Chapter 8 Molecular Shape Do we live in


















- Slides: 29

Chapter 8 “Molecular Shape” Do we live in a two dimensional world? How does molecular shape affect the properties of compounds in our world? T. Witherup 2006

OBJECTIVES l Identify the shapes of small molecules. l Describe and use the VSEPR theory. l Learn about hybrid orbitals. l Recognize the relationship between bond length and bond strengths. l Explain what determines the polarity of a molecule. l Explain why water is a polar molecule (a dipole).

8 -1 The Shape of Small Molecules l Just as architects use models to see how a building will look when finished, chemists use models to help visualize how atoms of molecules are arranged in space. ¡ l The shape of a molecule (molecular geometry) is described by the geometric figure formed when the atomic nuclei are imagined to be joined in straight lines. ¡ l Examples: Ball & stick, space filling, wire models. Examples: H----Cl, H----F, C----O, O----C----O The bond angle is the geometric angle between two adjacent bonds. ¡ Examples: H----Cl (180°); B----F angle in BF 3 (120°)

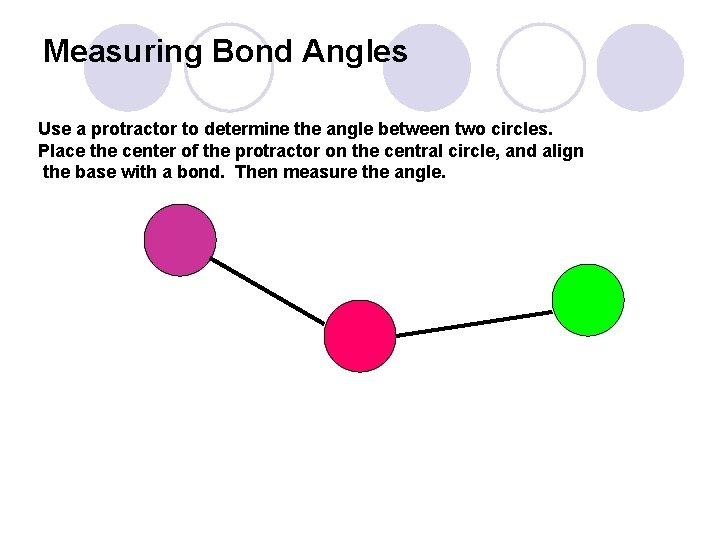
Measuring Bond Angles Use a protractor to determine the angle between two circles. Place the center of the protractor on the central circle, and align the base with a bond. Then measure the angle.

8 -1 The Shape of Small Molecules l Class Activity: In groups of two, follow the directions on “ 8 -1 Explore” with the following modification: ¡ l Use toothpicks in place of pipe cleaners. The purpose of this activity is to arrange the spheres around a central point so they are as far apart from each other as possible. ¡ ¡ Measure the angles carefully. This models the way atoms are arranged around a central atom.

Common Molecular Shapes l LINEAR (CO, HCl, CO 2) l TRIGONAL PLANAR (BCl 3, BF 3) l TETRAHEDRAL (CH 4, Si. Cl 4) l PYRAMIDAL (NH 3, PH 3) l BENT (H 2 O, H 2 S) l “T”-SHAPED (Cl. F 3) l SQUARE PLANAR (Xe. F 4) l TRIGONAL BIPYRAMIDAL (PF 5) l OCTAHEDRAL (SF 6)

VSEPR Theory l The VSEPR theory helps explain the shapes of simple molecules. l Valence Shell Electron Pair Repulsion (VSEPR) theory states that, in small molecules, the pairs of valence electrons are arranged as far apart from each other as possible (due to repulsion of electrons). l This results in geometries that minimize the energy of molecules.

VSEPR Theory: Electron Group Geometry & Molecular Geometry l Electron group geometry shows how groups of electrons are arranged around a central atom because of charge repulsion. l Molecular geometry shows how bonded atoms are arranged around that same central atom, again due to charge repulsion.

Examples of VSEPR Shapes (Balloons) (General Chemistry, Hill, Petrucci, Mc. Creary, Perry, 4 th Edition, Pearson Prentice Hall, 2005, p 389. ) 2 electron groups – linear 3 electron groups – trigonal planar 4 electron groups – tetrahedral 5 electron groups – trigonal bipyramidal 6 electron groups – octahedral (See textbook pages 256 -261. ) Let’s look at some examples of VSEPR Theory and structures at Georgia Southern University: http: //cost. georgiasouthern. edu/chemistry/general/molecule/vsepr. htm

Examples of VSEPR Shapes (Molecules) l Let’s take a look as a series of examples. ¡ ¡ l This link allows us to compare two similar structures: ¡ l Mark P. Heitz Slides (General Chemistry, 4 th Ed. , Hill, Petrucci, Mc. Creary & Perry, Chapter 10). Slides 6 – 13. http: //www. chem. purdue. edu/gchelp/vsepr/ The following link has a full tutorial about VSEPR Theory: ¡ http: //www. shef. ac. uk/chemistry/vsepr/

Bonding in Molecules l Remember that atomic orbital (quantum) theory was developed because the previous atomic theories were inadequate in explaining experimental data? l Likewise, atomic orbital theory (1 s, 2 p, etc. ) does not fully explain what happens to electrons in molecules, so quantum theory, too, needed to be modified. l This may be shown by examining the water molecule. ¡ ¡ ¡ Oxygen has six valence electrons in the 2 s 22 p 4 configuration. This means there are vacancies in two p-orbitals, each holding one electron that will bind with a hydrogen atom. Since all p-orbitals are 90° apart, we would predict water to have a right angle between its two hydrogen atoms. EXPERIMENTALLY WATER HAS A 104. 5° angle! We saw that VSEPR theory accounts for the observed shape of water, but it does not explain the bonding. Clearly something else must be occurring.

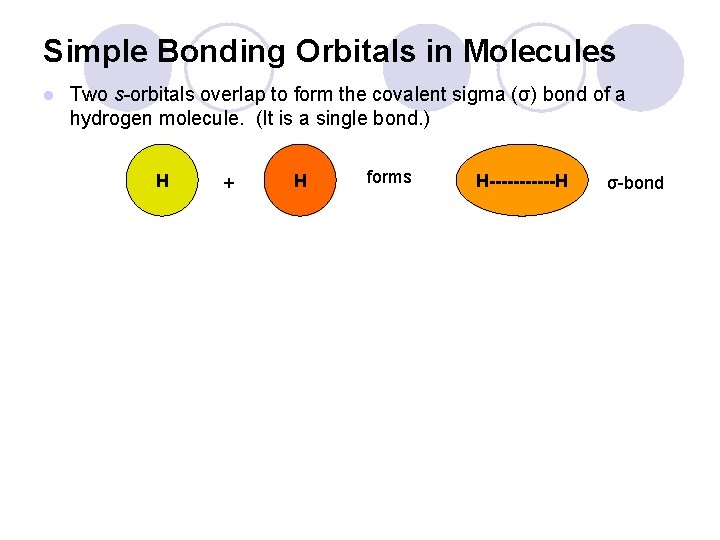
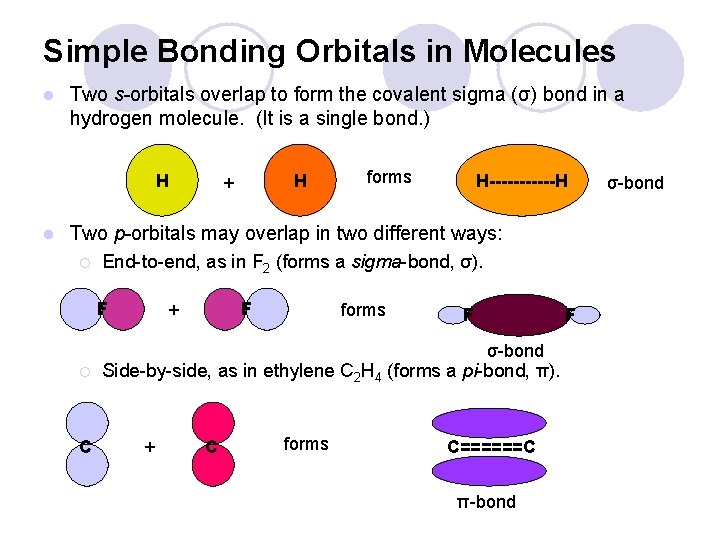
Simple Bonding Orbitals in Molecules l Two s-orbitals overlap to form the covalent sigma (σ) bond of a hydrogen molecule. (It is a single bond. ) H + H forms H------H σ-bond

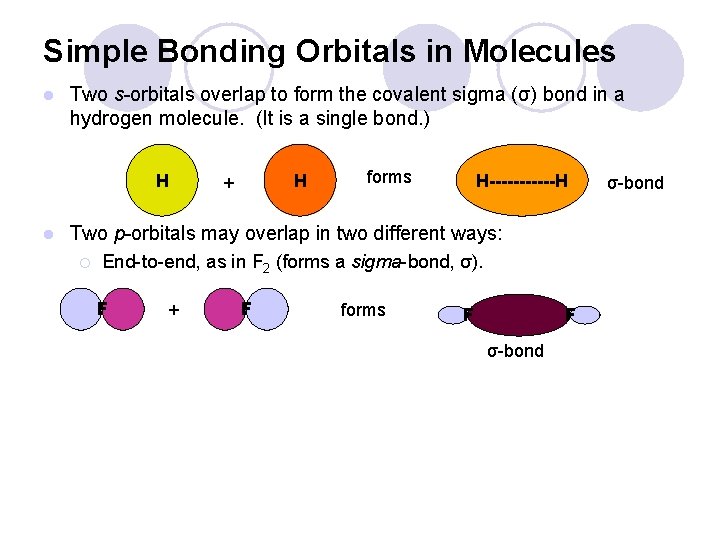
Simple Bonding Orbitals in Molecules l Two s-orbitals overlap to form the covalent sigma (σ) bond in a hydrogen molecule. (It is a single bond. ) H l H + forms H------H Two p-orbitals may overlap in two different ways: ¡ End-to-end, as in F 2 (forms a sigma-bond, σ). F + F forms F F σ-bond

Simple Bonding Orbitals in Molecules l Two s-orbitals overlap to form the covalent sigma (σ) bond in a hydrogen molecule. (It is a single bond. ) H l H + forms H------H Two p-orbitals may overlap in two different ways: ¡ End-to-end, as in F 2 (forms a sigma-bond, σ). F F + forms F F σ-bond ¡ C Side-by-side, as in ethylene C 2 H 4 (forms a pi-bond, π). + C forms C======C π-bond σ-bond

Bonding in Molecules - Hybridization l As two atoms approach to form a bond, their outer orbitals overlap and become changed (‘perturbed’). l Hybrid orbitals are used to describe this process, whereby the atomic orbitals mix together and have new properties. l Hybridization of atomic orbitals means that two or more orbitals may be mixed to form an equal number of identical ‘hybrid orbitals. ’ ¡ ¡ ¡ Consider what happens as you mix a gallon of yellow paint with a gallon of blue paint. What is the result? Two gallons of green paint! We may consider orbital mixing in a similar way, although the ‘mixing’ is really a mathematical solution to complex equations! To help us understand hybridization, we will look at some examples.

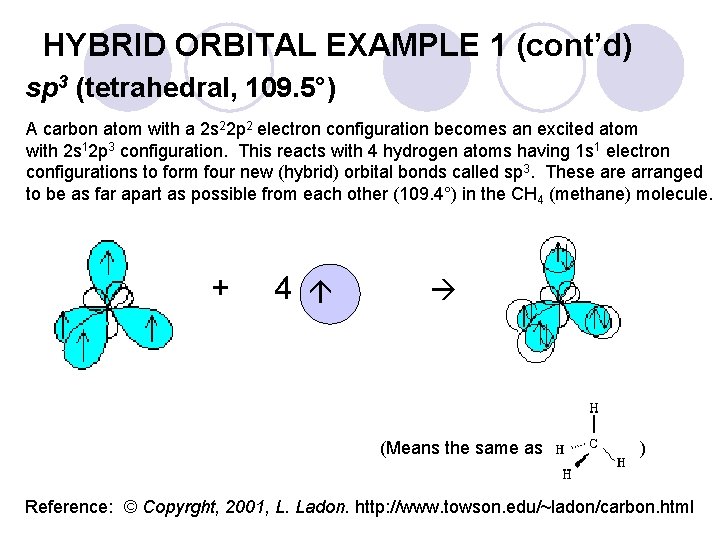
Hybrid Orbital Example 1 CH 4 is tetrahedral (by experiments), and VSEPR theory helps explain its shape, but let’s look at this in more detail. l Carbon has this electron configuration: l ¡ ¡ l C 1 s 2 2 p 2 Carbon should combine with TWO hydrogen atoms to make CH 2, not FOUR, as observed to make methane, CH 4. To explain this we may imagine that, as hydrogen atoms approach a carbon atom, the orbitals of carbon become mixed together in an ‘excited valence state’ of carbon. ¡ Carbon changes to C 1 s 2 2 s 1 2 p 3 or four equal 2 sp 3 valence orbitals, each containing only one electron, drawn as: C ¡ _ _ _ (four sp 3 orbitals) These electrons are free to pair (bond covalently in a σ-bond) with four hydrogen atoms by sharing, becoming: C ¡ _ _ _ (full sp 3 orbitals) To be as far apart as possible from each other, the hybrid orbitals and H atoms arrange as a tetrahedron around the carbon, as observed experimentally for CH 4.

HYBRID ORBITAL EXAMPLE 1 (cont’d) sp 3 (tetrahedral, 109. 5°) + 4 A carbon atom with a 2 s 22 p 2 electron configuration becomes an excited atom with 2 s 12 p 3 configuration. This reacts with 4 hydrogen atoms having 1 s 1 electron configurations to form four new (hybrid) orbital bonds called sp 3. These arranged to be as far apart as possible from each other (109. 4°) in the CH 4 (methane) molecule. (Means the same as ) Reference: © Copyrght, 2001, L. Ladon. http: //www. towson. edu/~ladon/carbon. html

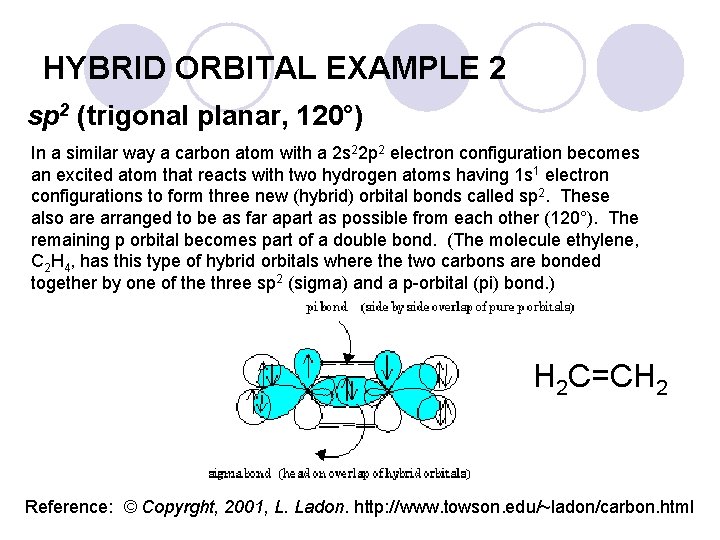
HYBRID ORBITAL EXAMPLE 2 sp 2 (trigonal planar, 120°) In a similar way a carbon atom with a 2 s 22 p 2 electron configuration becomes an excited atom that reacts with two hydrogen atoms having 1 s 1 electron configurations to form three new (hybrid) orbital bonds called sp 2. These also are arranged to be as far apart as possible from each other (120°). The remaining p orbital becomes part of a double bond. (The molecule ethylene, C 2 H 4, has this type of hybrid orbitals where the two carbons are bonded together by one of the three sp 2 (sigma) and a p-orbital (pi) bond. ) H 2 C=CH 2 Reference: © Copyrght, 2001, L. Ladon. http: //www. towson. edu/~ladon/carbon. html

HYBRID ORBITAL EXAMPLE 3 sp (linear, 180°) Also, a carbon atom with a 2 s 22 p 2 electron configuration becomes an excited atom that may react with a hydrogen atom having 1 s 1 electron configuration to form two new (hybrid) orbital bonds called sp. These also are arranged to be as far apart as possible from each other (180°). The remaining 2 p orbitals become part of a triple bond. (The molecule acetylene, C 2 H 2, has this type of hybrid orbitals where the two carbons are bonded together by a one sp (sigma) and two p-orbitals (pi). ) HCΞCH Reference: © Copyrght, 2001, L. Ladon. http: //www. towson. edu/~ladon/carbon. html

HYBRID ORBITALS of CARBON l sp 3 (tetrahedral, 109. 5°) (CH 4) ¡ l All single bonds (sigma, σ); end-to-end overlap. sp 2 (trigonal planar, 120°) (H 2 C=CH 2) Single bond skeleton (sigma, σ) (H-C & C-C). ¡ Double bond forms from the remaining p-orbital of carbon to form a pi bond (π); sideways overlap above & below the skeleton. ¡ l sp (linear, 180°) (H-CΞC-H) Single bond skeleton (sigma, σ) (H-C-C-H). ¡ Triple bond forms from the remaining two p-orbitals to form two pi bonds; sideways overlap is perpendicular to each other. ¡

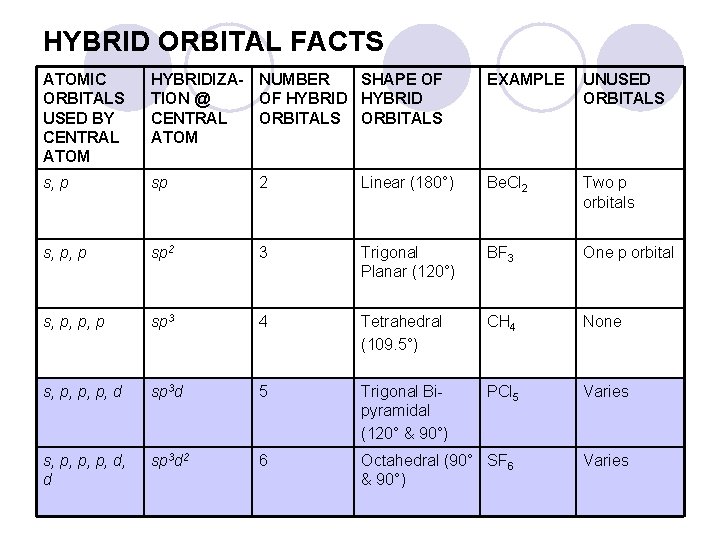
HYBRID ORBITAL FACTS ATOMIC ORBITALS USED BY CENTRAL ATOM HYBRIDIZA- NUMBER SHAPE OF TION @ OF HYBRID CENTRAL ORBITALS ATOM EXAMPLE UNUSED ORBITALS s, p sp 2 Linear (180°) Be. Cl 2 Two p orbitals s, p, p sp 2 3 Trigonal Planar (120°) BF 3 One p orbital s, p, p, p sp 3 4 Tetrahedral (109. 5°) CH 4 None s, p, p, p, d sp 3 d 5 Trigonal Bipyramidal (120° & 90°) PCl 5 Varies s, p, p, p, d, d sp 3 d 2 6 Octahedral (90° SF 6 & 90°) Varies

Examples of Molecules with Hybrid Orbitals (Let’s draw Lewis structures of these. ) l Ethane, C 2 H 6 l Benzene, C 6 H 6 l Formaldehyde, H 2 CO l Acetic acid, H 3 CCOOH l Propyne, H 3 CCCH

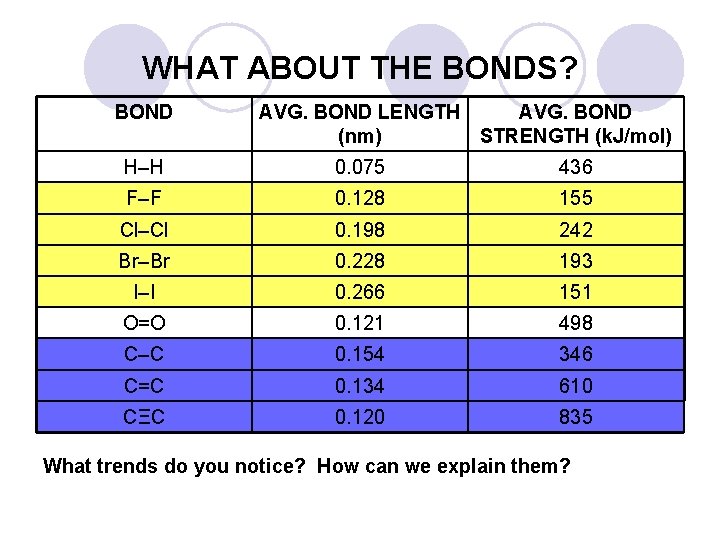
WHAT ABOUT THE BONDS? BOND AVG. BOND LENGTH AVG. BOND (nm) STRENGTH (k. J/mol) H–H 0. 075 436 F–F 0. 128 155 Cl–Cl 0. 198 242 Br–Br 0. 228 193 I–I 0. 266 151 O=O 0. 121 498 C–C 0. 154 346 C=C 0. 134 610 CΞC 0. 120 835 What trends do you notice? How can we explain them?

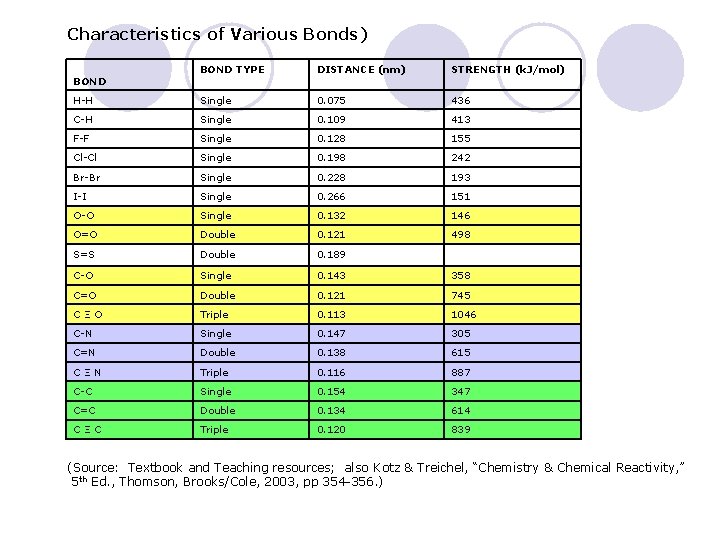
PROPERTIES OF BONDS In our models the bonds are all the same length, but this is NOT true in reality. l As one moves down a Group of the Periodic Table, the atoms form longer bonds. l ¡ l Atoms get larger moving down a Group. Multiple bonds are shorter and stronger than single bonds. The more electrons in a bond, the greater the attraction to the positive nuclei of a bond. ¡ Electrons act as the ‘electrical glue’ between the two nuclei. ¡ l Let’s look at some bond lengths in more detail. ¡ Look for the trends, and explain them.

Characteristics of Various Bonds) BOND TYPE DISTANCE (nm) STRENGTH (k. J/mol) H-H Single 0. 075 436 C-H Single 0. 109 413 F-F Single 0. 128 155 Cl-Cl Single 0. 198 242 Br-Br Single 0. 228 193 I-I Single 0. 266 151 O-O Single 0. 132 146 O=O Double 0. 121 498 S=S Double 0. 189 C-O Single 0. 143 358 C=O Double 0. 121 745 CΞO Triple 0. 113 1046 C-N Single 0. 147 305 C=N Double 0. 138 615 CΞN Triple 0. 116 887 C-C Single 0. 154 347 C=C Double 0. 134 614 CΞC Triple 0. 120 839 BOND (Source: Textbook and Teaching resources; also Kotz & Treichel, “Chemistry & Chemical Reactivity, ” 5 th Ed. , Thomson, Brooks/Cole, 2003, pp 354 -356. )

8 -2 POLARITY l Recall what we learned about polar & non-polar bonds. ¡ ¡ Are electrons shared equally in all bonds? What happens if they are not shared equally? See this link to find out: http: //cost. georgiasouthern. edu/chemistry/general/molecule /polar. htm Because of the polarity of bonds and how they are arranged around a central atom, molecules may also be polar or non-polar. l Dipole: a polar molecule, one that has a positive end a negative end. l What determines the polarity of a molecule? ¡ The shape of a molecule and the polarity of its bonds together determine if a molecule is polar or non-polar. l ¡ Let’s look at some examples.

POLARITY: EXAMPLES l Hydrogen Chloride: H Cl l Carbon Monoxide: C O l Carbon Dioxide: O C O l Formaldehyde: H 2 C O l Water: l Proteins: See http: //jmol. sourceforge. net/ ¡ H O H Arrows show the direction of negative charge in a bond, toward the most electronegative atom. Use δ+ and δ– to indicate the charges. Is water linear? Why or why not? (Go to the Jmol Demonstration Pages. )

Properties of Polar Molecules l Polar molecules align in an electric field. ¡ l Formaldehyde ¡ ¡ l ¡ Not polar because the effect of the two polar bonds cancel. Gas at room temperature due to lack of attraction between molecules; also soluble in water. Water ¡ ¡ l A dipole because of the imbalance of polar bonds. A gas, but very soluble in water. Carbon Dioxide ¡ l Similar to a compass aligning with Magnetic North. A dipole because the bent shape of the molecule does not cancel the polar bonds. The shape of the water molecule has a major impact on it’s properties, such as melting, boiling points, solubility and physical states at room temperature. Large Molecules ¡ ¡ Dipoles are critical to the functions of life. Proteins, DNA/RNA, carbohydrates, lipids, etc.

Did we cover the OBJECTIVES? Describe and use the VSEPR theory. l Identify the shapes of small molecules. l ¡ Linear, trigonal planar, tetrahedral, pyramidal, bent, Tshaped, square planar, trigonal bipyramidal, octahedral. Learn about hybrid orbitals. l Recognize the relationship between bond length and bond strengths. l Explain what determines the polarity of a molecule. l Explain why water is a polar molecule (a dipole). l