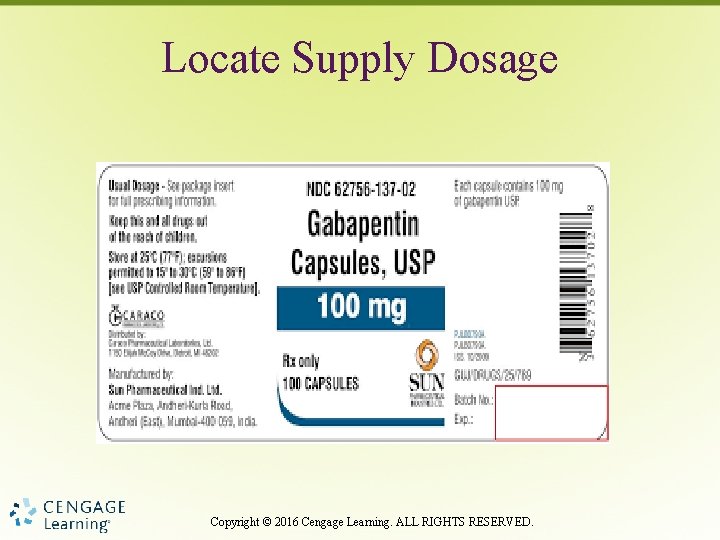
Locate Supply Dosage Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
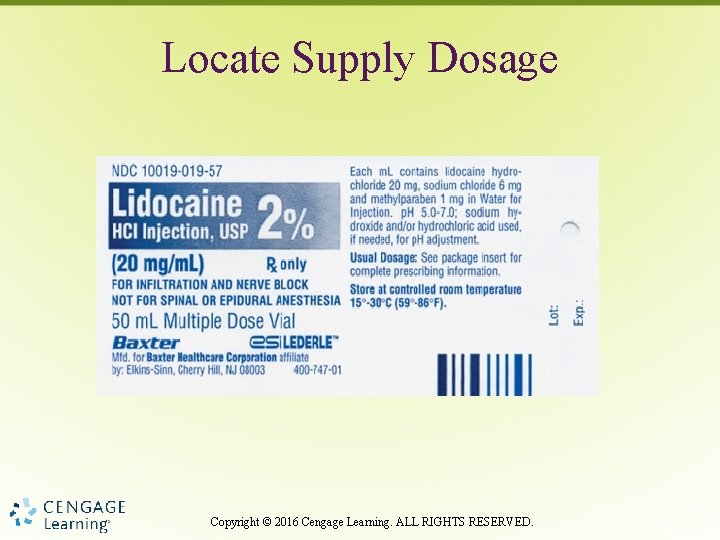
Locate Supply Dosage Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Supply Dose • Safety concerns with high-risk medications have led to a new way to express the supply dosage for single- and multiple-dose injectable drug products. • To prevent too large a dose, dosage is displayed based on the total amount in the container with the amount per m. L printed directly below in smaller print. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Supply Dose • Otherwise, the label provides the total volume per container, but not the supply dosage. • Until manufacturers comply by revising all medication labels and the supply of older labels is exhausted, extreme caution must be taken when locating the dosage strength. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Total Volume • Full quantity contained in package, bottle or vial § tablets and solids = total number of items contained in package, bottle, or vial § Liquids = total fluid volume Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
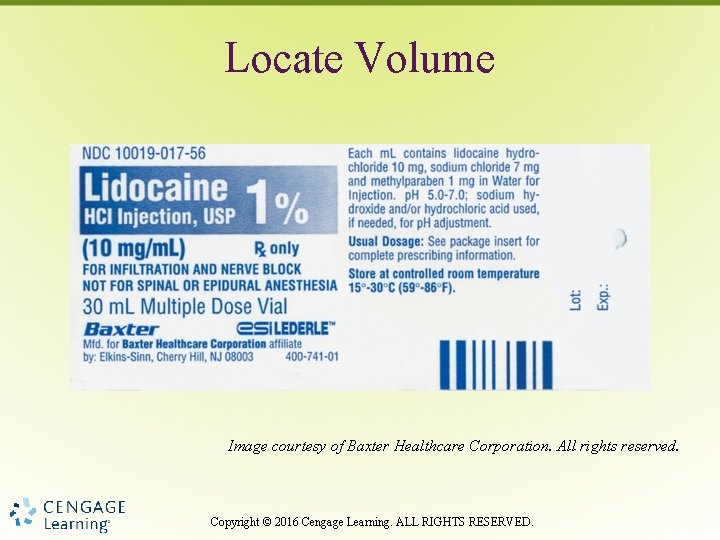
Locate Volume Image courtesy of Baxter Healthcare Corporation. All rights reserved. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

2009 FDA requirement: • Strength per total volume be prominent on the label of both single and multi-vial doses of injectable product labels • Followed in close proximity by the strength per m. L Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Administration Route • Site of body or method of drug delivery – Oral • Unless specified, tablets, capsules, and caplets intended for oral use – Enteral – Sublingual – Injection Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Administration Route • Site of body or method of drug delivery – – Otic Optic Topical Rectal Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Administration Route Image courtesy of Baxter Healthcare Corporation. All rights reserved. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Label Alerts • Warnings or special alerts • Manufacturers may print warnings • Pharmacists may issue special alerts – E. g. , refrigerate at all times, keep in a dry place, replace cap tightly, protect from light, shake well before dispensing… Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Expiration Date • Check prior to administration • If expired, discard or return to pharmacy Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Expiration Date Used with permission from Abbott Laboratories. Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Lot or Control Numbers • Required by Federal law • Important if drug is recalled for damage or tampering • Quickly identifies particular group of medication packages Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

National Drug Code (NDC) • Federal law requires every prescription med to have a unique ID number • Printed on every manufacturer’s label • Printed with the letters “NDC” followed by three groups of numbers Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Controlled Substance Schedule • Classifies drugs according to potential for use and abuse – Schedule I • Highest potential for abuse – Schedule V • Lowest potential for abuse Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Official Lists of Approved Drugs • • • 2 official national lists of approved drugs USP (UNITED STATES PHARMACOPEIA) NF (NATIONAL FORMULARY) Placed after generic name of the drug Do not mistake for other drug specific labels Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Unit or Single-Dose Labels • Dose strength understood as “per one” Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Combination Drugs • Different substances combined • Two or more drugs in one form • Usually prescribed by number of tablets, capsules, or milliliters – Not dosage strength Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Other Parts of Label • Manufacturer • National Drug Code • Bar code Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Locate Bar Code Used with permission from Abbott Laboratories Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Supply Dosage Expressed as a Ratio or Percent RULE: Ratio solutions express the number of grams of the drug per total milliliters of solution. RULE: Percentage (%) solutions express the number of grams of the drug per 100 milliliters of solution Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Checking Labels • Six Rights of medication administration: 1. 2. 3. 4. 5. 6. Right patient Right drug Right amount Right route Right time Right documentation Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Checking Labels • Check drug label three times before administering a medication to a patient: – Against medication order and the MAR – Before preparing medication – After preparing medication but before administering it Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.

Copyright © 2016 Cengage Learning. ALL RIGHTS RESERVED.
- Slides: 41