CHAPTER 8 Ohms law describes the relationship of

CHAPTER 8 Ohm’s law describes the relationship of current, voltage, and resistance

IN THIS CHAPTER… • Explain how electric current results from separation of charge and the movement of electrons • Apply the laws of static charge to electron flow in a circuit • Define voltage, current, and resistance • Draw circuit diagrams using appropriate symbols • Distinguish between potential and kinetic energy; static electricity; and conventional current and electron flow

CHAPTER 8 • In this chapter you will learn how electrical energy is transferred and transformed • Can you think of some examples where this electrical energy is transformed? • How about transferred?

8. 1 – ELECTRIC POTENTIAL ENERGY AND VOLTAGE

OBJECTIVES • Unlike charges gain electrical potential energy when they are moved farther apart • Voltage (potential difference) is the change in potential energy per coulomb of charge • Electrical energy depends on the amount of charge and the voltage

BATTERY VS LIGHTNING • Remember: Static Electricity is the build up of positive or negative charges • What is the difference between a battery and lightning in terms of charges? • They both have negative and positive charges • A lightning strike is an uncontrolled burst of electrical energy • A battery is can provide steady, controlled flow of electricity

BATTERIES • A battery is a combination of electrochemical cells connected together • These electrochemical cells convert chemical energy into electrical energy stored in charges • Electrochemical cells are most commonly referred to as cells or batteries • Electrochemical cells – converts chemical energy into electrical energy stored in charges. • Battery – a combination of one or more electrochemical cells.

THE BATTERY • Terminals are the end points where we make a connection • Extra electrons accumulate on one of the battery terminals making it negatively charged • The other terminal had lost these electrons and is therefore positively charged • So, when a battery is connected to a MP 3 player, electrons can travel through the wires • Because we can attract and repel electrons we can get them to move • Because we can move them we can get them to do work for us!!
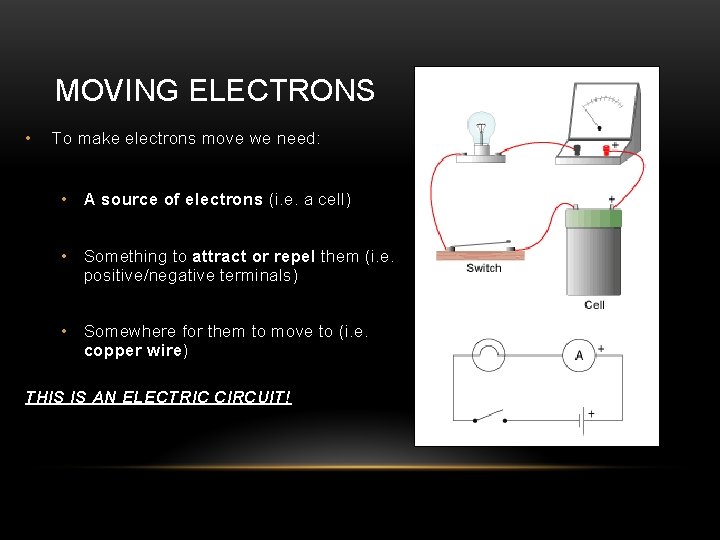
MOVING ELECTRONS • To make electrons move we need: • A source of electrons (i. e. a cell) • Something to attract or repel them (i. e. positive/negative terminals) • Somewhere for them to move to (i. e. copper wire) THIS IS AN ELECTRIC CIRCUIT!
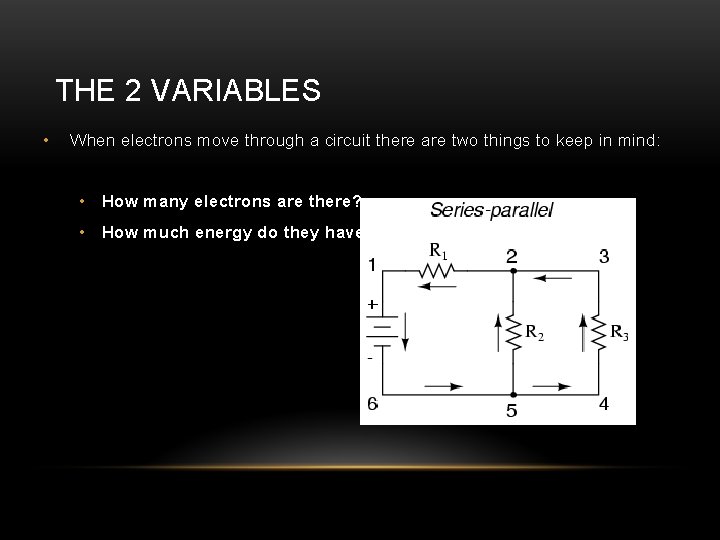
THE 2 VARIABLES • When electrons move through a circuit there are two things to keep in mind: • How many electrons are there? • How much energy do they have?

NUMBER OF ELECTRONS • The amount of electrons flowing through a circuit is called the CURRENT • Current in measured in Amps (A) by an Ammeter • 1 amp = 1 coulomb sec

ELECTRICAL POTENTIAL ENERGY • Energy - is the ability to do work • Potential Energy – energy stored in an object. • Kinetic Energy – energy of motion. • Electric Potential Energy – electrons have stored energy and the ability to do work after they leave the battery. • Electrons will help convert electrical energy into another type of energy (i. e. heat or light)

ELECTRICAL POTENTIAL DIFFERENCE • Voltage – the amount of electric potential energy per one coulomb of charge. • How far apart we move the charges • The charges want to move back to the atom • Unit of Voltage – volts (V) • Voltmeter – a device that measures the amount of potential difference between two locations of charge separation. • The voltage across a muscle cell in your body is about 70 millivolts (m. V) is one thousandth of a volt

COMPARING POTENTIAL ENERGY AND POTENTIAL DIFFERENCE • Potential Difference (voltage) – how far apart we move the charges. • The height of a rollercoaster. • Electric Potential Energy – how far apart we move charges and the amount of charge • The height of a rollercoaster + how many people are on the rollercoaster. • Very old video
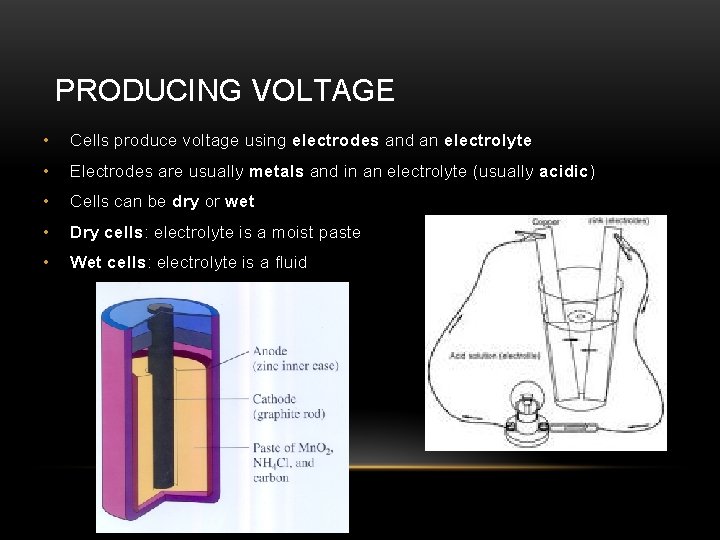
PRODUCING VOLTAGE • Cells produce voltage using electrodes and an electrolyte • Electrodes are usually metals and in an electrolyte (usually acidic) • Cells can be dry or wet • Dry cells: electrolyte is a moist paste • Wet cells: electrolyte is a fluid

MANY CELLS = BATTERY

MANY SOURCES OF ELECTRICAL ENERGY • Friction • Piezoelectric crystals (piezo = pressure or push) • Photo-electrochemical cells • Thermocouples • Generators • Video 1 • Hydroelectric • Nuclear Energy
- Slides: 17