Chapter 8 MOLECULAR STRUCTURE COVALENT BONDING THEORIES Two

Chapter 8 MOLECULAR STRUCTURE & COVALENT BONDING THEORIES

Two Simple Theories of Covalent Bonding � Valence Shell Electron Pair Repulsion Theory �VSEPR �R. J. Gillespie - 1950’s � Valence Bond Theory �Hybridized orbitals �L. Pauling - 1930’s & 40’s

VSEPR Theory � regions of high electron density around the central atom are as far apart as possible to minimize repulsions � five basic shapes �based on # of regions of high electron density � several modifications of these five basic shapes will also be examined
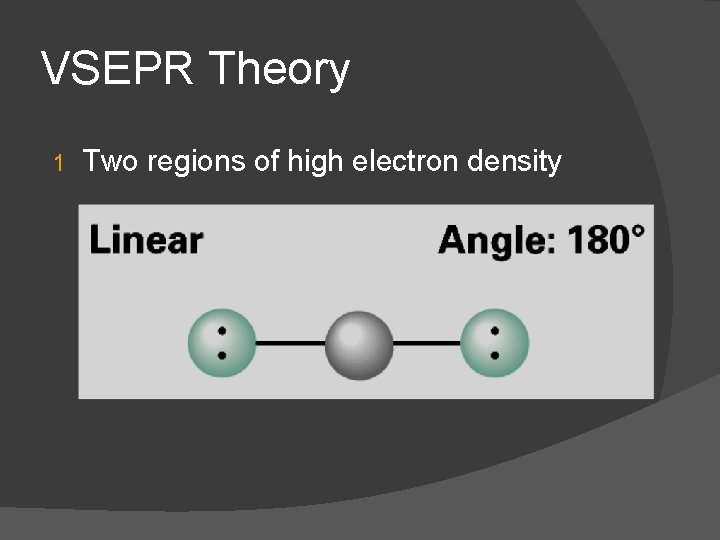
VSEPR Theory 1 Two regions of high electron density
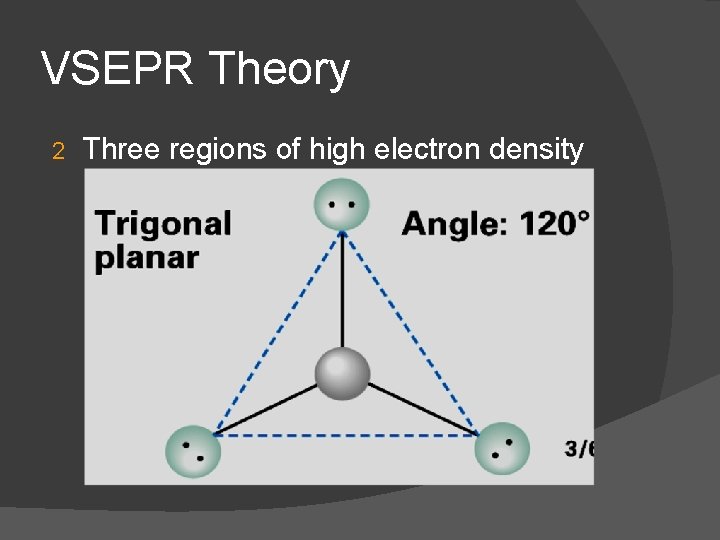
VSEPR Theory 2 Three regions of high electron density
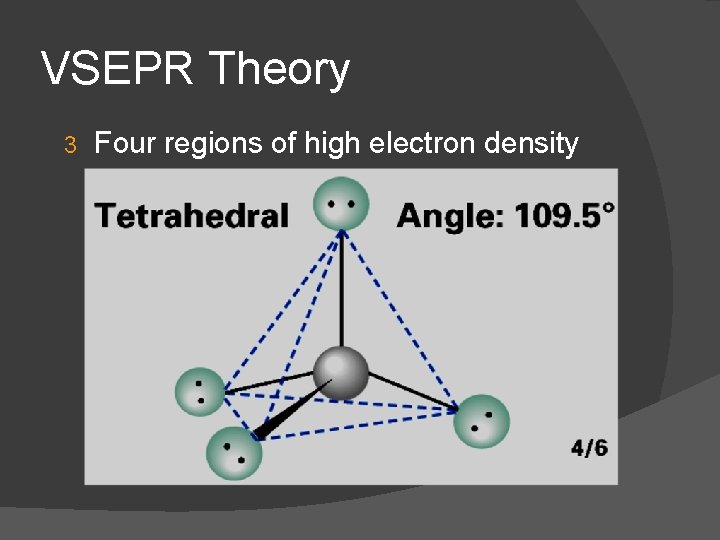
VSEPR Theory 3 Four regions of high electron density
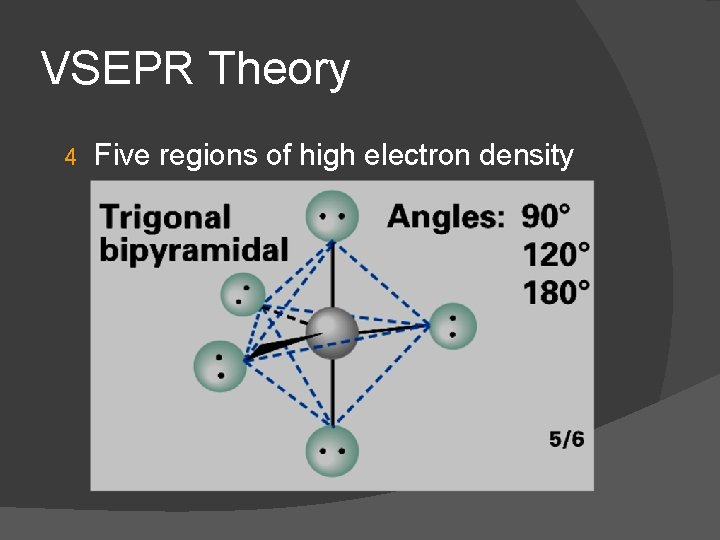
VSEPR Theory 4 Five regions of high electron density
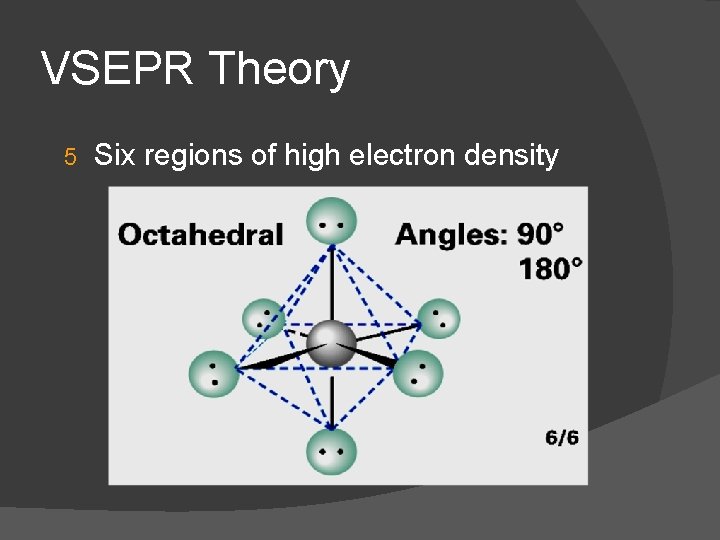
VSEPR Theory 5 Six regions of high electron density

VSEPR Theory � electronic geometry – the arrangement of the valence shell electrons around the central atom �determined by the locations of regions of high electron density around the central atom(s) � molecular geometry - determined by the arrangement of atoms around the central atom(s) �What the molecule really looks like electron pairs are not used in the molecular geometry determination
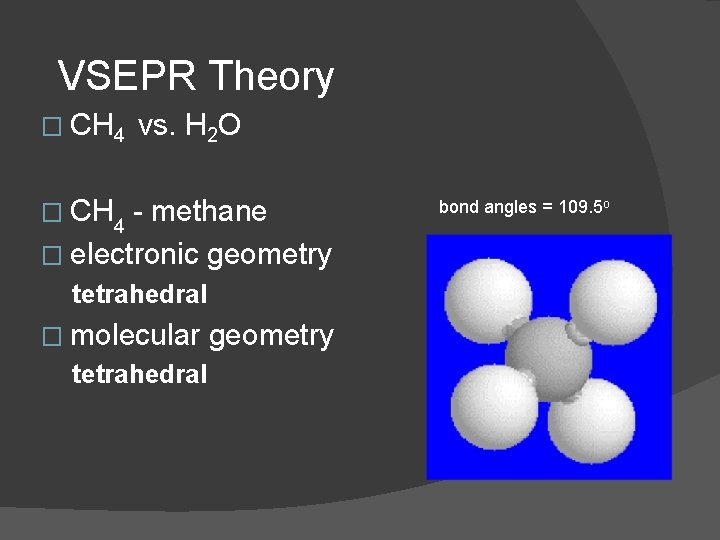
VSEPR Theory � CH 4 vs. H 2 O � CH 4 - methane � electronic geometry tetrahedral � molecular tetrahedral geometry bond angles = 109. 5 o
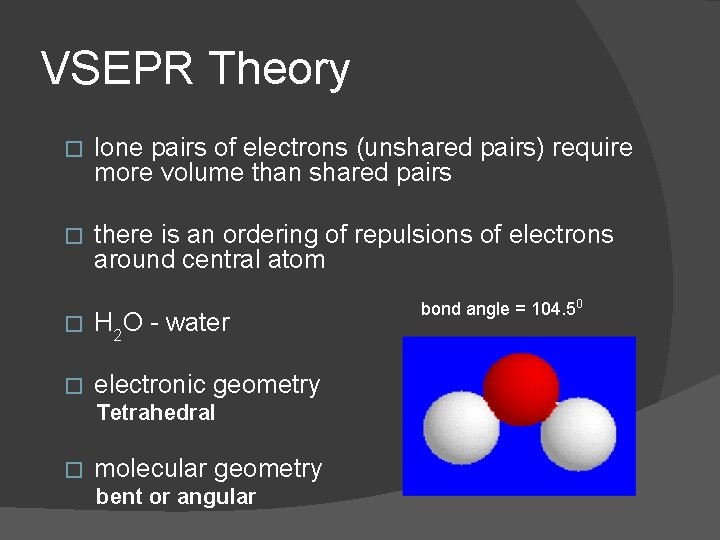
VSEPR Theory � lone pairs of electrons (unshared pairs) require more volume than shared pairs � there is an ordering of repulsions of electrons around central atom � H 2 O - water � electronic geometry Tetrahedral � molecular geometry bent or angular bond angle = 104. 50

VSEPR Theory 1. lone pair to lone pair repulsion is strongest 2. lone pair to bonding pair repulsion is intermediate 3. bonding pair to bonding pair repulsion is weakest � lone pair to lone pair repulsion is why bond angles in water are less than 109. 50

Valence Bond Theory � covalent bonds are formed by overlap of atomic orbitals � atomic orbitals on the central atom can mix and exchange their character – hybridization � common hybrids pink flowers, mules, corn, grass

Hybrid orbitals helps describe the same shapes as VSEPR – (hybridization – mixing of orbitals) From orbital diagrams Hybridization - CP s + p orbitals sp - 2 s + p orbitals sp 2 - 3 s + p + p orbitals sp 3 - 4 s + p + p + d orbitals sp 3 d - 5 s + p + p + d orbitals sp 3 d 2 - 6
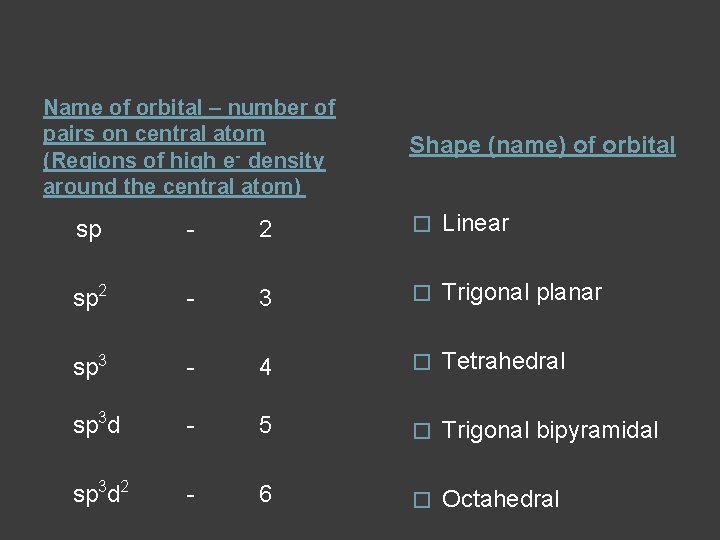
Name of orbital – number of pairs on central atom (Regions of high e- density around the central atom) Shape (name) of orbital sp - 2 � Linear sp 2 - 3 � Trigonal planar sp 3 - 4 � Tetrahedral sp 3 d - 5 � Trigonal bipyramidal sp 3 d 2 - 6 � Octahedral

Molecular Geometry Terminology � In the next sections the following terminology will be used A = central atom B = bonding pairs around central atom U = lone pairs around central atom For example: AB 3 U designates that there are 3 bonding pairs and 1 lone pair around the central atom (4 central pairs total)
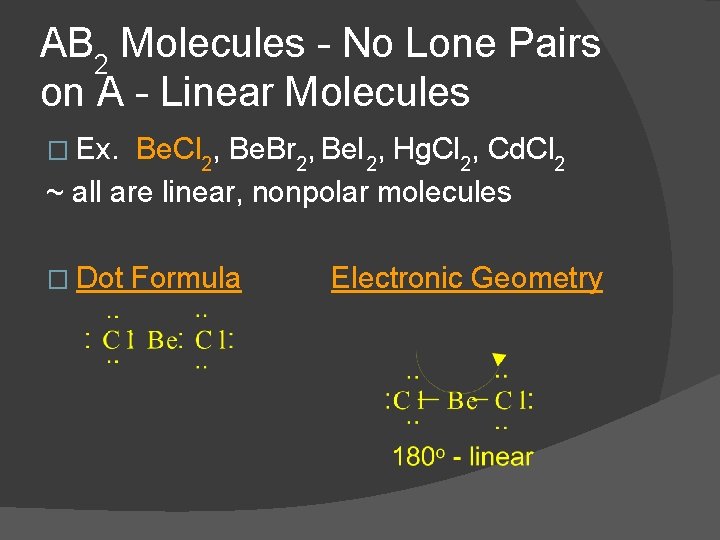
AB 2 Molecules - No Lone Pairs on A - Linear Molecules � Ex. Be. Cl 2, Be. Br 2, Be. I 2, Hg. Cl 2, Cd. Cl 2 ~ all are linear, nonpolar molecules � Dot Formula Electronic Geometry

AB 2 Molecules - No Lone Pairs on A - Linear Molecules � VSEPR Polarity
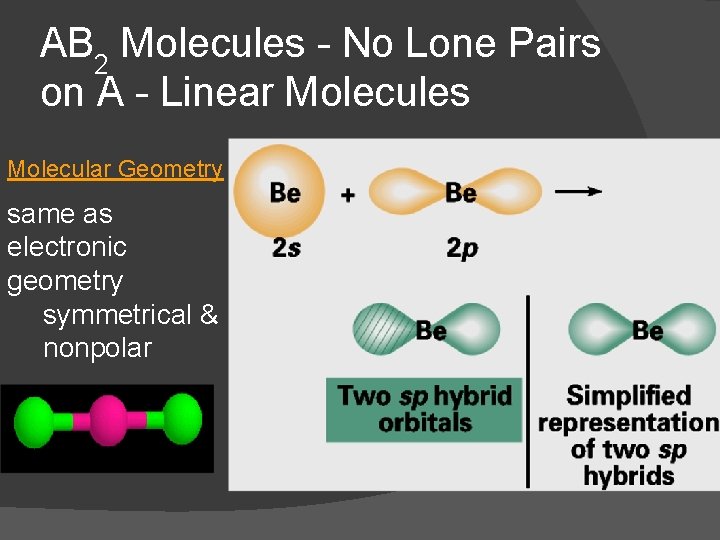
AB 2 Molecules - No Lone Pairs on A - Linear Molecules Molecular Geometry same as electronic geometry symmetrical & nonpolar
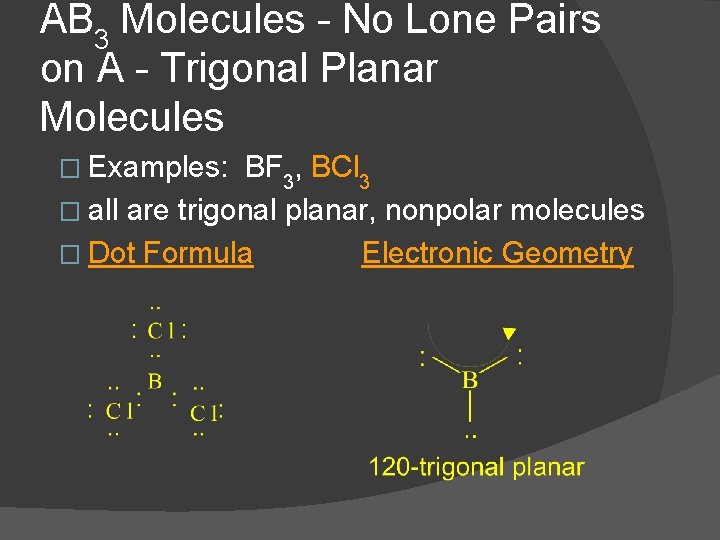
AB 3 Molecules - No Lone Pairs on A - Trigonal Planar Molecules � Examples: BF 3, BCl 3 � all are trigonal planar, nonpolar molecules � Dot Formula Electronic Geometry
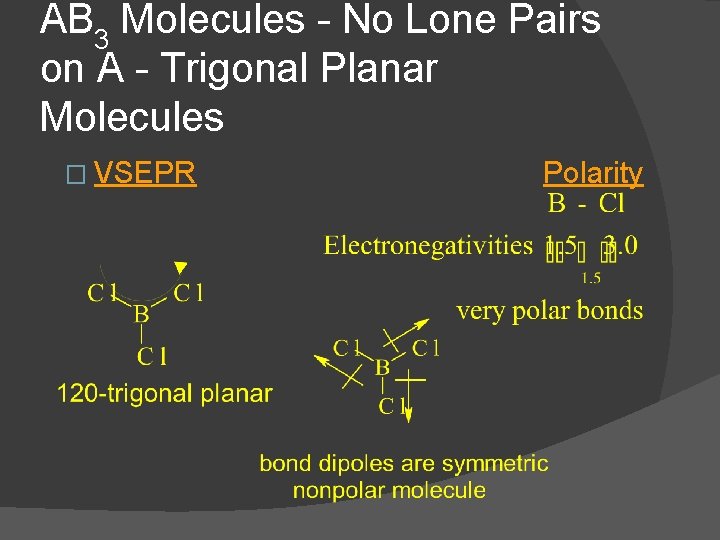
AB 3 Molecules - No Lone Pairs on A - Trigonal Planar Molecules � VSEPR Polarity
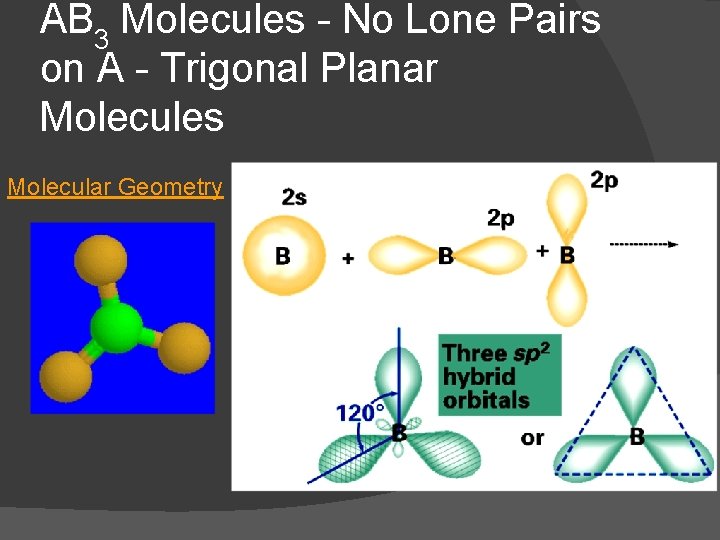
AB 3 Molecules - No Lone Pairs on A - Trigonal Planar Molecules Molecular Geometry
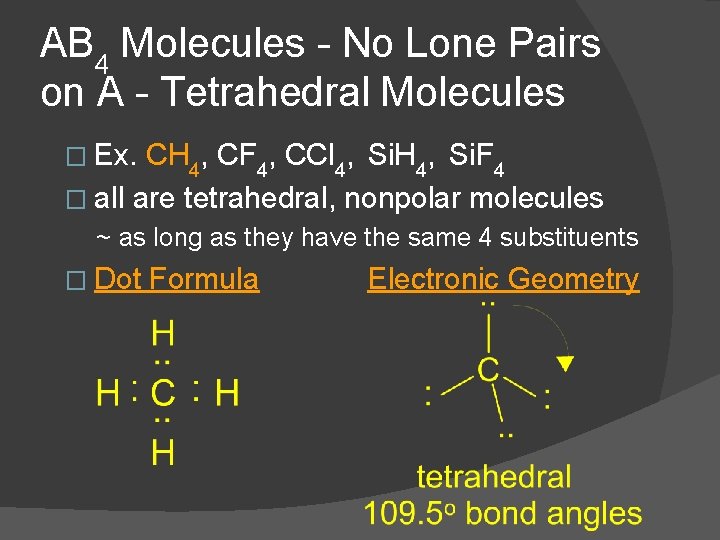
AB 4 Molecules - No Lone Pairs on A - Tetrahedral Molecules � Ex. CH 4, CF 4, CCl 4, Si. H 4, Si. F 4 � all are tetrahedral, nonpolar molecules ~ as long as they have the same 4 substituents � Dot Formula Electronic Geometry
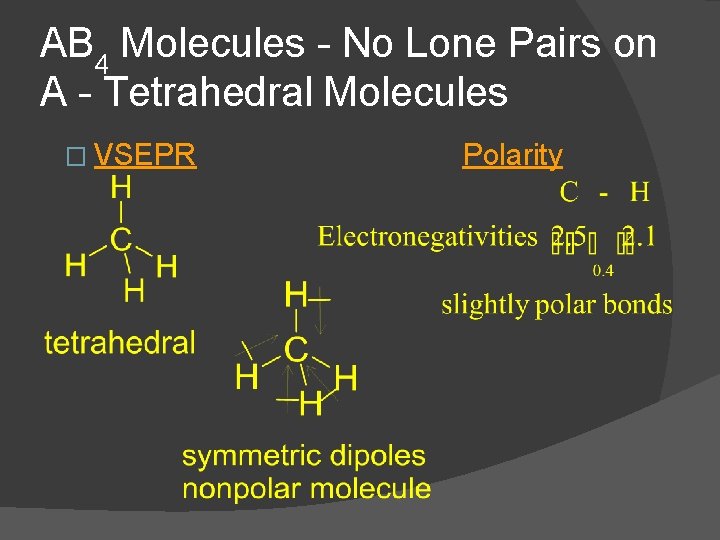
AB 4 Molecules - No Lone Pairs on A - Tetrahedral Molecules � VSEPR Polarity
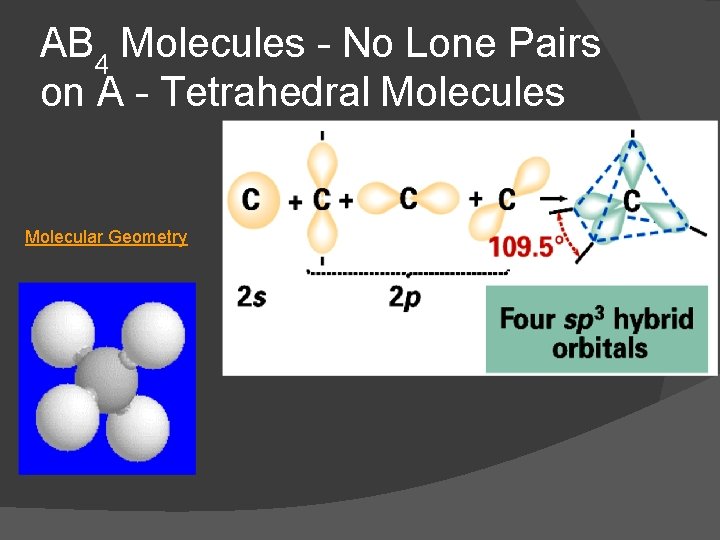
AB 4 Molecules - No Lone Pairs on A - Tetrahedral Molecules Molecular Geometry

AB 3 U Molecules - One Lone Pair - Pyramidal Molecules � examples NH 3, NF 3 � first example of lone pairs on the central atom �electronic and molecular geometry are different �all 3 substituents the same but molecule is polar � NH 3 and NF 3 are pyramidal, polar molecules
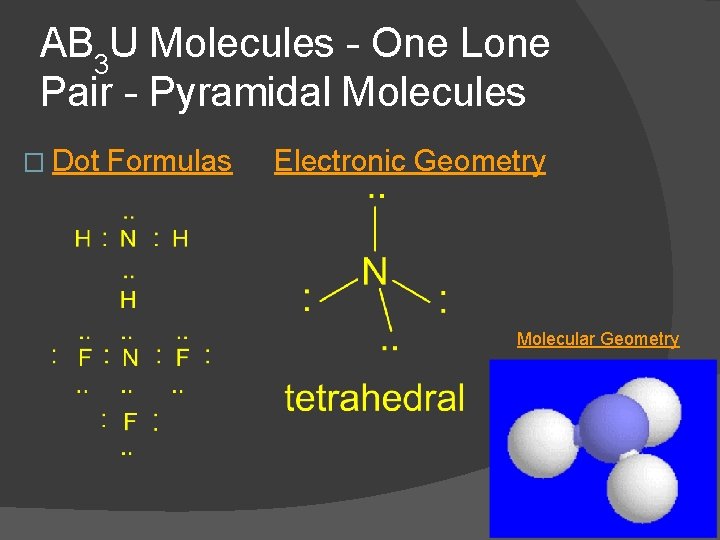
AB 3 U Molecules - One Lone Pair - Pyramidal Molecules � Dot Formulas Electronic Geometry Molecular Geometry
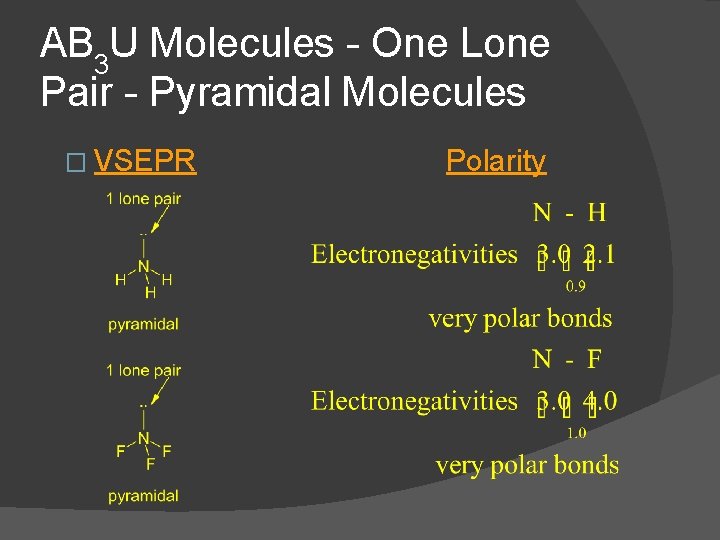
AB 3 U Molecules - One Lone Pair - Pyramidal Molecules � VSEPR Polarity
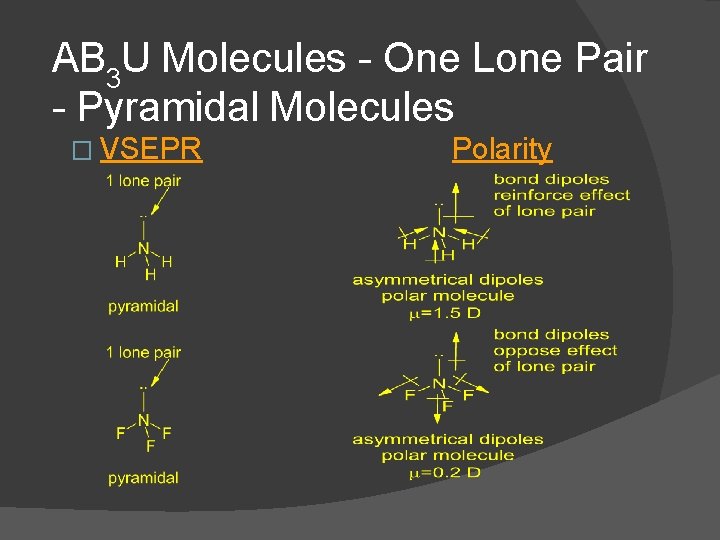
AB 3 U Molecules - One Lone Pair - Pyramidal Molecules � VSEPR Polarity
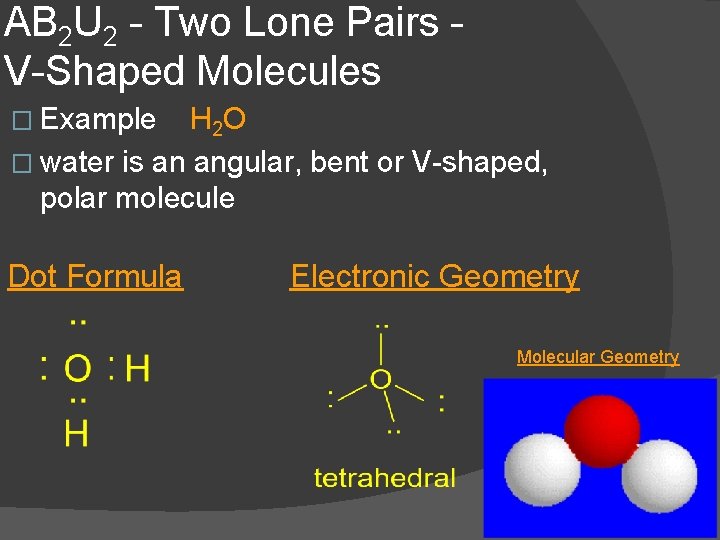
AB 2 U 2 - Two Lone Pairs V-Shaped Molecules � Example H 2 O � water is an angular, bent or V-shaped, polar molecule Dot Formula Electronic Geometry Molecular Geometry
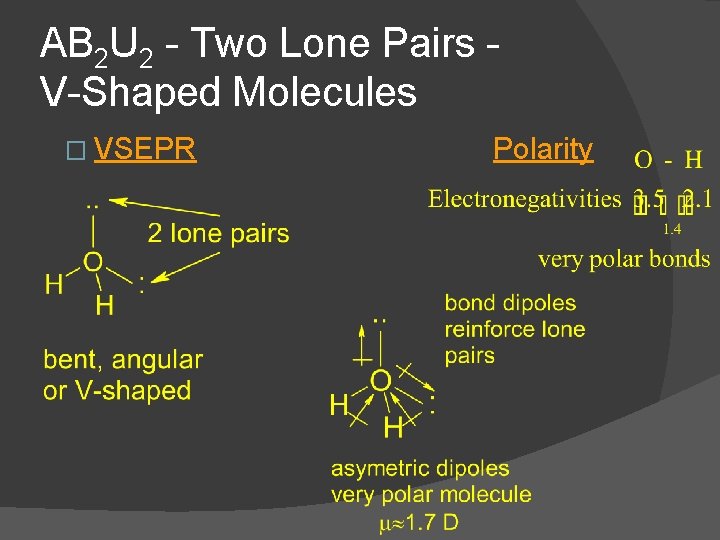
AB 2 U 2 - Two Lone Pairs V-Shaped Molecules � VSEPR Polarity
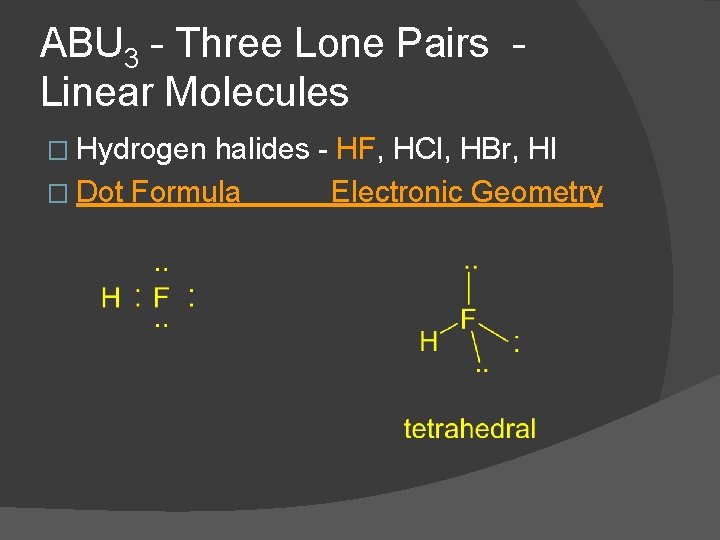
ABU 3 - Three Lone Pairs Linear Molecules � Hydrogen halides - HF, HCl, HBr, HI � Dot Formula Electronic Geometry
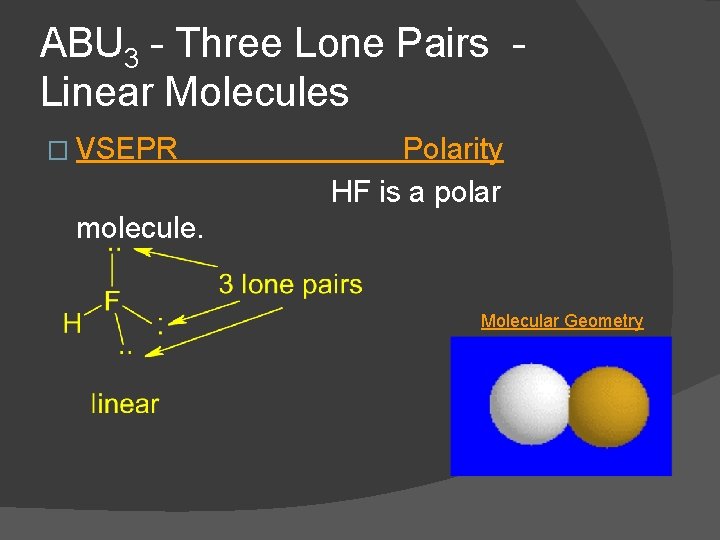
ABU 3 - Three Lone Pairs Linear Molecules � VSEPR Polarity HF is a polar molecule. Molecular Geometry
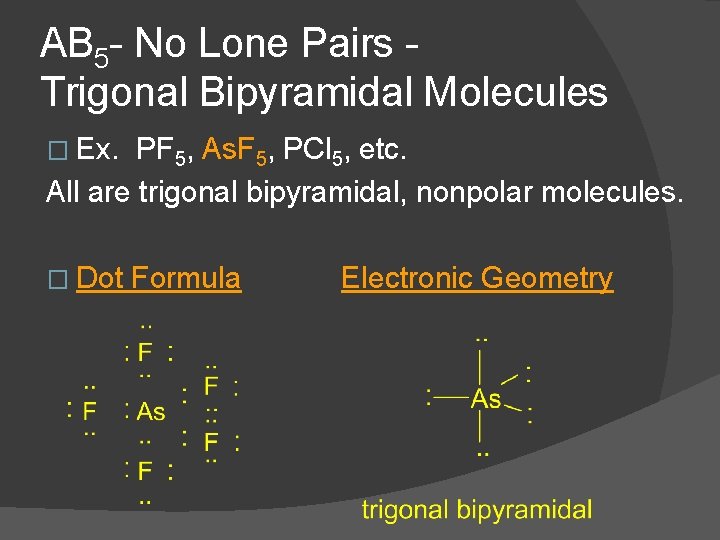
AB 5 - No Lone Pairs Trigonal Bipyramidal Molecules � Ex. PF 5, As. F 5, PCl 5, etc. All are trigonal bipyramidal, nonpolar molecules. � Dot Formula Electronic Geometry
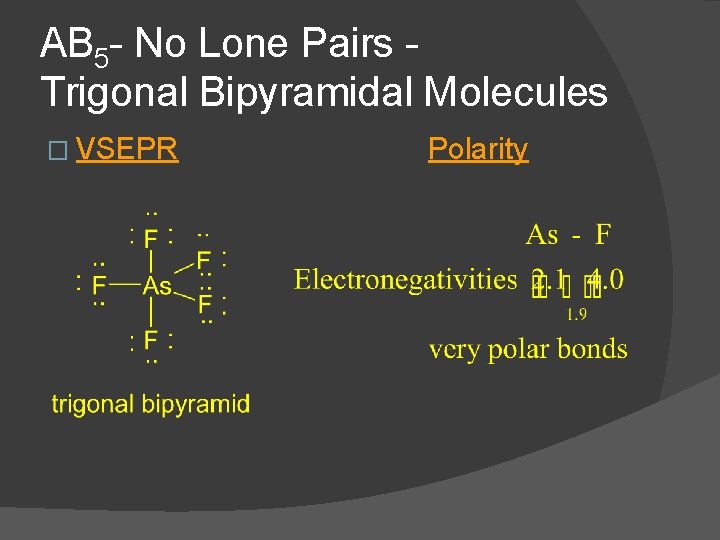
AB 5 - No Lone Pairs Trigonal Bipyramidal Molecules � VSEPR Polarity
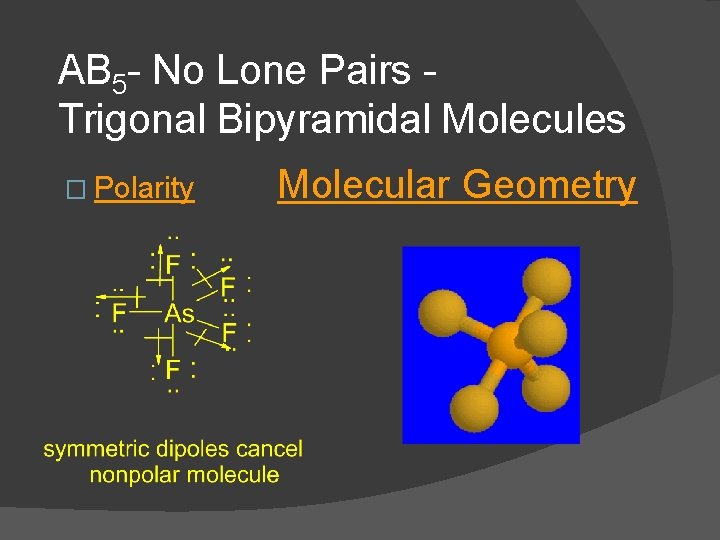
AB 5 - No Lone Pairs Trigonal Bipyramidal Molecules � Polarity Molecular Geometry
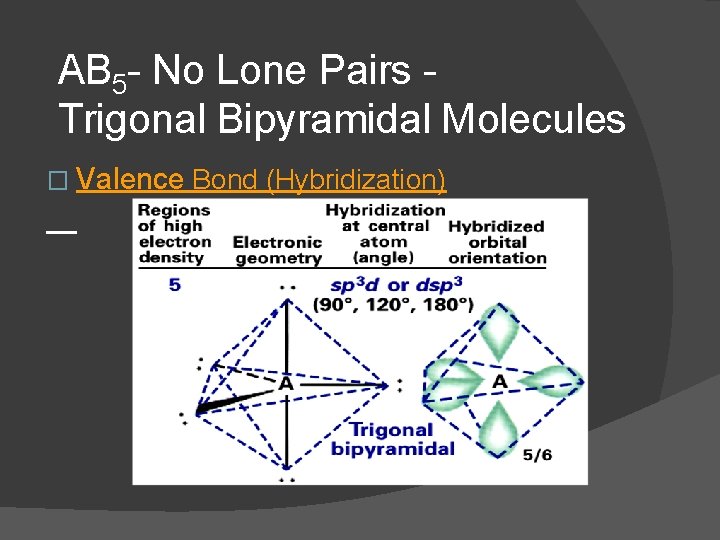
AB 5 - No Lone Pairs Trigonal Bipyramidal Molecules � Valence Bond (Hybridization)

AB 4 U- One Lone Pair Seesaw Molecules � For one lone pair an AB 4 U molecule results. � AB 4 U molecules have a seesaw shaped molecular geometry and are polar. SF 4 is an AB 4 U molecule lone pair occupies an equatorial position
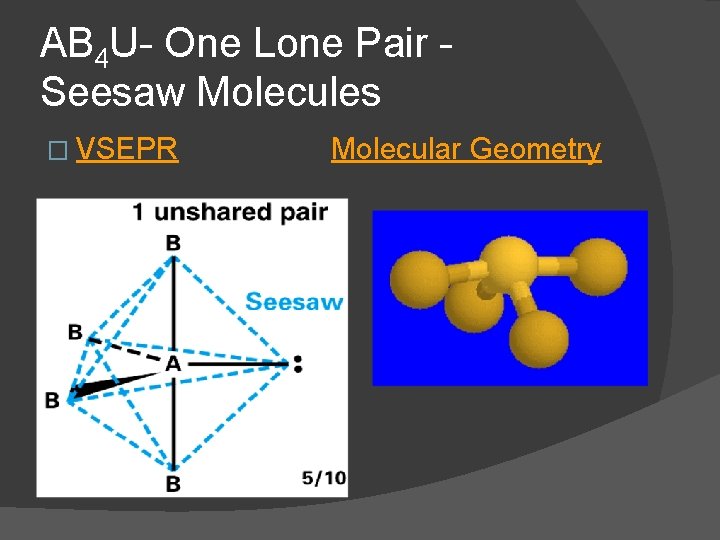
AB 4 U- One Lone Pair Seesaw Molecules � VSEPR Molecular Geometry

AB 3 U 2 - Two Lone Pairs T-shaped Molecules � For two lone pairs an AB 3 U 2 molecule results � AB 3 U 2 molecules have a T-shaped molecular geometry and are polar IF 3 is an AB 3 U 2 molecule two lone pairs occupy equatorial positions
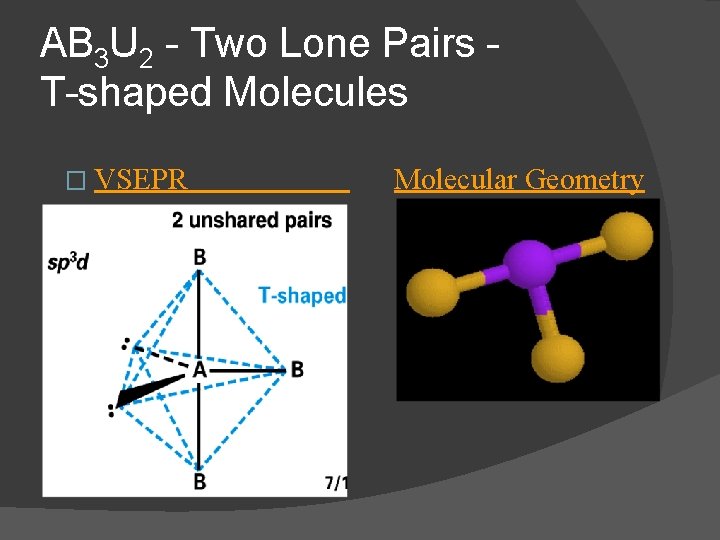
AB 3 U 2 - Two Lone Pairs T-shaped Molecules � VSEPR Molecular Geometry

AB 2 U 3 - Three Lone Pairs Linear Molecules � For three lone pairs an AB 2 U 3 molecule results � AB 2 U 3 molecules have a linear molecular geometry and are nonpolar Xe. F 2 is an AB 2 U 3 molecule three lone pairs occupy equatorial positions
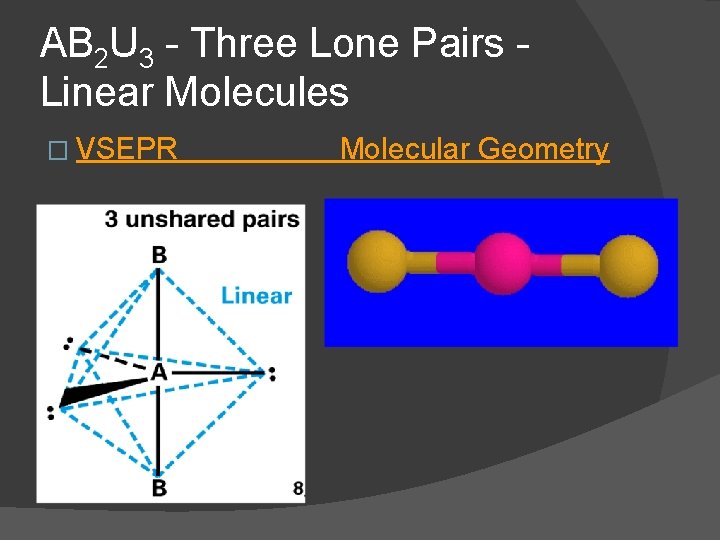
AB 2 U 3 - Three Lone Pairs Linear Molecules � VSEPR Molecular Geometry

AB 6 - No Lone Pairs Octahedral Molecules � Ex. SF 6, Se. F 6, SCl 6, etc. � These are octahedral and nonpolar molecules if all 6 substituents are the same � Dot Formula Electronic Geometry Molecular Geometry
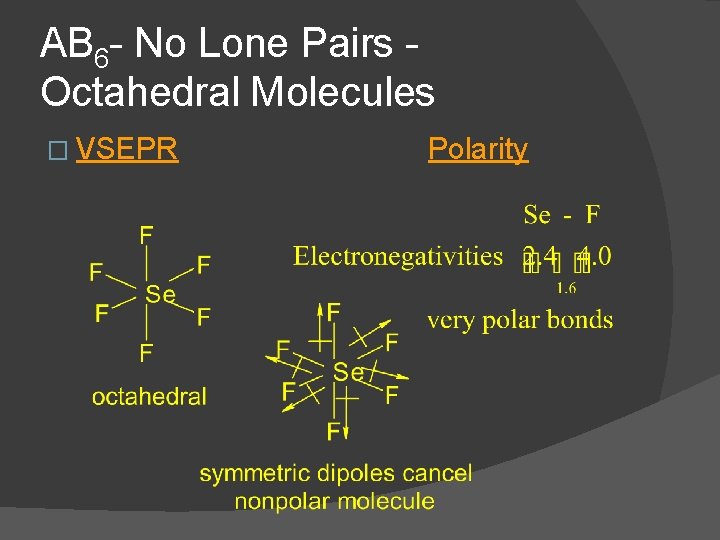
AB 6 - No Lone Pairs Octahedral Molecules � VSEPR Polarity
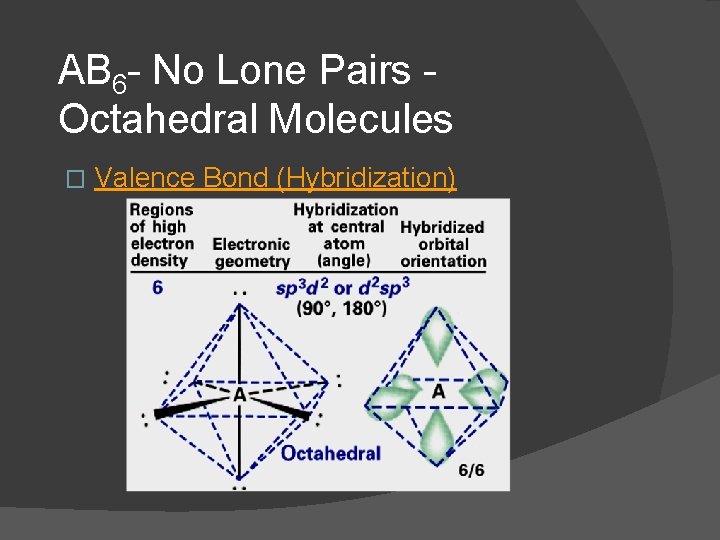
AB 6 - No Lone Pairs Octahedral Molecules � Valence Bond (Hybridization)

AB 5 U- One Lone Pair Square Pyramidal Molecules � For one lone pair an AB 5 U molecule results. � AB 5 U molecules have a square pyramidal molecular geometry and are polar. IF 5 is an AB 5 U molecule lone pair occupies an axial position
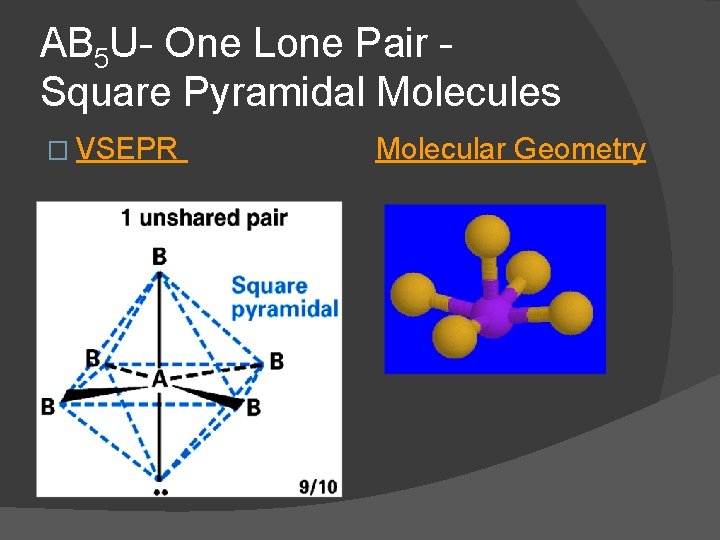
AB 5 U- One Lone Pair Square Pyramidal Molecules � VSEPR Molecular Geometry

AB 4 U 2 - Two Lone Pairs Square Planar Molecules � For two lone pairs an AB 4 U 2 molecule results. � AB 4 U 2 molecules have a square planar molecular geometry and are nonpolar. Xe. F 4 is an AB 4 U 2 molecule lone pairs occupy axial positions
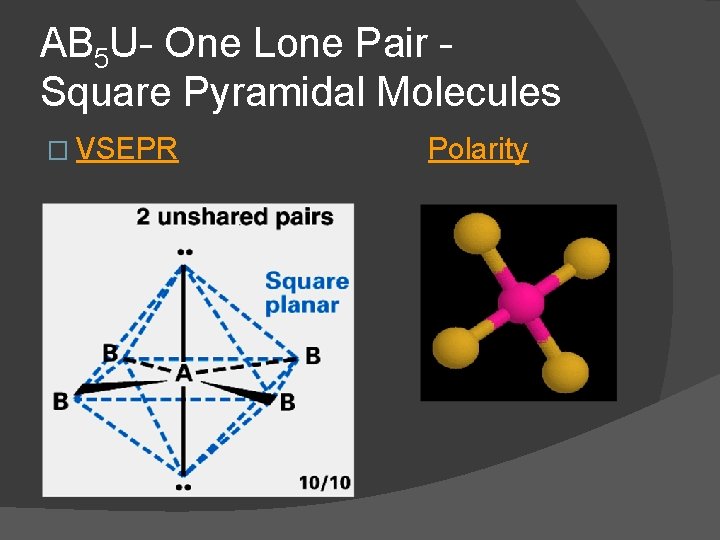
AB 5 U- One Lone Pair Square Pyramidal Molecules � VSEPR Polarity
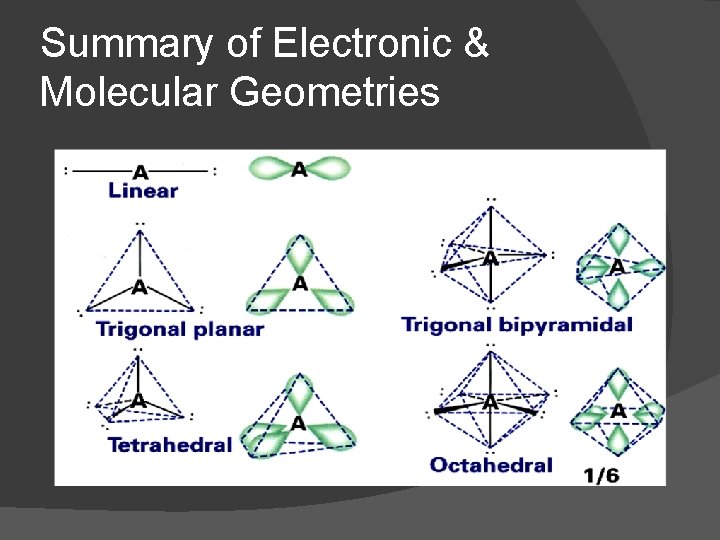
Summary of Electronic & Molecular Geometries
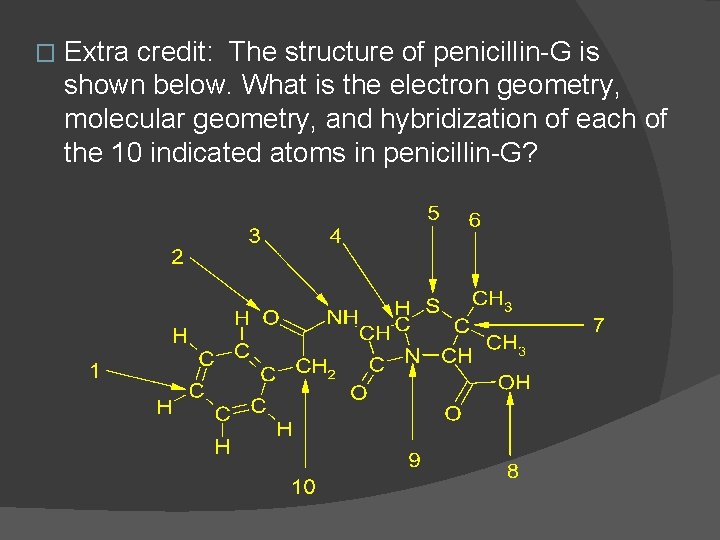
� Extra credit: The structure of penicillin-G is shown below. What is the electron geometry, molecular geometry, and hybridization of each of the 10 indicated atoms in penicillin-G?
- Slides: 52