Chapter 8 Metabolism Metabolism Metabolism The totality of

Chapter 8: Metabolism

Metabolism • Metabolism: The totality of an organisms chemical reactions – Manages the material and energy resources of the cell – Begin with molecule and end with product • Catabolic pathway: – Release of energy by breakdown of complex molecules to simpler compounds – When digestive enzymes break down food • Anabolic pathway: – Consume energy to build complex molecules from simpler ones – Building muscle fibers from amino acids in response to exorcise

Complexity of Metabolic Pathways

Anabolic vs Catabolic

Energy • • Energy is the capacity to do work Objects in motion possess kinetic energy Objects at rest possess potential energy Chemical energy: potential energy stored in the form of chemical bonds

Energy • Kinetic energy is energy associated with motion • Heat (thermal energy) is kinetic energy associated with random movement of atoms or molecules • Potential energy is energy that matter possesses because of its location or structure • Chemical energy is potential energy available for release in a chemical reaction

Energy

Thermo Dynamics • The study of energy transformations that occur in matter • Closed systems verses open systems • Organisms are open systems • The two laws – NUMERO UNO: principal of conservation of energy – Energy can be transferred and transformed, but it cannot be created or destroyed – NUMERO DOS: • Every energy transformation or transfer increases the entropy (disorder) of the universe

Thermodynamics • During energy transfer energy is often lost in the form of heat, sound, friction, and so on. • Think of all the energy lost as a car accelerates

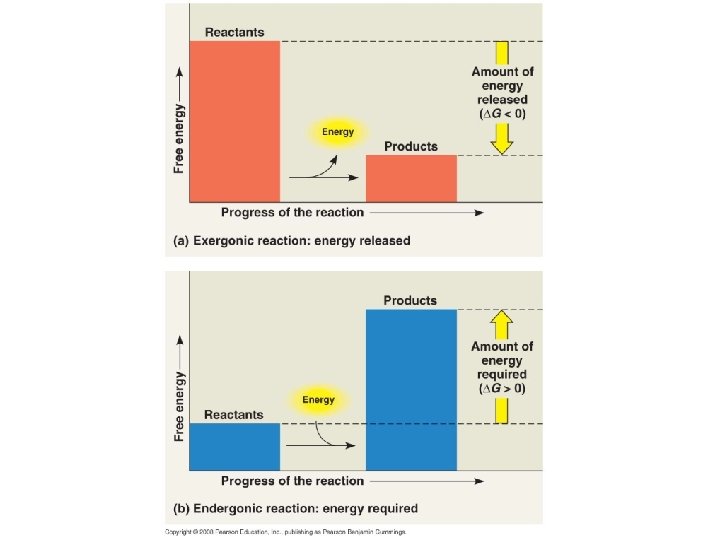
Free Energy • Free energy: the part of a systems energy that is able to perform work when the temperature of a system is uniform. – EXERgonic reaction: Energy is released • Occur spontaneously • Release free energy into the system – ENDERgonic reaction: Requires energy • Absorbs free energy from the system

• The change in free energy (∆G) during a process is related to the change in enthalpy, or change in total energy (∆H), change in entropy (∆S), and temperature in Kelvin (T): ∆G = ∆H – T∆S • Only processes with a negative ∆G are spontaneous • Spontaneous processes can be harnessed to perform work • Can use the free energy equation to determine if a reaction will occur spontaneously or not • Very important when studying metabolism and trying to determine which reactions will happen spontaneously.

Free Energy • Free energy • measure of a system’s instability, tendency to change to more stable state • During a spontaneous change, free energy decreases and stability of system increases • Equilibrium: state of max stability • A process is spontaneous and can perform work only when it is moving toward equilibrium
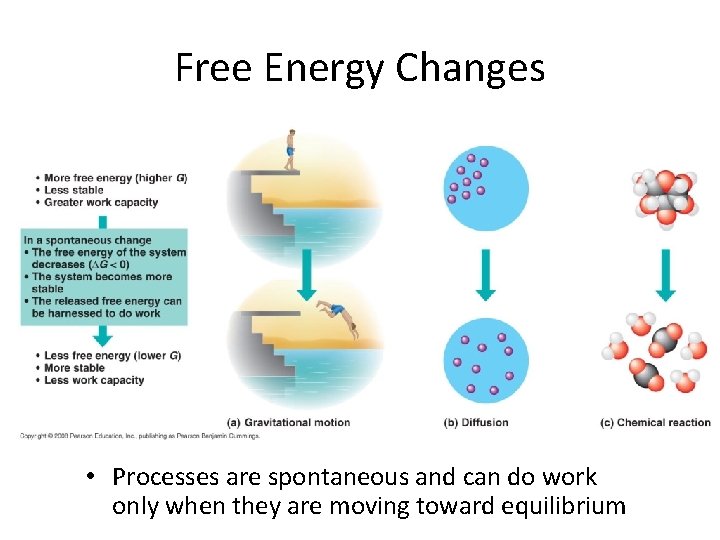
Free Energy Changes • Processes are spontaneous and can do work only when they are moving toward equilibrium
- Slides: 14