Chapter 8 Covalent Bonding Covalent bonding Usually forms
Chapter 8 Covalent Bonding

Covalent bonding ……. • Usually forms between two nonmetals (takers) • Neither want to give up their electrons but are willing to share electrons

Exception: Hydrogen • Hydrogen has one electron, but only needs one more to be “happy” Hydrogen and a nonmetal …. . neither wants to give up their electrons and neither has enough pull to take the other’s so they share a pair of electrons
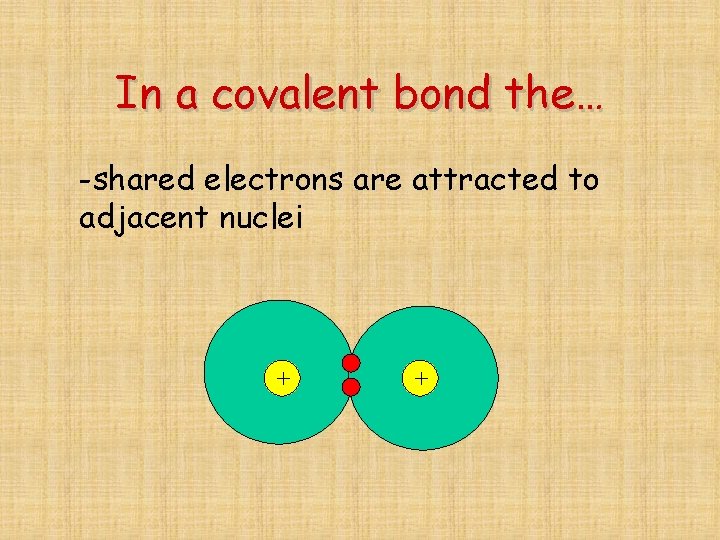
In a covalent bond the… -shared electrons are attracted to adjacent nuclei + +
Covalent Bonding

Covalent Bonds form molecules Molecule = smallest part of a covalent compound (* remember: Ionic Compounds formula units)

That’s why……. Covalent compounds are also called molecular compounds

Naming Covalent Compounds Binary (2 -element) compounds -similar to ionic, but indicate the number of atoms using prefixes
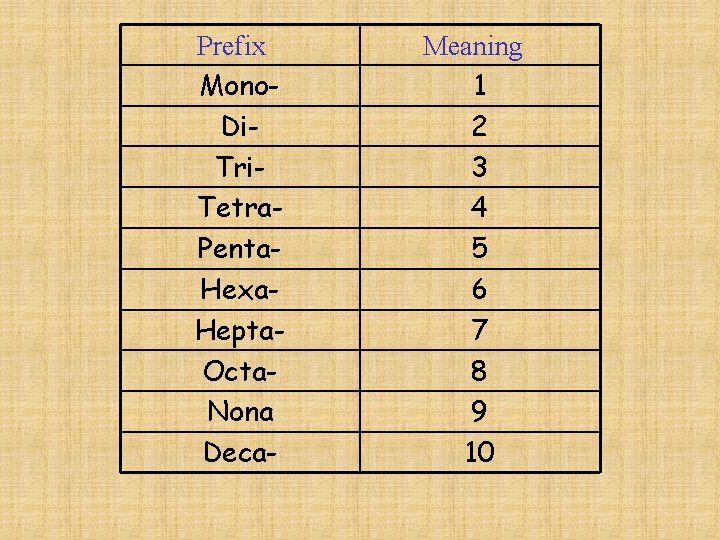
Prefix Mono. Di. Tri. Tetra. Penta. Hexa. Hepta. Octa. Nona Deca- Meaning 1 2 3 4 5 6 7 8 9 10

-if only 1 atom of 1 st element, then no prefix is used Ex. CO Carbon monoxide CO 2 Carbon dioxide

-if more than 1 atom of 1 st element, then use prefix N 2 O 4 Dinitrogen tetraoxide

Ws. Naming Molecular Compounds Ws. Name the following Molecular Compounds

Hydrogen Compounds can form Acids • Remember…. . Hydrogen is the exception to most rules • Hydrogen has 1 electron but only needs 1 more to be happy so it acts like a nonmetal

• Hydrogen doesn’t want to give up it’s one electron but it is willing to share it. • When hydrogen combines with an anion a covalent compound is formed

When these compounds are dissolved in water…. . they become acids.

What makes them an acid? • Hydronium ions are formed

How to Name Acids • Start with ionic compound name. • Then follow the 3 rules

Naming Acids Rule #1 (Binary Acids) • When anion ends in “ide”, the acid name begins with “hydro” and then ends in “ic”, then “acid” • Ex. Hydrogen chloride (HCl)= hydrochloric acid Hydrogen sulfide (H 2 S) =hydrosulfuric acid

Naming Acids Rule #2 (Tertiary Acids) • If anion ends in “ate”, the acid name is the stem of the anion with the suffix “-ic” followed by the word “acid” • Ex. Hydrogen nitrate (HNO 3) = nitric acid

Naming Acids Rule #3 (Tertiary Acids) • When anion ends in “ite” , the acid name is the stem of the anion with suffix “-ous” followed by the word “acid” • Ex. Hydrogen sulfite (H 2 SO 3) = sulfurous acid


Ws. Name the Following Acids Ws. Formula Writing
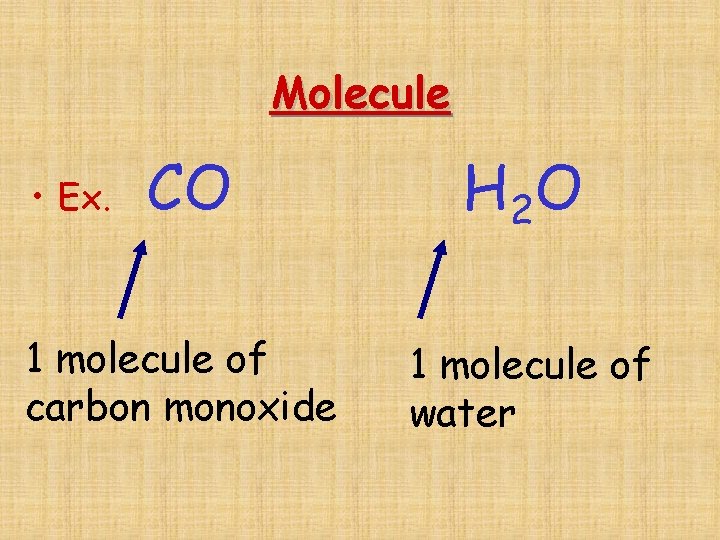
Molecule CO H 2 O 1 molecule of carbon monoxide 1 molecule of water • Ex.
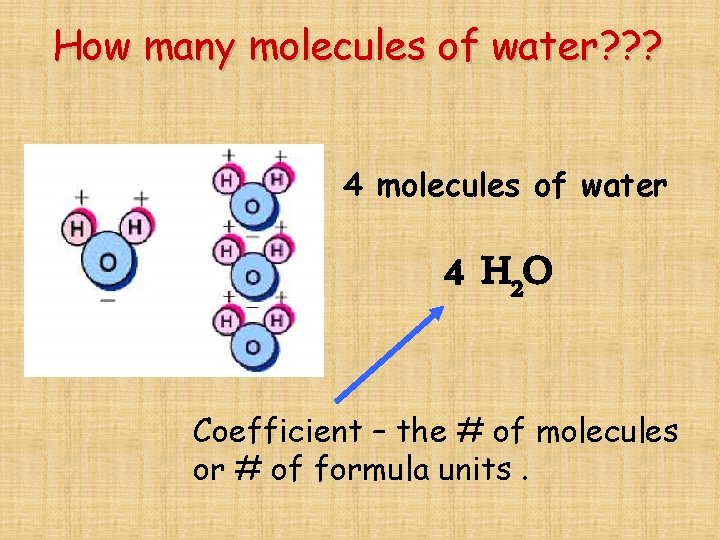
How many molecules of water? ? ? 4 molecules of water 4 H 2 O Coefficient – the # of molecules or # of formula units.

The number covalent bonds that will form depends on the # of valence electrons *Remember each atom is trying to achieve noble gas configuration (full outer shell) 7 valence electrons wants to share 1 pair 6 valence electrons wants to share 2 pair 5 valence electrons wants to share 3 pair

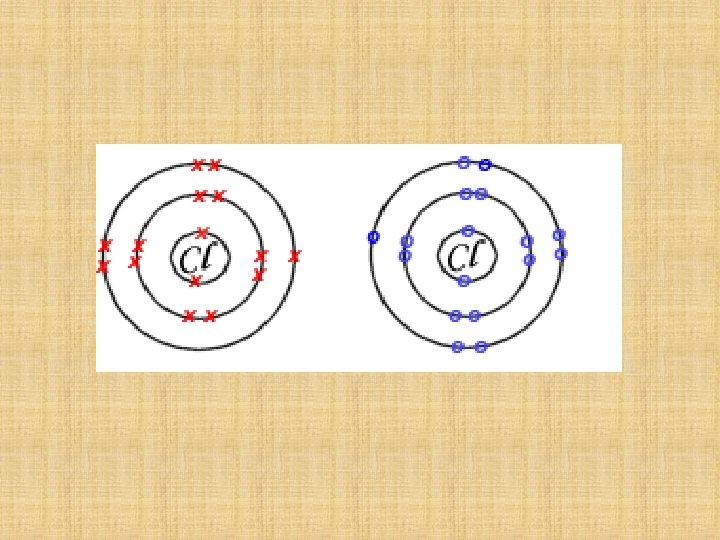
Chlorine atoms each have 7 valence electrons …. they each want one more so they will share one pair of electrons

Formation of Bonds
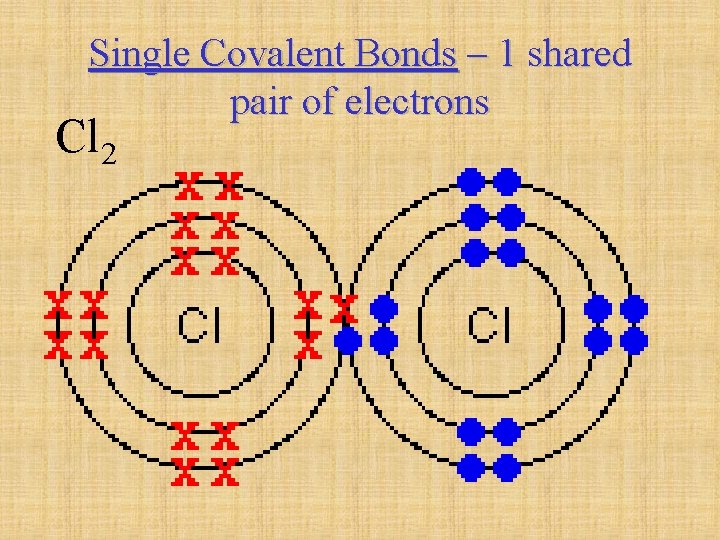
Single Covalent Bonds – 1 shared pair of electrons Cl 2

Single bond- 1 shared pair of electrons F and F F F Double bond-2 shared pairs of electrons O and O O O Triple bond-3 shared pairs of electrons N and N N N

Lewis Dot Diagram of Covalent Compounds -you show the shared pair(s) of electrons between symbols Ex. Cl 2 Cl Cl
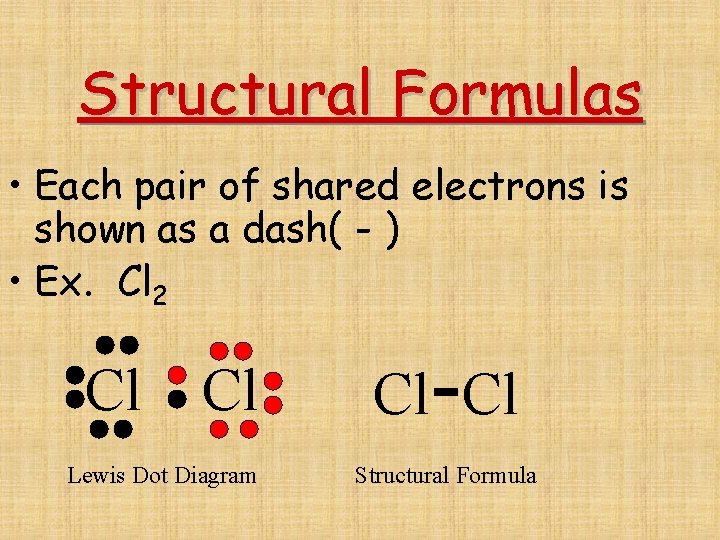
Structural Formulas • Each pair of shared electrons is shown as a dash( - ) • Ex. Cl 2 Cl Lewis Dot Diagram - Cl Cl Structural Formula

Be careful with Hydrogen (H 2) *Remember hydrogen only needs to share one pair
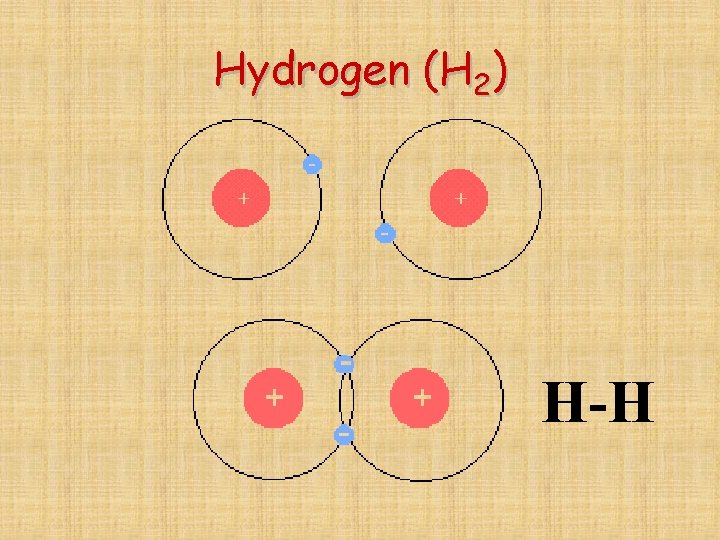
Hydrogen (H 2) H-H

Diatomic Molecules • Two different atoms covalently bonded • Ex. CO OH

Diatomic Elements -when atoms of same element covalently bond Ex. Cl and Cl Cl Cl

Diatomic Elements -elements that exist in pairs when not bonded to other atoms (“Br. INCl. HOF”) Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2 bromine iodine nitrogen chlorine hydrogen oxygen fluorine

More examples of single covalent bonds…
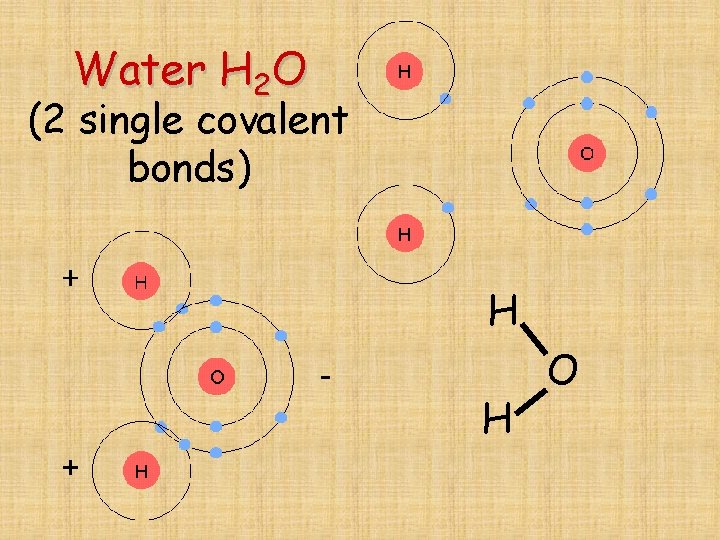
Water H 2 O (2 single covalent bonds) H H O
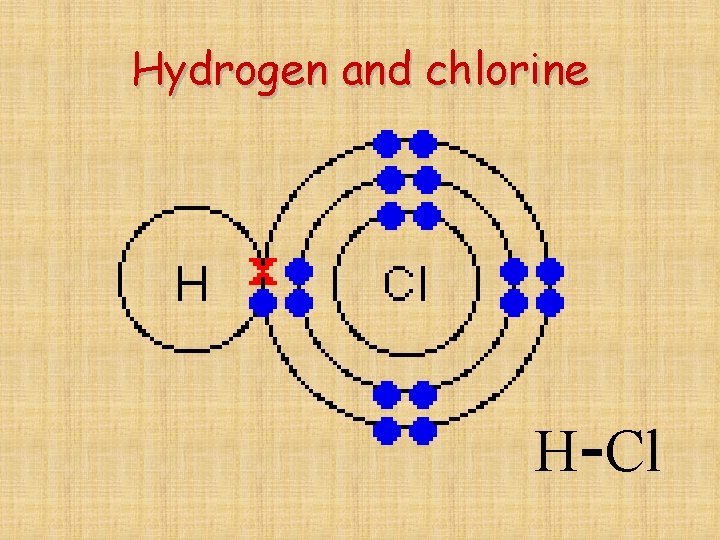
Hydrogen and chlorine H-Cl
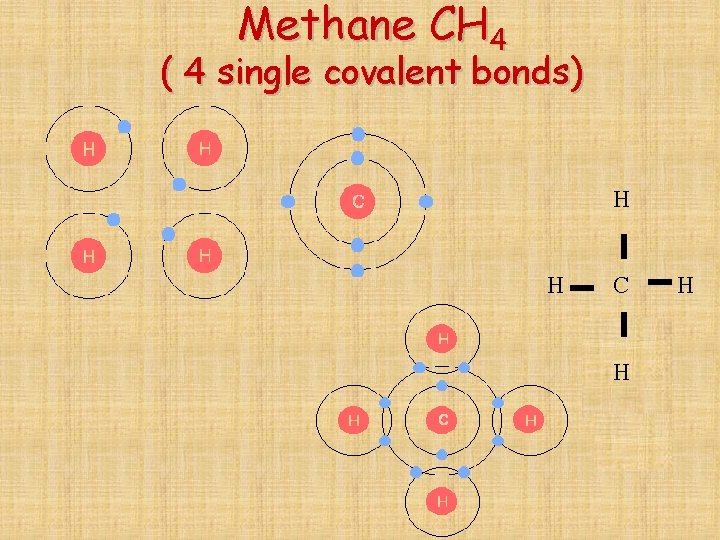
Methane CH 4 ( 4 single covalent bonds) H H C H
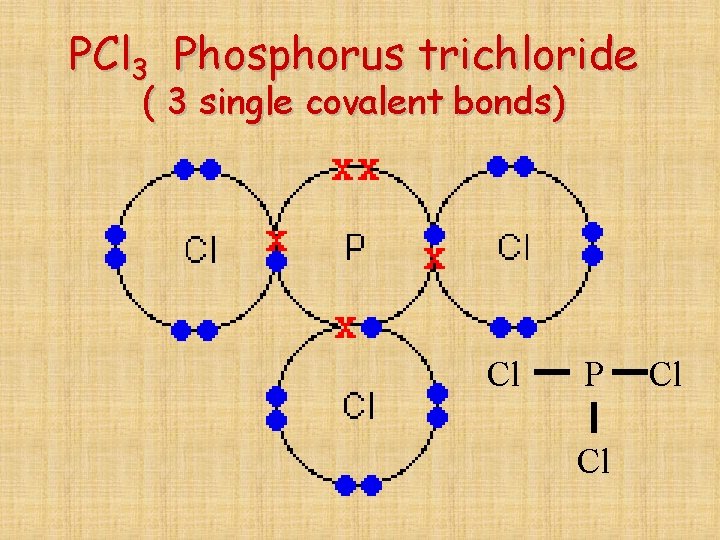
PCl 3 Phosphorus trichloride ( 3 single covalent bonds) Cl P Cl
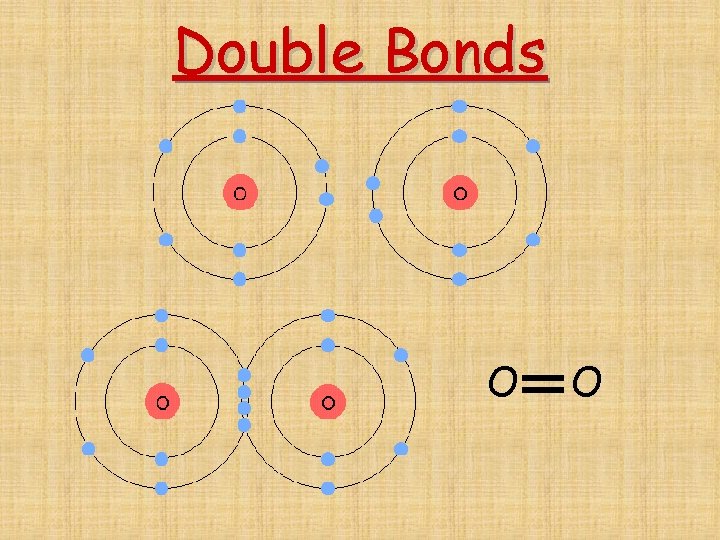
Double Bonds O O
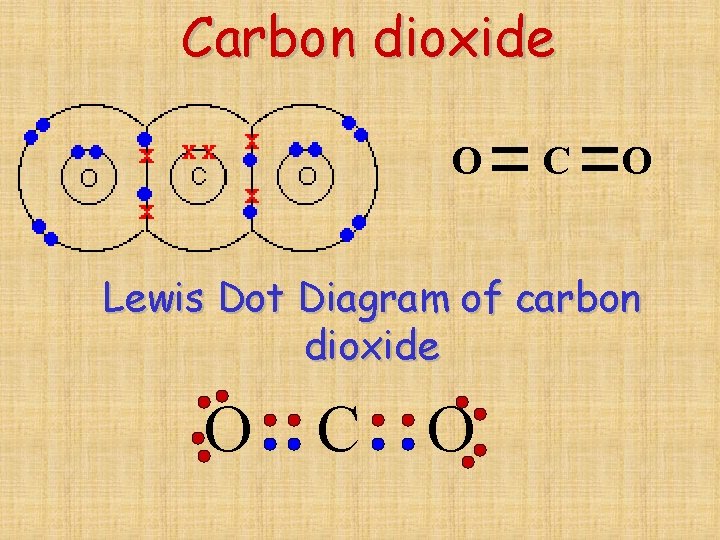
Carbon dioxide O C O Lewis Dot Diagram of carbon dioxide O C O
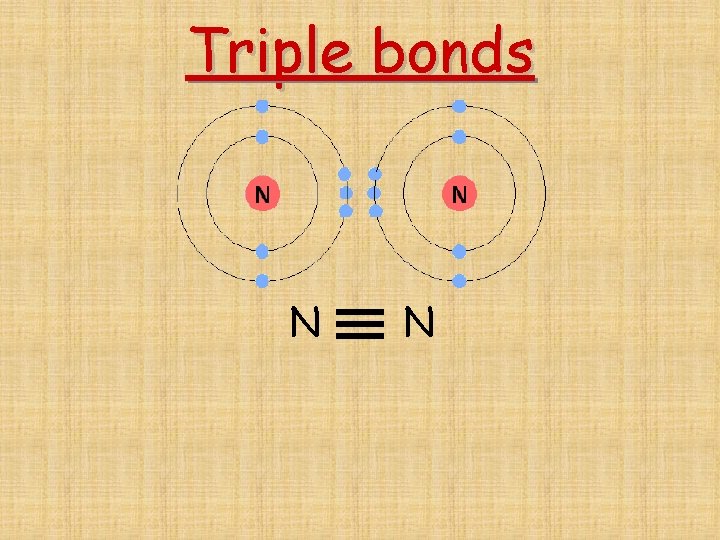
Triple bonds N N

Coordinate Covalent Bonds • A coordinate covalent bond is formed when one atom donates both electrons to be shared.
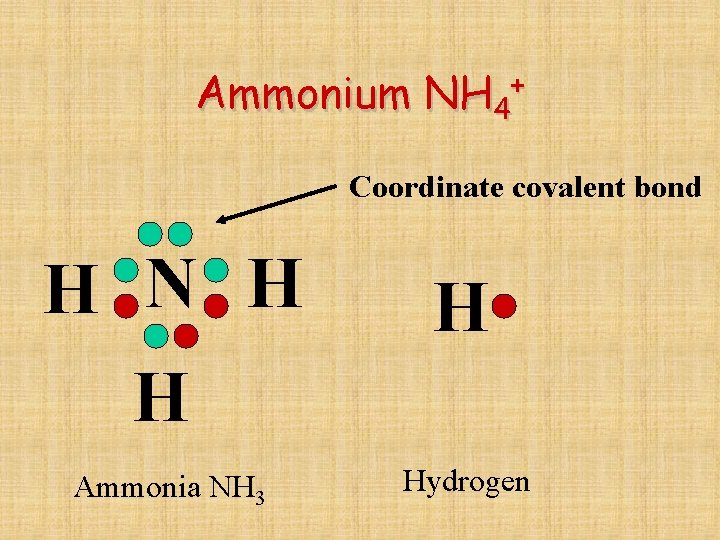
Ammonium + NH 4 Coordinate covalent bond H N H H H Ammonia NH 3 Hydrogen

In coordinate bond structural formula…… An arrow points from the atom donating the pair of electrons toward the atom receiving them. H H N H H

When drawing electron dot diagrams……. . • if an atom is “short” 2 electrons – try a coordinate covalent bond

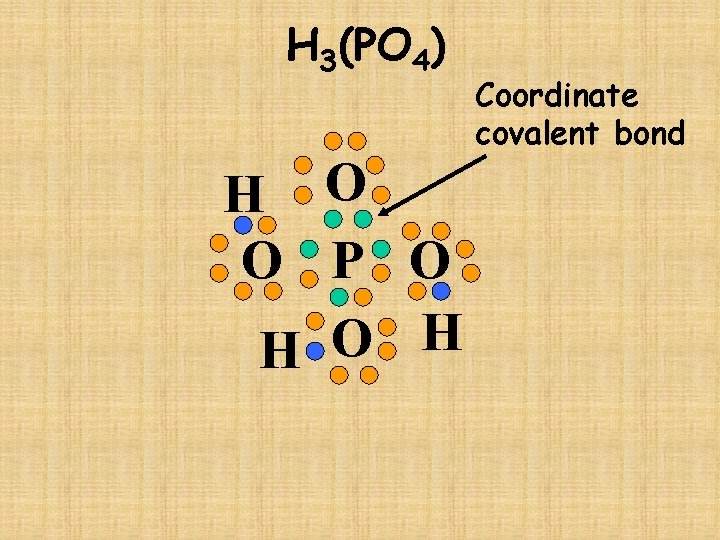
H 3(PO 4)
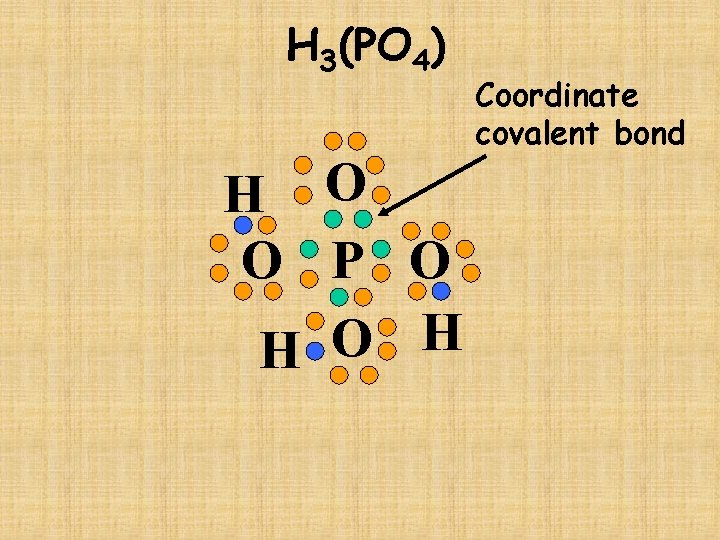
H 3(PO 4) H O O P O H Coordinate covalent bond

Bond Polarity • Nonpolar - When bonding electrons are equally shared (same electronegativity) • Ex. Diatomic elements • Polar – When one atom has higher electronegativity ( attractive force) and pulls electron closer to it. It gains a slight negative charge and the other atom gains a slight positive charge • Ex. H 2 O

Ws. What is Electronegativity?
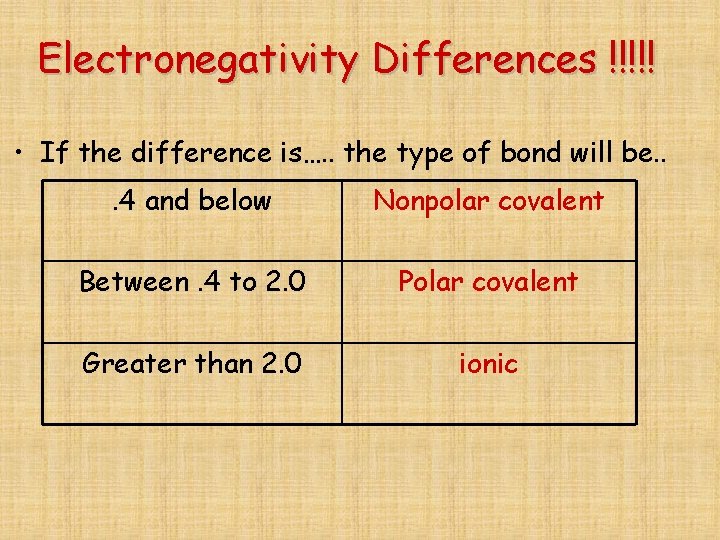
Electronegativity Differences !!!!! • If the difference is…. . the type of bond will be. . . 4 and below Nonpolar covalent Between. 4 to 2. 0 Polar covalent Greater than 2. 0 ionic

Describing each bond in the molecule as …. • Even pull= Nonpolar Uneven pull = polar Strong uneven pull = ionic

Showing polarity of individual bonds Use differences in electronegativities Indicated with δ ( lower case Greek letter delta) and charge.
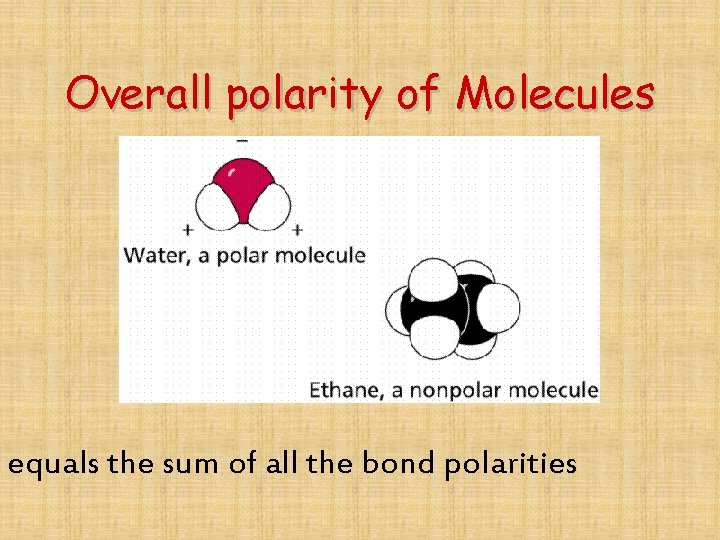
Overall polarity of Molecules equals the sum of all the bond polarities
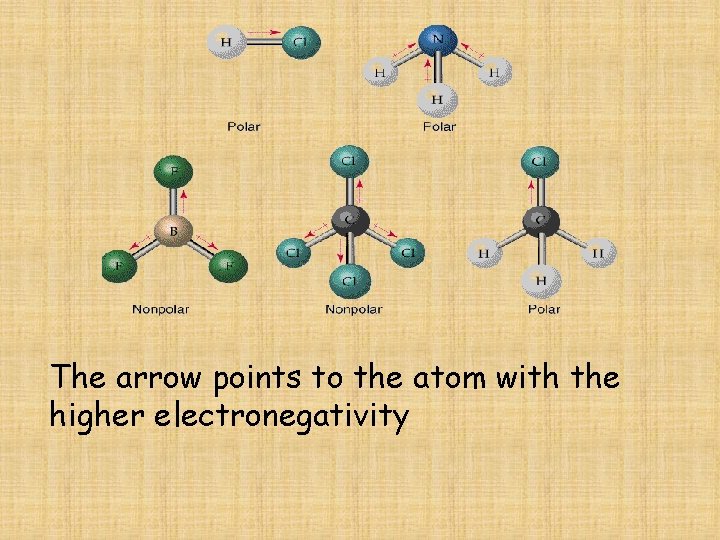
The arrow points to the atom with the higher electronegativity

Symmetry in Molecules • Symmetrical molecules are usually nonpolar. Polar bonds cancel each other out • Ex. CO 2
Carbon Tetrachloride
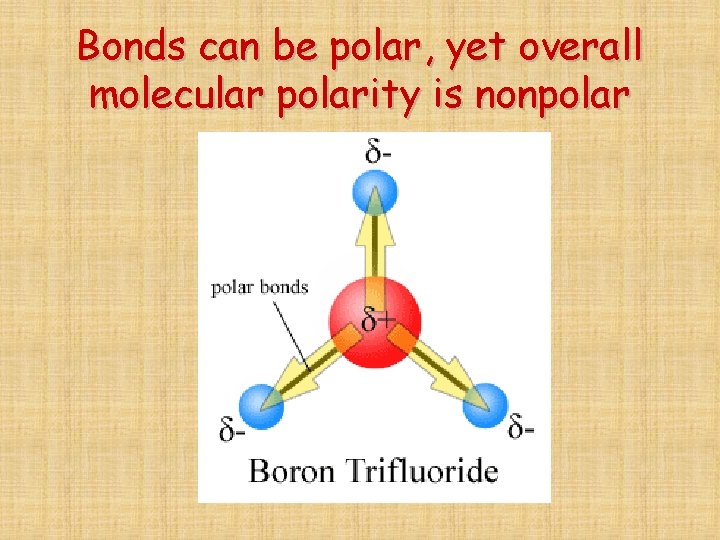
Bonds can be polar, yet overall molecular polarity is nonpolar

Asymmetrical Molecules • Asymmetrical molecules are polar if there are polar bonds in the structure. • Ex. H 2 O
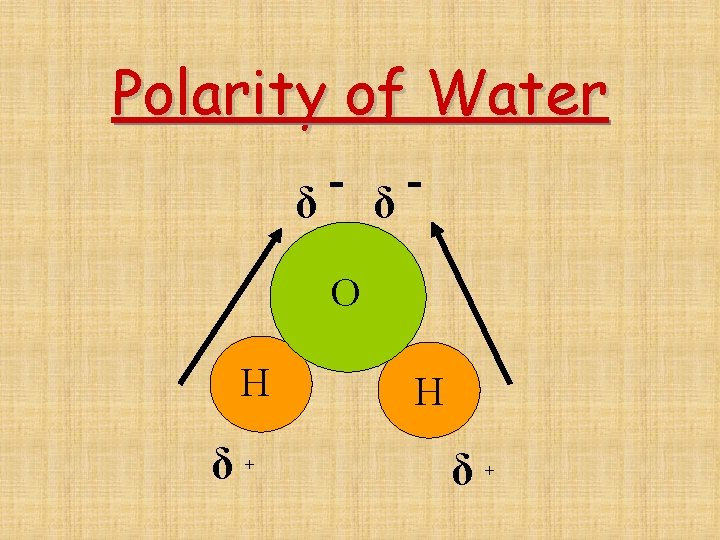
Polarity of Water δ δ O H δ+ H δ+

VSEPR Theory • Valence shell electron-pair repulsion theory • Because electron pairs repel, molecules adjust shapes so that pairs are as far away from each other as possible

Domains • • Single bond Double bond Triple bond Lone unshared pair

The # of Electron Domains determines the Basic Electron Domain shape • • • Linear – 1 or 2 domains Trigonal Planar – 3 domains Tetrahedral – 4 domains Trigonal bipyramidal – 5 domains Octrahedral – 6 domains
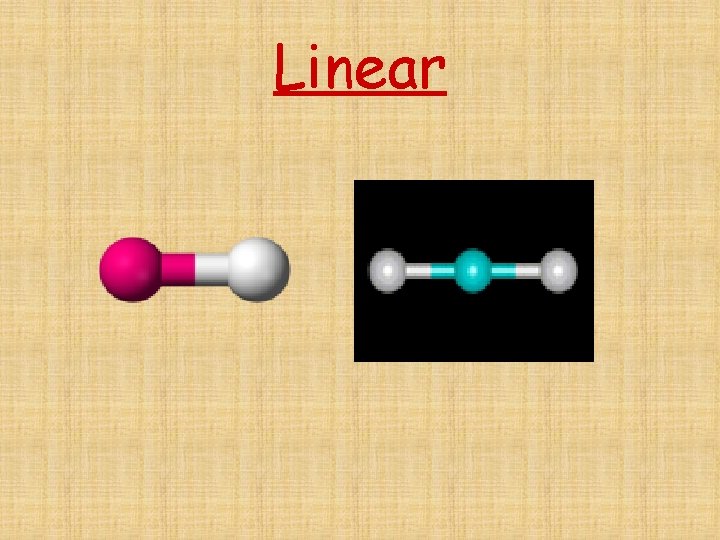
Linear
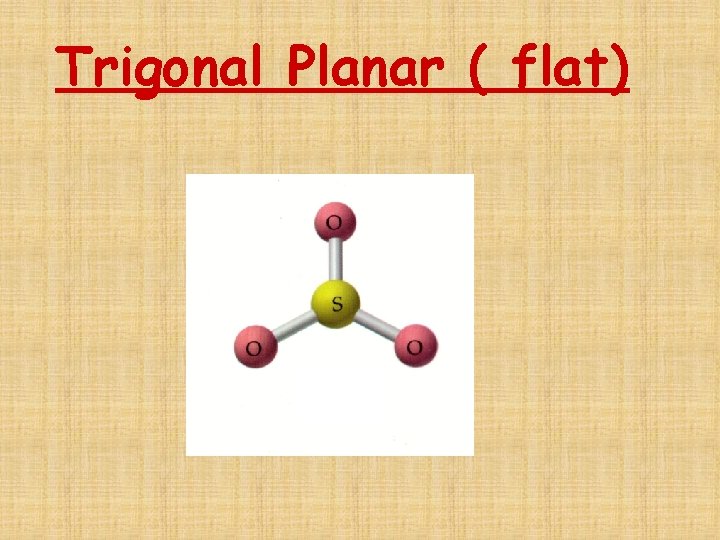
Trigonal Planar ( flat)
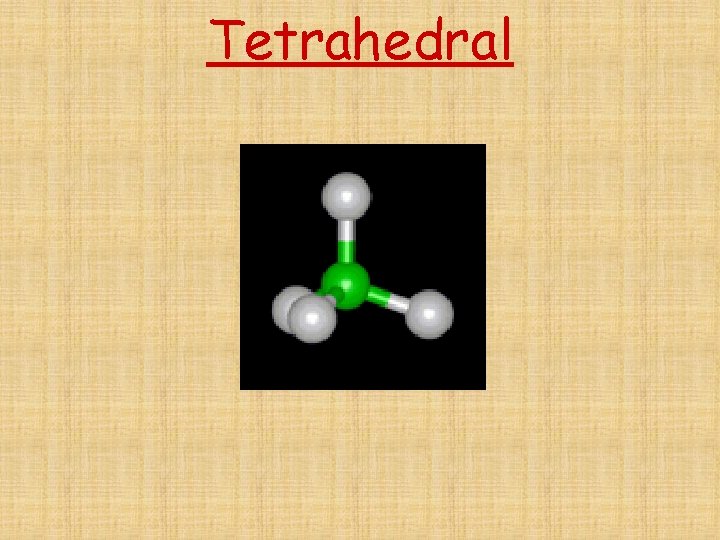
Tetrahedral
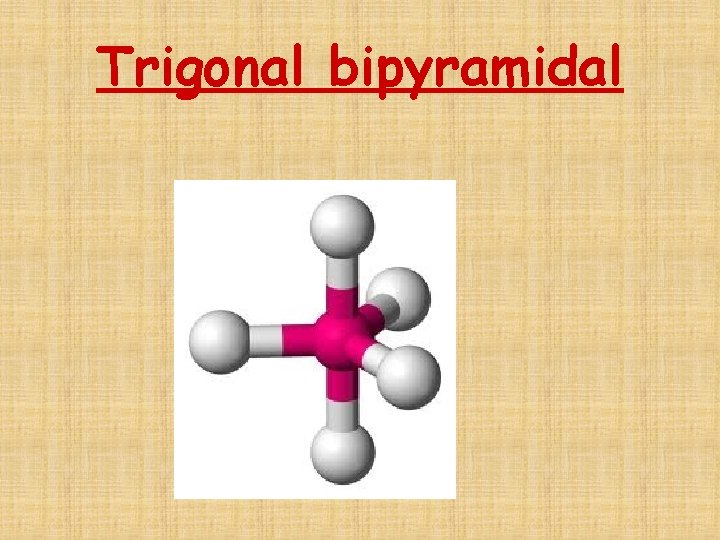
Trigonal bipyramidal
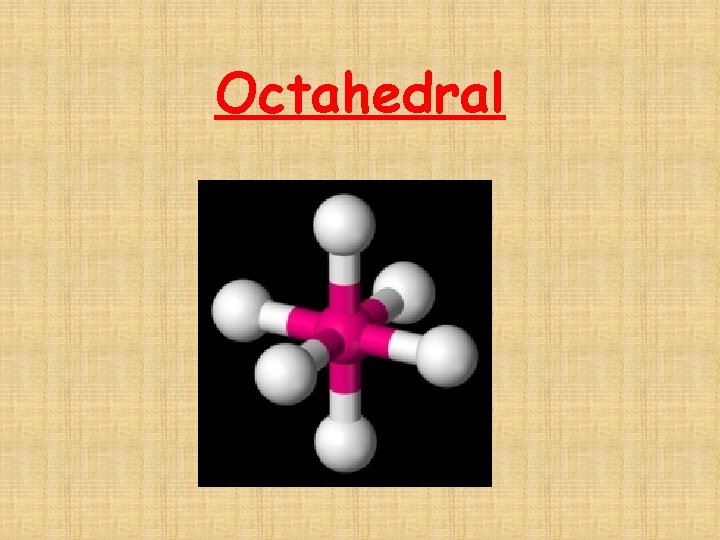
Octahedral

Determining the Electron Domain Shape • Count up all valence electrons • Start with central atom (C, or Lowest EN, never H) • Place other atoms around with maximum repulsion • Put 8 electrons around outside atoms • Any extra electrons go around the central atom • Count the number of domains to determine the basic shape • **** some compounds do NOT follow the octet rule

Let’s try some • • • CS 2 Be. H 2 CO 2 CH 4 CCl 4 NO 3 NH 4+ Si. F 4 SF 4

Molecular geometry • Shapes are altered if there are non- bonding pairs of electrons surrounding the central atom
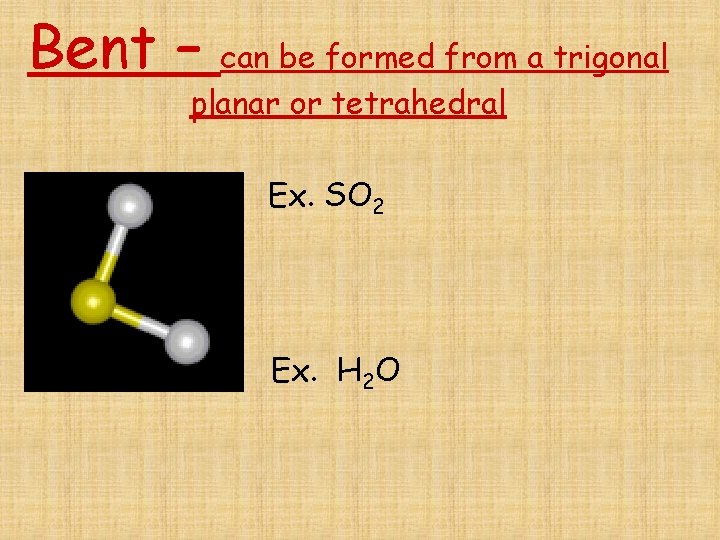
Bent – can be formed from a trigonal planar or tetrahedral Ex. SO 2 Ex. H 2 O
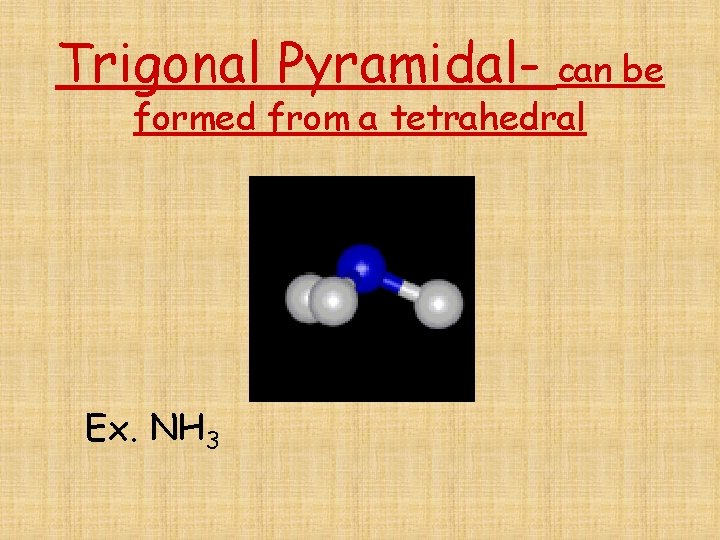
Trigonal Pyramidal- can be formed from a tetrahedral Ex. NH 3

Ws. Electron Domain and Molecular Geometries
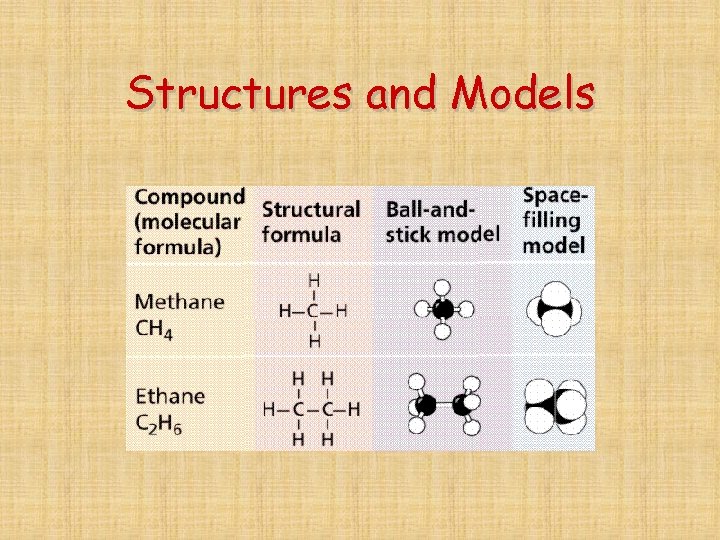
Structures and Models

Covalent Bonding: Molecular Model Building Lab

Model Colors • • • Black – Carbon family Blue – Nitrogen family Red - Oxygen family Green or Purple - Halogens Yellow - Hydrogen

• Use sticks to show single bonds • Use springs to show multiple bonds
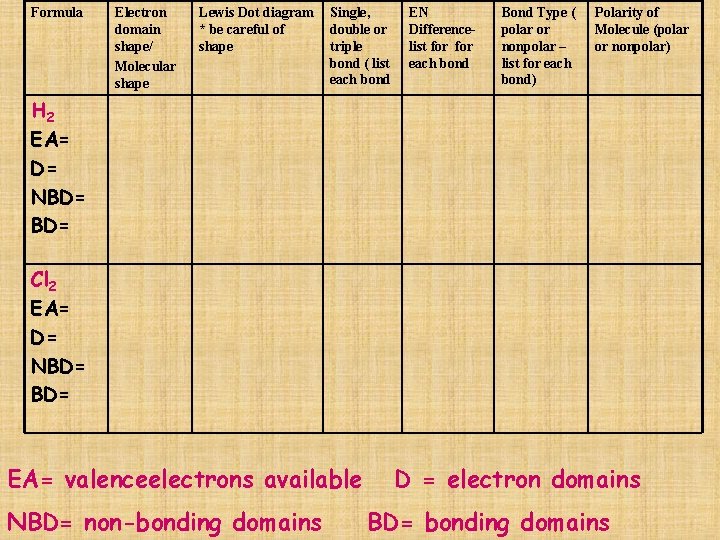
Formula Electron domain shape/ Molecular shape Lewis Dot diagram * be careful of shape Single, double or triple bond ( list each bond EN Differencelist for each bond Bond Type ( polar or nonpolar – list for each bond) Polarity of Molecule (polar or nonpolar) H 2 EA= D= NBD= Cl 2 EA= D= NBD= EA= valenceelectrons available NBD= non-bonding domains D = electron domains BD= bonding domains

Electron –Domain Basic Bond Shapes • • • Linear Bent Trigonal Planar Trigonal Pyramidal Tetrahedral

Hints for Lab

Start with Central Atom • If it has carbon, use “C” as central atom • If no carbon, use single atom or atom with lowest EN as central atom • Never put hydrogen as central atom

Hints about the Acids • Do polyatomic ion groups first, then add the hydrogens • Use single atom, except Hydrogen, as central atom • Usually contain coordinate covalent bonds

Last Hint !!!! • If an atom is “short” 2 electrons – try a coordinate covalent bond
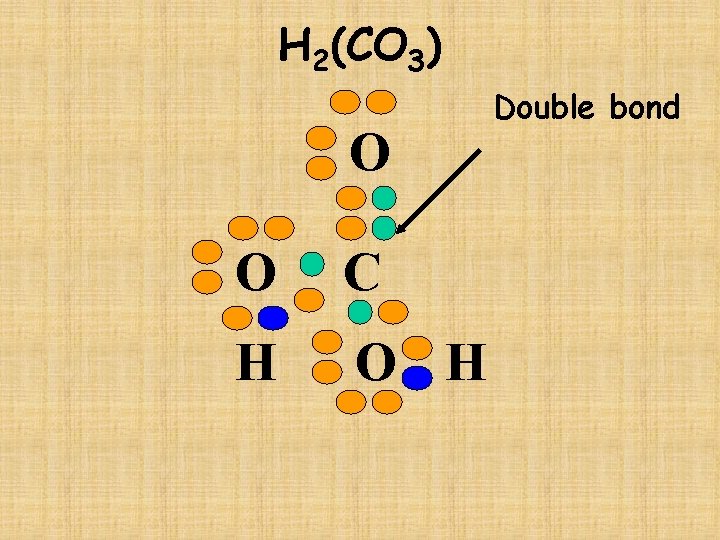
H 2(CO 3) O O C H O H Double bond
H 3(PO 4) H O O P O H Coordinate covalent bond
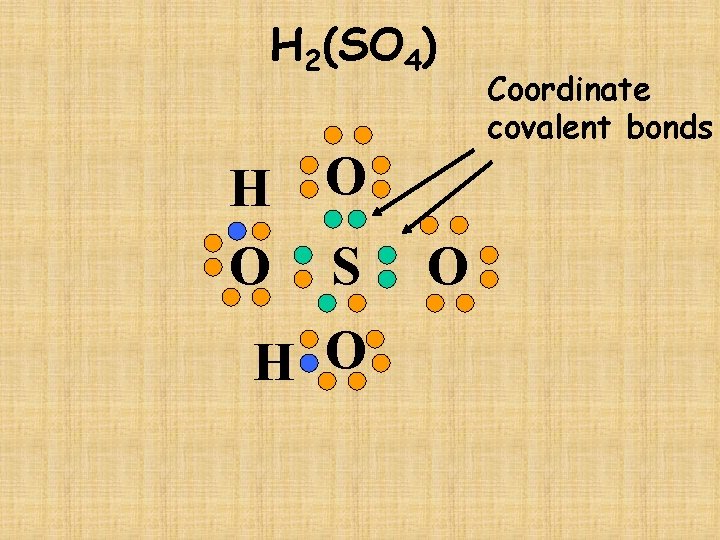
H 2(SO 4) H O O S H O O Coordinate covalent bonds
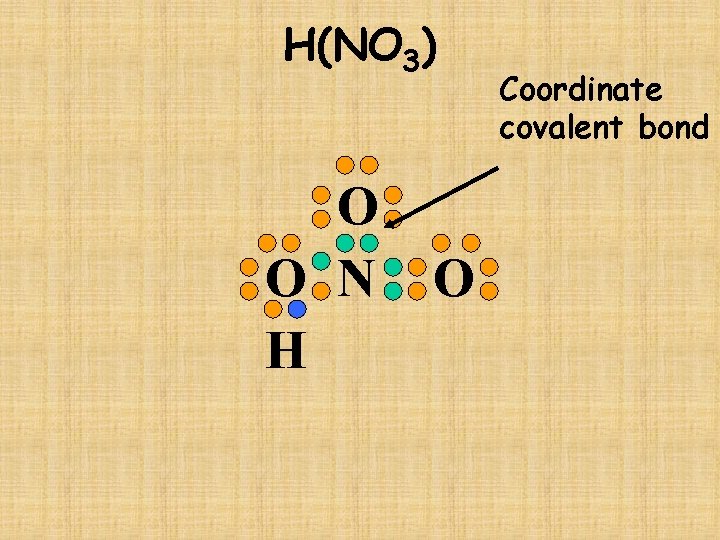
H(NO 3) O O N H O Coordinate covalent bond

Properties of Covalent Compounds • Low melting points (usually below 300 o. C) • Boiling points usually lower than ionic compounds • Many are liquids or gases at room temperature • High to low solubility • Poor to non-conducting

Covalent Compounds form Network Solids • Most covalently bonds substances melt easily because you just have to break the weak attractions between molecule and molecule • Ex. Ice – Water – Sugar (C 12 H 22 O 11)

Covalent crystals • If covalent compound forms crystals (repeating pattern)……. . • Requires a lot of energy to break all the covalent bonds • Have high melting points • Ex. diamonds
A diamond ( pure carbon)

Solubility of Covalent Compounds • “Like dissolves Like” • Polar compounds will dissolve in polar solvents • Nonpolar compounds will dissolve in nonpolar solvents

Why don’t water and oil mix? -Water is polar, oil is nonpolar. -Water is more attracted to itself than to the oil. Why do detergents cause water and oil to mix? -detergents are long molecules with one polar and one nonpolar end. -the water is attracted to the polar end and the oil is attracted to the nonpolar end.
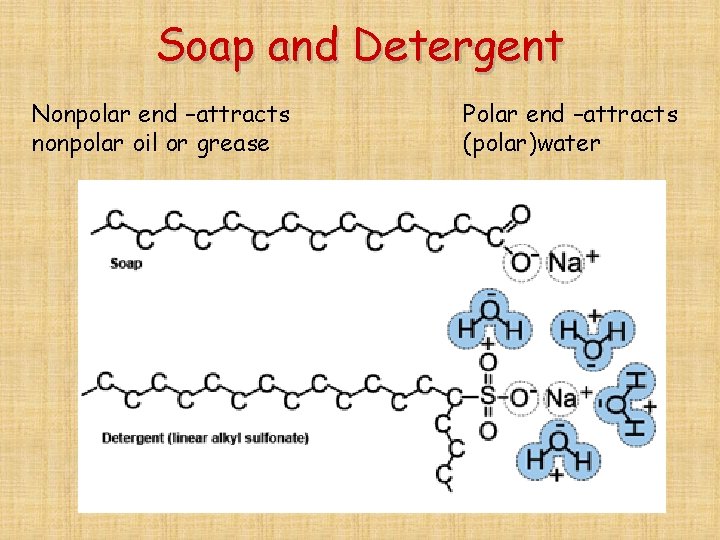
Soap and Detergent Nonpolar end –attracts nonpolar oil or grease Polar end –attracts (polar)water

Chapter Review Covalent Bonding
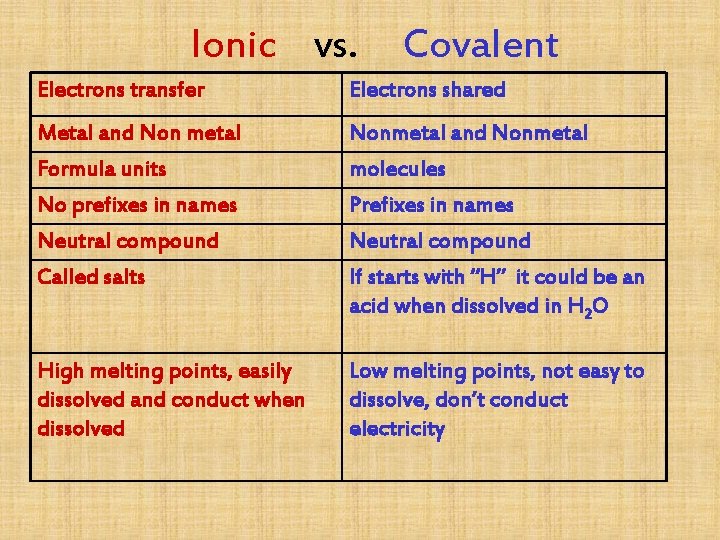
Ionic vs. Covalent Electrons transfer Electrons shared Metal and Non metal Formula units No prefixes in names Neutral compound Nonmetal and Nonmetal molecules Prefixes in names Neutral compound Called salts If starts with “H” it could be an acid when dissolved in H 2 O High melting points, easily dissolved and conduct when dissolved Low melting points, not easy to dissolve, don’t conduct electricity

Ws. Bond Prediction Ws. Naming Compounds Ws. Name the Following Compounds Ws. Naming Compounds Reference Sheet

Diatomics = 2 atoms of same element bonded to each other Br. INCl. HOF Diatomic element is linear and Nonpolar

Covalent Bonds – 4 Types • Single- 1 shared pair of electrons (1 from each atom) • Double – 2 shared pairs of electrons (two from each atom) • Triple- 3 shared pairs of electrons, (three from each atom) • Coordinate covalent – both electrons of shared pair come from 1 atom

Structural Formula shows arrangement of bonds • • Cl Cl single O O double N N triple C O coordinate covalent

Electron Dot Diagrams • Every atom MUST have 8 electrons drawn around it except “H”. • “H” needs 2 electrons

Polar vs. Nonpolar – depends on EN ( electronegativity) difference • Nonpolar – shared evenly – EN difference below. 4 • Polar – shared unevenly – EN. 4 – 2. 0 • Ionic – pulls electron off of atom. EN above 2. 0

Polarity of Bond • Depends on electronegativity difference

Polarity of Molecule • Look at symmetry, then polarity of the bonds

Know how to name ionic compounds, covalent compounds and acids

The End
- Slides: 110