Chapter 8 Covalent Bonding Jennie L Borders Section
Chapter 8 – Covalent Bonding Jennie L. Borders
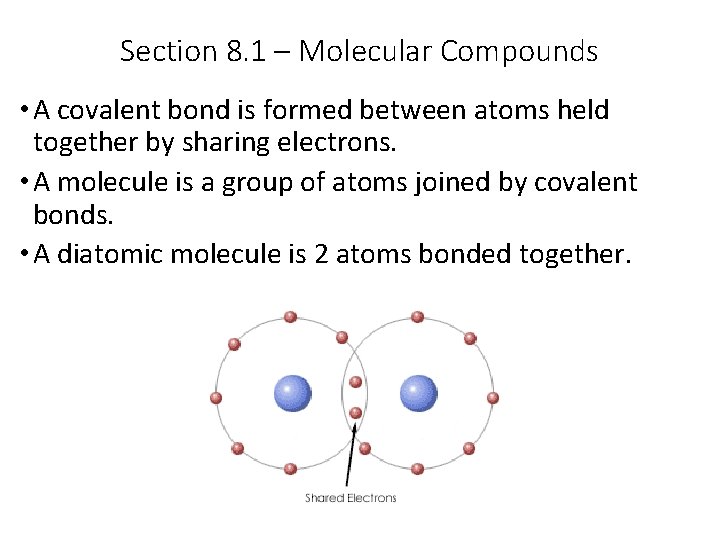
Section 8. 1 – Molecular Compounds • A covalent bond is formed between atoms held together by sharing electrons. • A molecule is a group of atoms joined by covalent bonds. • A diatomic molecule is 2 atoms bonded together.
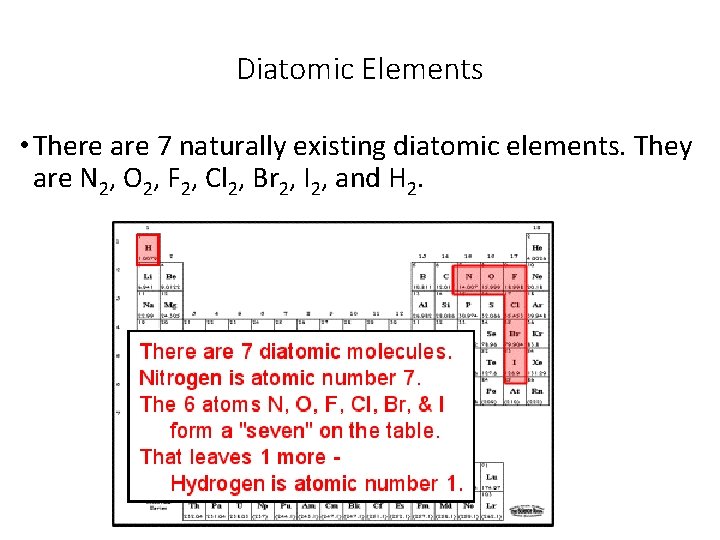
Diatomic Elements • There are 7 naturally existing diatomic elements. They are N 2, O 2, F 2, Cl 2, Br 2, I 2, and H 2.

Properties of Molecular Compounds • Low melting points • Tend to be gases or liquids • Made of nonmetals • Made of covalent bonds • Poor conductors Ionic Compounds high melting points crystalline solids metal and nonmetal ionic bonds conductor when molten or aqueous

Molecular Compounds • When covalent bonding is dominant, most compounds exist as molecules, have low melting and boiling points, and exhibit nonelectrolyte behavior. • When ionic bonding is dominant, most compounds are brittle solids, have a crystal lattice structure, have high melting points, and exhibit strong electrolyte behavior.

Ionic or Covalent? • A simple approach to decide whether a bond is ionic or covalent is to assume that a bond between a metal and a nonmetal is ionic and a bond between two nonmetals is covalent. • The problem with this approach is that there are many exceptions. Ex: Sn. Cl 4 = metal and nonmetal = covalent

Ionic or Covalent? • Another approach to tell if a bond is ionic or covalent is to compare the electronegativity difference between the two bonded atoms. Bond Ionic Polar Nonpolar Electronegativity Difference >2 0. 5 – 2. 0 >0. 5

Transition Metals • The problem with this method is that there is no difference in the electronegativity values for the transition metals that have multiple charges. • The rule of thumb is that the higher the charge (+4 or higher) on a transition metal, the more covalent the bond.

Molecular Formulas • A molecular formula is the chemical formula of a molecular compound. • A molecular formula shows how many atoms of each element a molecule contains. • A molecular formula shows the actual number of atoms while a formula unit shows the lowest wholenumber ratio of ions.

Section 8. 1 Assessment 1. How are the melting points and boiling points of molecular compounds usually different from those of ionic compounds? 2. What information does a molecular formula provide? 3. What are the only elements that exist in nature as uncombined atoms? What term is used to describe such elements? 4. Describe how the molecule whose formula is NO is different from the molecule whose formula is N 2 O.

Section 8. 1 Assessment 5. Give an example of a diatomic molecule found in Earth’s atmosphere.

Section 8. 2 – The Nature of Covalent Bonding • In ionic bonding, atoms transfer electrons to achieve noble gas configuration. • In covalent bonding, atoms share electrons to achieve noble gas configuration. • Most atoms share electrons until they have a total of 8 valence electrons (octet rule). However, hydrogen only needs 2 electrons to be stable.
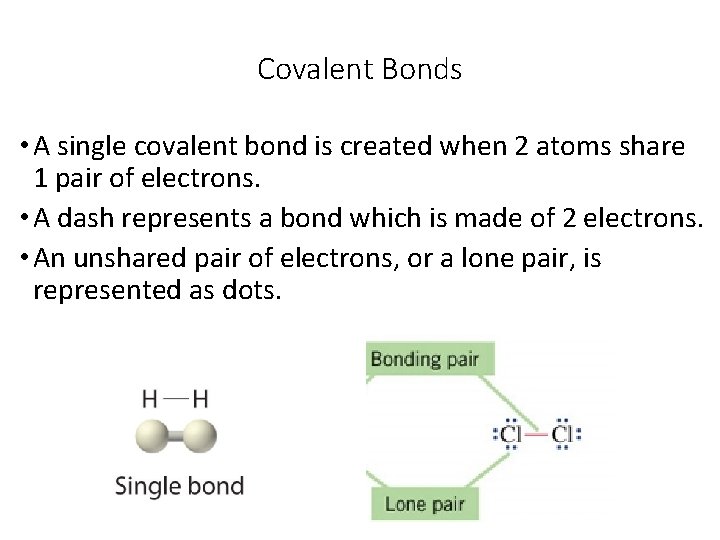
Covalent Bonds • A single covalent bond is created when 2 atoms share 1 pair of electrons. • A dash represents a bond which is made of 2 electrons. • An unshared pair of electrons, or a lone pair, is represented as dots.

Sample Exercise • Given the Lewis symbols for the elements nitrogen and fluorine, predict the stable binary compound (a compound composed of two elements) formed when nitrogen reacts with fluorine, and draw its Lewis structure.

Practice Exercise • Compare the Lewis symbol for neon with the Lewis structure for methane, CH 4. In what important way are the electron arrangements about neon and carbon alike? In what important respect are they different?

Rules for Writing Lewis Dot Structures 1. Add up the total number of valence electrons. 2. Bond the atoms with single bonds. (Single atoms go in the middle. ) 3. Add electrons until each atom has a full octet and each hydrogen has a duet (2 electrons). 4. Add up total valence electrons in Lewis dot structure and compare to the total from step 1.

Sample Problems • Write the Lewis dot structure for the following: • H 2 • Cl 2 • H 2 O • C 2 H 6

Practice Problems • Write the Lewis dot structure for the following: • Br 2 • CH 2 Cl 2 (hint: C is in the middle) • NH 3
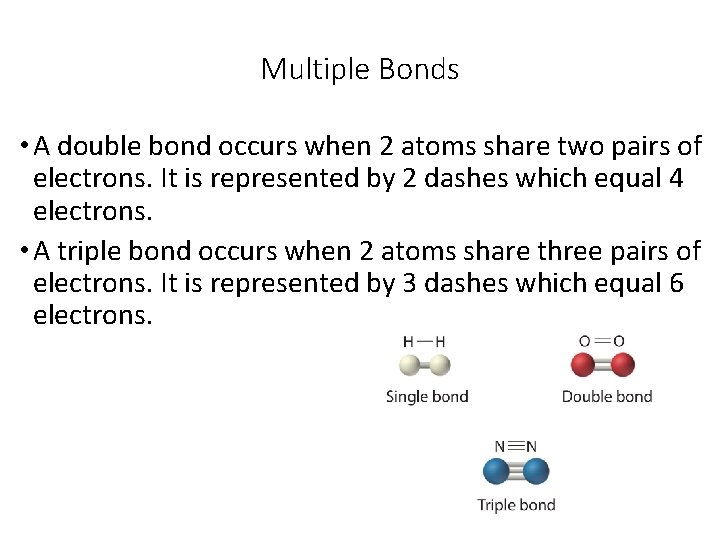
Multiple Bonds • A double bond occurs when 2 atoms share two pairs of electrons. It is represented by 2 dashes which equal 4 electrons. • A triple bond occurs when 2 atoms share three pairs of electrons. It is represented by 3 dashes which equal 6 electrons.

Lewis Dot Structures with Multiple Bonds • When writing the Lewis dot structures, following the 4 steps we learned. • When you add up the total number of electrons in your Lewis dot structure, sometimes it will not equal the total from step 1. • For every extra electron pair you have, you need to add 1 more bond in your structure.

Sample Problem • Write the Lewis dot structure for the following: • O 2 • CO 2

Practice Problems • Write the Lewis dot structures for the following: • N 2 • HCN • C 2 H 4
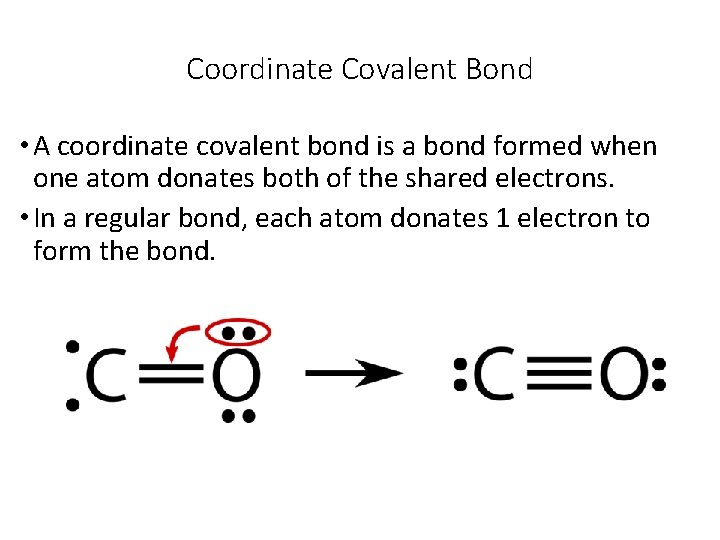
Coordinate Covalent Bond • A coordinate covalent bond is a bond formed when one atom donates both of the shared electrons. • In a regular bond, each atom donates 1 electron to form the bond.

Polyatomic Ions • When writing the Lewis dot structure for a polyatomic ion, you have to take into account the charge when you add up the number of valence electrons in step 1. • After you draw the Lewis dot structure, you have to put the whole structure in brackets and write the charge.

Sample Problems • Write the Lewis dot structure for the following: • Br. O 3 - • OH-

Practice Problems • Write the Lewis dot structure for the following: • PO 4 -3 • NO+ • CN-
Bond Dissociation Energy • The bond dissociation energy is the energy needed to break a bond. • As the number of bonds increases, the bond dissociation energy increases. Single < Double < Triple
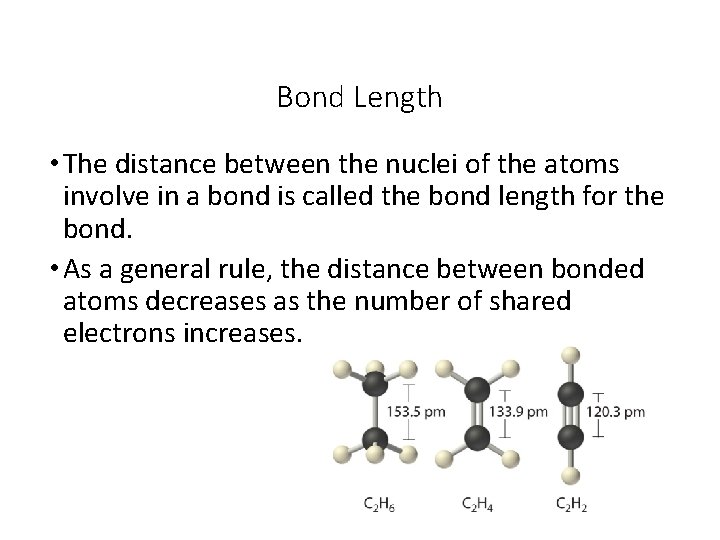
Bond Length • The distance between the nuclei of the atoms involve in a bond is called the bond length for the bond. • As a general rule, the distance between bonded atoms decreases as the number of shared electrons increases.
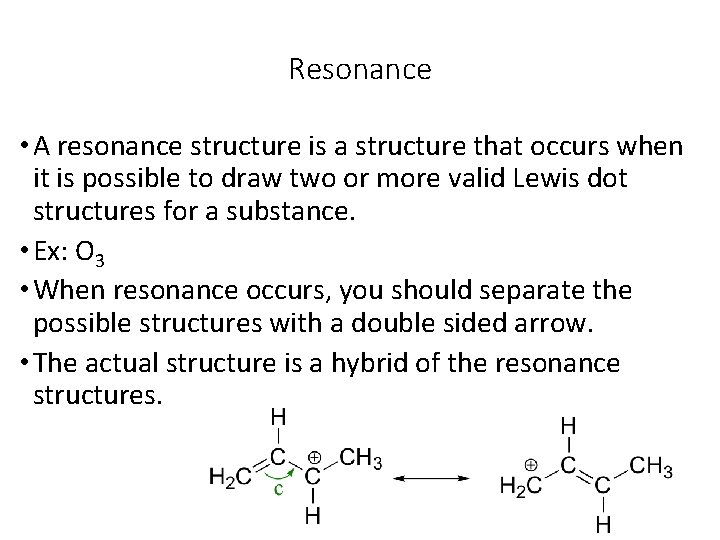
Resonance • A resonance structure is a structure that occurs when it is possible to draw two or more valid Lewis dot structures for a substance. • Ex: O 3 • When resonance occurs, you should separate the possible structures with a double sided arrow. • The actual structure is a hybrid of the resonance structures.

Sample Problems • Write the Lewis dot structures for the following: • O 3 • Which is predicted to have the shorter sulfur-oxygen bonds, SO 3 or SO 32 -?

Practice Problems • Write the Lewis dot structures for the following: • NO 3 - • HCO 2 -

Formal Charge • The formal charge of an atom in a molecule is the charge the atom would have if all the atoms in the molecule had the same electronegativity. (**Remember that formal charges are not real charges!) Formal charge = valence electrons – electrons in Lewis structure

Formal Charge • When more than one Lewis structure can be drawn for a molecule, formal charges can be used to pick the most accepted structure. 1. We generally choose the Lewis structure in which the atoms bear formal charges closest to zero. 2. We generally choose the Lewis structure in which any negative charges reside on the more electronegative atoms.

Sample Exercise • The following are three possible Lewis structures for the thiocyanate ion, NCS-: a. Determine the formal charges of the atoms in each structure. . . [: N – C ≡ S: ]. . . [N = C = S]. . . [: N ≡ C –. . S: ]- b. Which is the preferred structure?

Practice Exercise The cyanate ion (NCO-) has three possible Lewis structures. Draw all three Lewis structures, label the formal charges on the atoms, and indicate the preferred structure.

Exceptions to the Octet Rule • Some molecules are exceptions to the octet rule. • ODD NUMBER - Some atoms have an odd number of electrons. This usually occurs with nitrogen. The odd electron goes to the central atom. • LESS THAN 8 – Some atoms have less than 8 electrons. This usually happens with elements 1 -5. Be will have 4 e-, and B will have 6 e-. • MORE THAN 8 – Some atoms have more than 8 electrons. This usually happens with S, P, the halogens, and some noble gases.

Sample Problem • Write the Lewis dot structure for the following: • NO 2 • BF 3 • ICl 4 -

Practice Problem • Write the Lewis dot structure for the following: • Be. Cl 2 • Xe. F 2 • Cl. O 2

Strengths of Covalent Bonds • The stability of a molecule is related to the strengths of the covalent bonds it contains. • The strength of a covalent bond between two atoms is determined by the energy required to break that bond. • The bond enthalpy is the enthalpy change, DH, for the breaking of a particular bond in one mole of a gaseous substance.

Bond Enthalpy • We use the designation D(bond type) to represent the bond enthalpies. • When molecules contain multiple identical bonds, then we use the average bond enthalpy.

Bond Enthalpy • The bond enthalpy is always a positive quantity; energy is always required to break chemical bonds. • The greater the bond enthalpy is, the stronger the bond. • A molecule with strong chemical bonds generally has less tendency to undergo chemical change than does one with weak bonds.

Bond Enthalpy and Reactions • When calculating the DH of a reaction using bond enthalpies you use the following equation: DHrxn = S(bonds broken) – S(bonds formed)

Sample Exercise • Using Table 8. 4, estimate DH for the following reaction: H H I I H – C – H(g) + 7/2 O 2(g) 2 O = C = O(g) + 3 H – O – H(g) I I H H

Practice Exercise • Using Table 8. 4, estimate DH for the following reaction: H – N – H(g) N ≡ N(g) + 2 H – H(g) I I H H

Bond Enthalpy and Bond Length • As the number of bonds between the carbon atom increases, the bond enthalpy increases and the bond length decreases; that is, the carbon atoms are held more closely and more tightly together. • In general, as the number of bonds between two atoms increases, the bond grows shorter and stronger.

Sample Integrative Exercise Phosgene has the following elemental composition: 12. 41% C, 16. 17% O, and 71. 69% Cl by mass. Its molar mass is 98. 9 g/mol. b. Draw three Lewis structures for the molecule that satisfy the octet rule for each atom.

Sample Integrative Exercise c. Using formal charges, determine which Lewis structure is the most important one.

Sample Integrative Exercise d. Using average bond enthalpies, estimate DH for the formation of gaseous phosgene from CO(g) and Cl 2(g).

Section 8. 2 Assessment 1. What electron configurations do atoms usually achieve by sharing electrons to form covalent bonds? 2. When are two atoms likely to form a double bond between them? A triple bond? 3. How is a coordinate covalent bond different from other covalent bonds? 4. How is the strength of a covalent bond related to its bond dissociation energy?

Section 8. 2 Assessment 5. What kinds of information does a structural formula reveal about the compound it represents? 6. Draw the electron dot structures for the following molecules. a. H 2 S b. PH 3 c. Cl. F
Section 8. 3 – Bonding Theories • The VSEPR (valence shell electron pair repulsion) theory states that the repulsion between electron pairs causes molecular shapes to adjust so that the valence electron pairs stay as far apart as possible. • The overall shape of a molecule is determined by its bond angles. • Lone pair electrons alter the shape more than bonding electrons due to the fact that they spread out more.

VSEPR 5 Basic Shapes In general, each nonbonding pair, single bond, or multiple bond produces an electron domain around the central atom.
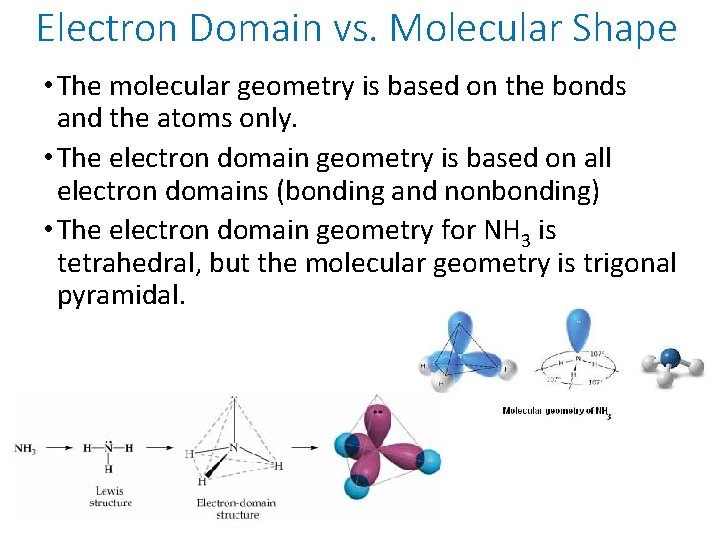
Electron Domain vs. Molecular Shape • The molecular geometry is based on the bonds and the atoms only. • The electron domain geometry is based on all electron domains (bonding and nonbonding) • The electron domain geometry for NH 3 is tetrahedral, but the molecular geometry is trigonal pyramidal.
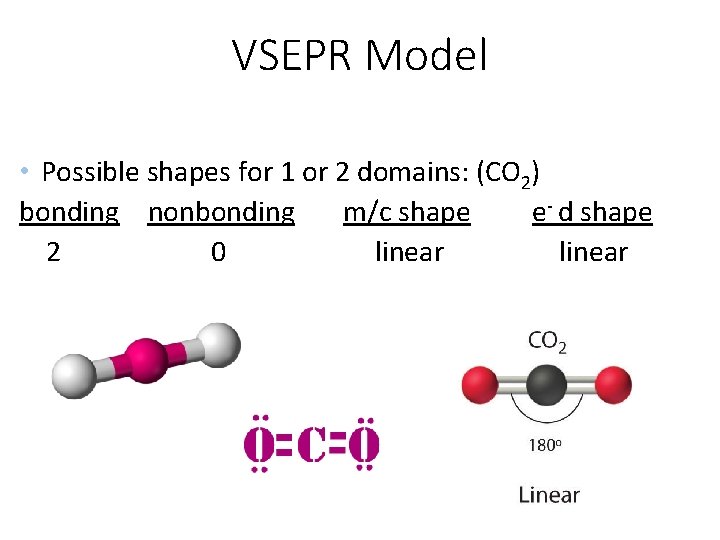
VSEPR Model • Possible shapes for 1 or 2 domains: (CO 2) bonding nonbonding m/c shape e- d shape 2 0 linear
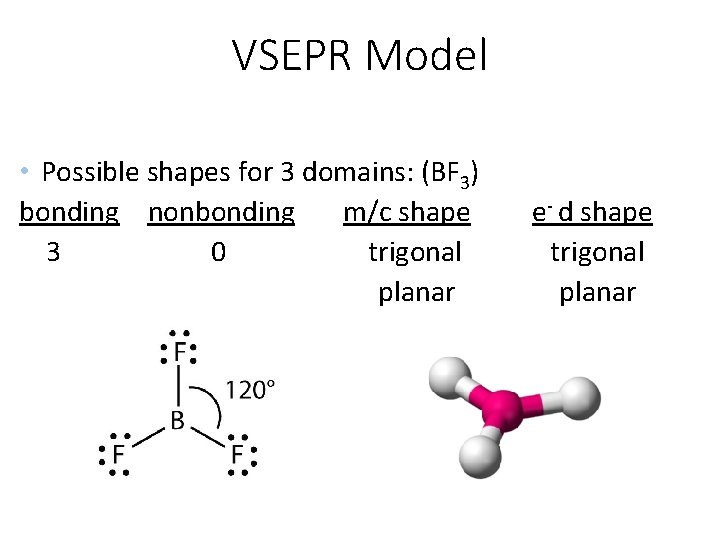
VSEPR Model • Possible shapes for 3 domains: (BF 3) bonding nonbonding m/c shape 3 0 trigonal planar e- d shape trigonal planar
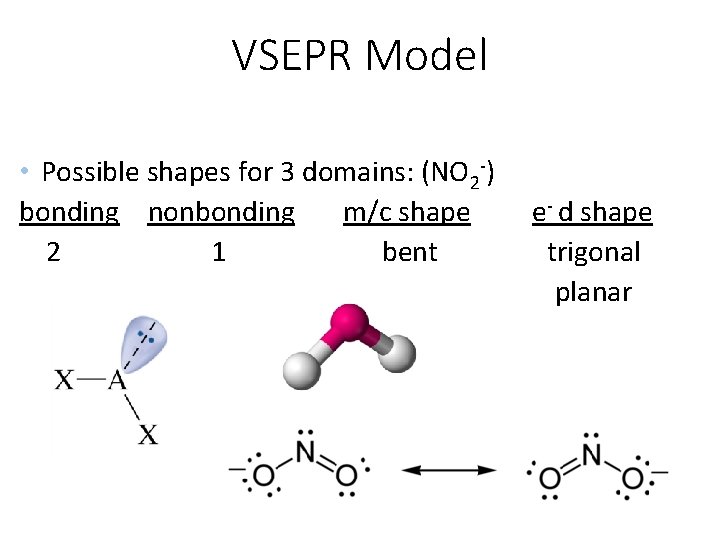
VSEPR Model • Possible shapes for 3 domains: (NO 2 -) bonding nonbonding m/c shape 2 1 bent e- d shape trigonal planar
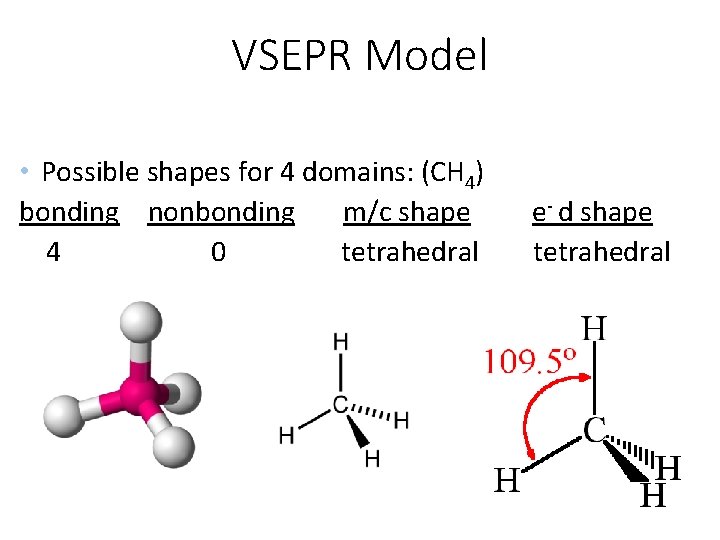
VSEPR Model • Possible shapes for 4 domains: (CH 4) bonding nonbonding m/c shape 4 0 tetrahedral e- d shape tetrahedral
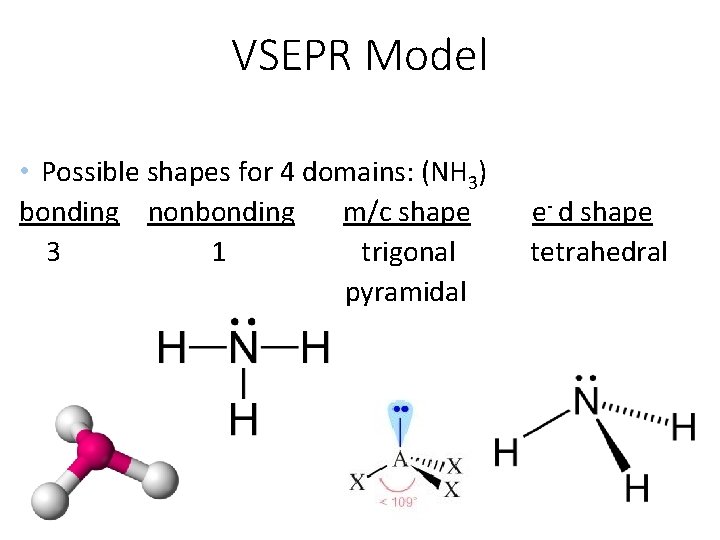
VSEPR Model • Possible shapes for 4 domains: (NH 3) bonding nonbonding m/c shape 3 1 trigonal pyramidal e- d shape tetrahedral
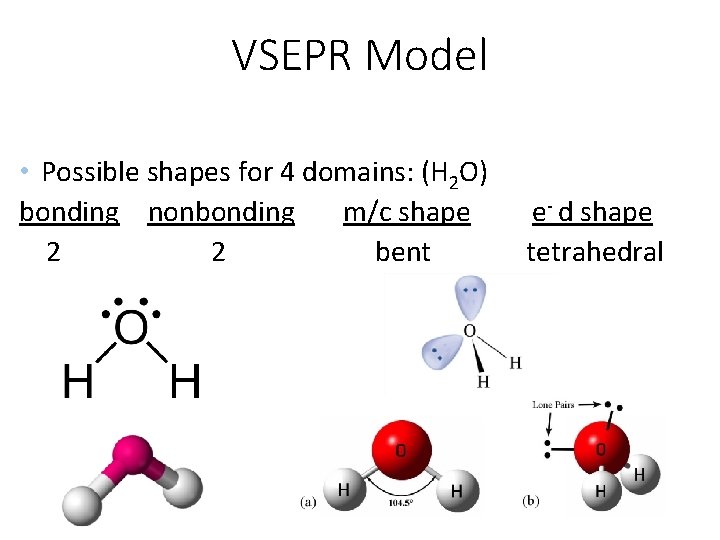
VSEPR Model • Possible shapes for 4 domains: (H 2 O) bonding nonbonding m/c shape 2 2 bent e- d shape tetrahedral
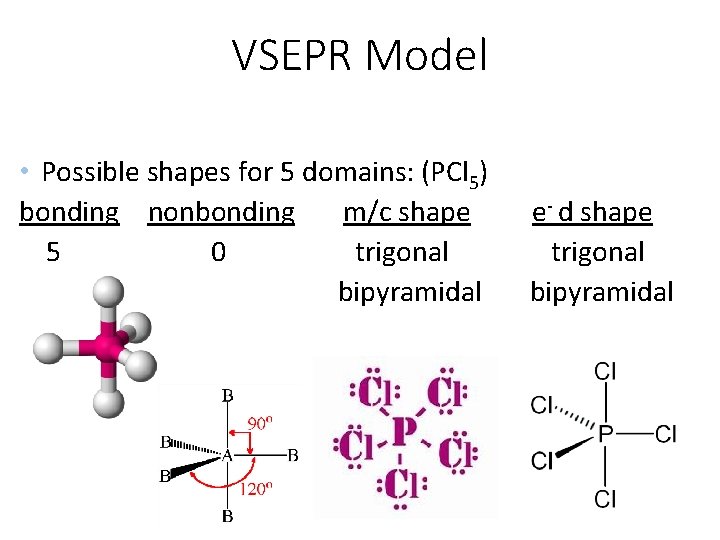
VSEPR Model • Possible shapes for 5 domains: (PCl 5) bonding nonbonding m/c shape 5 0 trigonal bipyramidal e- d shape trigonal bipyramidal
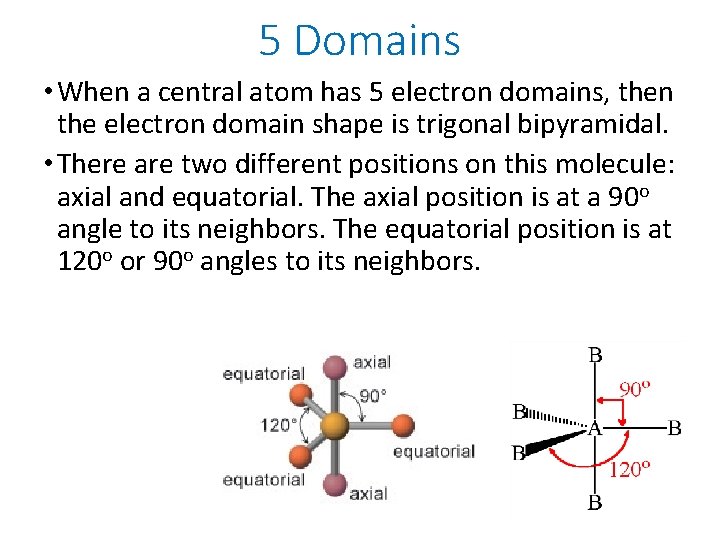
5 Domains • When a central atom has 5 electron domains, then the electron domain shape is trigonal bipyramidal. • There are two different positions on this molecule: axial and equatorial. The axial position is at a 90 o angle to its neighbors. The equatorial position is at 120 o or 90 o angles to its neighbors.
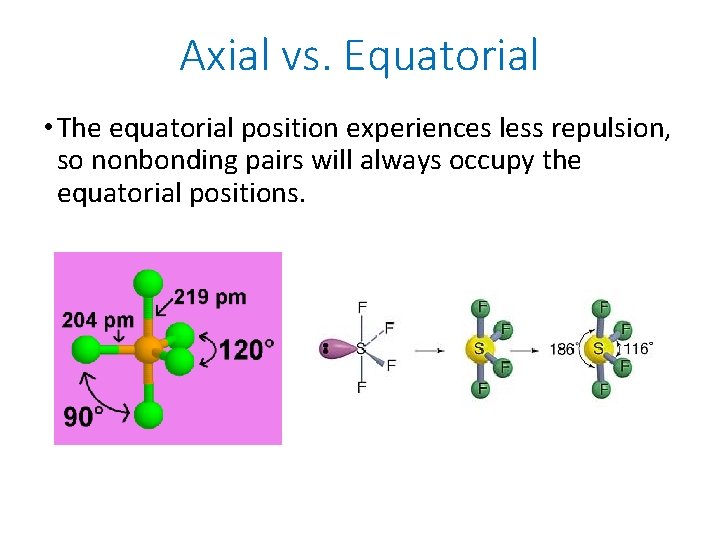
Axial vs. Equatorial • The equatorial position experiences less repulsion, so nonbonding pairs will always occupy the equatorial positions.
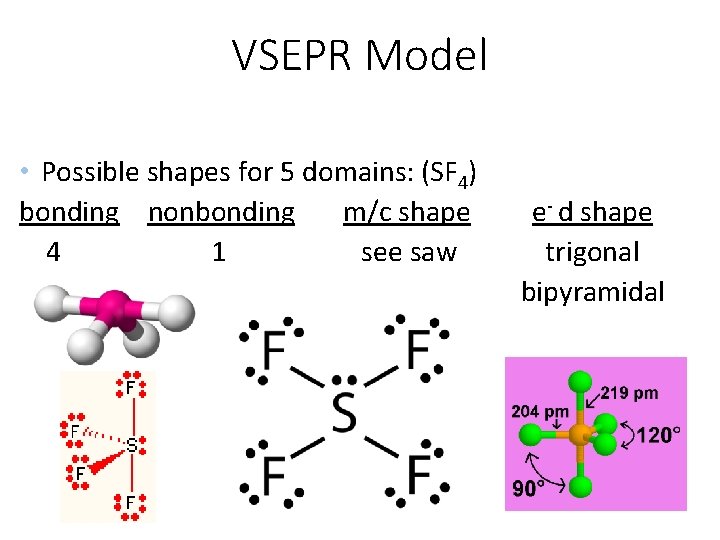
VSEPR Model • Possible shapes for 5 domains: (SF 4) bonding nonbonding m/c shape 4 1 see saw e- d shape trigonal bipyramidal
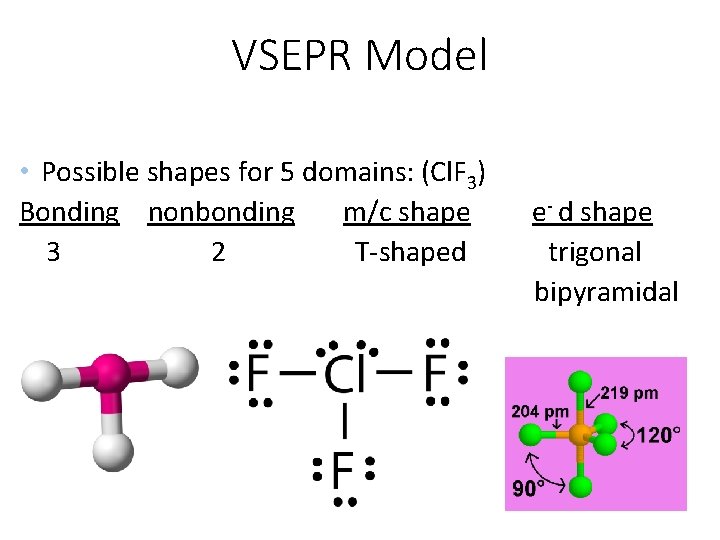
VSEPR Model • Possible shapes for 5 domains: (Cl. F 3) Bonding nonbonding m/c shape 3 2 T-shaped e- d shape trigonal bipyramidal
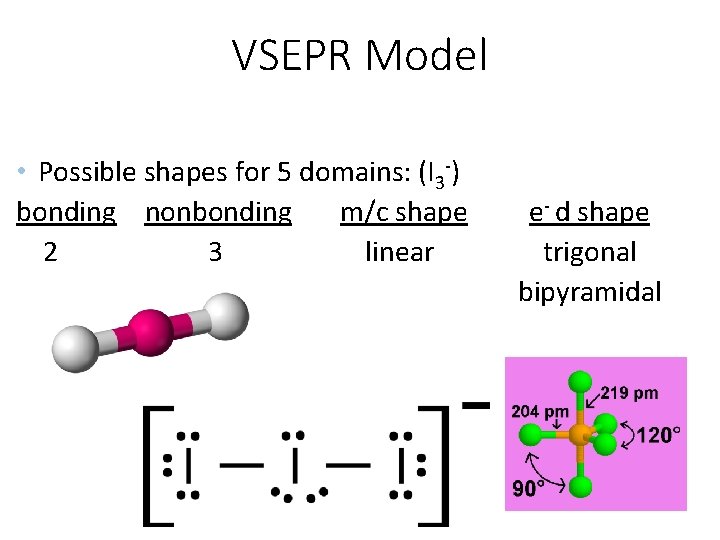
VSEPR Model • Possible shapes for 5 domains: (I 3 -) bonding nonbonding m/c shape 2 3 linear e- d shape trigonal bipyramidal
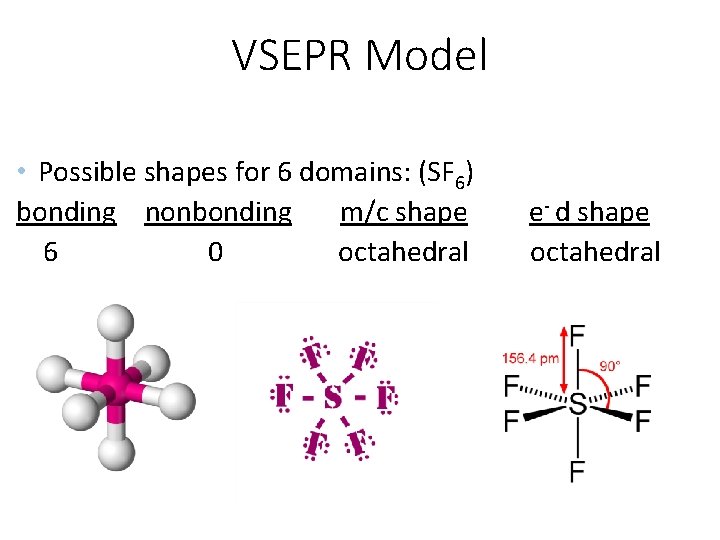
VSEPR Model • Possible shapes for 6 domains: (SF 6) bonding nonbonding m/c shape 6 0 octahedral e- d shape octahedral
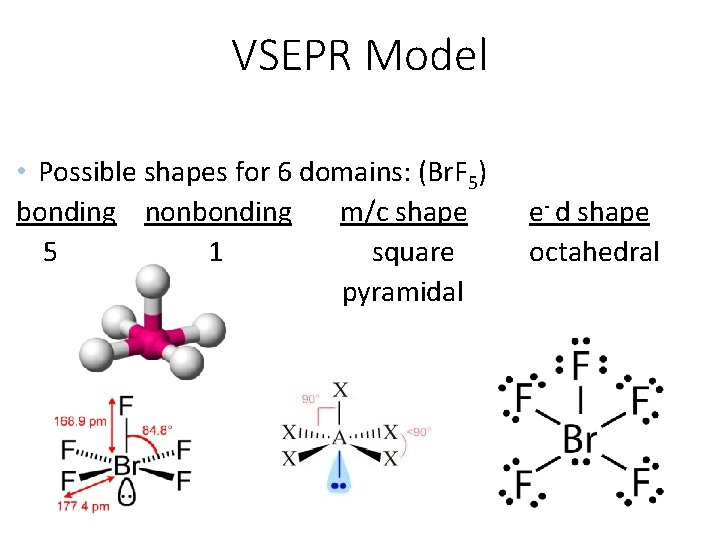
VSEPR Model • Possible shapes for 6 domains: (Br. F 5) bonding nonbonding m/c shape 5 1 square pyramidal e- d shape octahedral
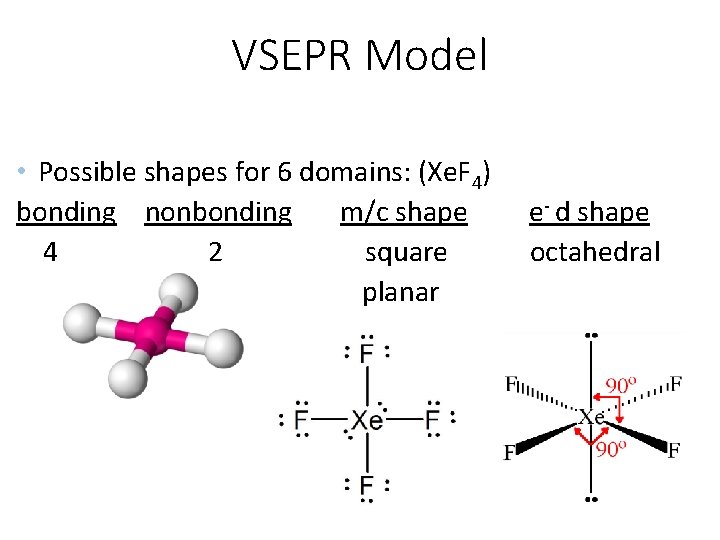
VSEPR Model • Possible shapes for 6 domains: (Xe. F 4) bonding nonbonding m/c shape 4 2 square planar e- d shape octahedral

Sample Exercise • Use the VSEPR model to predict the molecular geometry of a. O 3 b. Sn. Cl 3 -

Practice Exercise • Predict the electron-domain geometry and the molecular geometry for a. Se. Cl 2 b. CO 32 -

Sample Exercise • Use the VSEPR model to predict the molecular geometry of a. SF 4 b. IF 5

Sample Exercise • Predict the electron-domain geometry and molecular geometry of a. Cl. F 3 b. ICl 4 -

Bond Angles • Because a nonbonding pair experiences less nuclear attraction than bonding pairs, its electron domain is more spread out. • As a result, the electron domains for nonbonding electron pairs exert greater repulsive forces on adjacent electron domains and tend to compress the bond angles.

Bond Angles • Notice the difference in the bond angles of CH 4, NH 3, and H 2 O, all of which have a tetrahedral electron domain geometry.
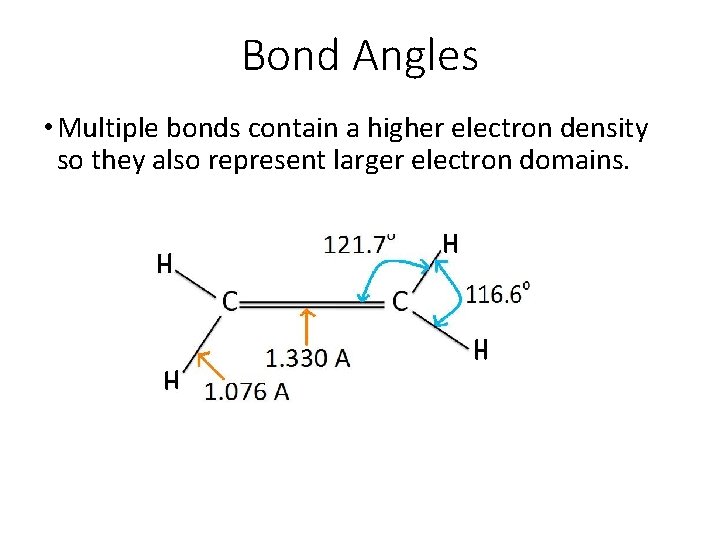
Bond Angles • Multiple bonds contain a higher electron density so they also represent larger electron domains.

Sample Exercise • Eyedrops used for dry eyes usually contain the water-soluble polymer called poly(vinylalcohol), which is based on the unstable organic molecule called vinyl alcohol: Predict the approximate values for the H – O – C and the O – C bond angles.
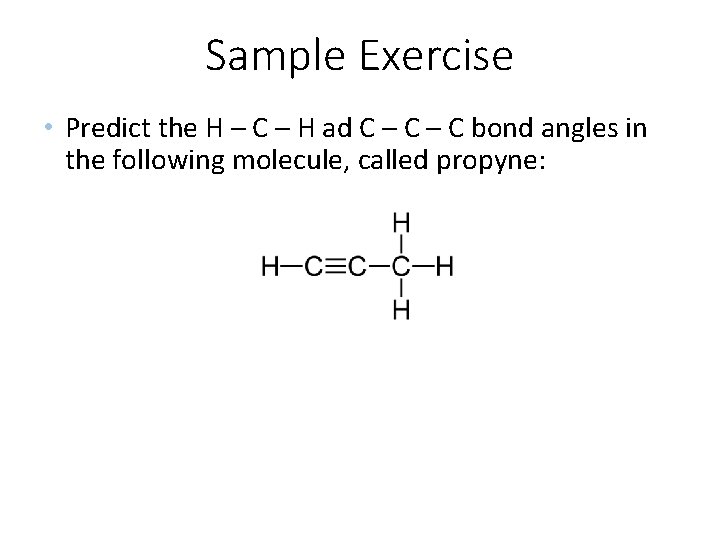
Sample Exercise • Predict the H – C – H ad C – C bond angles in the following molecule, called propyne:

Section 8. 4 – Polar Bonds and Molecules • Covalent bonds involve sharing electrons between atoms. • When the atoms in the bond pull equally, the bonding electrons are shared equally, and the bond is nonpolar. • When the atoms in the bond pull unequally, the bonding electrons are pulled closer to one atom, and the bond is polar.

Polarity • An atom’s “strength” is measured by the electronegativity (the ability to attract electrons). • The larger the electronegativity the more strongly an atom attracts electrons. • The more electronegative elements gets a d- (partial negative) charge and the less electronegative element gets a d+ (partial positive) charge. • The greater the difference in electronegativity between the two atoms, the more polar the bond.

Sample Problem • Determine the polarity of the following bonds: • H – Cl • F - P

Practice Problem • Determine the polarity of the following bonds. • Cl – C • O - S
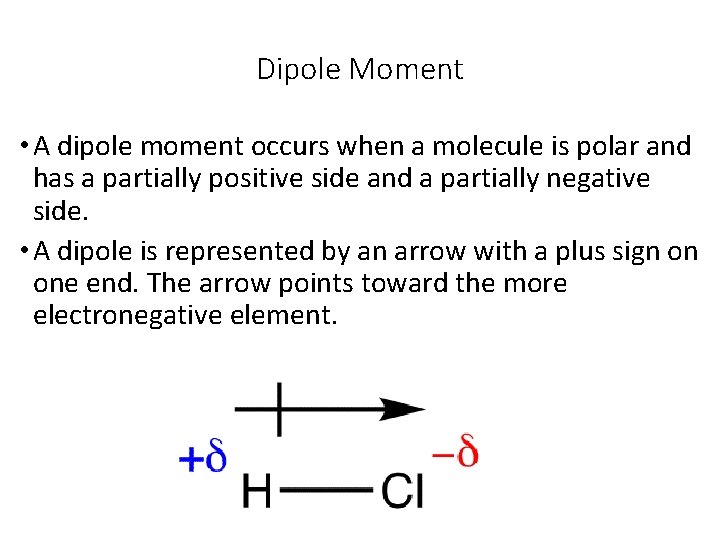
Dipole Moment • A dipole moment occurs when a molecule is polar and has a partially positive side and a partially negative side. • A dipole is represented by an arrow with a plus sign on one end. The arrow points toward the more electronegative element.

Sample Problem • Draw the dipole moments for the following: • H – F • H 2 O

Practice Problem • Draw the dipole moment for the following: • H – S • N - H

Dipole Moment • Whenever a distance separates two electrical charges of equal magnitude but opposite sign, a dipole is established. • The quantitative measure of the magnitude of a dipole is called the dipole moment (m). m = Qr m = dipole moment (debyes (D) = 3. 34 x 10 -30 coulomb-meters) Q = charge (units of 1. 60 x 10 -19 C) r = distance (m)
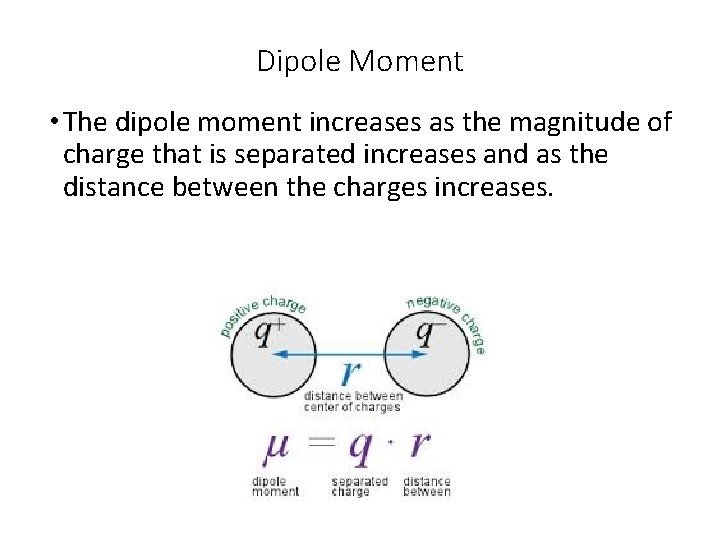
Dipole Moment • The dipole moment increases as the magnitude of charge that is separated increases and as the distance between the charges increases.

Sample Exercise • The bond length in the HCl molecule is 1. 27Å. a. Calculate the dipole moment, in debyes, that would result if the charges on the H and Cl atoms were 1+ and 1 -, respectively. b. The experimentally measured dipole moment of HCl(g) is 1. 08 D. What magnitude of charge, in units of e, on the H and Cl atoms would lead to this dipole moment?

Practice Exercise • The dipole moment of chlorine monofluoride, Cl. F(g), is 0. 88 D. The bond length of the molecule is 1. 63Å. a. Which atom is expected to have the partial negative charge? b. What is the charge on that atom, in units of e?

Bond Length • As we proceed from HF to HI, the electronegativity difference decreases and the bond length increases.

Polar Molecules • When polar molecules are placed between oppositely charged plates, the partially negative side is attracted to the positive plate and the partially positive side is attracted to the negative plate.
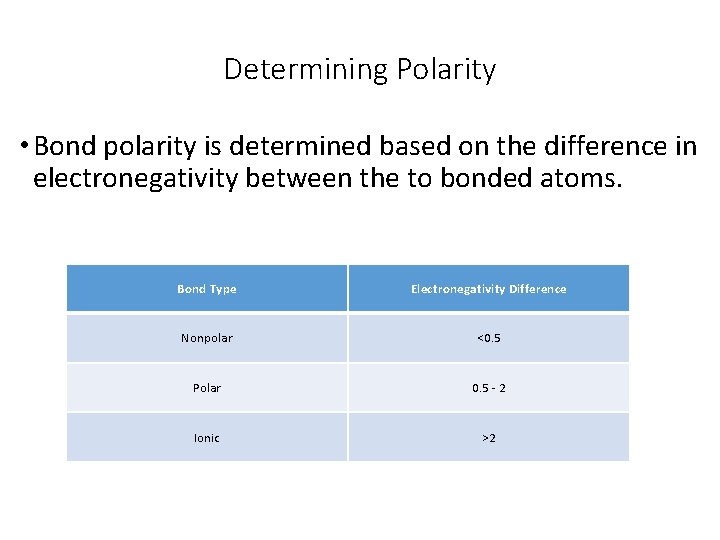
Determining Polarity • Bond polarity is determined based on the difference in electronegativity between the to bonded atoms. Bond Type Electronegativity Difference Nonpolar <0. 5 Polar 0. 5 - 2 Ionic >2

Sample Problem • Are the following bonds nonpolar, or ionic? • N – H • F – F • Ca - Cl

Practice Problem • Are the following bonds polar, nonpolar, or ionic? • H – Br • C – O • Li - O

Sample Exercise • In each case, which bond is more polar? Indicate in each case which atom has the partial negative charge. a. B-Cl or C-Cl b. P-F or P-Cl

Practice Exercise • Which of the following bonds is most polar: S-Cl, S -Br, Se-Cl, or Se-Br?
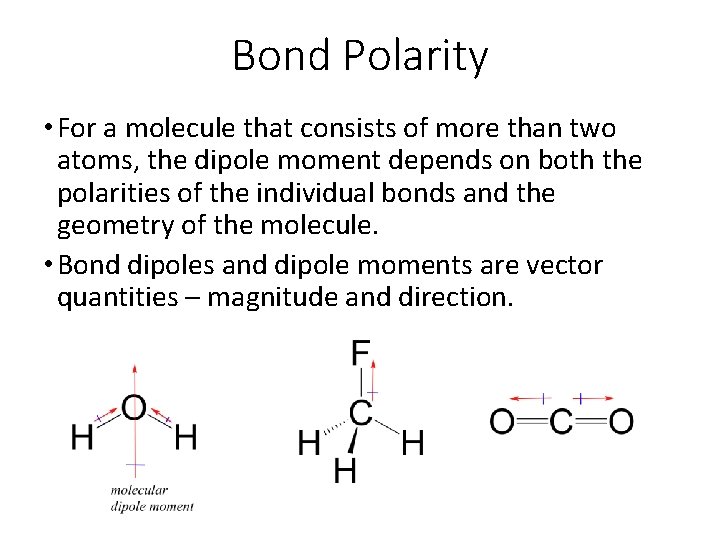
Bond Polarity • For a molecule that consists of more than two atoms, the dipole moment depends on both the polarities of the individual bonds and the geometry of the molecule. • Bond dipoles and dipole moments are vector quantities – magnitude and direction.

Polarity • The overall dipole moment is the vector sum of its bond dipoles. • If the dipoles are of equal magnitude but in opposite directions, then they cancel out making the molecule nonpolar.

Polarity • If the dipoles do not cancel out, then the molecule is polar. Be sure to consider shape before deciding polarity.

Sample Exercise • Predict whether the following molecules are polar or nonpolar: a. Br. Cl b. SO 2 c. SF 6

Practice Exercise • Determine whether the following molecules are polar or nonpolar: a. NF 3 b. BCl 3
Attractions • Intramolecular forces are the attractive forces within a single molecule. Ex: the bonds • Intermolecular forces are the attractive forces that exist between multiple molecules. Ex: dipole attractions

London Dispersion Forces • London dispersion forces are the weakest intermolecular forces that exist between ALL molecules. • These are the only forces in nonpolar molecules. • London dispersion forces tend to increase in strength with increasing molecular weight. • These forces also increase as the molecule chain length increases.

Dipole Forces • Dipole-dipole attractions exist between polar molecules. • An ion-dipole force exists between an ion and the partial charge on the end of a polar molecule. • The magnitude of attraction increases either as the charge of the ion in or the magnitude of the dipole increases.
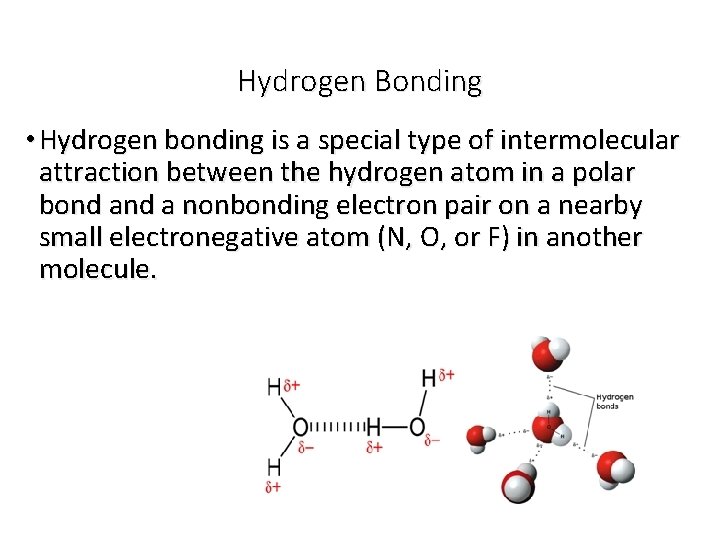
Hydrogen Bonding • Hydrogen bonding is a special type of intermolecular attraction between the hydrogen atom in a polar bond a nonbonding electron pair on a nearby small electronegative atom (N, O, or F) in another molecule.
Strength of Intermolecular Forces London Dispersion < Dipole-Dipole < Hydrogen Bonding Intermolecular forces of different substance vary over a wide range, but are generally weaker than an actual bond.

Intermolecular Forces • When comparing the strengths of intermolecular attractions in two substances, consider the following generalizations: 1. For molecules of similar molecular weights and shapes, dispersion forces are equal unless there is a difference in the polarity. 2. For molecules of different molecular weights, the substance with more mass has the strongest attractions.

Sample Problem • Determine the intermolecular forces that exist in the following molecules. • SO 2 • CH 4 • HF

Practice Problems • Determine the intermolecular forces that exist between the following molecules. • NH 3 • O 2 • PCl 3

Sample Exercise • The dipole moments of CH 3 CN is 3. 9 D and CH 3 I is 1. 62 D. a. Which of these substances has greater dipole attractions among its molecules? b. Which of these substances has greater London dispersion attractions?

Sample Exercise con’t c. The boiling points of CH 3 CN and CH 3 I are 354. 8 K and 315. 6 K, respectively. Which substance has the greater overall attractive forces?

Practice Exercise • Of Br 2, HCl, HBr, and N 2, which is likely to have a. the largest intermolecular dispersion forces? b. the largest dipole-dipole attractive forces?

Sample Exercise • In which of the following substances is hydrogen bonding likely to play an important role in determining physical properties: methane (CH 4), hydrazine (H 2 NNH 2), methyl fluoride (CH 3 F), or hydrogen sulfide (H 2 S)?

Practice Exercise • In which of the following substances is significant hydrogen bonding possible: methylene chloride (CH 2 Cl 2), phosphine (PH 3), hydrogen peroxide (HOOH), or acetone (CH 3 COCH 3)?

Intermolecular Forces • The molecules of a liquid must overcome their attractive forces to separate and form a vapor. The stronger the attractive forces are, the higher the boiling point. • The melting points of solids also increases as the strengths of the intermolecular forces increase. • In general, the boiling point increases with increasing intermolecular forces. • Remember that intermolecular forces are additive.

Sample Exercise • List the substances Ba. Cl 2, H 2, CO, HF, and Ne in order of increasing boiling points.

Practice Exercise a. Identify the intermolecular attractions present in the following substances: CH 3, CH 3 OH, and CH 3 CH 2 OH. b. Select which substance has the highest boiling point.

Cohesive vs. Adhesive • Intermolecular forces that bind similar molecules together, such as hydrogen bonding in water, are called cohesive forces. • Intermolecular forces that bind a substance to a surface are called adhesive forces.

Meniscus • The meniscus of water is U-shaped because the adhesive forces between the water and glass are greater than the cohesive forces between the water molecules. • The meniscus of mercury curves downward because the cohesive forces are greater than the adhesive forces.

Capillary Action • The rise of liquids up very narrow tubes is called capillary action. • Adhesive forces are responsible for capillary action.

Network Solids • A network solid is a solid in which every atom is covalently bonded to one another. • A network solid is extremely strong and has a very high melting point. Ex: diamond and Si. O 2

Section 8. 4 Assessment 1. How do electronegativity values determine the charge distribution in a polar covalent bond? 2. What happens when polar molecules are between oppositely charged metal plates? 3. Compare the strengths of intermolecular attractions to the strengths of ionic bonds and covalent bonds. 4. Not every molecule with polar bonds is polar. Use CO 2 as an example.

Section 8. 4 Assessment 5. Draw the Lewis dot structure for each molecule. Identify the partial positive and partial negative atoms in each molecule. a. HOOH b. Br. Cl c. HBr d. H 2 O
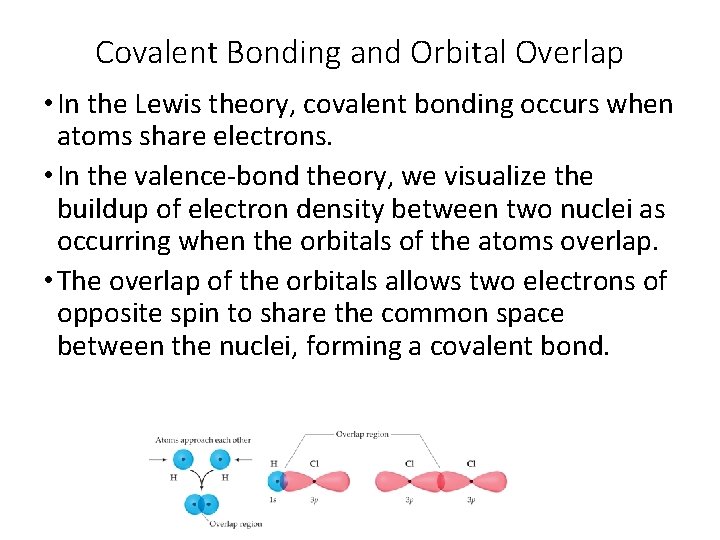
Covalent Bonding and Orbital Overlap • In the Lewis theory, covalent bonding occurs when atoms share electrons. • In the valence-bond theory, we visualize the buildup of electron density between two nuclei as occurring when the orbitals of the atoms overlap. • The overlap of the orbitals allows two electrons of opposite spin to share the common space between the nuclei, forming a covalent bond.
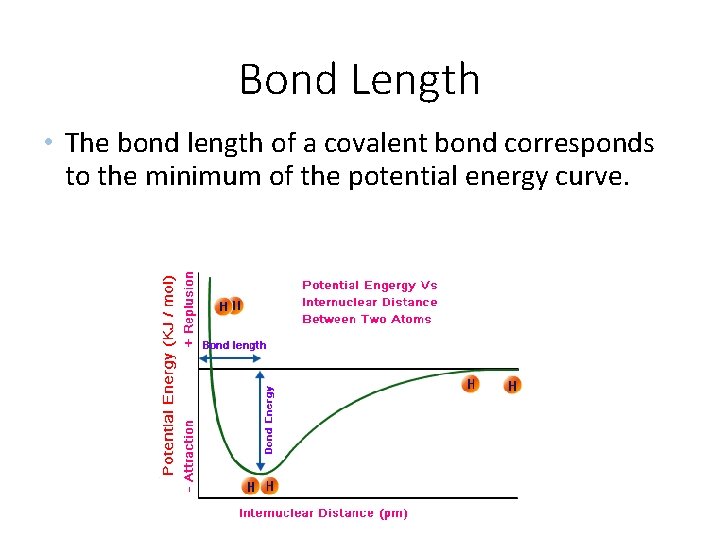
Bond Length • The bond length of a covalent bond corresponds to the minimum of the potential energy curve.

Hybrid Orbitals • To explain geometries, we assume that the atomic orbitals on an atom mix to form new orbitals called hybrid orbitals. • The process of mixing atomic orbitals is called hybridization. • The number of hybrid orbitals must equal the number of atomic orbitals.

sp Hybrid Orbitals • Be. F 2 • F = ___ ___ ___ 1 s 2 s 2 p • This shows that F can bond by filling its 2 p sublevel. • Be = ___ ___ ___ • 1 s 2 s 2 p

sp Hybrid Orbitals • Since Be has no unpaired electrons, it should not form bonds, so one of the electrons is “promoted” to a 2 p orbital. ___ • Be = ___ ___ 1 s 2 s 2 p • However, this arrangement would not make the two Be – F bonds equal because one would involve and s orbital and one would involve a p orbital.
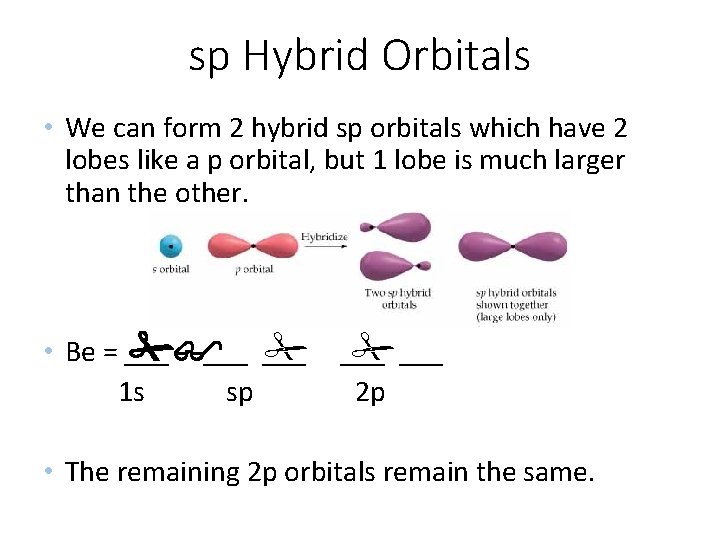
sp Hybrid Orbitals • We can form 2 hybrid sp orbitals which have 2 lobes like a p orbital, but 1 lobe is much larger than the other. • Be = ___ ___ 1 s sp ___ 2 p • The remaining 2 p orbitals remain the same.
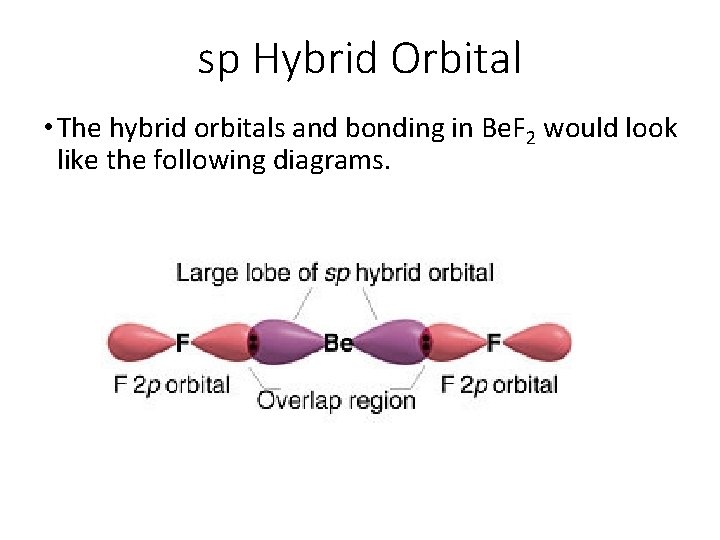
sp Hybrid Orbital • The hybrid orbitals and bonding in Be. F 2 would look like the following diagrams.

Hybrid Orbitals • Whenever we mix a certain number of atomic orbitals, we get the same number of hybrid orbitals. • Each of the hybrid orbitals is equivalent to the others but points in a different direction. • Thus mixing one 2 s and one 2 p orbital yields two equivalent sp hybrid orbitals that point in opposite directions.
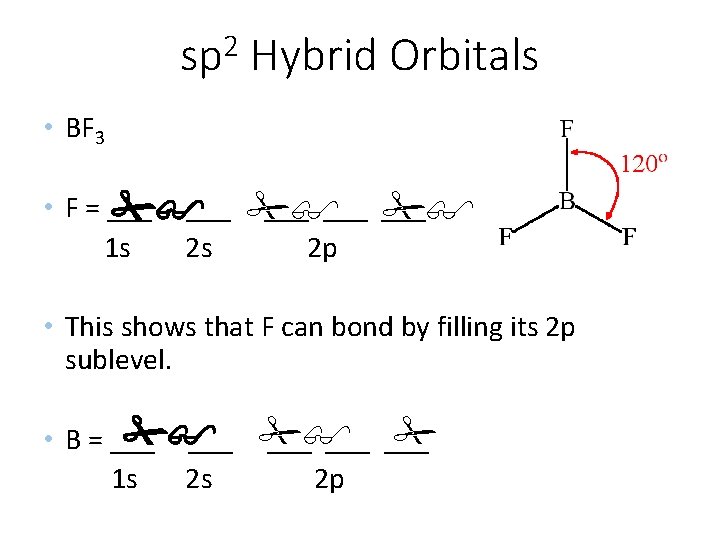
2 sp Hybrid Orbitals • BF 3 • F = ___ ___ 1 s 2 s 2 p • This shows that F can bond by filling its 2 p sublevel. • B = ___ ___ ___ 1 s 2 s 2 p

2 sp Hybrid Orbitals • Since B has one unpaired electron, it should only form one bond, so one of the electrons is “promoted” to a 2 p orbital to allow for 3 bonds. • B = ___ ___ ___ 1 s 2 s 2 p • However, this arrangement would not make three B – F bonds equal because one would involve and s orbital and two would involve p orbitals.

2 sp Hybrid Orbitals • We can form 3 hybrid sp 2 orbitals which have 2 lobes like a p orbital, but 1 lobe is much larger than the other. ___ • B = ___ ___ 1 s sp 2 2 p • The remaining 2 p orbital remains the same.
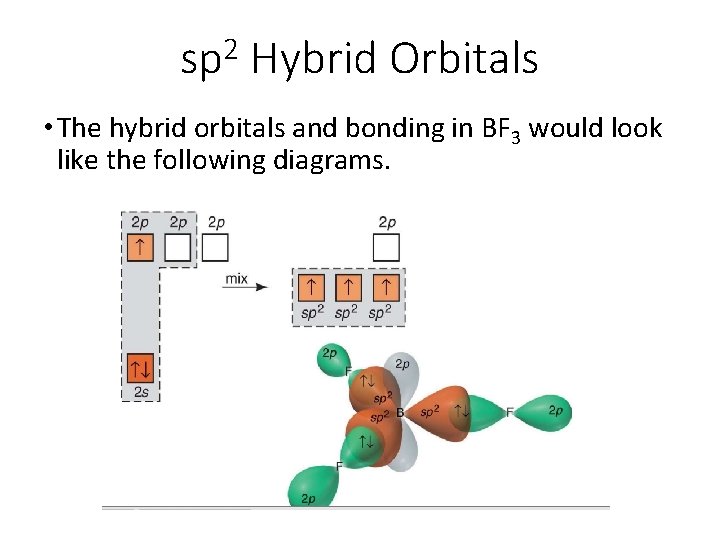
2 sp Hybrid Orbitals • The hybrid orbitals and bonding in BF 3 would look like the following diagrams.
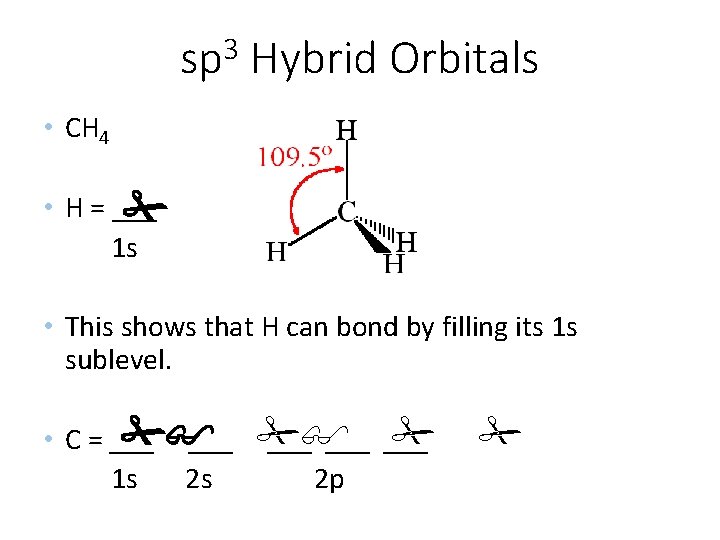
3 sp Hybrid Orbitals • CH 4 • H = ___ 1 s • This shows that H can bond by filling its 1 s sublevel. • C = ___ ___ ___ 1 s 2 s 2 p

3 sp Hybrid Orbitals • Since C has two unpaired electrons, it should only form two bonds, so one of the electrons is “promoted” to a 2 p orbital to allow for 4 bonds. • C = ___ ___ ___ 1 s 2 s 2 p • However, this arrangement would not make the four C – H bonds equal because one would involve and s orbital and three would involve p orbitals.

3 sp Hybrid Orbitals • We can form 4 hybrid sp 3 orbitals which have 2 lobes like a p orbital, but 1 lobe is much larger than the other. • C = ___ ___ 1 s sp 3
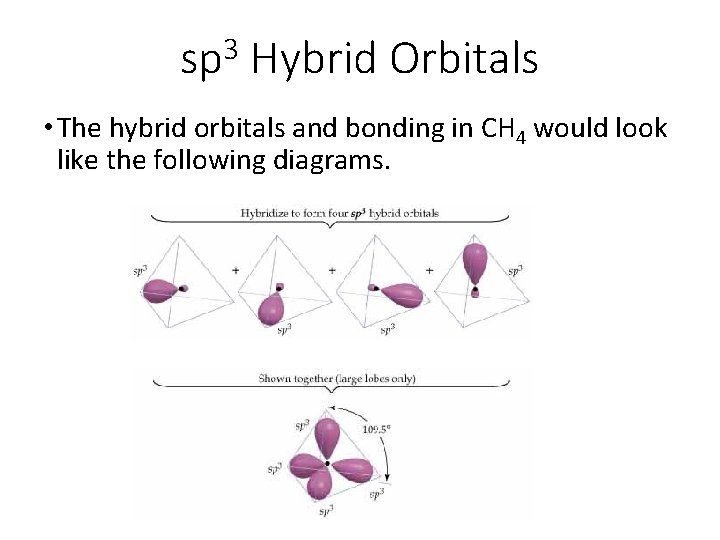
3 sp Hybrid Orbitals • The hybrid orbitals and bonding in CH 4 would look like the following diagrams.
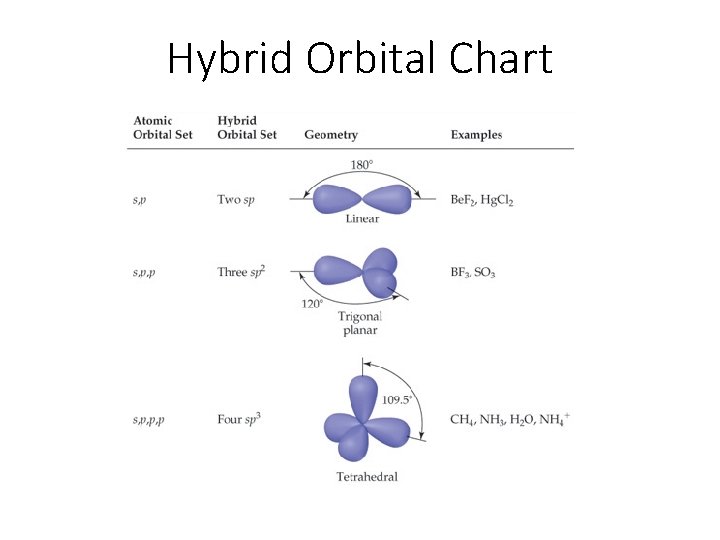
Hybrid Orbital Chart

Sample Exercise • Indicate the hybridization of orbitals employed by the central atom in NH 2 -.

Practice Exercise • Predict the electron-domain geometry and the hybridization of the central atom in SO 32 -.
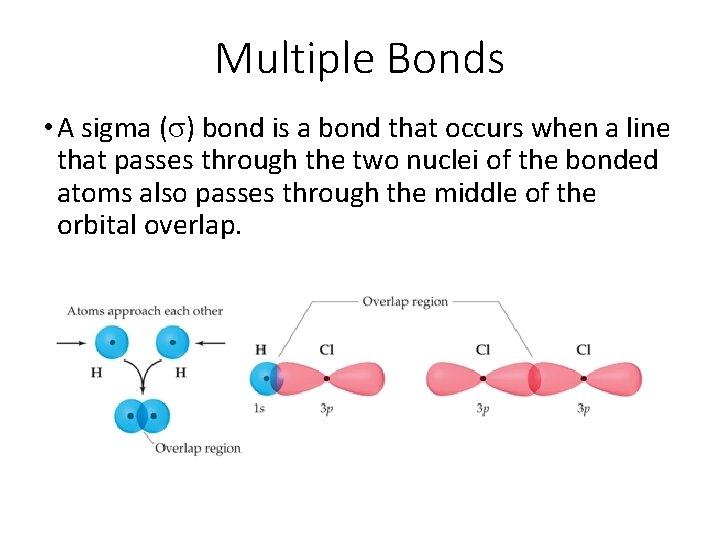
Multiple Bonds • A sigma (s) bond is a bond that occurs when a line that passes through the two nuclei of the bonded atoms also passes through the middle of the orbital overlap.
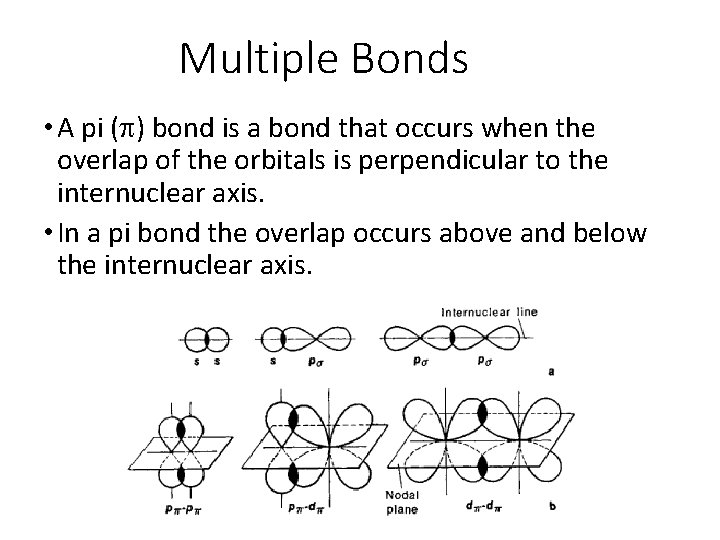
Multiple Bonds • A pi (p) bond is a bond that occurs when the overlap of the orbitals is perpendicular to the internuclear axis. • In a pi bond the overlap occurs above and below the internuclear axis.

Pi vs. Sigma • Unlike a sigma bond, in a pi bond there is no probability of finding the electron on the internuclear axis. • Since the p orbitals in a pi bond overlap sideways rather than directly facing each other, the total overlap in a pi bond tends to be less than in a sigma bond. This means that pi bonds tend to be weaker than sigma bonds.
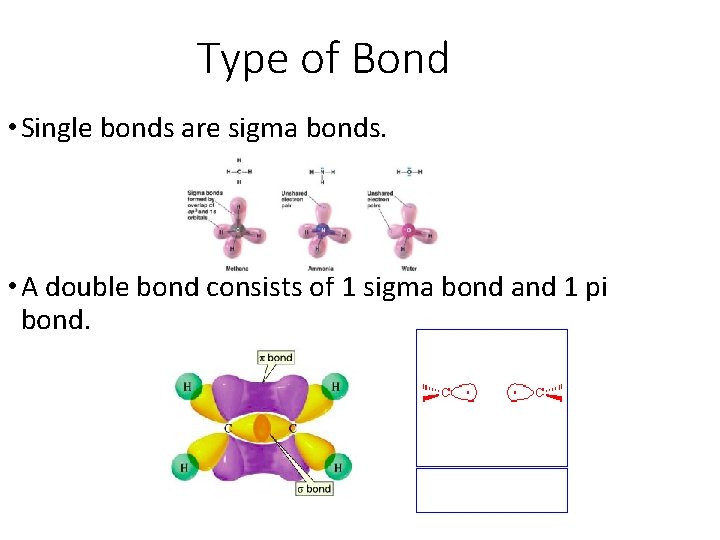
Type of Bond • Single bonds are sigma bonds. • A double bond consists of 1 sigma bond and 1 pi bond.
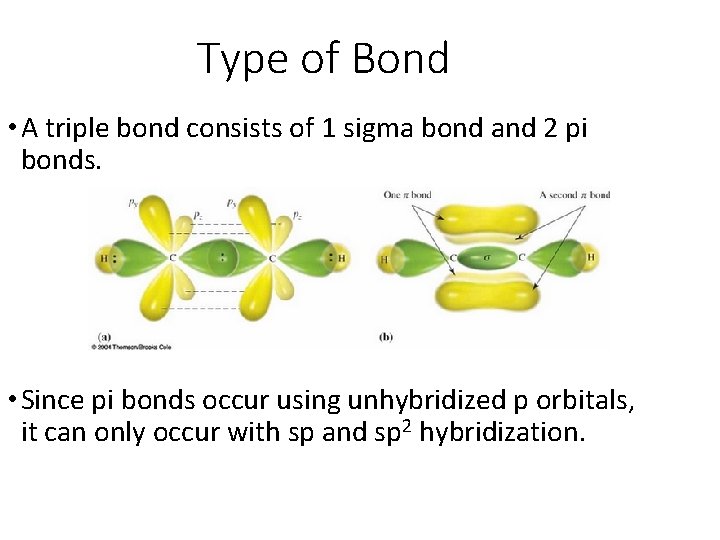
Type of Bond • A triple bond consists of 1 sigma bond and 2 pi bonds. • Since pi bonds occur using unhybridized p orbitals, it can only occur with sp and sp 2 hybridization.

Sample Exercise • Formaldehyde has the Lewis structure Describe how the bonds in formaldehyde are formed in terms of overlaps of appropriate hybridized and unhybridized orbitals.

Practice Exercise • Consider the acetonitrile molecule: a. Predict the bond angles around each carbon atom.

Practice Exercise Con’t b. Describe the hybridization at each carbon atom. c. Determine the total number of s and p bonds in the molecule.
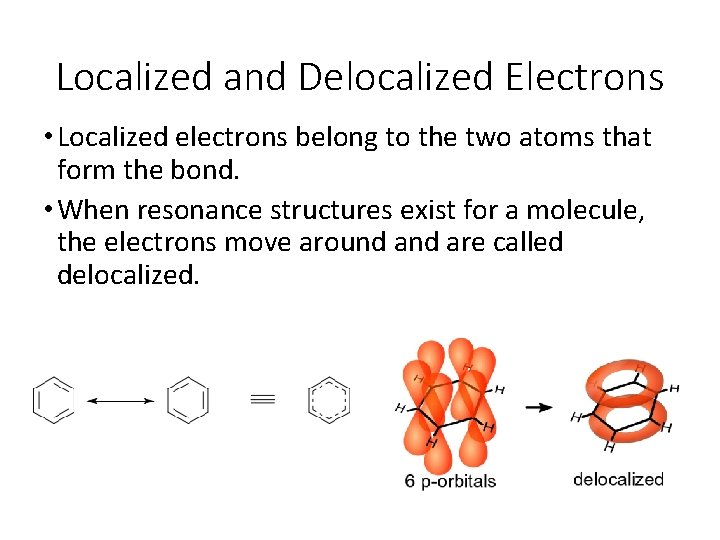
Localized and Delocalized Electrons • Localized electrons belong to the two atoms that form the bond. • When resonance structures exist for a molecule, the electrons move around are called delocalized.

Sample Exercise • Describe the bonding in the nitrate ion, NO 3 -. Does this ion have delocalized p bonds?

Practice Exercise • Which of the following molecules or ions will exhibit delocalized bonding: SO 3, SO 32 -, H 2 CO, O 3, NH 4+?

Section 9. 3 – Naming with Nonmetals • Nonmetals are to the right of the stair-step line on the periodic table. • When naming with nonmetals, you name the first element and add –ide to the second element. • You also must add prefixes to indicate the number of atoms of each element. • The only time that you do not need a prefix is when the first element only has one atom.
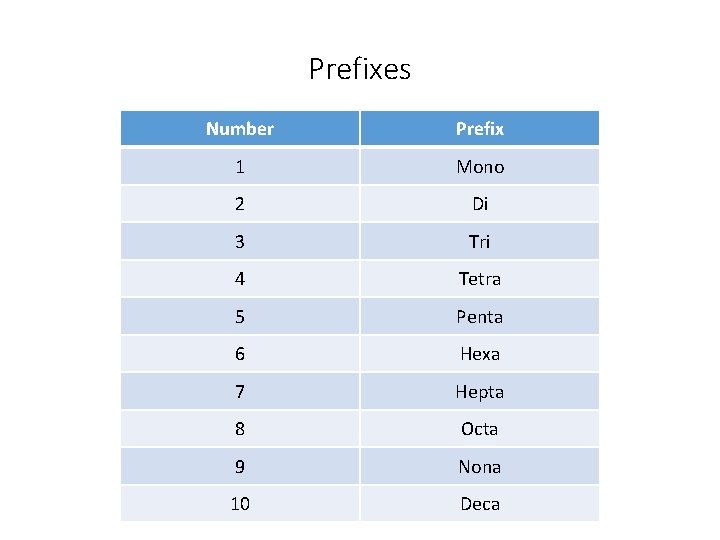
Prefixes Number Prefix 1 Mono 2 Di 3 Tri 4 Tetra 5 Penta 6 Hexa 7 Hepta 8 Octa 9 Nona 10 Deca

Sample Problem • Write the name of the following molecules. • H 2 O • CO 2 • CO

Practice Problem • Write the name of the following molecules. • Si. Br 4 • N 2 O 5 • NH 3

Writing Formulas with Nonmetals • When writing the formula of a compound that starts with a nonmetal, you do not need to balance charges because the prefixes tell you the number of atoms.

Sample Problem • Write the formula for the following compounds. • Carbon tetrachloride • Sulfur hexafluoride

Practice Problem • Write the formula for the following molecules. • Diphosphorous trioxide • Bromine monoiodide

Section 9. 3 Assessment 1. What information do prefixes tell you? 2. Write the names for the following molecules. a. b. c. d. e. f. g. NI 3 BCl 3 Cl 2 O 7 SO 3 N 2 H 4 N 2 O 3 CS 2

Section 9. 3 Assessment 3. Write the formula for the following compounds: a. carbon tetrabromide b. phosphorus pentachloride c. iodine heptafluoride d. chlorine trifluoride e. iodine dioxide 4. The name a student gives for the molecular compound Si. Cl 4 is monosilicon tetrachloride. Is this correct?

Sample Integrative Exercise • Elemental sulfur is a yellow solid that consists of S 8 molecules. The structure of the S 8 molecule is an eight-membered ring. Heating elemental sulfur to high temperatures produces gaseous S 2 molecules: S 8(s) 4 S 2(g) a. With respect to electronic structure, which element in the second row of the periodic table is most similar to sulfur?

Sample Integrative Exercise b. Use the VSEPR model to predict the S – S bond angles in S 8, and the hybridization at S in S 8.

Sample Integrative Exercise d. Use average bond enthalpies (Table 8. 4) to estimate the enthalpy change for the reaction just described. Is the reaction exothermic or endothermic?
- Slides: 164