Chapter 8 Quantities in Chemical Reactions Products are


























- Slides: 42

Chapter 8 Quantities in Chemical Reactions Products are carbon dioxide and water Octane in gas tank Octane mixes with oxygen 2006, Prentice Hall

CHAPTER OUTLINE § § § § Stoichiometry Molar Ratios Mole-Mole Calculations Mass-Mass Calculations Limiting Reactant Percent Yield 2

Global Warming: Too Much Carbon Dioxide • The combustion of fossil fuels such as octane (shown here) produces water and carbon dioxide as products. • Carbon dioxide is a greenhouse gas that is believed to be responsible for global warming.

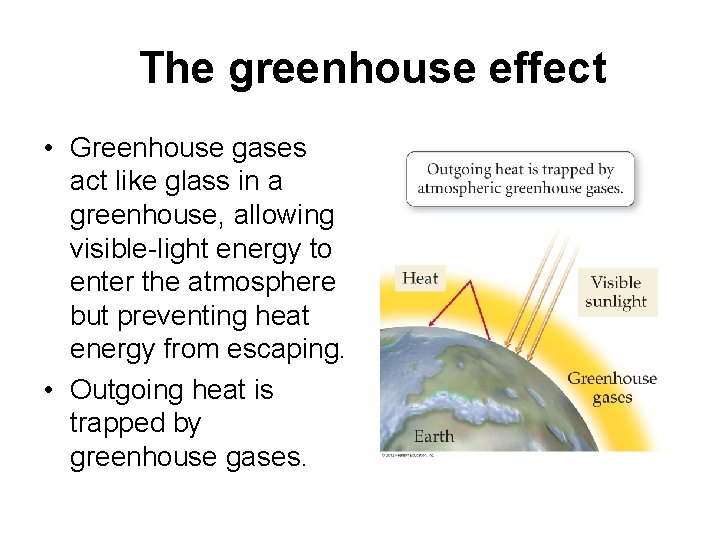
The greenhouse effect • Greenhouse gases act like glass in a greenhouse, allowing visible-light energy to enter the atmosphere but preventing heat energy from escaping. • Outgoing heat is trapped by greenhouse gases.

Combustion of fossil fuels produces CO 2. • Consider the combustion of octane (C 8 H 18), a component of gasoline: 2 C 8 H 18(l) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(g) • The balanced chemical equation shows that 16 mol of CO 2 are produced for every 2 mol of octane burned.

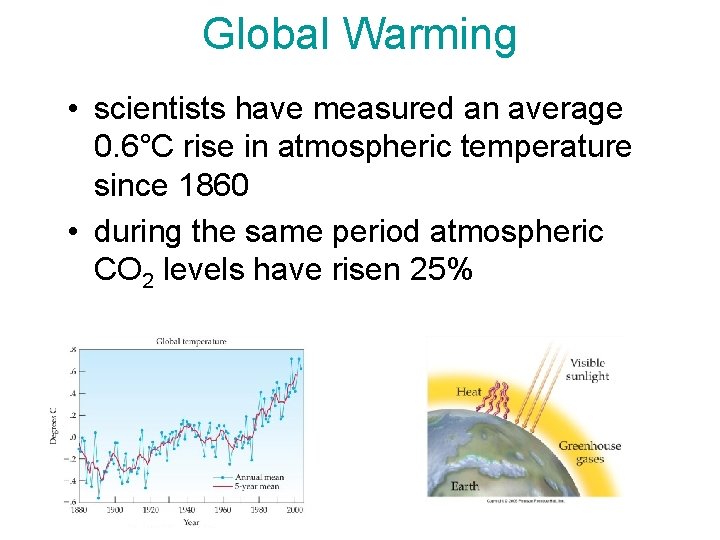
Global Warming • scientists have measured an average 0. 6°C rise in atmospheric temperature since 1860 • during the same period atmospheric CO 2 levels have risen 25%

The Source of Increased CO 2 • the primary source of the increased CO 2 levels are combustion reactions of fossil fuels we use to get energy (methane and octane) – 1860 corresponds to the beginning of the Industrial Revolution in the US and Europe

STOICHIOMETRY q Stoichiometry is the quantitative relationship between the reactants and products in a balanced chemical equation. q A balanced chemical equation provides several important information about the reactants and products in a chemical reaction. 8

MOLAR RATIOS For example: 1 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) This is the molar 2 molecules 3 molecules ratios between 100 molecules the 300 reactants and 200 molecules products 6 molecules 106 molecules 3 x 10 2 x 106 molecules 1 molecule 1 mole 3 moles 2 moles 9

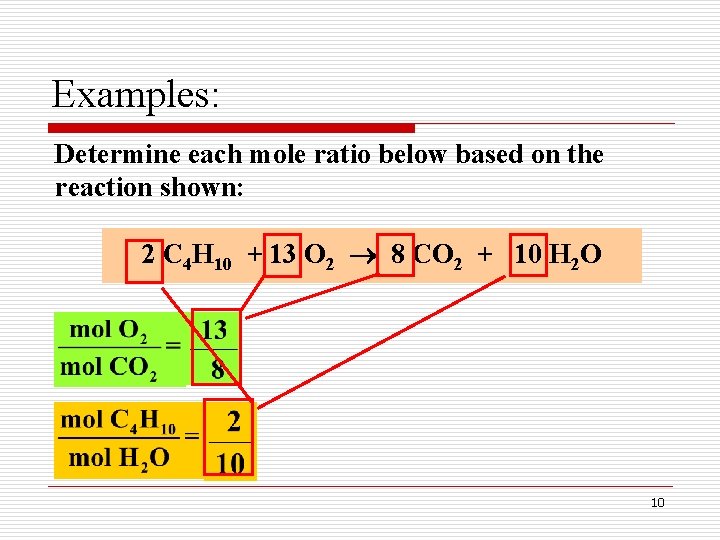
Examples: Determine each mole ratio below based on the reaction shown: 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O 10

Stoichiometry - Allows us to predict products that form in a reaction based on amount of reactants. The amount of each element must be the same throughout the overall reaction. For example, the amount of element H or O on the reactant side must equal the amount of element H or O on the product side. 2 H 2 + O 2 2 H 2 O

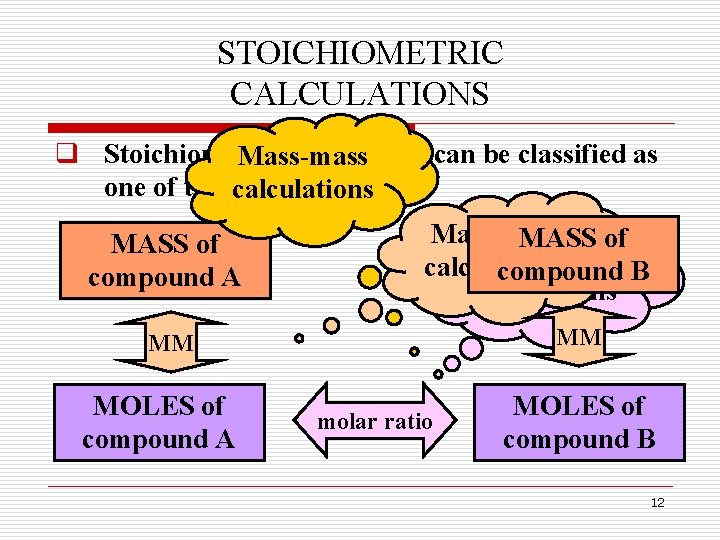
STOICHIOMETRIC CALCULATIONS q Stoichiometric calculations can be classified as Mass-mass one of the following: calculations MASS of compound A Mass-mole MASS of Mole-mole calculations compound B calculations MM MM MOLES of compound A molar ratio MOLES of compound B 12

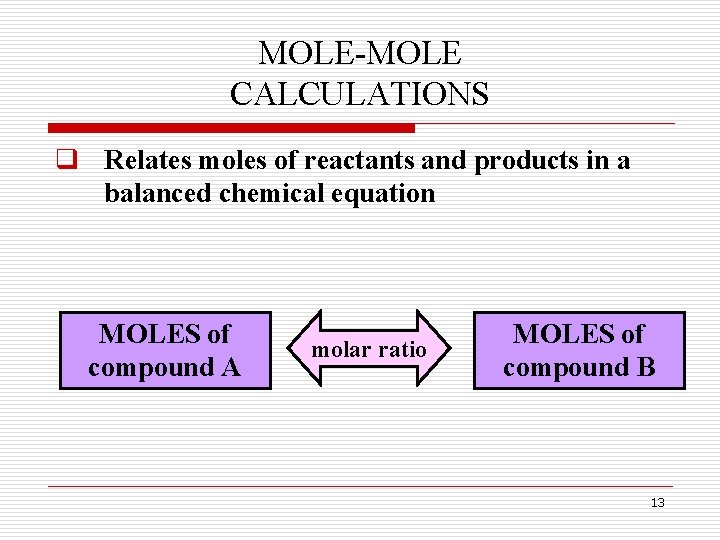
MOLE-MOLE CALCULATIONS q Relates moles of reactants and products in a balanced chemical equation MOLES of compound A molar ratio MOLES of compound B 13

Example 1: How many moles of ammonia can be produced from 32 moles of hydrogen? (Assume excess N 2 present) 1 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) 32 mol H 2 2 3 = 21 mol NH 3 Mole ratio 14

Example 2: In one experiment, 6. 80 mol of ammonia are prepared. How many moles of hydrogen were used up in this experiment? 1 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) 6. 80 mol NH 3 3 2 = 10. 2 mol H 2 Mole ratio 15

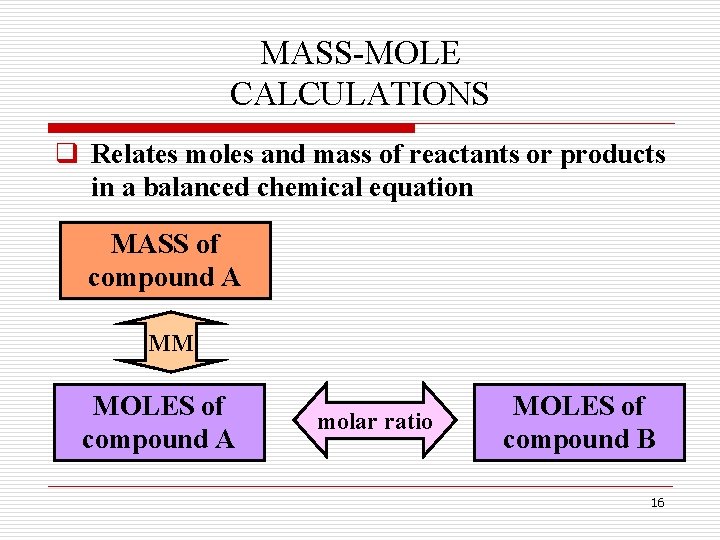
MASS-MOLE CALCULATIONS q Relates moles and mass of reactants or products in a balanced chemical equation MASS of compound A MM MOLES of compound A molar ratio MOLES of compound B 16

Example 1: How many moles of ammonia can be produced from the reaction of 125 g of nitrogen? 1 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) 125 g N 2 1 28. 0 Molar mass 2 1 = 8. 93 mol NH 3 Mole ratio 17

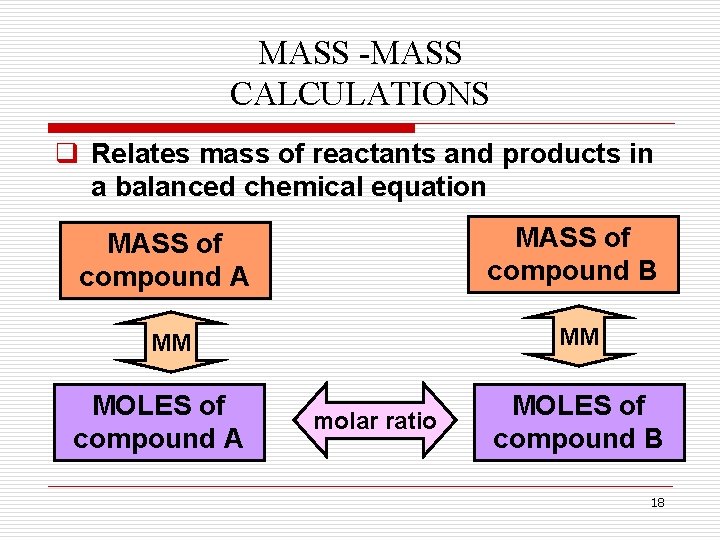
MASS -MASS CALCULATIONS q Relates mass of reactants and products in a balanced chemical equation MASS of compound A MASS of compound B MM MM MOLES of compound A molar ratio MOLES of compound B 18

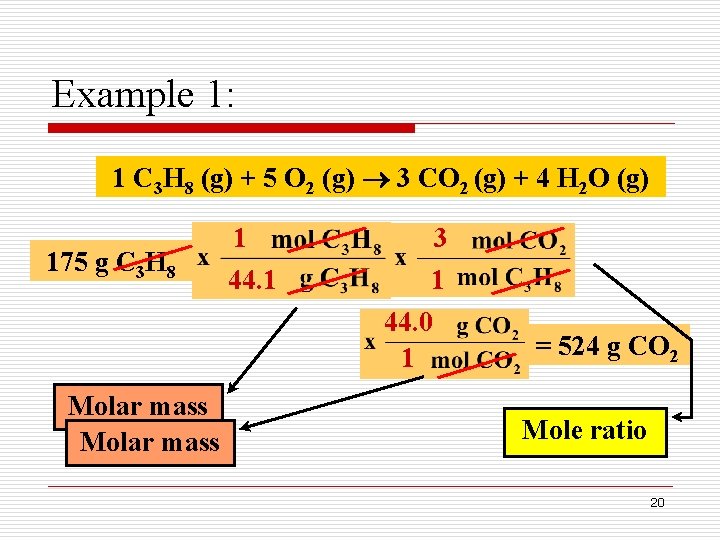
Example 1: What mass of carbon dioxide will be produced from the reaction of 175 g of propane, as shown? 1 C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (g) Mass of propane Moles of carbon dioxide Mass of carbon dioxide 19

Example 1: 1 C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (g) 175 g C 3 H 8 Molar mass 1 44. 1 3 1 44. 0 1 = 524 g CO 2 Mole ratio 20

LIMITING REACTANT q When 2 or more reactants are combined in nonstoichiometric ratios, the amount of product produced is limited by the reactant that is not in excess. q This reactant is referred to as limiting reactant. q When doing stoichiometric problems of this type, the limiting reactant must be determined first before proceeding with the calculations. 21

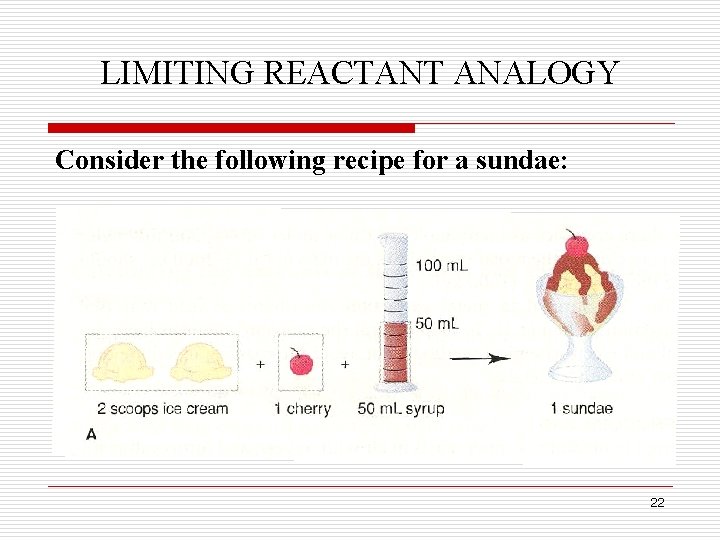
LIMITING REACTANT ANALOGY Consider the following recipe for a sundae: 22

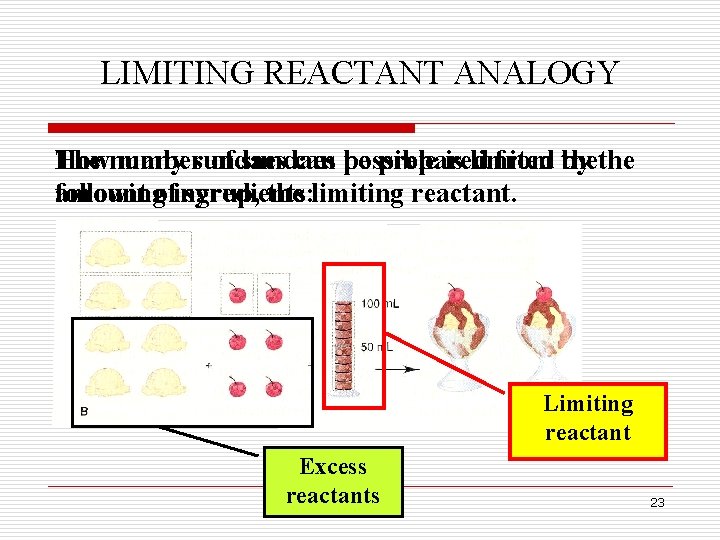
LIMITING REACTANT ANALOGY The number How many sundaes of sundaes can possible be prepared is limited from by thethe followingofingredients: amount syrup, the limiting reactant. Limiting reactant Excess reactants 23

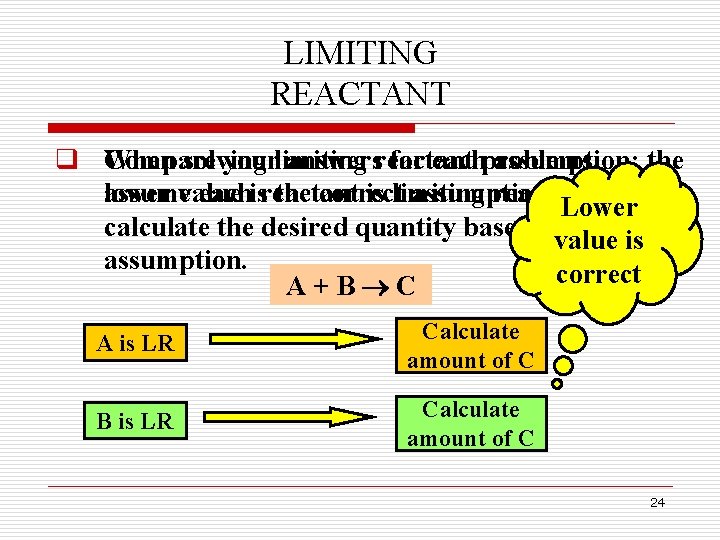
LIMITING REACTANT q When Compare solving yourlimiting answersreactant for eachproblems, assumption; the assumevalue lower eachisreactant the correct is limiting assumption. reactant, and Lower calculate the desired quantity based on that value is assumption. correct A+B C A is LR Calculate amount of C B is LR Calculate amount of C 24

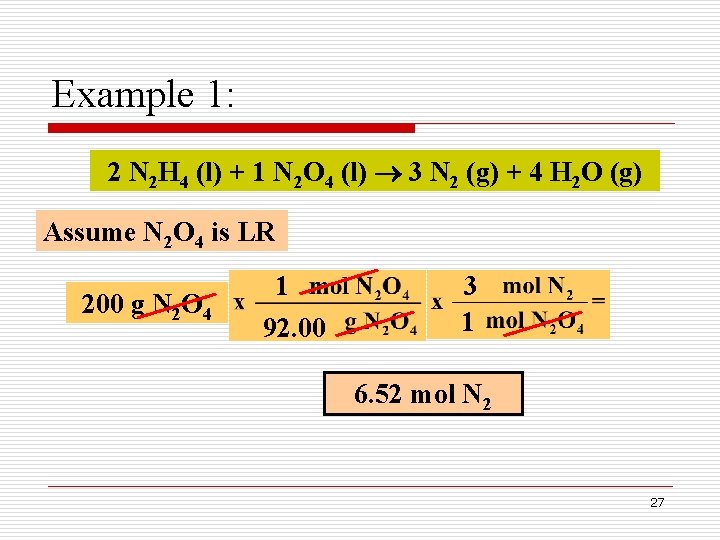
Example 1: A fuel mixture used in the early days of rocketry was a mixture of N 2 H 4 and N 2 O 4, as shown below. How many grams of N 2 gas is produced when 100 g of N 2 H 4 and 200 g of N 2 O 4 are mixed? 2 N 2 H 4 (l) + 1 N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (g) Limiting reactant Mass-mass calculations 25

Example 1: 2 N 2 H 4 (l) + 1 N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (g) Assume N 2 H 4 is LR 100 g N 2 H 4 1 32. 04 3 2 4. 68 mol N 2 26

Example 1: 2 N 2 H 4 (l) + 1 N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (g) Assume N 2 O 4 is LR 200 g N 2 O 4 1 92. 00 3 1 6. 52 mol N 2 27

Example 1: 2 N 2 H 4 (l) + 1 N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (g) Assume N 2 H 4 is LR 4. 68 mol N 2 Assume N 2 O 4 is LR 6. 52 mol N 2 H 4 is LR Correct amount 28

Example 1: 2 N 2 H 4 (l) + 1 N 2 O 4 (l) 3 N 2 (g) + 4 H 2 O (g) Calculate mass of N 2 4. 68 mol N 2 28. 0 1 = 131 g N 2 29

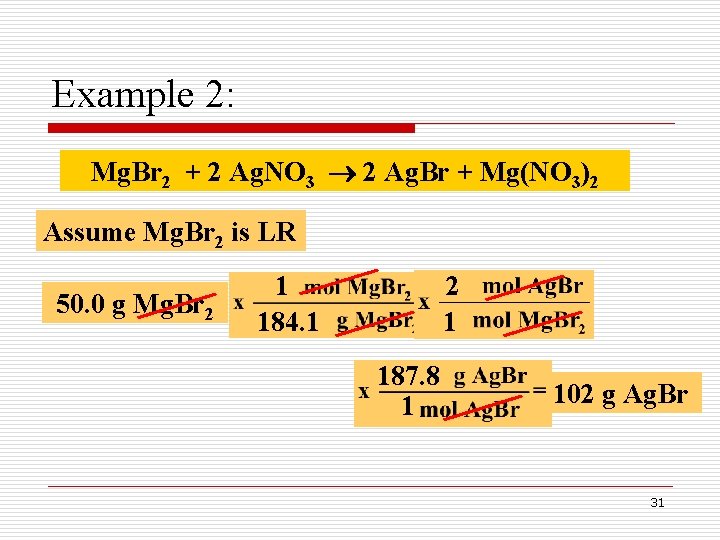
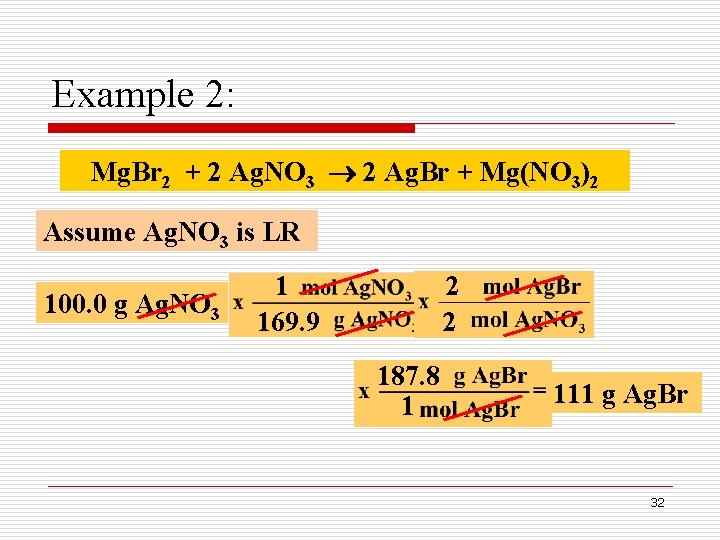
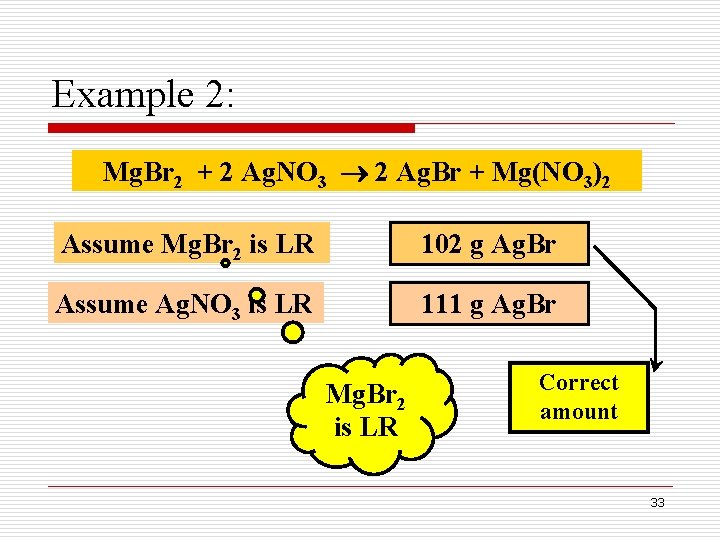
Example 2: How many grams of Ag. Br can be produced when 50. 0 g of Mg. Br 2 is mixed with 100. 0 g of Ag. NO 3, as shown below: Mg. Br 2 + 2 Ag. NO 3 2 Ag. Br + Mg(NO 3)2 Limiting Reactant 30

Example 2: Mg. Br 2 + 2 Ag. NO 3 2 Ag. Br + Mg(NO 3)2 Assume Mg. Br 2 is LR 50. 0 g Mg. Br 2 1 184. 1 2 1 187. 8 1 102 g Ag. Br 31

Example 2: Mg. Br 2 + 2 Ag. NO 3 2 Ag. Br + Mg(NO 3)2 Assume Ag. NO 3 is LR 100. 0 g Ag. NO 3 1 169. 9 2 2 187. 8 1 111 g Ag. Br 32

Example 2: Mg. Br 2 + 2 Ag. NO 3 2 Ag. Br + Mg(NO 3)2 Assume Mg. Br 2 is LR 102 g Ag. Br Assume Ag. NO 3 is LR 111 g Ag. Br Mg. Br 2 is LR Correct amount 33

PERCENT YIELD q The amount of product calculated through stoichiometric ratios are the maximum amount product that can be produced during the reaction, and is thus called theoretical yield. q The actual yield of a product in a chemical reaction is the actual amount obtained from the reaction. 34

PERCENT YIELD q The percent yield of a reaction is obtained as follows: 35

Example 1: In an experiment forming ethanol, theoretical yield is 50. 0 g and the actual yield is 46. 8 g. What is the percent yield for this reaction? 92. 7 % 36

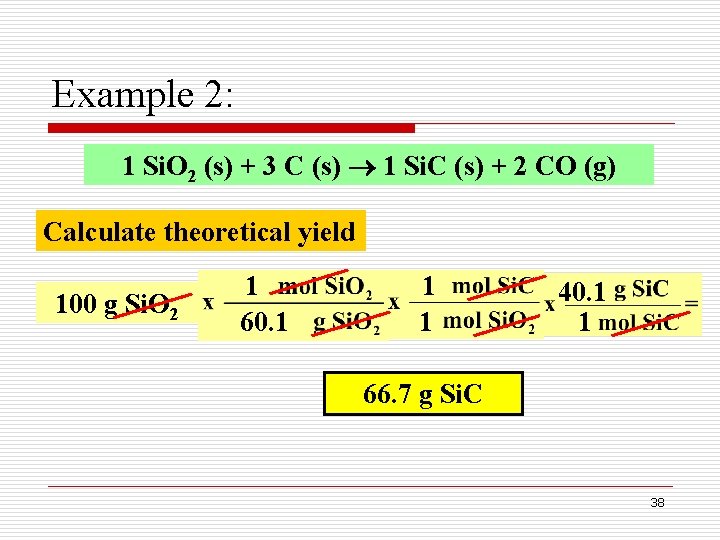
Example 2: Silicon carbide can be formed from the reaction of sand (Si. O 2) with carbon as shown below: 1 Si. O 2 (s) + 3 C (s) 1 Si. C (s) + 2 CO (g) When 100 g of sand are processed, 51. 4 g of Si. C is produced. What is the percent yield of Si. C in this reaction? Actual yield 37

Example 2: 1 Si. O 2 (s) + 3 C (s) 1 Si. C (s) + 2 CO (g) Calculate theoretical yield 100 g Si. O 2 1 60. 1 1 1 40. 1 1 66. 7 g Si. C 38

Example 2: Calculate percent yield 77. 1 % 39

Theoretical and Actual Yield • In order to determine theoretical yield, we use reaction stoichiometry to determine the amount of product each of our reactants could make. • The theoretical yield will always be the least possible amount of product. – The theoretical yield will always come from the limiting reactant. • Because of both controllable and uncontrollable factors, the actual yield of product will always be less than theoretical yield.

Chap. 8 terms you should know 1. Limiting reactant - the reactant that limits the amount of product produced in a chemical reaction. The reactant that makes the least amount of product. 2. Theoretical yield - the amount of product that can be made in a chemical reaction based on the amount of limiting reactant. 3. Actual yield - the amount of product actually produced by a chemical reaction. 4. Percent yield - The percent of theoretical yield that was actually obtained. actual yield % yield = theoretical yield x 100

THE END 42