Chapter 8 Covalent Bonding The Nature of Covalent

Chapter 8: Covalent Bonding The Nature of Covalent Bonding (Part 1)

The Octet Rule in Covalent Bonding l In ionic bonding, electrons are transferred so that each ion reaches the noble gas configuration. l In covalent bonds, electron sharing usually occurs so that atoms attain the electron configurations of noble gases.

The Octet Rule in Covalent Bonding l Example: ¡ Each hydrogen atom has one electron. ¡ A pair of hydrogen atoms share these two electrons when they form a covalent bond in the hydrogen molecule. ¡ Thus, each hydrogen atom attains the electron configuration of helium, a noble gas with two electrons.

Single Covalent Bonds l The hydrogen atoms in a hydrogen molecule are held together mainly by the attraction of the shared electrons to the positive nuclei. l Two atoms held together by sharing a pair of electrons are joined by a single covalent bond.

Single Covalent Bond l Example: H· + H· → H: H Hydrogen Atom Hydrogen Molecule Shared pair of electrons

Single Covalent Bond l An electron dot structure such as H: H represents the shared pair of electrons of the covalent bond by two dots. . : F·. . + ·F: . . → : F: F: . . Flourine Atom Flourine Molecule
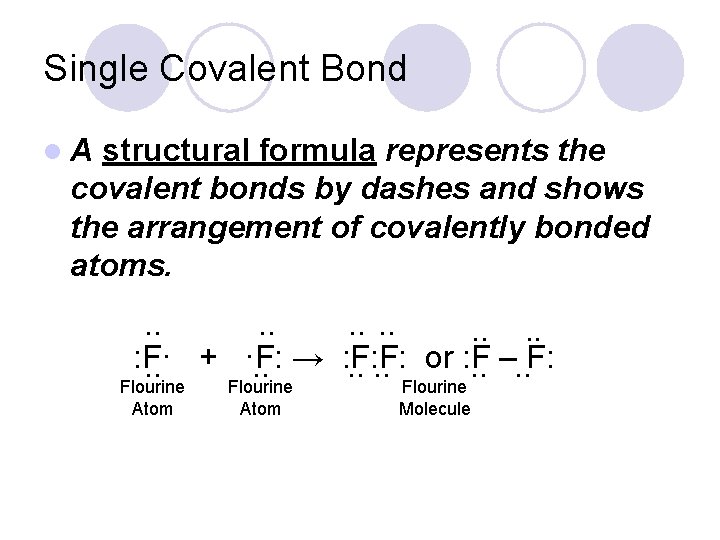
Single Covalent Bond l. A structural formula represents the covalent bonds by dashes and shows the arrangement of covalently bonded atoms. . . : F·. . + ·F: . . → : F: F: . . or : F. . –. . F: Flourine Atom Flourine Molecule

Single Covalent Bond l. A pair of valence electrons that are not shared between atoms is called an unshared pair. – . . . 2 H· + : O· → : O: H or : O – H. . . Hydrogen Oxygen Atoms H H Atom Water Molecule

Checkpoint l What does a structural formula represent? l What is the electron dot structure of a methane molecule? l Complete page 220. Practice Problems 7 & 8 on

Double & Triple Covalent Bonds l Atoms form double or triple covalent bonds if they can attain a noble gas structure by sharing two pairs or three pairs of electrons.

Double & Triple Covalent Bonds l. A bond that involves two shared pairs of electrons is a double covalent bond. l A bond formed by sharing three pairs of electrons is a triple covalent bond. . . . : O: + : O: → O: : O or. . . Oxygen Atoms Oxygen Molecule . . . O=O. . .

8. 2 Section Assessment l Complete questions 13, 14, 15, 20, 21.
- Slides: 12