Chapter 8 p 142 160 METABOLISM ENZYMATIC REACTIONS

Chapter 8 (p. 142 -160) METABOLISM & ENZYMATIC REACTIONS

Thermodynamics First Law Second Law • Energy can be transferred and transformed, but it cannot be created or destroyed • Every energy transfer or transformation increases the entropy (disorder) of the universe © 2011 Pearson Education, Inc.

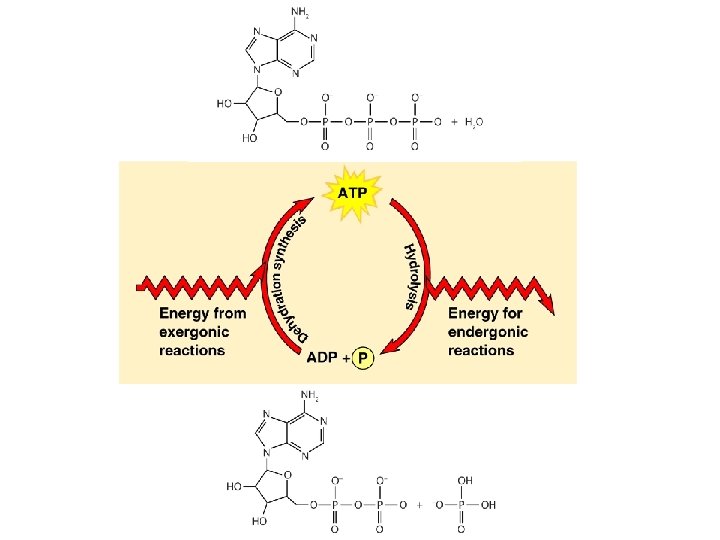
Free-Energy Change, G • Energy that can do work when temperature and pressure are uniform, as in a living cell ∆G = ∆H – T∆S © 2011 Pearson Education, Inc.
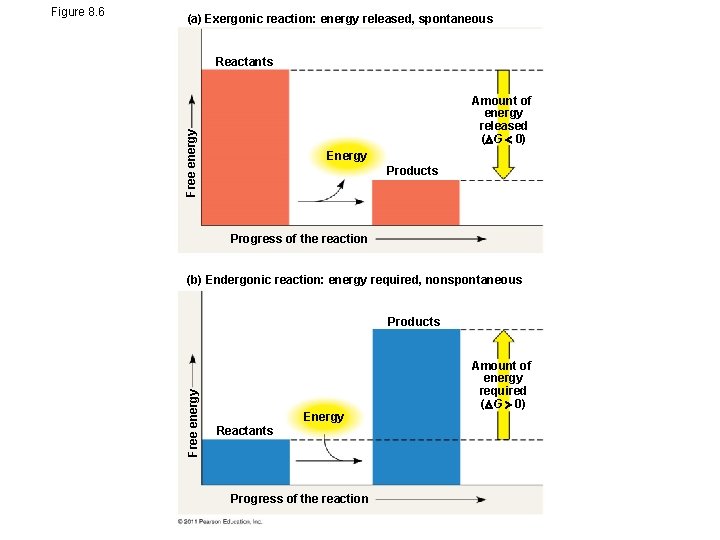
(a) Exergonic reaction: energy released, spontaneous Reactants Free energy Amount of energy released ( G 0) Energy Products Progress of the reaction (b) Endergonic reaction: energy required, nonspontaneous Products Free energy Figure 8. 6 Energy Reactants Progress of the reaction Amount of energy required ( G 0)
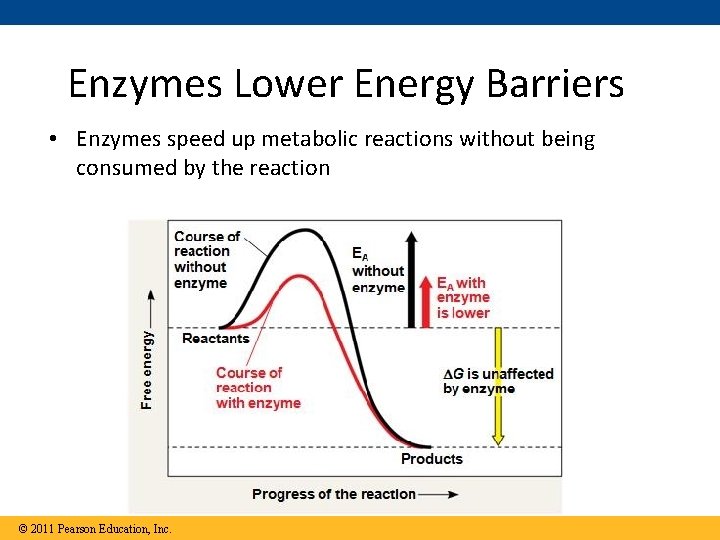
Enzymes Lower Energy Barriers • Enzymes speed up metabolic reactions without being consumed by the reaction © 2011 Pearson Education, Inc.
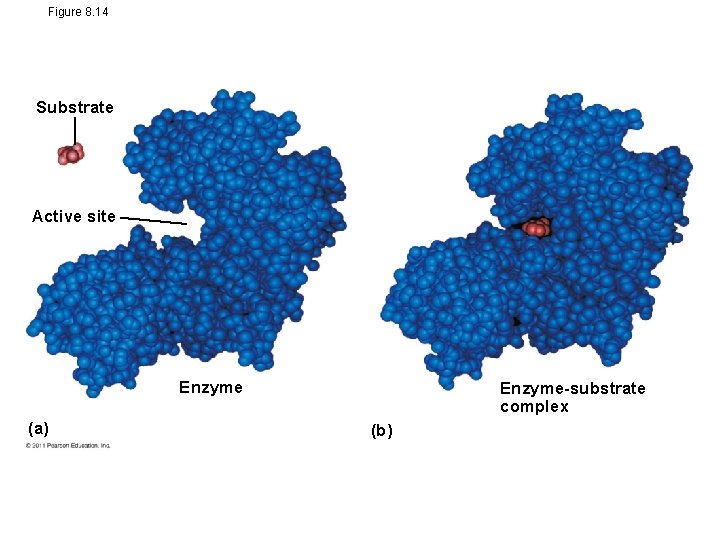
Figure 8. 14 Substrate Active site Enzyme (a) Enzyme-substrate complex (b)
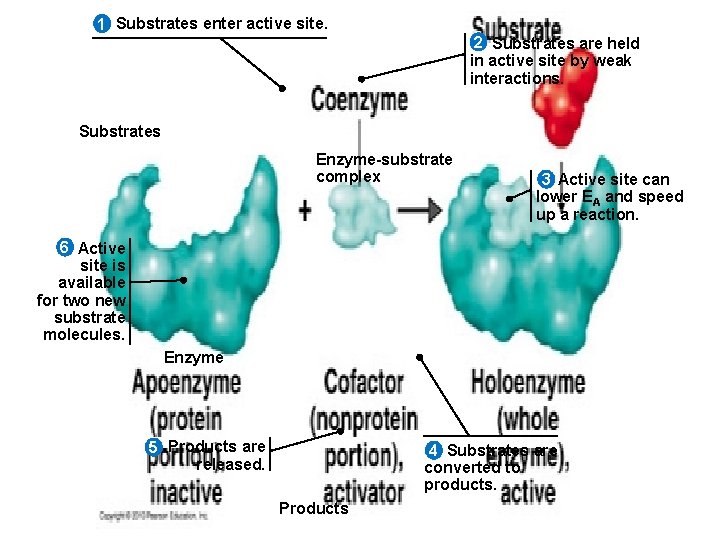
1 Substrates enter active site. 2 Substrates are held in active site by weak interactions. Substrates Enzyme-substrate complex 3 Active site can lower EA and speed up a reaction. 6 Active site is available for two new substrate molecules. Enzyme 5 Products are released. 4 Substrates are converted to products. Products
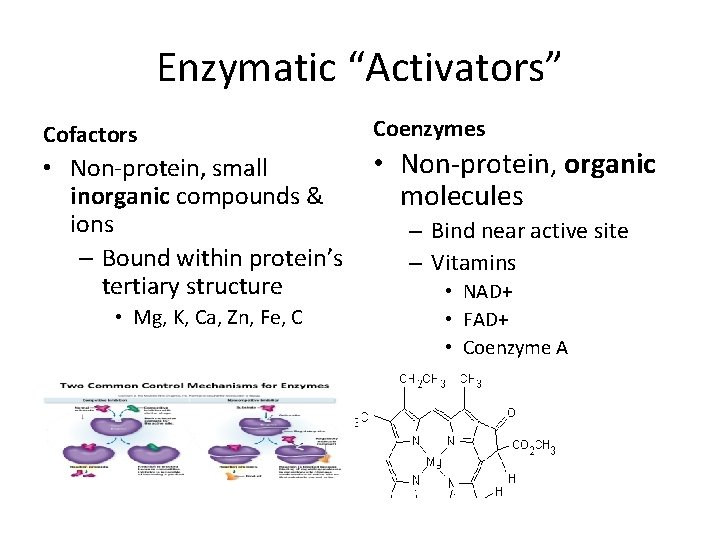
Enzymatic “Activators” Cofactors • Non-protein, small inorganic compounds & ions – Bound within protein’s tertiary structure • Mg, K, Ca, Zn, Fe, C Coenzymes • Non-protein, organic molecules – Bind near active site – Vitamins • NAD+ • FAD+ • Coenzyme A


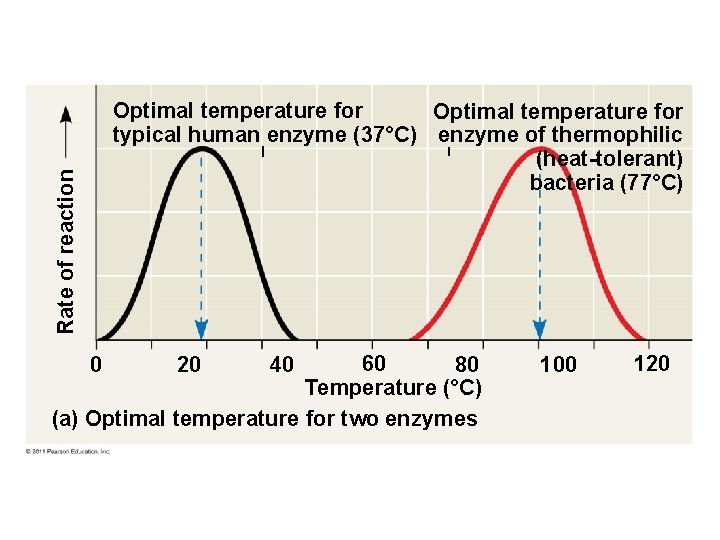
Rate of reaction Optimal temperature for typical human enzyme (37°C) enzyme of thermophilic (heat-tolerant) bacteria (77°C) 60 80 Temperature (°C) (a) Optimal temperature for two enzymes 0 20 40 100 120

Figure 8. 16 b Rate of reaction Optimal p. H for pepsin (stomach enzyme) 0 5 p. H (b) Optimal p. H for two enzymes 1 2 3 4 Optimal p. H for trypsin (intestinal enzyme) 6 7 8 9 10
- Slides: 14