Chapter 8 Heat Transfer 8 1 Kinetic Molecular












- Slides: 14

Chapter 8 & Heat Transfer

8. 1 Kinetic Molecular Theory n n All matter is made up of atoms or molecules (groups of atoms). These can be considered the “fundamental particles” of matter. The degree to which these particles are bound together by attractive forces determines the state of the matter.

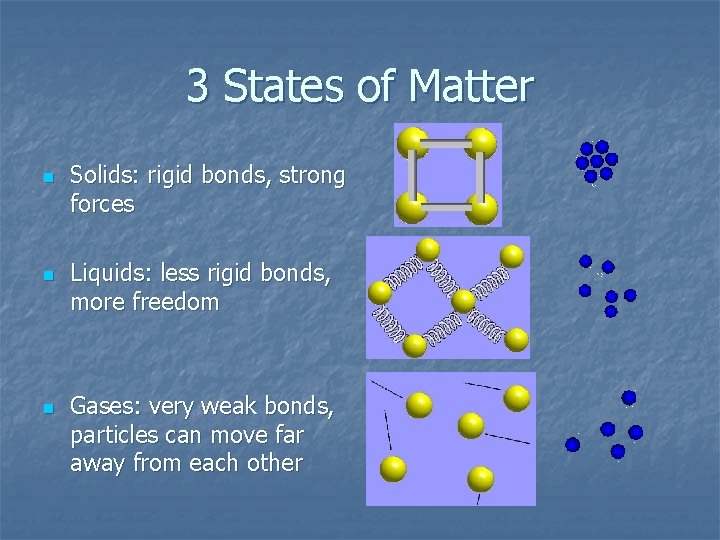
3 States of Matter n n n Solids: rigid bonds, strong forces Liquids: less rigid bonds, more freedom Gases: very weak bonds, particles can move far away from each other

Temperature vs. Heat n n n Particles that are in random motion have kinetic energy The average kinetic energy of particles in a sample of matter is the temperature of the sample The total kinetic energy of ALL the particles in a sample of matter is the heat energy of the sample.

Which object is at a higher temperature?

Temperature Scales n A sample has zero average kinetic energy (all particles are stopped) at -273 o C (celsius) or 0 o K (kelvin) {also known as absolute zero} n n Water freezes (turns into solid) at 0 o C or +273 o. K Water boils (turns into gas) at 100 o C or +373 o. K

8. 2 Thermal Expansion n n As matter gains heat energy, particles speed up and the average distance between particles increase. The result: its volume e x p a n d s e. g. ; as the liquid in a thermometer is heated, thermal expansion causes the volume to increase. The result: the liquid rises up the glass tube.

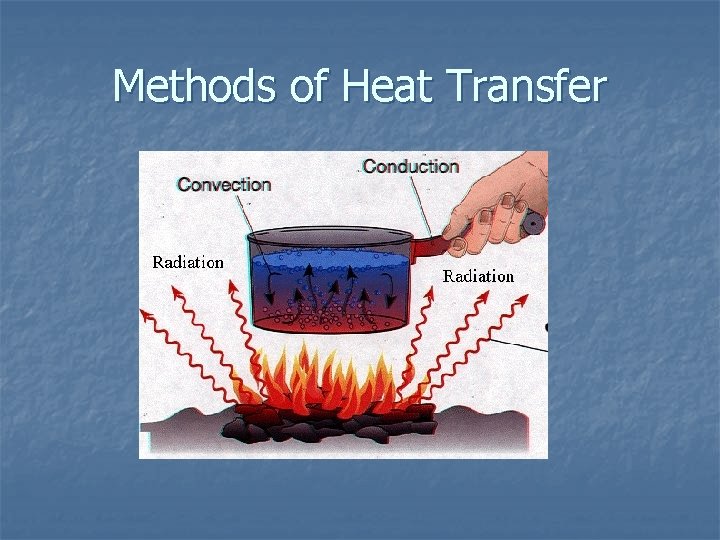
8. 3 Heat: Thermal Energy Transfer n Conduction: Heat transfer by particle collisions in a solid. Heat conductor example: silver, copper (metals) Heat insulator example: cork, air (see table 8. 3 on page 256 in textbook) n Convection: Heat transfer in liquids & gases by a circulating path of fluid (water cycle) (see figure 8. 11 on page 257 in textbook)

n Radiation: electromagnetic waves travel through space. When these heat waves (infared radiation) hit matter, they may be reflected, transmitted or absorbed. If absorbed, the particles in the matter may increase their movement and the overall average kinetic energy (temperature) increases.

Methods of Heat Transfer

Methods of Heat Transfer

8. 4 Specific Heat Capacity n Heat transfer depends on 3 things: 1. Temperature Difference 2. Mass of substance 3. Type of substance

n n n Each type of substance has its own property with respect to heat transfer, called its specific heat capacity. Definition: Specific heat capacity is the amount of energy that is needed to increase the temperature of 1 kg of a particular substance by 1 o C Specific heat capacity is measured in [J/kg. C]

Mathematical Definition of Heat Capacity n Mathematical definition: n ∆EH = mc∆T ∆EH : heat energy transferred [ J ] c: specific heat capacity [ J/kgo C ] m: mass [kg] ∆T: =change in temperature [o C]