Chapter 8 Covalent Bonds Section 8 1 Matter

Chapter 8 Covalent Bonds Section 8. 1
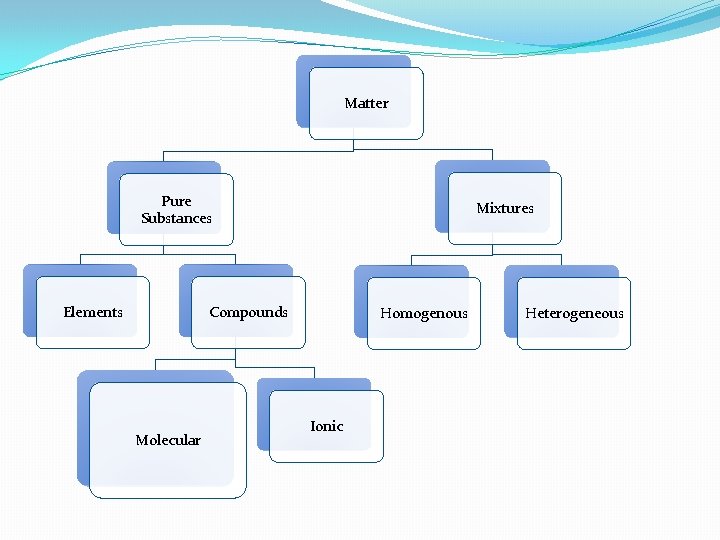
Matter Pure Substances Elements Mixtures Compounds Molecular Homogenous Ionic Heterogeneous

Common Molecules �www. reciprocalnet. org/edumodules/commonmolecul es/element/list. html#simple

Molecules and Molecular compounds �Covalent bond- atoms held together by the sharing of electrons �What is the difference between an ionic compound a molecular compound?

Examine this picture of Na. Cl in H 2 O

�Ionic compounds are made of + and – charged ions arranged in repeating 3 -D patterns ex: Na. Cl �Molecular compound is made of elements chemically combined ex: H 2 O �Molecular compounds have lower melting and boiling points than ionic compounds

Molecular formulas �Molecular formula-chemical formula of a molecular compound that shows how many atoms of each element are in a molecule �NOTE: this reflects actual # of atoms-not whole number ratio �Ex: H 2 O �Subscript (2) after the H indicates there are 2 hydrogen atoms and one oxygen atom How many carbon and oxygen atoms are in a molecule of CO 2?

�Chocolate chip cookie recipe: � 2 eggs � 1 cup sugar � 1 cup brown sugar � 2 cups chocolate chips � 1 tsp vanilla � 2 cups flour � If I change the recipe-add peanut butter chips instead of chocolate chips, the cookies are different � In a molecular formula, the subscripts tell you the number of atoms of each element in a compound
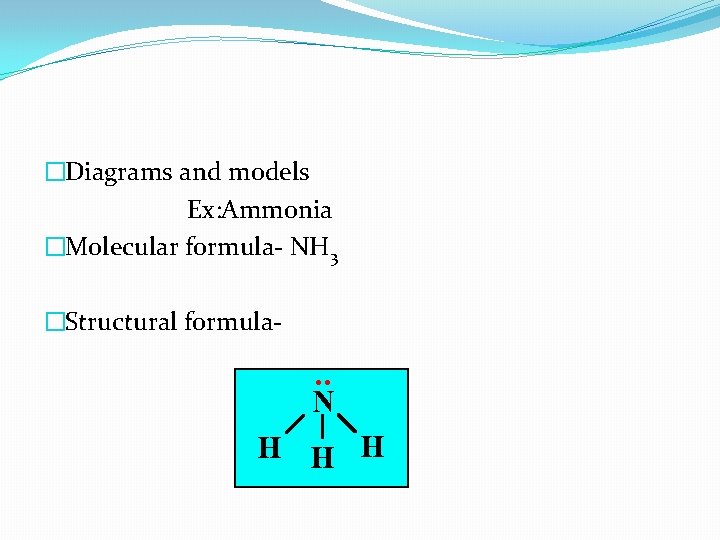
�Diagrams and models Ex: Ammonia �Molecular formula- NH 3 �Structural formula-
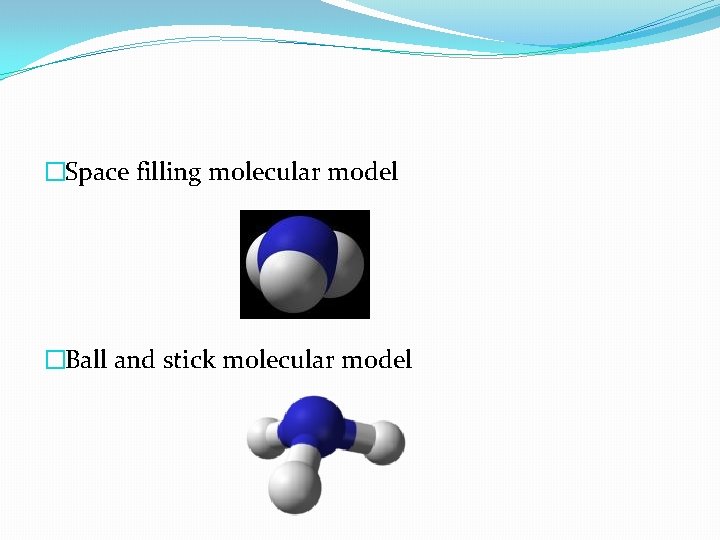
�Space filling molecular model �Ball and stick molecular model
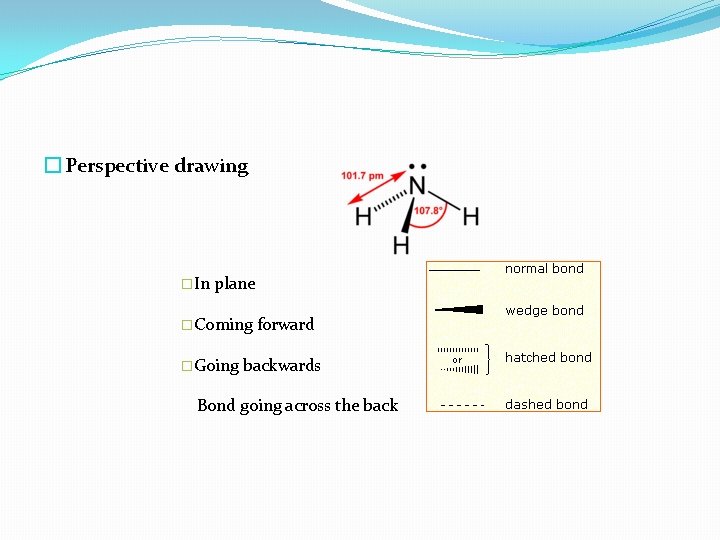
� Perspective drawing �In plane �Coming �Going forward backwards Bond going across the back

�Molecule-a neutral group of atoms joined together by covalent bonds �Diatomic molecule- a molecule consisting of two of the same atoms

�Carbon is black, oxygen is red…. �What is the formula of this molecule?
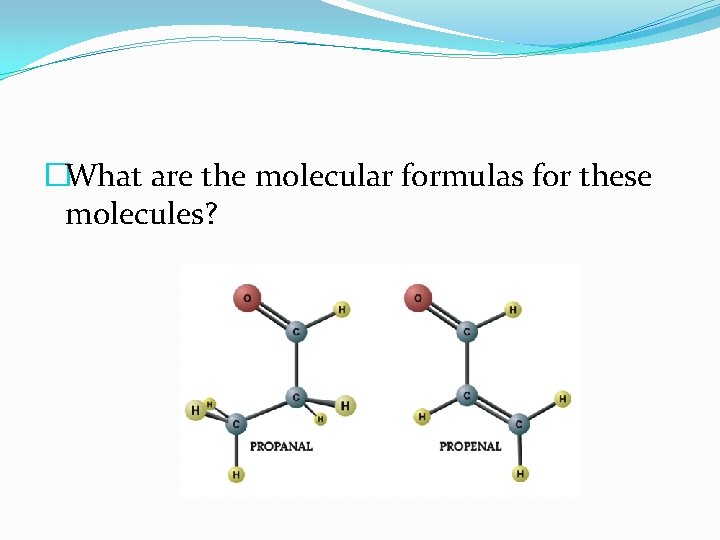
�What are the molecular formulas for these molecules?

TRUE OR FALSE: �All compounds contain molecules.

�FALSE

�There are no elements that exist as molecules.

�FALSE

�Most molecular compounds are composed of two or more nonmetallic elements.

�TRUE

�Atoms in molecular compounds share electrons.

�TRUE

�Molecular compounds tend to have higher melting and boiling points than ionic compounds

�FALSE

�Which of the following formulas represent molecules? �Br 2 �KBr �CH 4 �SO 3 �N 2 H 2 �Ne 2

�Answer: Br 2, CH 4, SO 3, N 2 H 2

�Which of the following formulas represent compounds? �Br 2 �KBr �CH 4 �SO 3 �N 2 H 2 �Ne 2

�Answer: KBr, CH 4, SO 3, N 2 H 2
- Slides: 28