Chapter 8 The Colliodal Fraction Chapter 9 Soil














- Slides: 33

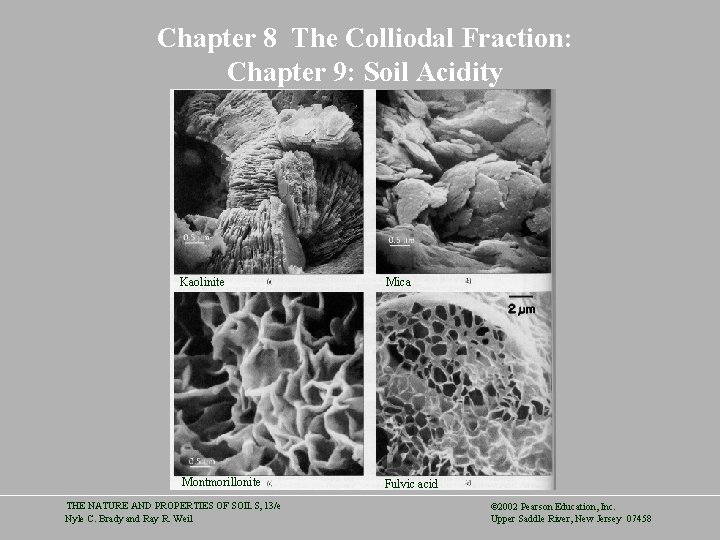
Chapter 8 The Colliodal Fraction: Chapter 9: Soil Acidity Kaolinite Mica Montmorillonite Fulvic acid THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

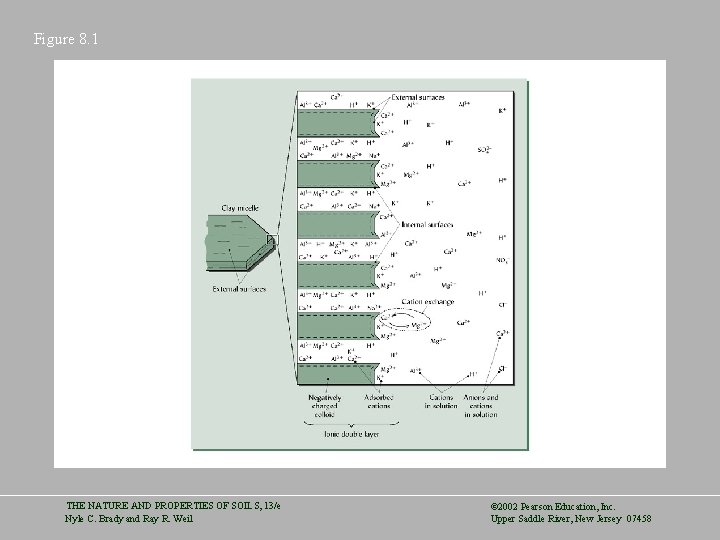
Figure 8. 1 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

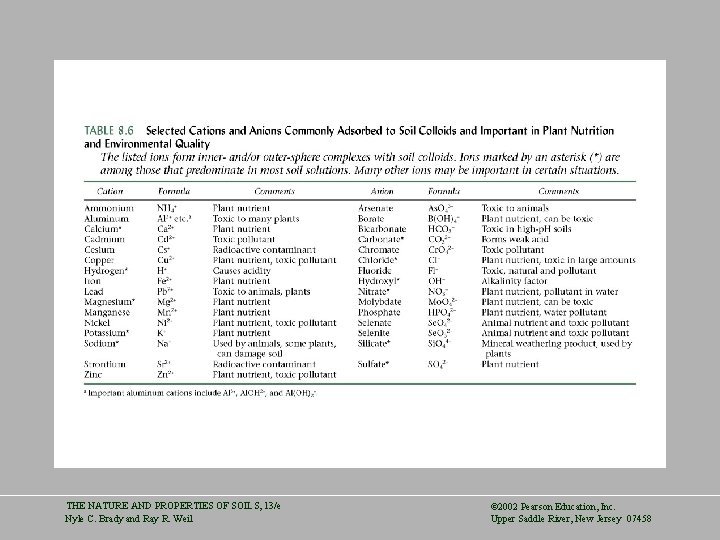
THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

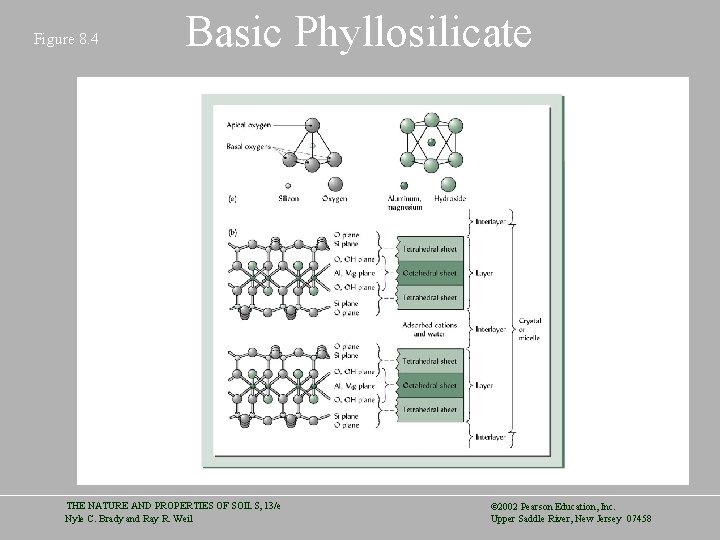
Figure 8. 4 Basic Phyllosilicate THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Important Clays Minerals • Kaolinite – Most common in soils • Smectite – Shrink-Well Clays – Montmorillonite • Vermiculite • Micas – Fine-grained micas – Illite • Chlorite • Humus – Humic acid and organic matter THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Figure 8. 27 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

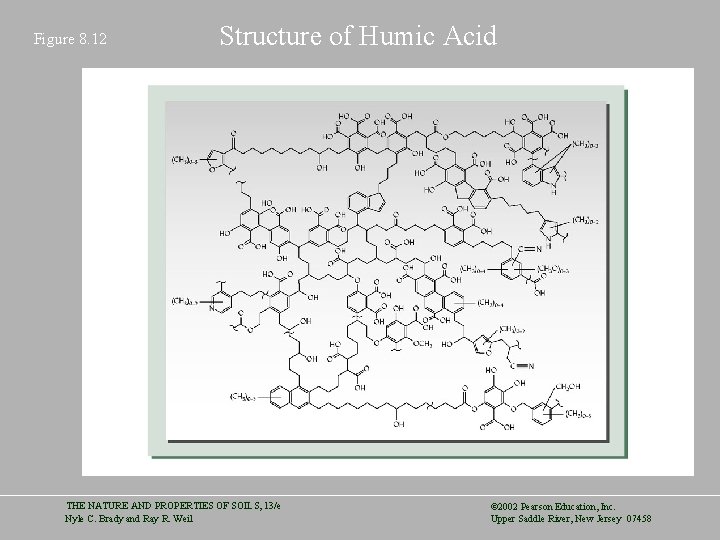
Figure 8. 12 Structure of Humic Acid THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

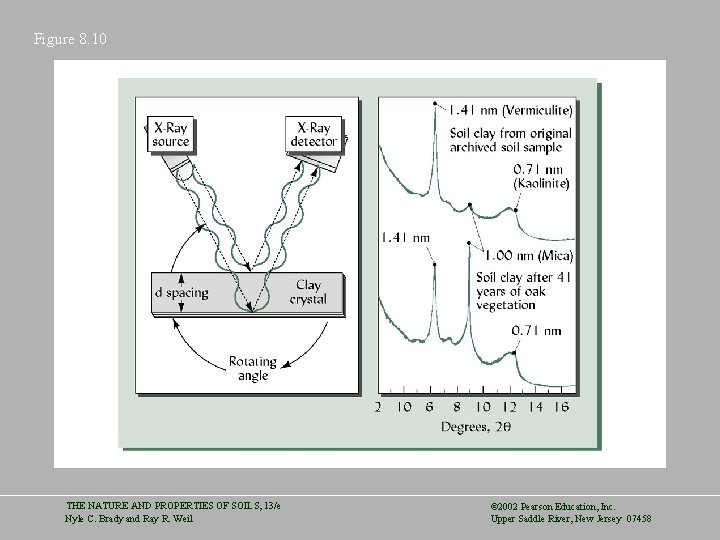
Figure 8. 10 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

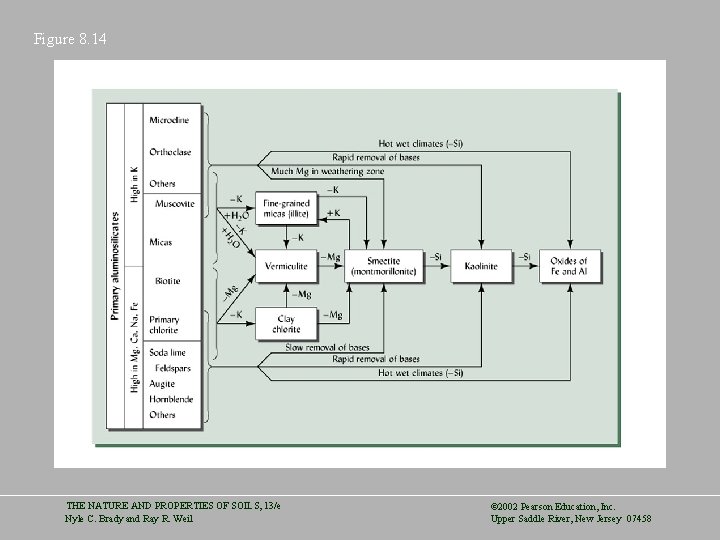
Genesis of Soil Colloids • Alteration – An example would be the change from muscovite mica to fine-grained mica through physical breakdown, loss of some elements and addition of others. • Recrystallization – Results from the complete breakdown of the original crystal structure and regrowth of clay minerals. • Relative Stages of Weathering – Fine-grained micas are an early stage while kaolinite and iron oxides are late stages THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Figure 8. 14 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

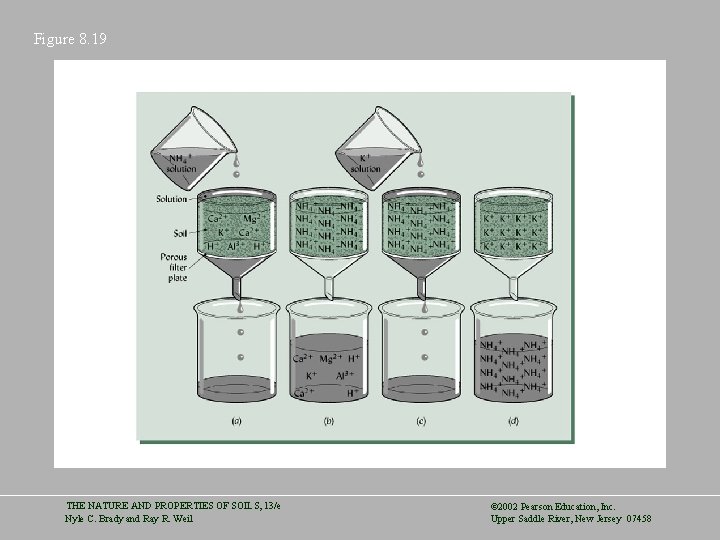
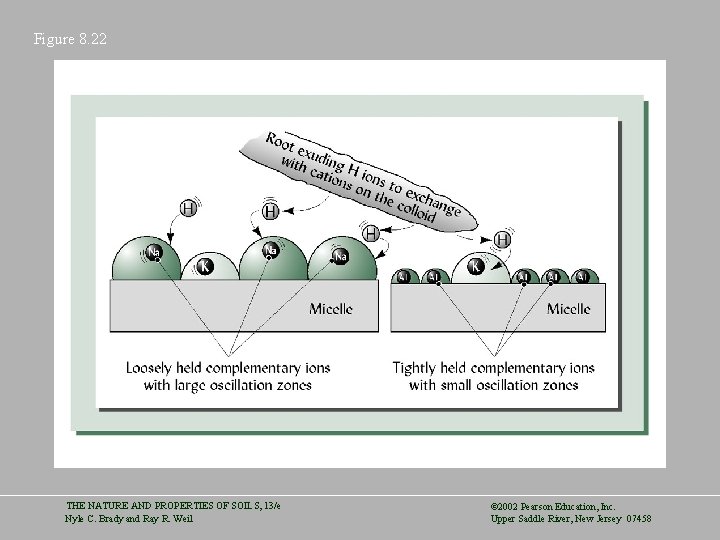
Principles Governing Cation Exchange Reactions • Reversibility – A cation is usually replaced by a hydrogen ion and this reaction is reversible Micelle*Na+ + H+ = Micelle*H+ + Na+ • Charge Equivalence – Exchanges take place on a charge-by-charge basis Micelle*Ca 2+ + 2 H+ = Micelle*2 H+ + Ca 2+ • Ratio Law – The amount of exchangeable cation in the soil solution will affect the ion’s equilibrium THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Principles Governing Cation Exchange Reactions • Mass Action – If a product precipitates, volatilizes, or strongly associates with an anion, the reaction is not reversible. • Cation Selectivity – Higher charge and hydrated radius affect the exchangeability of cations Al 3+ > Ca 2+ > Mg 2+ > K+ = NH 4+ > Na+ THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Figure 8. 19 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Figure 8. 22 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

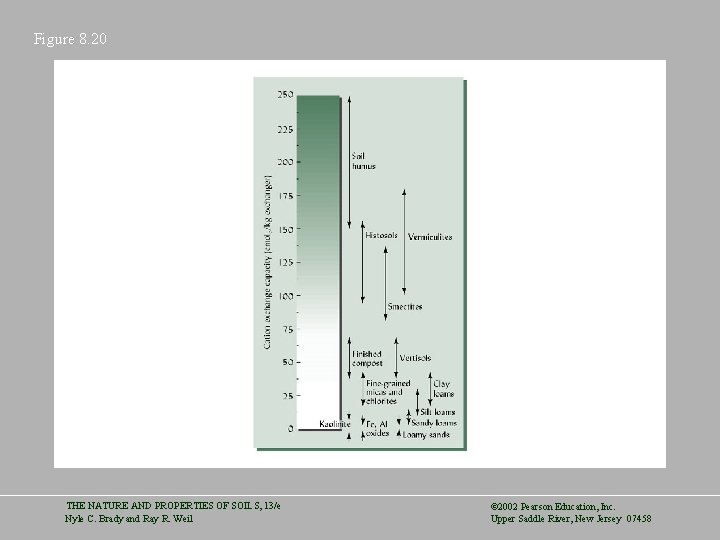
Figure 8. 20 THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

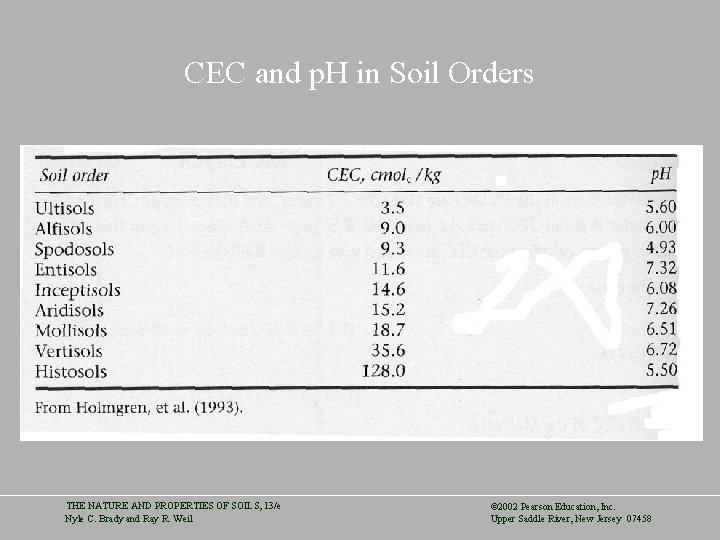
CEC and p. H in Soil Orders THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

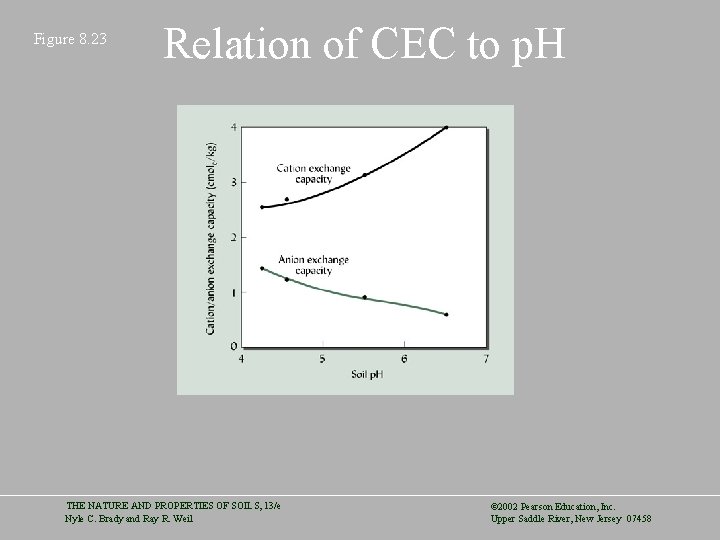
Figure 8. 23 Relation of CEC to p. H THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

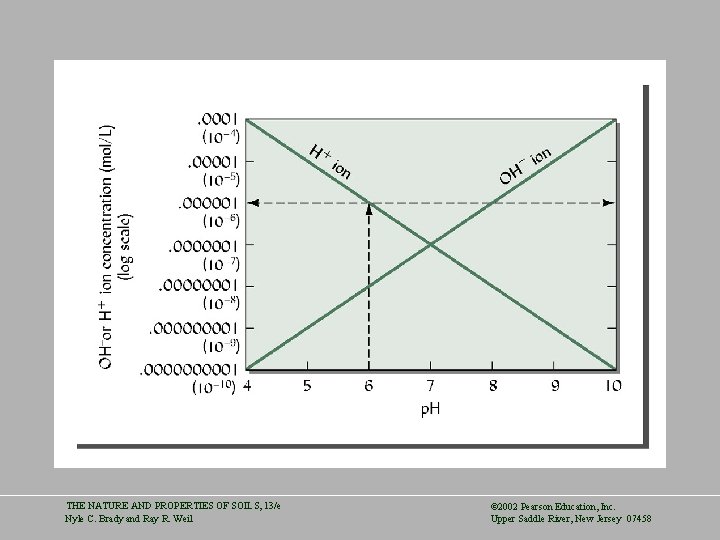
THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

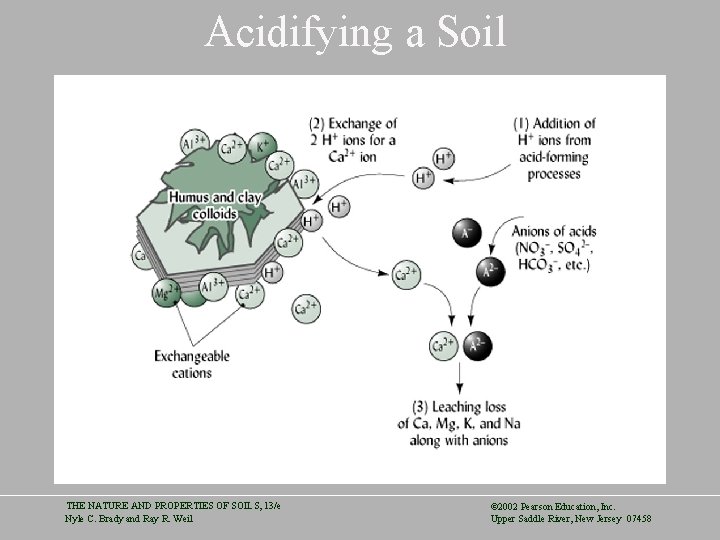
Acidifying a Soil THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

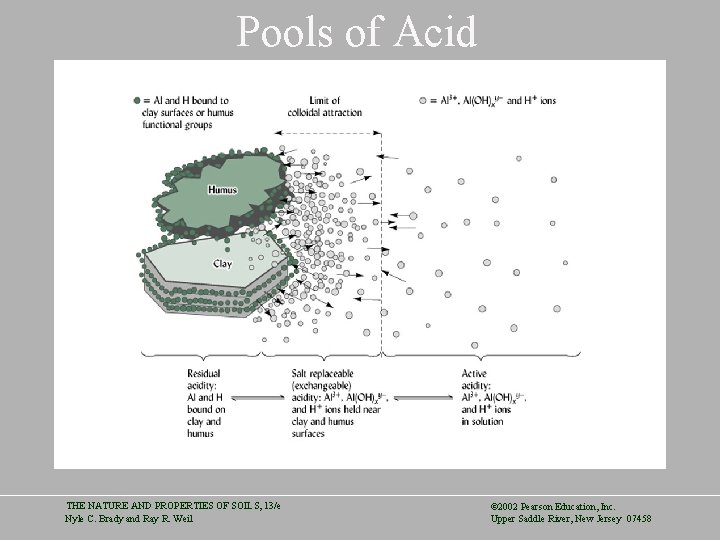
Pools of Soil Acidity • Active Acidity – H+ ion activity in the soil solution. • Exchangeable (Salt Replaceable) Acidity – Associated with exchangeable aluminum and hydrogen ions in the parent material. – The take the place of beneficial cations. • Residual Acidity – Hydrogen and Aluminum ions bound to organic matter and clays. May free-up Cation exchange locations with liming – May be 100, 000 times greater acid input than the other pools THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Pools of Acid THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

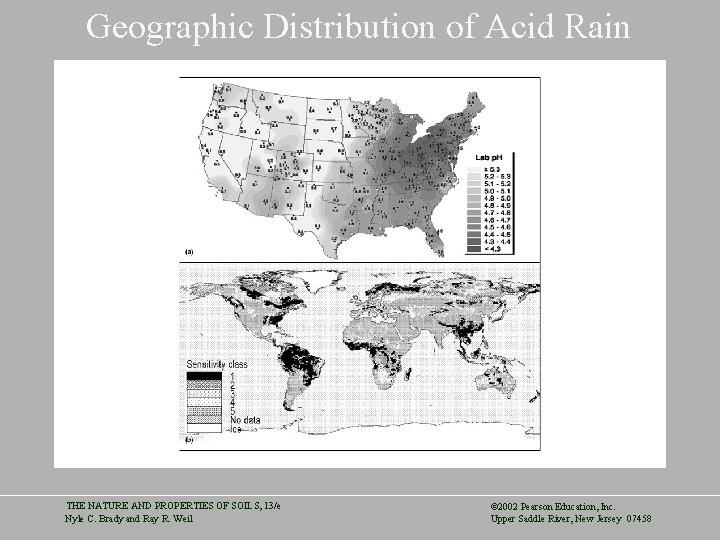
Acid Rain THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Geographic Distribution of Acid Rain THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Effect of Soil p. H on Root Growth THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

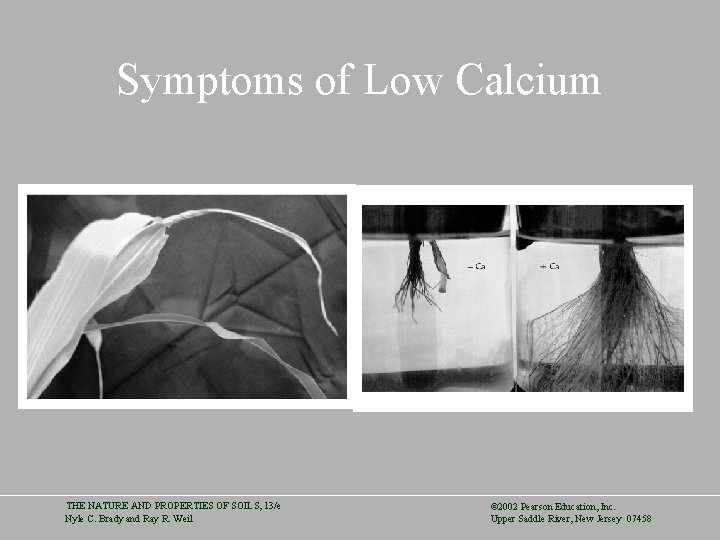
Plant Macronutrients • Calcium and Magnesium are essential to plants and are tied to soil acidity • Calcium – Calcium is second only to N and K as a plant macronutrient – Used in cell walls, cell elongation, membrane permeability, and enzymes – Taken up exclusively by young root tips and redistributed in the plant with transpiration water – Symptoms: Young leaves not unfolding, lack of root development, blossom-end rot, empty peanut shells THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Symptoms of Low Calcium THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

Plant Macronutrients • Magnesium – – Taken up in less quantity than Calcium Used in the Chlorophyll molecule Used in the synthesis of oils, proteins, and enzymes Symptoms: Mottled green and yellow coloring in the older leaves. • Both Calcium and Magnesium deficiencies can be remedied by applying lime (Ca. O) or Gypsum (Ca. So 4*2 H 2 O) THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

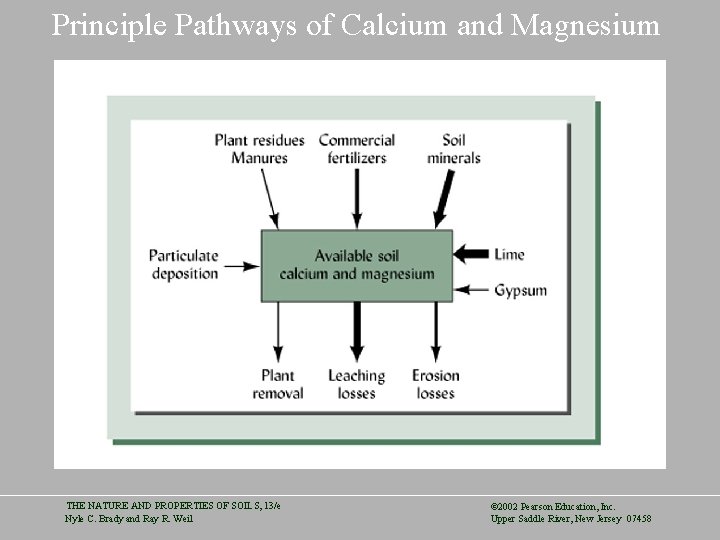
Principle Pathways of Calcium and Magnesium THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

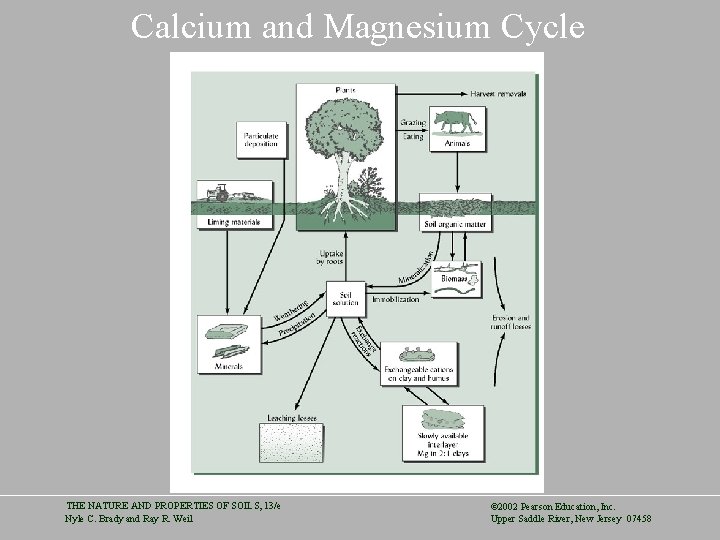
Calcium and Magnesium Cycle THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458

THE NATURE AND PROPERTIES OF SOILS, 13/e Nyle C. Brady and Ray R. Weil © 2002 Pearson Education, Inc. Upper Saddle River, New Jersey 07458