Chapter 8 Covalent Bonding 8 1 The Covalent
Chapter 8 – Covalent Bonding 8. 1 The Covalent Bond 8. 2 Naming Molecules 8. 3 Molecular Structures 8. 4 Molecular Shape 8. 5 Electronegativity and Polarity Topic is Lewis Structures (combination of material found in 2 sections)

Sections 8. 1/8. 3 Covalent Bond/Molecular Structures Atoms gain stability when they share electrons and form covalent bonds. The sharing can be described by the Lewis structure of the compound. • Apply the octet rule to atoms that form covalent bonds. • Describe the similarities and differences between ionic and covalent bonding. • Describe the forces that act and energy changes that occur when atoms form a molecule.

Sections 8. 1/8. 3 Covalent Bond/Molecular Structures • Categorize valance electrons as bonding or nonbonding. • List the basic steps used to draw Lewis structures. • Describe the formation of single, double, and triple covalent bonds using Lewis structures. • Explain why resonance occurs, and identify resonance structures. • Draw valid Lewis structures for molecules, including those involving multiple bonds, resonance, expanded octets, and electron deficient molecules.

Why Atoms Bond Noble gas configuration especially stable • ns 2 np 6 (except for He) • Full outer energy level • Octet of electrons Atoms bond to achieve a lower energy state (more stability)

Ionic vs Covalent Bonding In ionic bonding, electrons transferred to achieve octet for each ion • Number of ionic compounds small compared to total number of known compounds In covalent bonding, electrons shared to achieve octet (mostly) for each atom

Covalent Bonding In covalent bonding, electrons shared to achieve octet (mostly) for each atom • Sharing occurs when electronegativities of atoms same or similar • Majority of covalent bonds formed between nonmetallic elements • Electronegativity difference < 1. 7 (see next slide) – bond will have more covalent character than ionic character
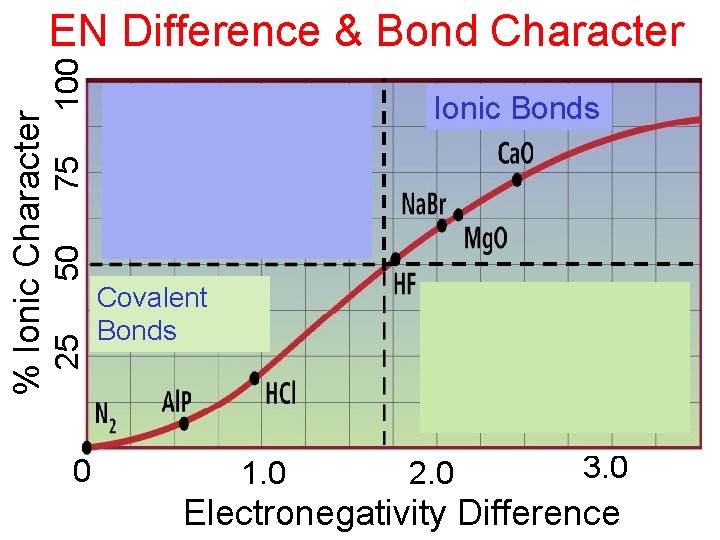
100 75 Ionic Bonds 50 25 % Ionic Character EN Difference & Bond Character 0 Covalent Bonds 1. 0 2. 0 3. 0 Electronegativity Difference

Covalent Bonding In covalent bonding, electrons shared to achieve octet (mostly) for each atom Molecule formed when 2 or more atoms bond covalently
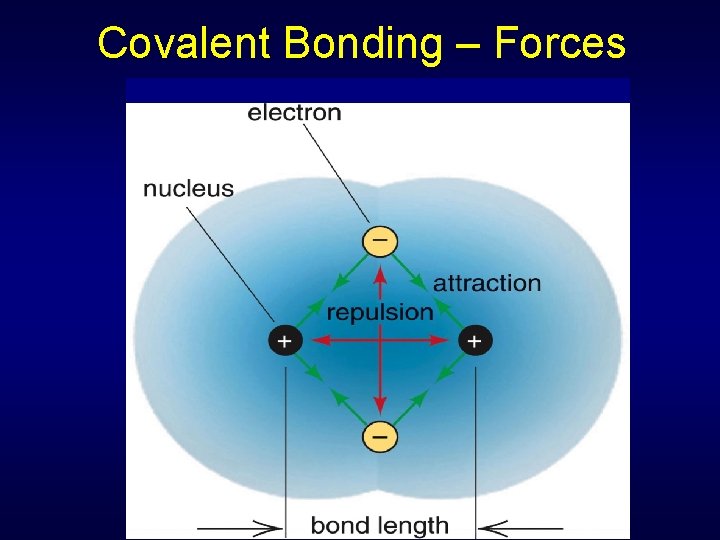
Covalent Bonding – Forces

Covalent Bonding – Forces No interaction Nucleus attracted to other atoms electrons – Not optimum distance Nucleus attracted to other atoms electrons – optimum distance Net repulsion from positive nuclei
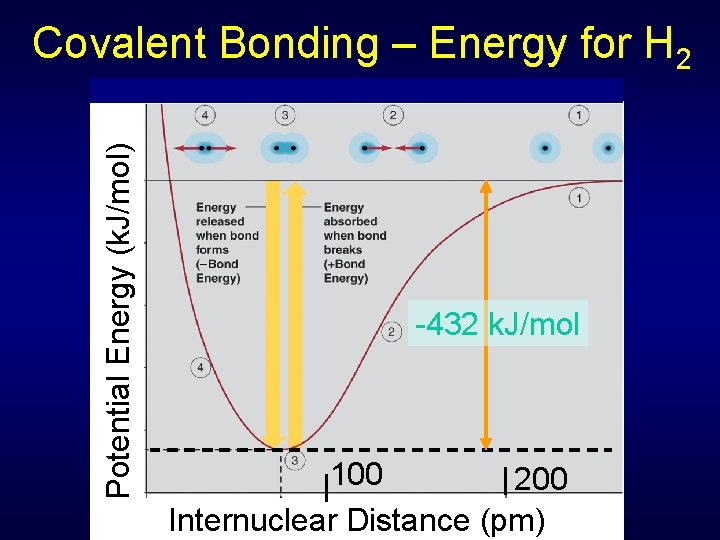
Potential Energy (k. J/mol) Covalent Bonding – Energy for H 2 -432 k. J/mol 100 200 Internuclear Distance (pm)

Lewis Structures - Atoms (Electron Dot Diagrams) Way of keeping track of valence electrons To write for atom • Write symbol for element • Put one dot for each valence electron • Don’t pair up until you have to (Hund’s rule) X

Lewis Structure - Covalent Molecules Valence electrons of each element in molecule are divided into 2 categories: • Bonding – pair of electrons shared by two atoms to form the covalent bond ² Shared pair represented by a line connecting the element symbols H—H • Nonbonding – called lone pairs ² A few molecules have odd # total electrons – have unpaired nonbonding electron
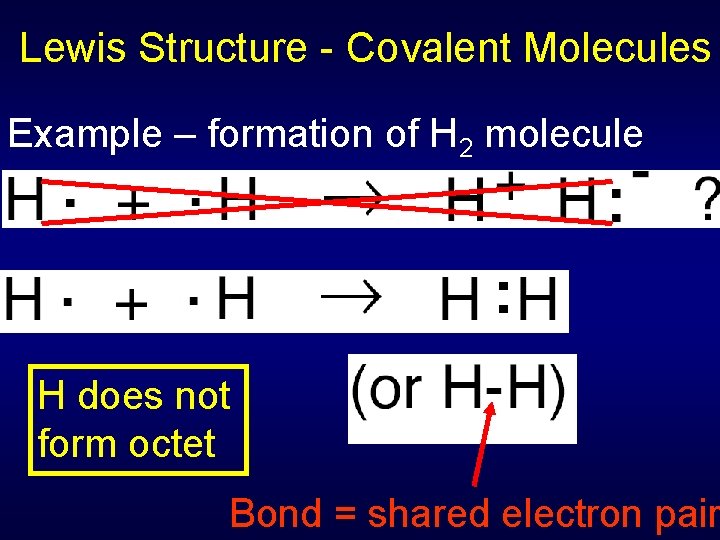
Lewis Structure - Covalent Molecules Example – formation of H 2 molecule H does not form octet Bond = shared electron pair
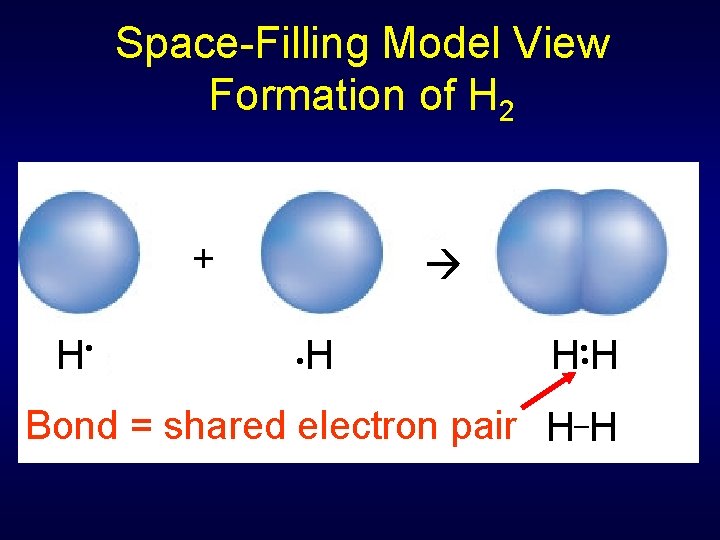
Space-Filling Model View Formation of H 2 + H • • H _ Bond = shared electron pair H H
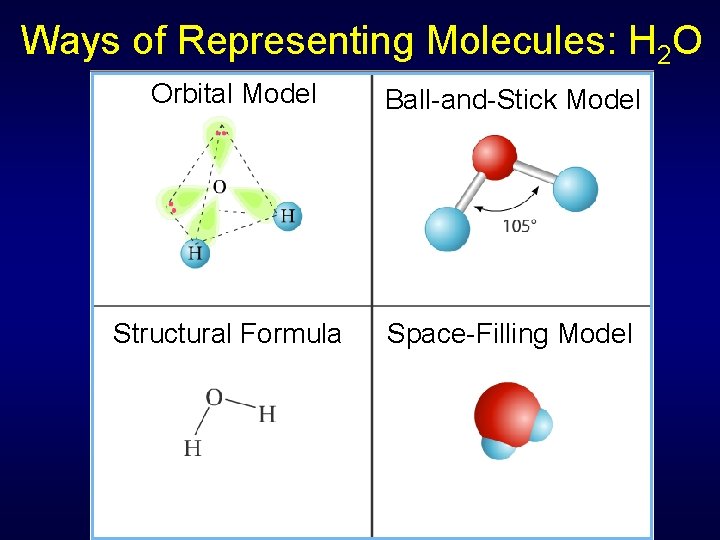
Ways of Representing Molecules: H 2 O Orbital Model Ball-and-Stick Model Structural Formula Space-Filling Model
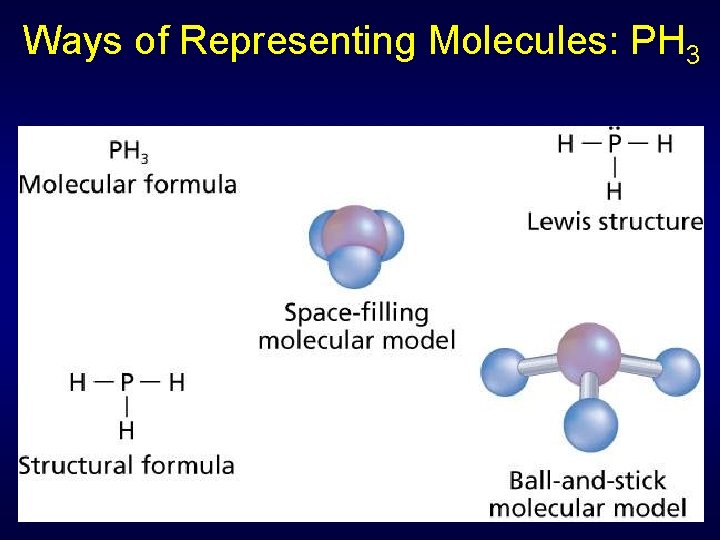
Ways of Representing Molecules: PH 3

Covalent Bonding – F 2 F 1 s 22 p 5 7 valence electrons Forms F 2 molecule Each F shares 1 valence electron Molecule is more stable than individual atoms
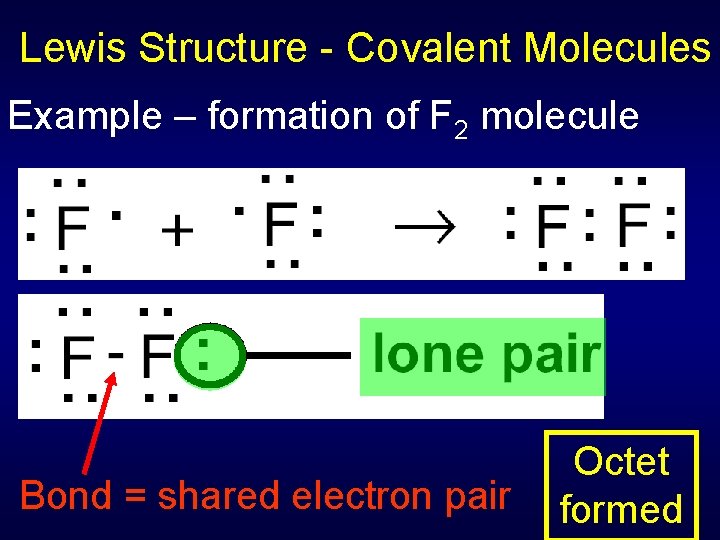
Lewis Structure - Covalent Molecules Example – formation of F 2 molecule Bond = shared electron pair Octet formed
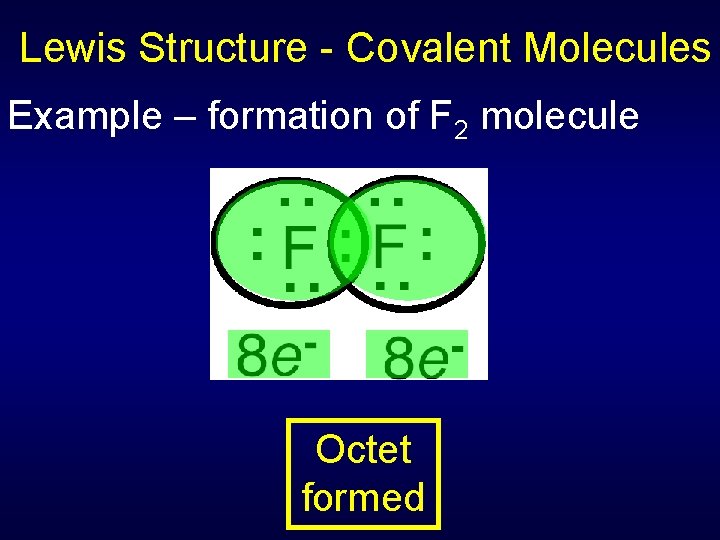
Lewis Structure - Covalent Molecules Example – formation of F 2 molecule Octet formed
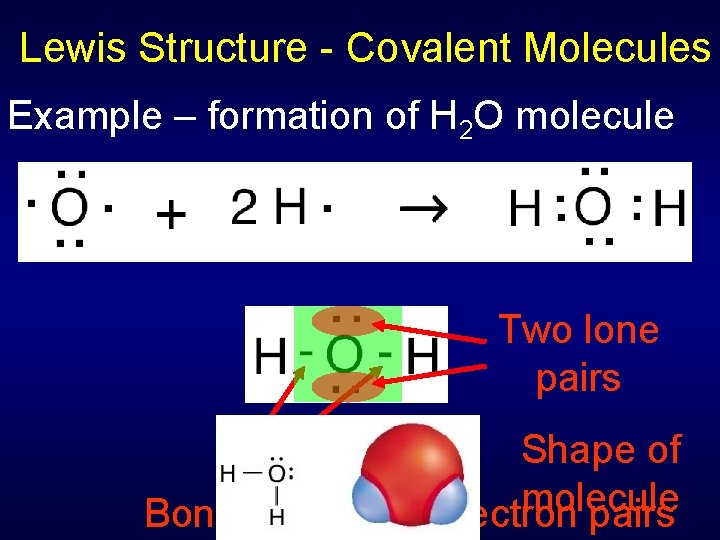
Lewis Structure - Covalent Molecules Example – formation of H 2 O molecule Two lone pairs Shape of Octet molecule formed Bonds = shared electron pairs
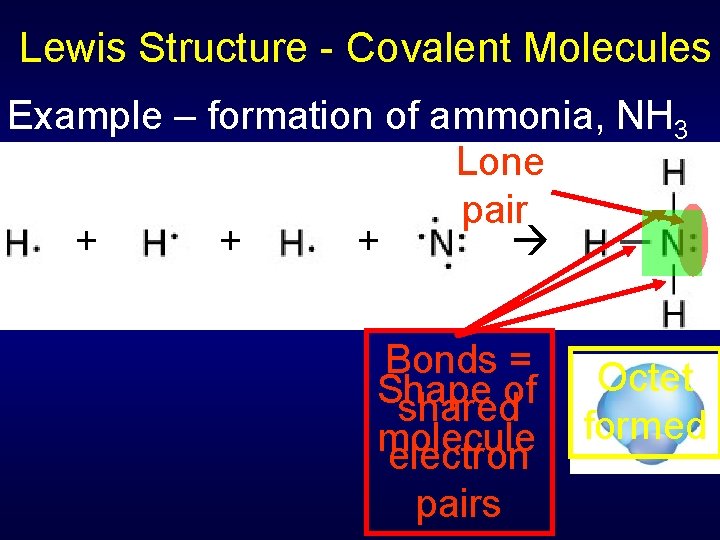
Lewis Structure - Covalent Molecules Example – formation of ammonia, NH 3 Lone pair + + + Bonds = Shape of shared molecule electron pairs Octet formed
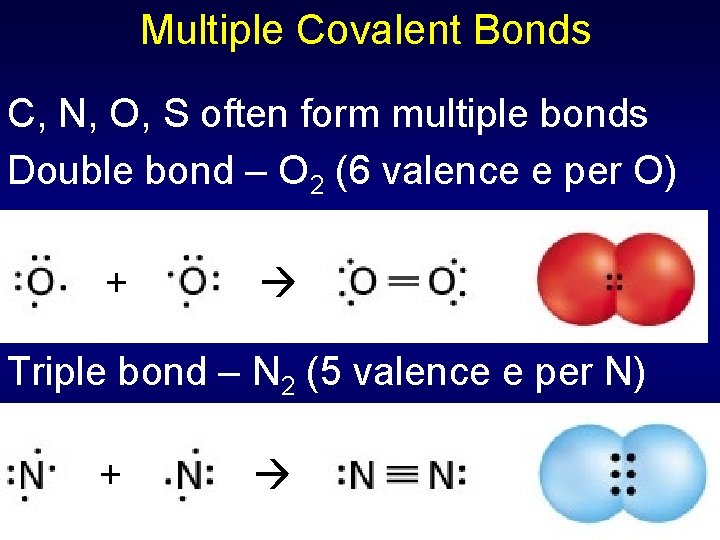
Multiple Covalent Bonds C, N, O, S often form multiple bonds Double bond – O 2 (6 valence e per O) + Triple bond – N 2 (5 valence e per N) +

Guide for Writing Lewis Structures Similar to procedure on p. 254, but without # of bonding pairs Step 1 – Write skeletal structure • least electronegative atom usually occupies central position Step 2 – Count total number of valence electrons • polyatomic anions, add # of - charges ² e. g. CO 32 - add 2 electrons to total • polyatomic cations, subtract # of + charges

Step 3 – Place single bond between central atom and surrounding atoms Step 4 – Complete octet for terminal atoms (not for H) Step 5 – Add remaining to central atom Step 6 – If octet rule not satisfied for central atom, add multiple (double, triple) bonds between terminal and central atom, using the lone pairs from the terminal atoms
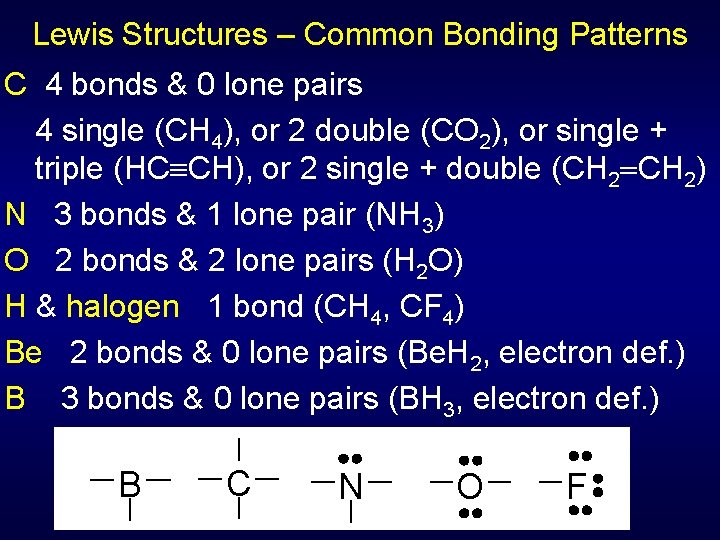
Lewis Structures – Common Bonding Patterns C 4 bonds & 0 lone pairs 4 single (CH 4), or 2 double (CO 2), or single + triple (HC CH), or 2 single + double (CH 2) N 3 bonds & 1 lone pair (NH 3) O 2 bonds & 2 lone pairs (H 2 O) H & halogen 1 bond (CH 4, CF 4) Be 2 bonds & 0 lone pairs (Be. H 2, electron def. ) B 3 bonds & 0 lone pairs (BH 3, electron def. ) B C N O F

Lewis Structure Examples Total Valence Electrons a, HF 1+7= 8 Draw Single Bonds H-F Calculate Number of Electrons Remaining 6 Use Remaining Check Electrons to Achieve Noble Number of Electrons Gas Configuration H, 2 F, 8 b, N 2 5 + 5 = 10 c, NH 3 5 + 3(1) = 8 N-N 8 N, 8 2 H, 2 N, 8 d, CH 4 4 + 4(1) = 8 0 H, 2 C, 8 e, CF 4 4 + 4(7) = 32 24 F, 8 C, 8 N, 8 f, NO+ 5 + 6 - 1 = 10 N-O 8 O, 8

Practice Problems 1 -5 page 244 Problems 37 -38, page 255 Problems 39 -40, page 256 (mult bonds) Problems 41 -42 page 257 (ions) Problems 104(a-d), page 275 Problems 1(a-d), page 979 Problems 4(a-e) page 980

Lewis Structure Example: NO 3─ 1. Write skeletal structure N central because it is least electronegative 2. Count valence electrons N=5 3 O = 3 x 6 = 18 (-) = 1 Total = 24 e-

Example NO 3 3. Attach atoms with single bonds (pairs of electrons) & subtract from total Electrons Start 24 Used 6 Left 18 ─, Continued

Example NO 3─ , Continued 4. Complete octets, outside -in Keep going until all atoms have an octet or you run out of electrons Electrons Start 18 Used 18 Left 0

Example NO 3 ─, 5. If central atom does not have octet, bring in electron pairs from outside atoms to share If structure is an ion, use brackets and indicate the charge 6. For this ion an extra step is needed – draw resonance structures Continued -1
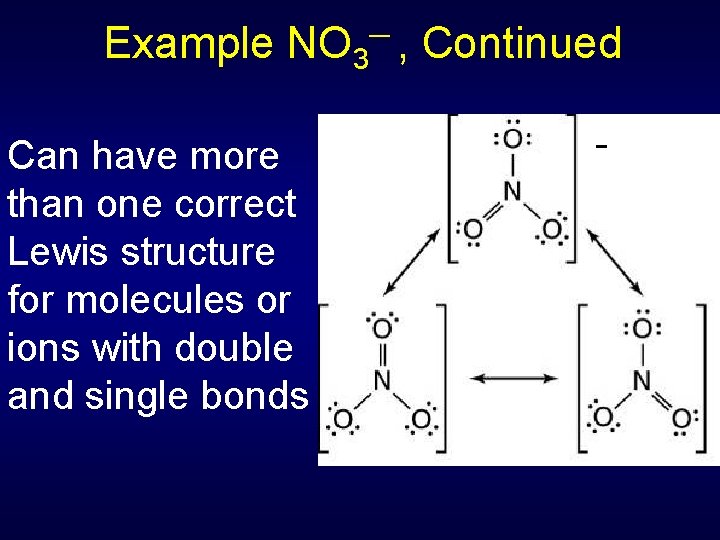
Example NO 3─ , Continued Can have more than one correct Lewis structure for molecules or ions with double and single bonds -

Resonance Structures Resonance structures differ only in position of electron pairs, never the atom positions Molecule behaves as if it had only one structure (the average one) • NO 3 - has all bond lengths identical -

Practice (Resonance Structures) Problems 43 -46 page 258 Problems 101, 103 pages 274 -5 Problems 5, 6 page 980
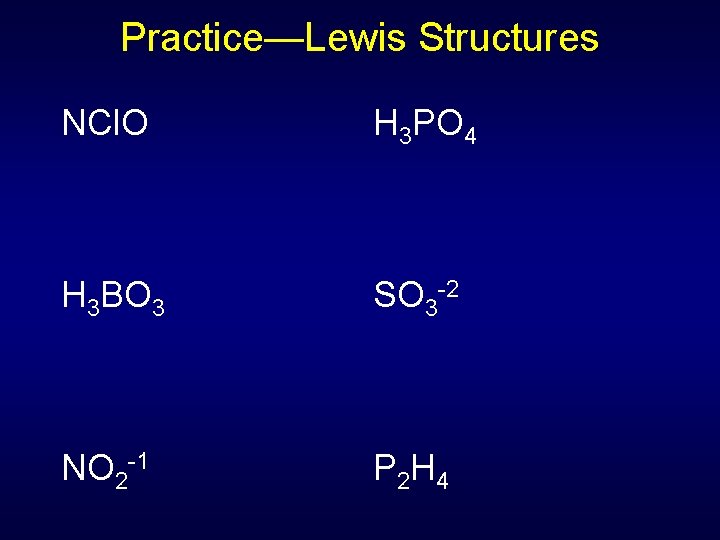
Practice—Lewis Structures NCl. O H 3 PO 4 H 3 BO 3 SO 3 -2 NO 2 -1 P 2 H 4
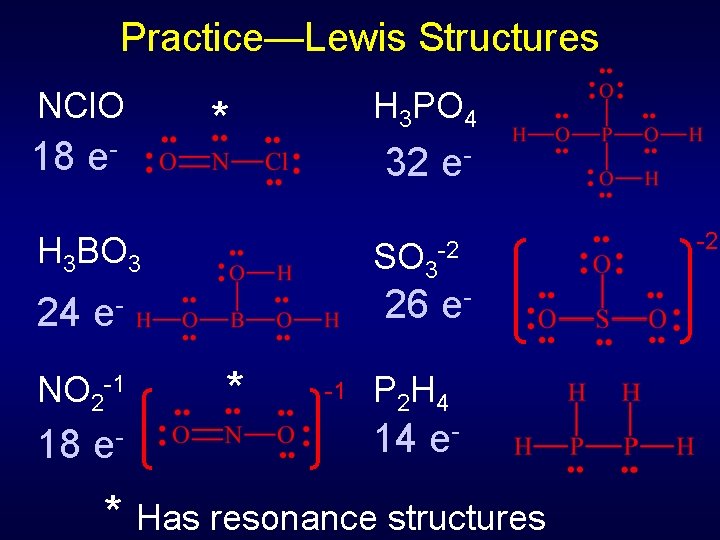
Practice—Lewis Structures NCl. O 18 e- H 3 PO 4 * 32 e- H 3 BO 3 24 26 e- e- NO 2 -1 18 SO 3 e- -2 * -1 P 2 H 4 14 e- * Has resonance structures -2

Exceptions to Octet Rule Molecules with odd number of total valence electrons NO 2 – 17 valence electrons Also Cl. O 2, NO

Exceptions to Octet Rule Electron deficient – form with fewer than 8 electrons around atom • Be, B • Rare Tend to form coordinate covalent bonds – both electrons in shared pair donated by single atom +
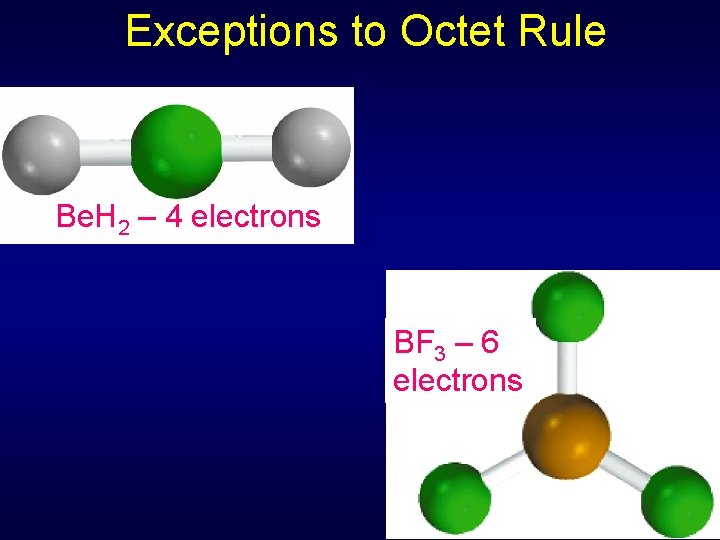
Exceptions to Octet Rule Be. H 2 – 4 electrons BF 3 – 6 electrons

Exceptions to Octet Rule More than 8 valence electrons = expanded octet PCl 5 SF 6 d orbitals involved • Only can occur for period 3 and higher, not periods 1 or 2

Practice (Octet Exceptions) Problems 47 - 49 page 260 Problems 102 (a-d), 104(a-d) page 273 Problem 7, page 980

Chapter 8 – Covalent Bonding 8. 1 8. 2 8. 3 8. 4 8. 5 The Covalent Bond Naming Molecules Molecular Structures Molecular Shape Electronegativity and Polarity

Section 8. 2 Naming Molecules Specific rules are used when naming binary molecular compounds, binary acids, and oxyacids. • Translate molecular formulas into binary molecular compound names and also the reverse process. • Name acidic solutions

Naming Binary Covalent Compounds First element named first, using entire element name Second element named using same procedure as for ionic compounds – root of element name + ide ending Use prefixes except if first element = 1 • Drop final letter in prefix if precedes vowel • Carbon monoxide , not monooxide
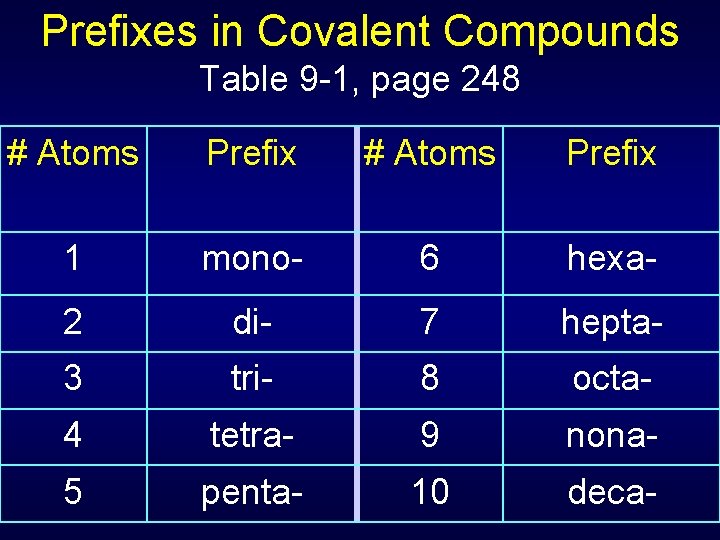
Prefixes in Covalent Compounds Table 9 -1, page 248 # Atoms Prefix 1 mono- 6 hexa- 2 di- 7 hepta- 3 tri- 8 octa- 4 tetra- 9 nona- 5 penta- 10 deca-

Naming Binary Covalent Compounds Name of Al. Cl 3 ? Aluminum chloride Name of PCl 3 ? Phosphorus trichloride Name of Al 2 O 3? Aluminum oxide Name of P 2 O 5 ? Diphosphorus pentoxide The naming systems for ionic and covalent compounds are different!!!

Common Names Table 9 -2, page 249 Form ula Common Name Molecular Compound Name H 2 O Water Dihydrogen monoxide NH 3 Ammonia Nitrogen trihydride N 2 H 4 Hydrazine Dinitrogen tetrahydride N 2 O Nitrous oxide Dinitrogen monoxide (laughing gas) NO Nitric oxide Nitrogen monoxide

Naming Acids For our purposes, acids are what result when molecules dissolved in water produce H+ (hydrogen ions) • HCl(g) in water H+(aq) + Cl-(aq) • Product is hydrochloric acid Two common types • Binary – H and one other element • Oxyacid – H and an oxyanion

Naming True Binary Acids Use prefix hydro- to name hydrogen part of compound For remainder, use a “form of the root” of 2 d element plus suffix –ic followed by word acid HCl – hydrochloric acid H 2 S – hydrosulfuric acid • Root of S for acid name not “sulf” as in Na 2 S (sodium sulfide)

Naming Acids Similar to Binary Acids (Rare) If second part of compound is a polyatomic anion that does not contain oxygen (rare), use same system as for a true binary acid employing the root name for the anion CN- – cyanide anion HCN – hydrocyanic acid

Naming Oxyacids Name is based solely on the anion “A form of the root name of the anion” + suffix + acid Anion suffix Acid Suffix -ate -ic -ite -ous HNO 3 Nitric acid NO 3 - = nitrate HNO 2 Nitrous acid NO 2 - = nitrite

Naming Molecular Compounds Flow Chart, Fig 9 -9, page 251
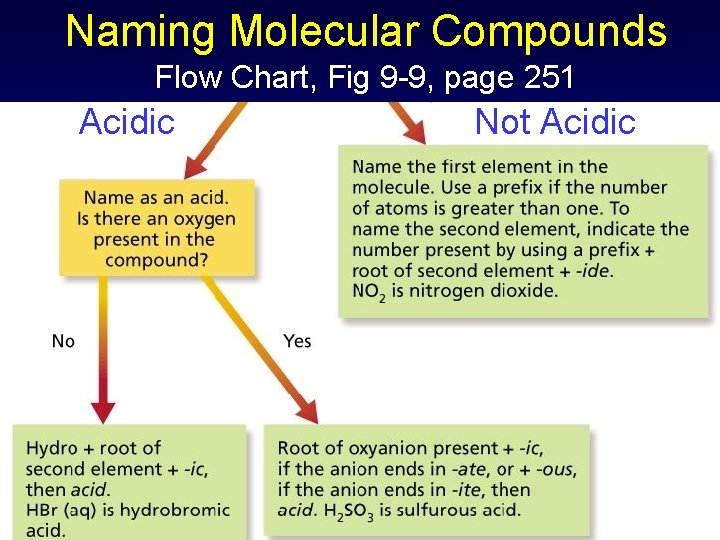
Naming Molecular Compounds Flow Chart, Fig 9 -9, page 251 Acidic Not Acidic

Practice Problems 13 -17 page 249 (binary covalent) Problems 18 -22 page 250 (acids) Problems 27 -29 page 251 (mixed) Problems 94 -96(all a-d) page 273 Problems 97 -98(all a-d) page 273 Problems 2 (a-f) page 874 (binary cov) Problem 3 page 875 (acids)

Chapter 8 – Covalent Bonding 8. 1 8. 2 8. 3 8. 4 8. 5 The Covalent Bond – Bond Strength Naming Molecules Molecular Structures Molecular Shape Electronegativity and Polarity

Section 8. 1 The Covalent Bond • Relate the strength of a covalent bond to its bond length, bond order, and bond dissociation energy. • Describe how the overall energy of a reaction (i. e. , whether it is an endo- or exothermic reaction) is related to the bond energies of the reactant and product molecules.

Potential Energy (k. J/mol) Covalent Bonding – Energy for H 2 -432 k. J/mol 100 200 Internuclear Distance (pm)

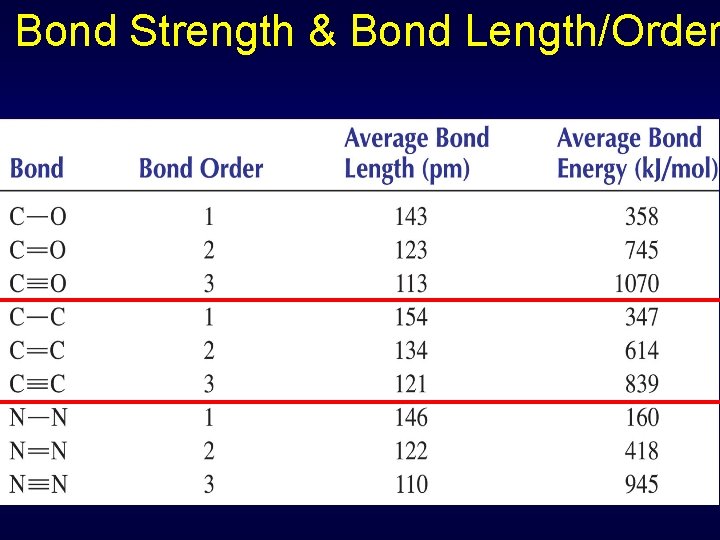
Bond Strength & Bond Length/Order Distance between bonding nuclei at position of max attraction = bond length Scale of bond length: ~10 -10 m =100 pm Bond order: Single 1 Double 2 Triple 3
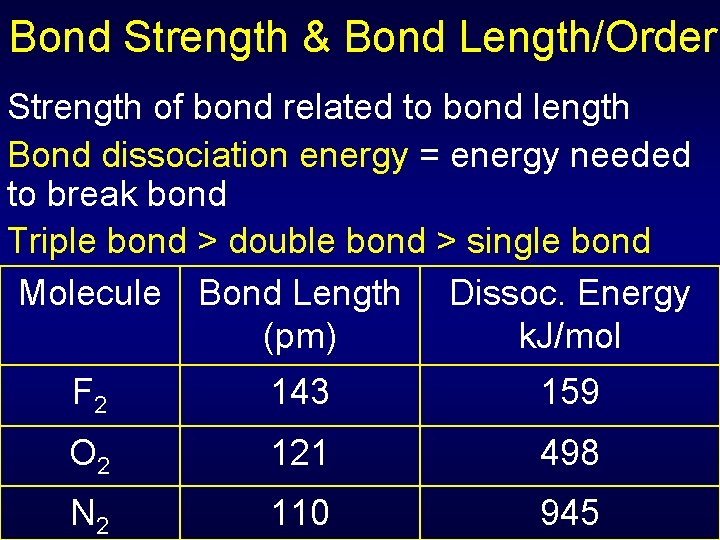
Bond Strength & Bond Length/Order Strength of bond related to bond length Bond dissociation energy = energy needed to break bond Triple bond > double bond > single bond Molecule Bond Length Dissoc. Energy (pm) k. J/mol F 2 143 159 O 2 121 498 N 2 110 945
Bond Strength & Bond Length/Order

Reaction Energies & Bond Energies Chemical reaction Bonds in reactant molecules broken New bonds formed in product molecules CH 4 + 2 O 2 2 H 2 O + CO 2 Breaking C-H bonds and O=O bonds Making O-H bonds and C=O bonds

Reaction Energies & Bond Energies CH 4(g) + 2 O 2(g) 2 H 2 O(g) + CO 2(g) Total energy change determined by difference of energy of bonds broken (reactant side) and formed (product side) • Endothermic – need more energy to break than get back in formation • Exothermic – bond formation energy larger than energy needed to break bonds
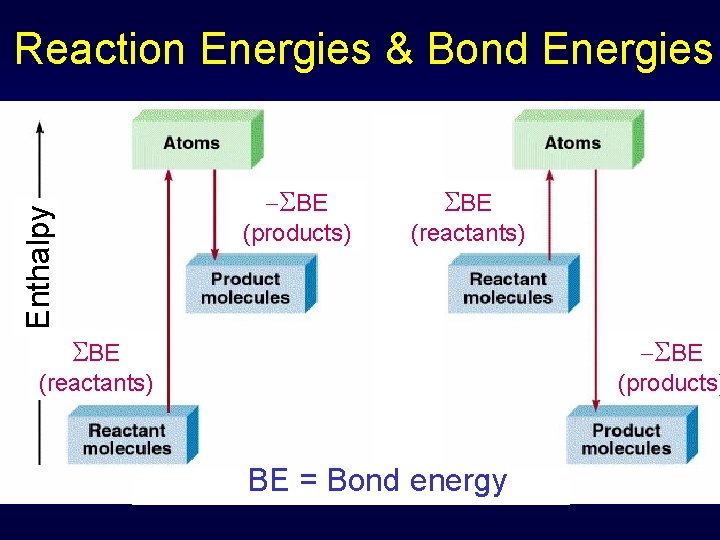
Reaction Energies & Bond Energies Enthalpy SBE (products) SBE (reactants) SBE (products) (reactants) BE = Bond energy

Chapter 8 – Covalent Bonding 8. 1 8. 2 8. 3 8. 4 8. 5 The Covalent Bond Naming Molecules Molecular Structures Molecular Shape Electronegativity and Polarity

Section 8. 4 Molecular Shapes The VSEPR model is used to determine molecular shape. • Summarize the VSEPR bonding theory, including the role of bonding and nonbonding pairs of electrons. • Predict the shape of, and the bond angles in, a molecule using VSEPR theory.

2 Simple Theories Related to Covalent Bonding Valence Shell Electron Pair Repulsion Theory (VSEPR) • Use Lewis structures to predict shape Valence Bond Theory • Extends Lewis bonding model to focus on orbitals, particularly hybridized orbitals

VSEPR Valence Shell Electron Pair Repulsion Theory - allows us to predict geometry Lewis structures tell us how the atoms are connected to each other Lewis structures don’t tell us anything about shape Shape of a molecule can greatly affect its properties

Molecular Shape & Biological Sensors For some biological systems, a response is generated or a chemical change is initiated when a molecular key fits into correspondingly shaped molecular lock • Key is typically small molecule • Lock is typically large molecule with a shaped receptor site v Only interacts with key of a specific shape

Lewis Structure (a) & Tetrahedral Geometry (b) for Methane (CH 4)

VSEPR Molecules take a shape that puts electron pairs as far away from each other as possible (electron pair repulsion) Have to draw the Lewis structure to determine categories of electron pairs • bonding • nonbonding lone pair Lone pair take more space Multiple bonds count as one pair

Balloon Analogy for the Mutual Repulsion of Electron Groups Two Three Four Five Six Number of Electron Groups

VSEPR The number of pairs determines • bond angles • underlying structure The number and position of atoms determines • actual molecular shape
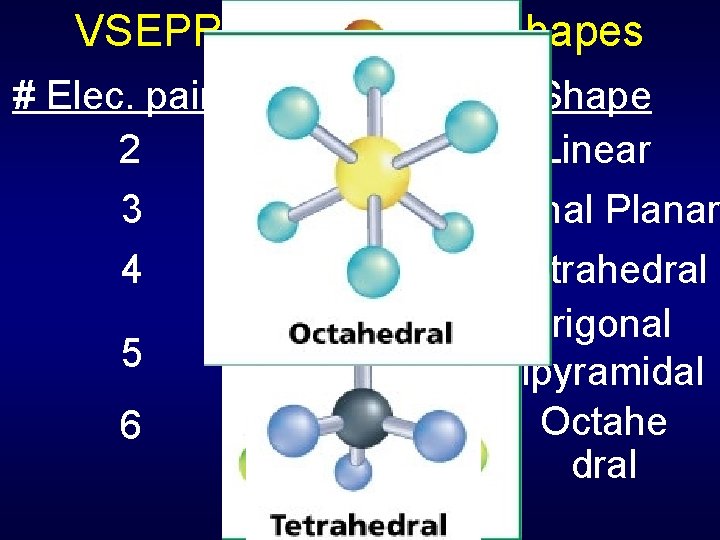
VSEPR – Underlying Shapes # Elec. pairs Bond Angles Shape 2 180° Linear 120° Trigonal Planar 3 4 5 6 109. 5° 90° & 120° 90° Tetrahedral Trigonal Bipyramidal Octahe dral

Actual Molecular Shapes Non. Electron Bonding Pairs Shape 2 3 3 4 4 4 2 3 2 4 3 2 0 0 1 2 linear trigonal planar bent tetrahedral trigonal pyramidal bent

Actual Molecular Shapes Non. Electron Bonding Pairs Shape 5 5 5 4 3 2 0 1 2 3 trigonal bipyrimidal See-saw T-shaped linear
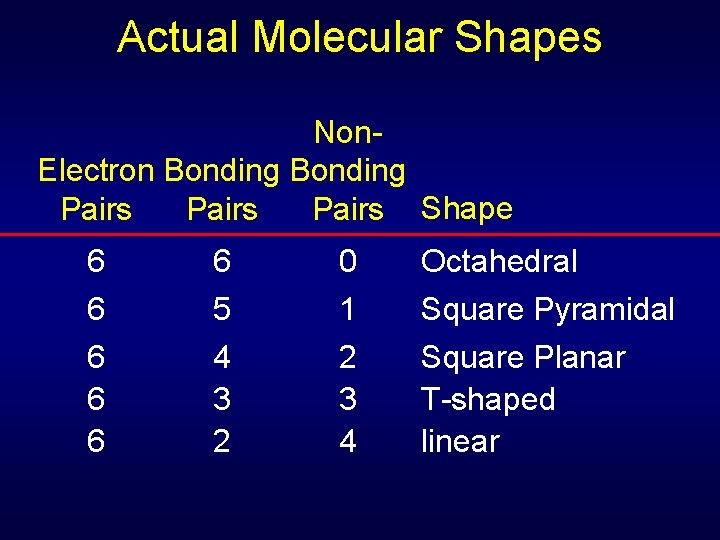
Actual Molecular Shapes Non. Electron Bonding Pairs Shape 6 6 6 5 4 3 2 0 1 2 3 4 Octahedral Square Pyramidal Square Planar T-shaped linear
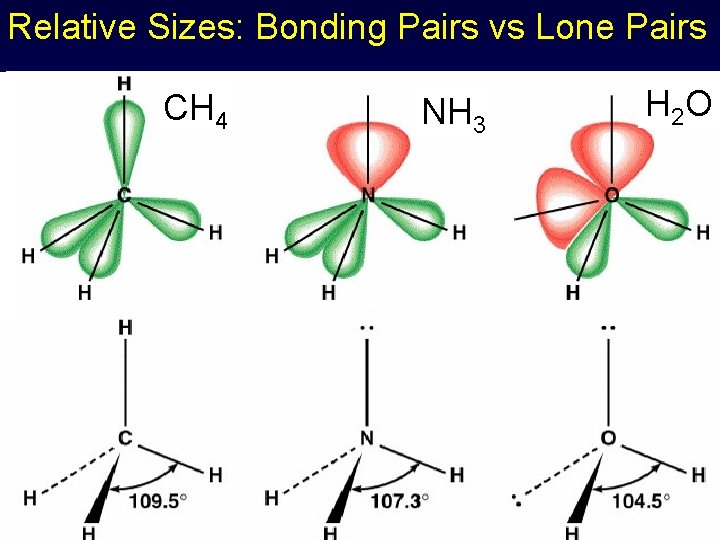
Relative Sizes: Bonding Pairs vs Lone Pairs CH 4 NH 3 H 2 O
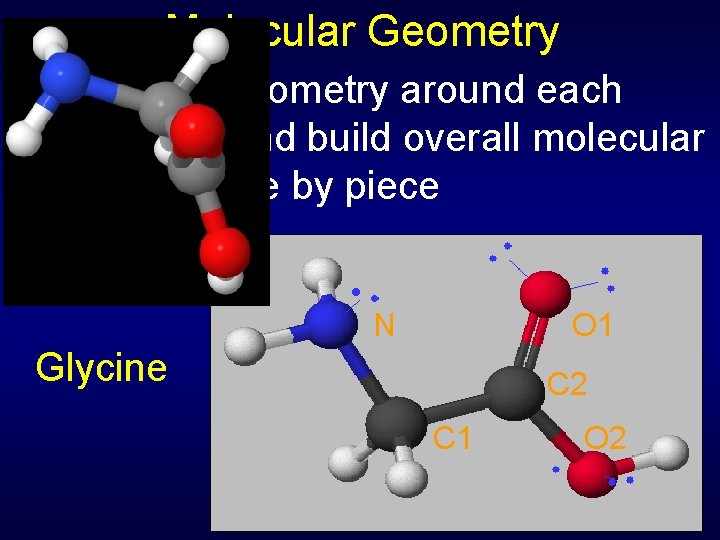
Molecular Geometry Can predict geometry around each atom center and build overall molecular geometry piece by piece N O 1 Glycine C 2 C 1 O 2

Chapter 8 – Covalent Bonding 8. 1 8. 2 8. 3 8. 4 The Covalent Bond Naming Molecules Molecular Structures Molecular Shape (extension of book) Valence Bond Theory - Orbital Overlap Hybrid Orbitals Quantum mechanical calculations 8. 5 Electronegativity and Polarity

Section 8. 4 Molecular Shapes The Valence Bond model is used to determine molecular shape via the concept of overlap of orbitals, particularly hybrid orbitals. • Describe the valence bond model of bonding • Explain the similarities and differences between the Lewis and valence bond models of chemical bonds. • Describe sigma and pi bonds and identify these bonds within molecules. • Define hybridization.

Section 8. 4 Molecular Shapes • Relate the type of hybridization (sp 3, sp 2, etc. ) to the VSEPR geometry of a molecule • Identify the specific type of hybridization that occurs within a given molecule and identify the specific orbitals (hybrid or non-hybrid) that are involved in each sigma and pi bond. • Explain how quantum mechanics and the wave function concept can be applied to a molecule.

Valence Bond Theory Lewis structures indicate status of electrons • Shared in bond • Lone pair No information about orbitals involved Valence bond theory • Bonds are formed by overlap of half-filled atomic orbitals • Orbital geometry can give direct information about molecular shape

Sigma Bonds Single covalent bonds = sigma bond • Symbol Greek letter Occurs when electron pair shared in area centered between two atoms Atomic orbitals overlap end to end, forming a bonding orbital • Localized region where bonding electrons will most likely be found
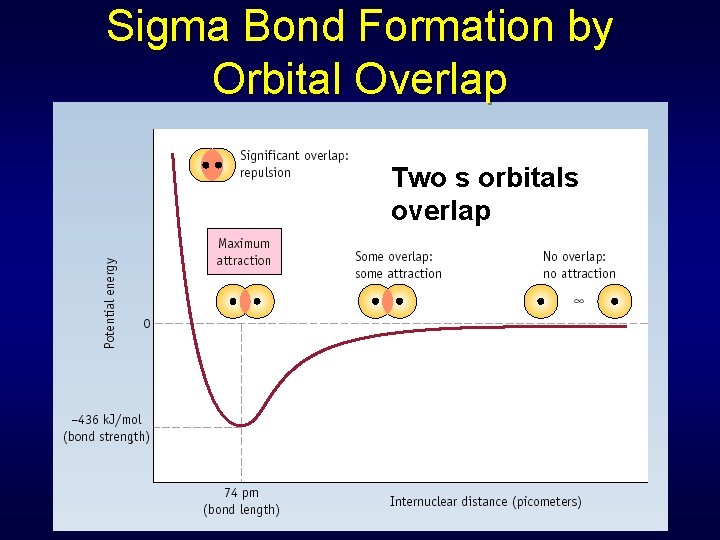
Sigma Bond Formation by Orbital Overlap Two s orbitals overlap
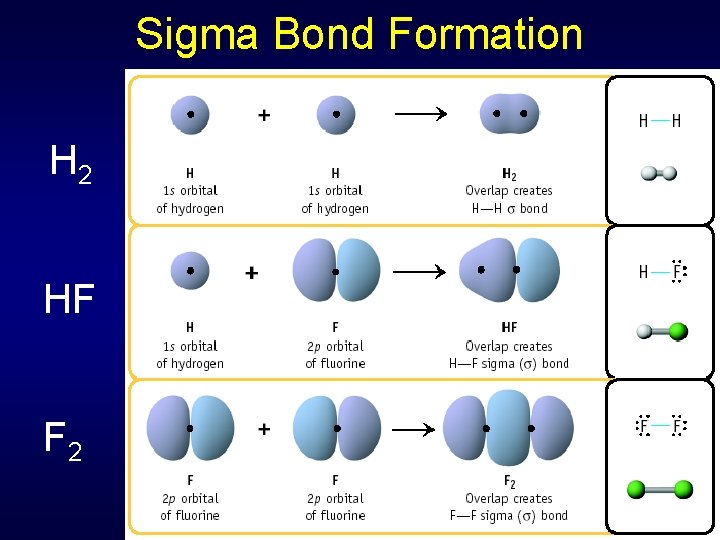
Sigma Bond Formation H 2 HF F 2 Two s orbitals overlap Two p orbitals overlap

Sigma Bonding – F 2 px 1 py 2 pz 2 —F F Area of overlap for atomic p 1 p 2 p 2 x y z orbitals
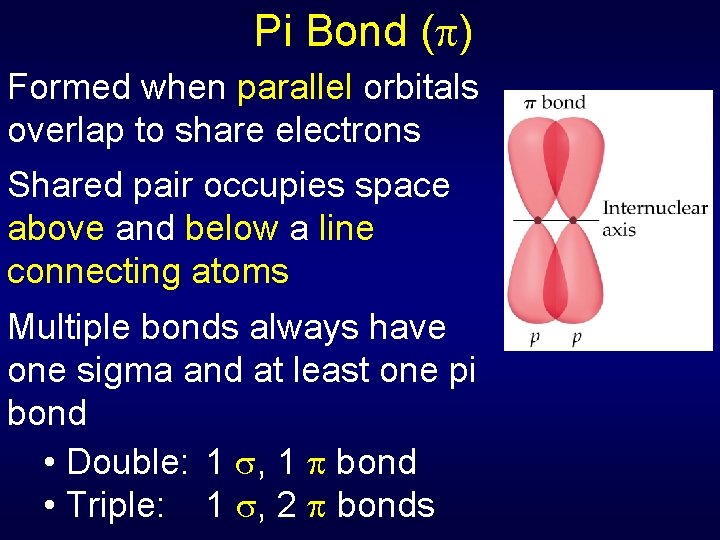
Pi Bond ( ) Formed when parallel orbitals overlap to share electrons Shared pair occupies space above and below a line connecting atoms Multiple bonds always have one sigma and at least one pi bond • Double: 1 , 1 bond • Triple: 1 , 2 bonds
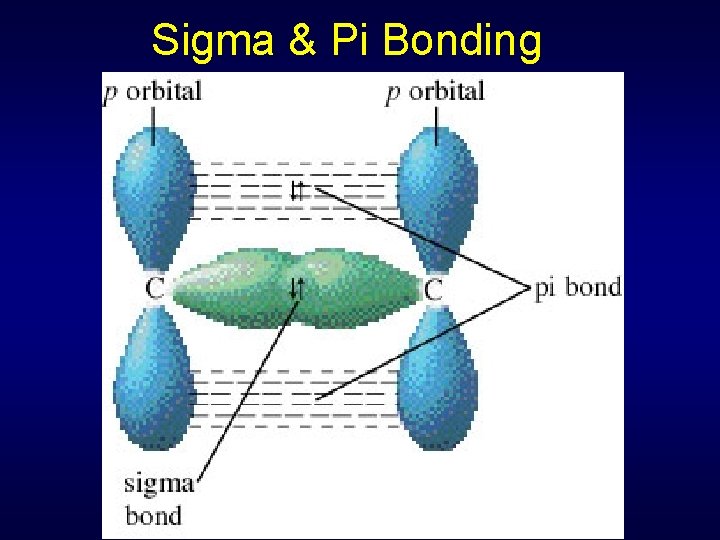
Sigma & Pi Bonding
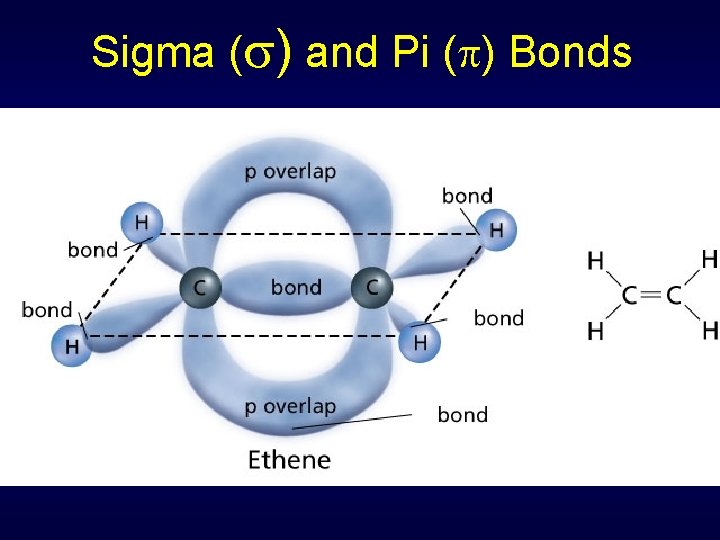
Sigma ( ) and Pi ( ) Bonds

Hybrid Orbitals For correct geometry of polyatomic molecules using the valence bond model, have to use concept of hybrid orbitals • CH 4 has 109. 5 angles, but atomic p orbitals are at right angles to each other

Hybrid Orbitals Hybrid orbitals – orbitals obtained when 2 or more nonequivalent orbitals combine to form an equal number of identical, degenerate orbitals Hybridization – mixing of atomic orbitals in an atom (usually a central atom) to generate a set of hybrid orbitals Use VSEPR logic to determine geometry of hybrid orbitals formed
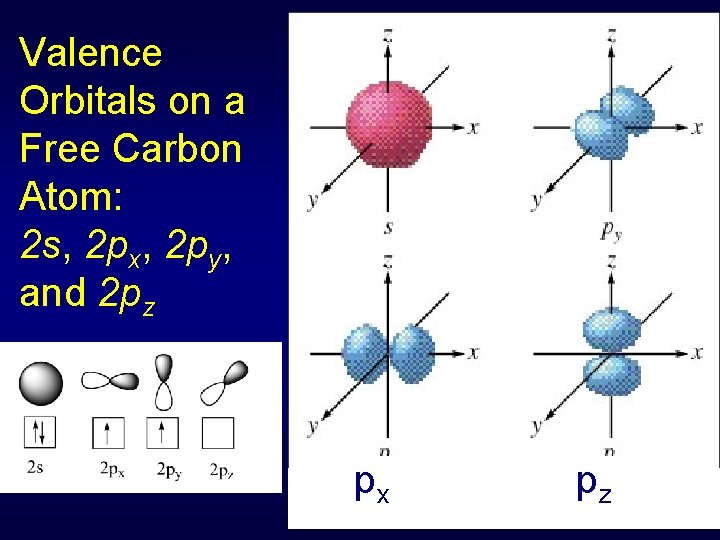
Valence Orbitals on a Free Carbon Atom: 2 s, 2 px, 2 py, and 2 pz s py px pz

Formation of sp 3 Hybrid Orbitals From Original Valence Orbitals Hybrid ization
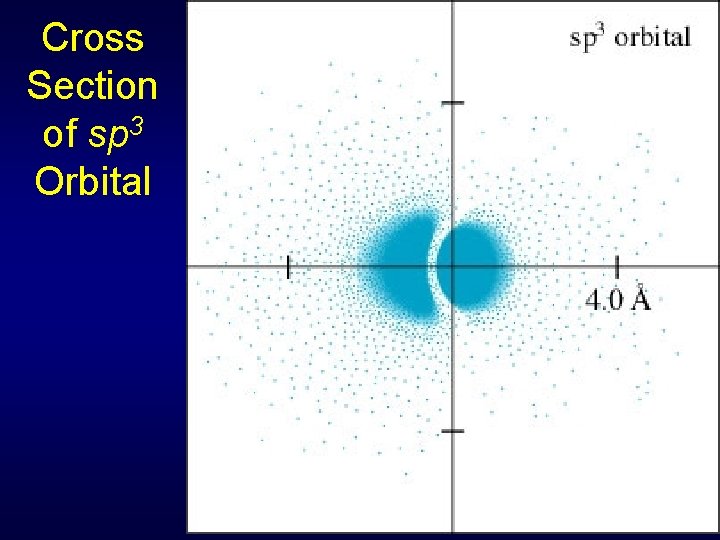
Cross Section of sp 3 Orbital
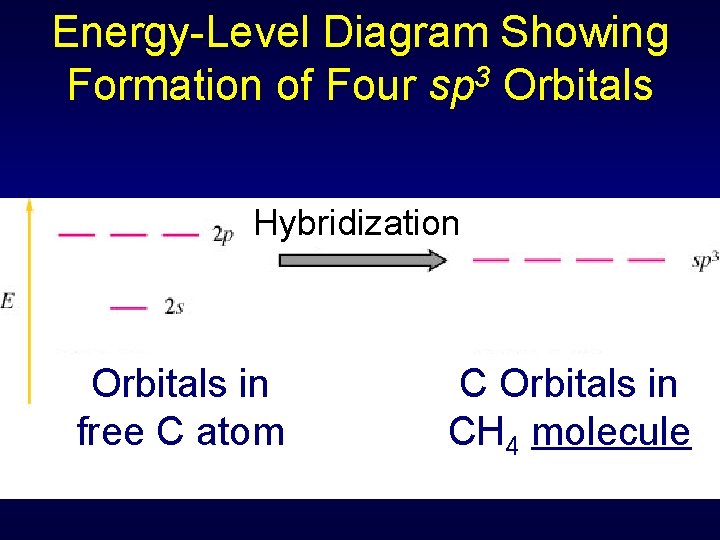
Energy-Level Diagram Showing Formation of Four sp 3 Orbitals Hybridization Orbitals in free C atom C Orbitals in CH 4 molecule
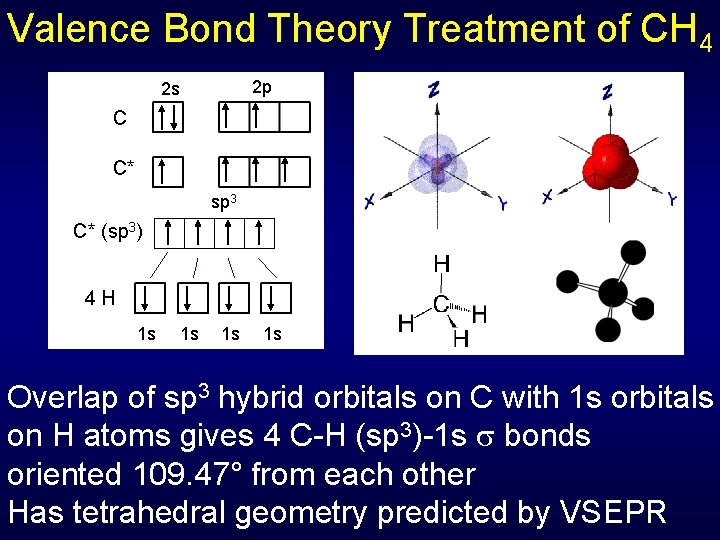
Valence Bond Theory Treatment of CH 4 2 p 2 s C C* sp 3 C* (sp 3) 4 H 1 s 1 s Overlap of sp 3 hybrid orbitals on C with 1 s orbitals on H atoms gives 4 C-H (sp 3)-1 s bonds oriented 109. 47° from each other Has tetrahedral geometry predicted by VSEPR
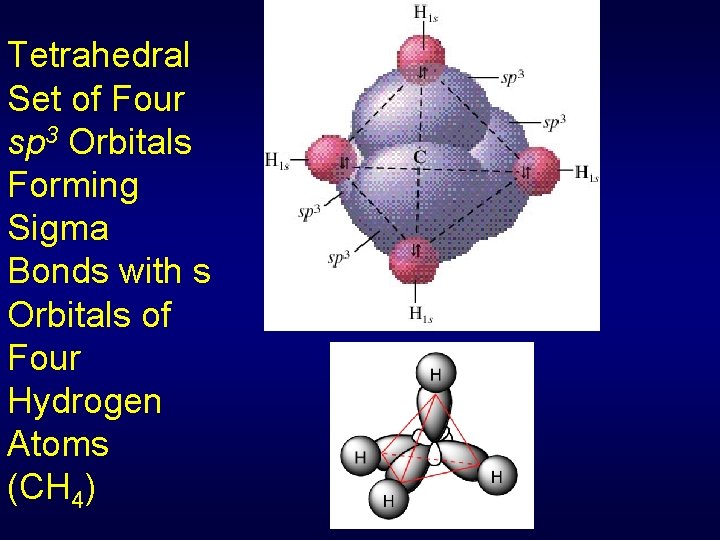
Tetrahedral Set of Four sp 3 Orbitals Forming Sigma Bonds with s Orbitals of Four Hydrogen Atoms (CH 4)
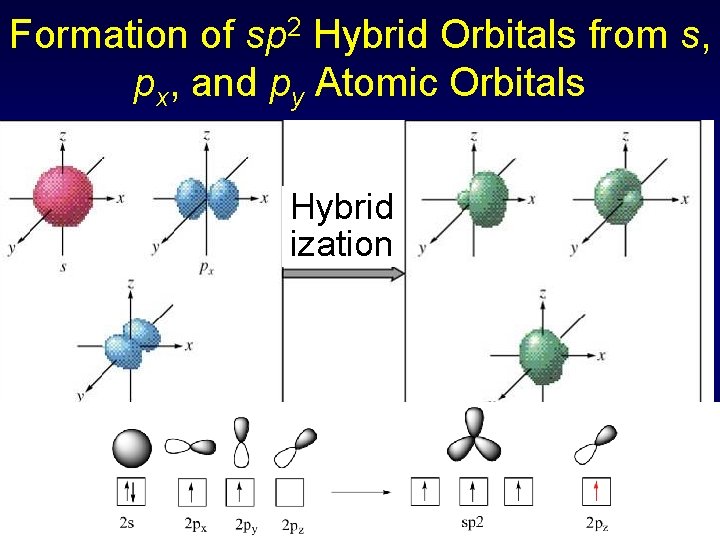
Formation of sp 2 Hybrid Orbitals from s, px, and py Atomic Orbitals Hybrid ization
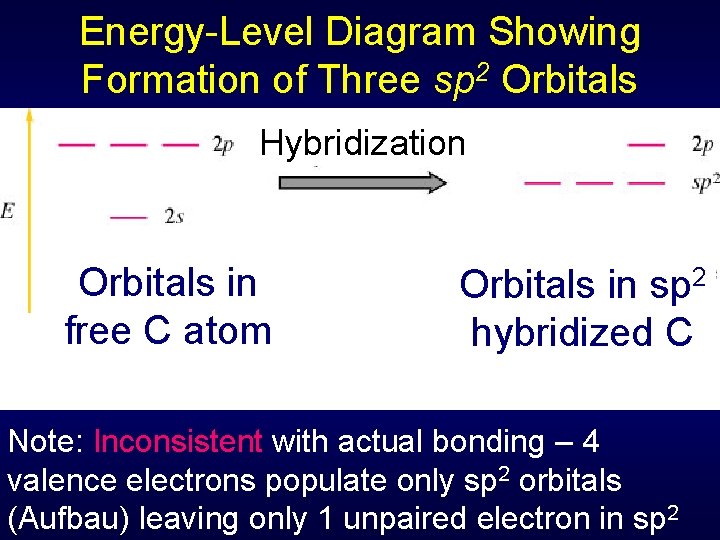
Energy-Level Diagram Showing Formation of Three sp 2 Orbitals Hybridization Orbitals in free C atom Orbitals in sp 2 hybridized C Note: Inconsistent with actual bonding – 4 valence electrons populate only sp 2 orbitals (Aufbau) leaving only 1 unpaired electron in sp 2
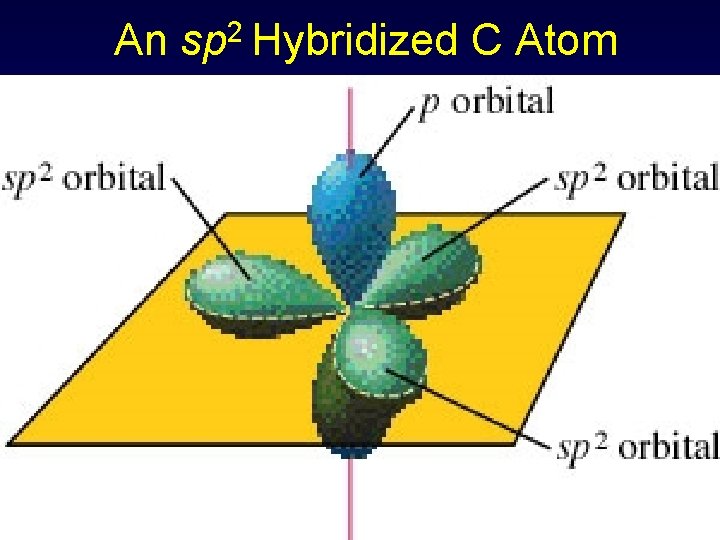
An sp 2 Hybridized C Atom
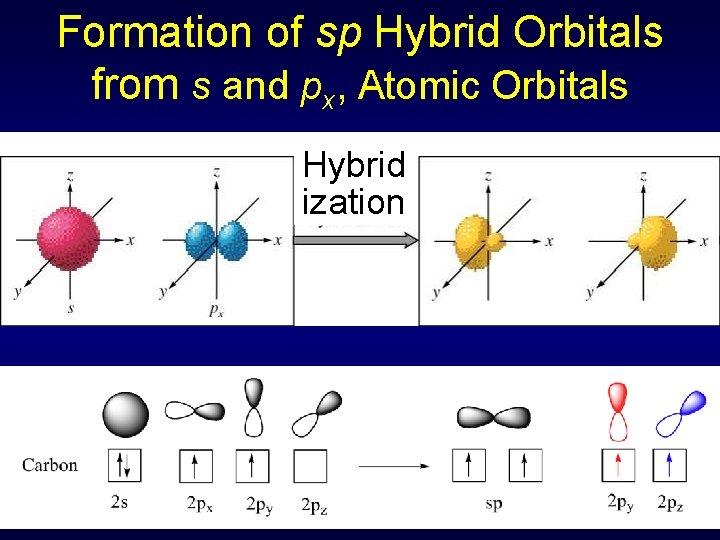
Formation of sp Hybrid Orbitals from s and px, Atomic Orbitals Hybrid ization

Energy-Level Diagram Showing Formation of Two sp Hybrid Orbitals Hybridization Orbitals in free C atom Orbitals in sp hybridized C Note: Inconsistent with actual bonding – 4 valence electrons should populate only sp orbitals (Aufbau) leaving no unpaired electrons
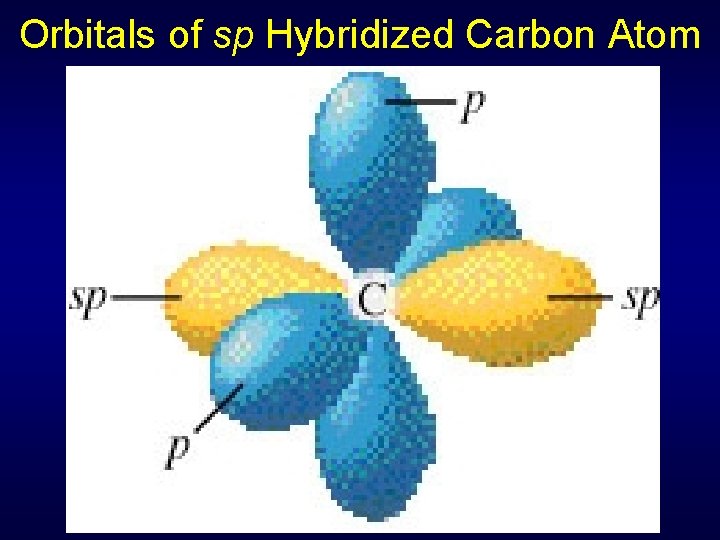
Orbitals of sp Hybridized Carbon Atom
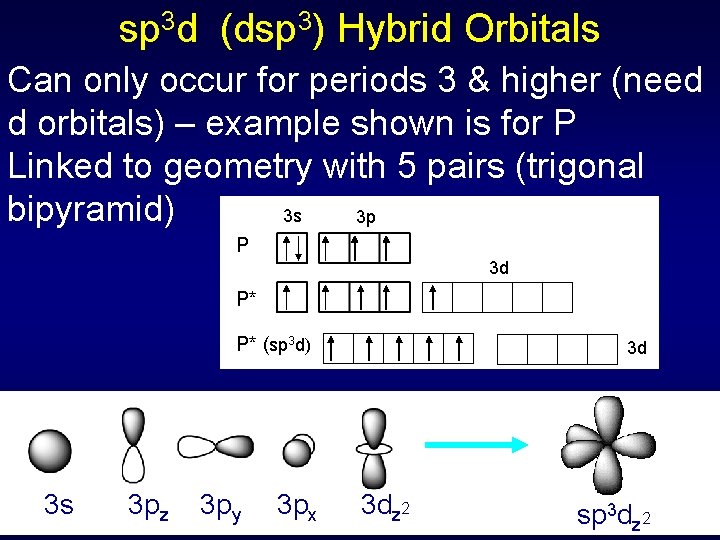
sp 3 d (dsp 3) Hybrid Orbitals Can only occur for periods 3 & higher (need d orbitals) – example shown is for P Linked to geometry with 5 pairs (trigonal bipyramid) 3 s 3 p P 3 d P* P* (sp 3 d) 3 s 3 pz 3 py 3 px 3 d 3 dz 2 sp 3 dz 2

Set of dsp 3 Hybrid Orbitals on a Phosphorus Atom

sp 3 d 2 (d 2 sp 3) Hybrid Orbitals Can only occur for periods 3 & higher (need d orbitals) Linked to geometry with 6 pairs (octahedral) Example on next slide for S

S - Octahedral Set of d 2 sp 3 Orbitals

Relationship among the number of effective pairs, geometry, and the hybrid orbital set required to obtain this geometry shown on the following two slides
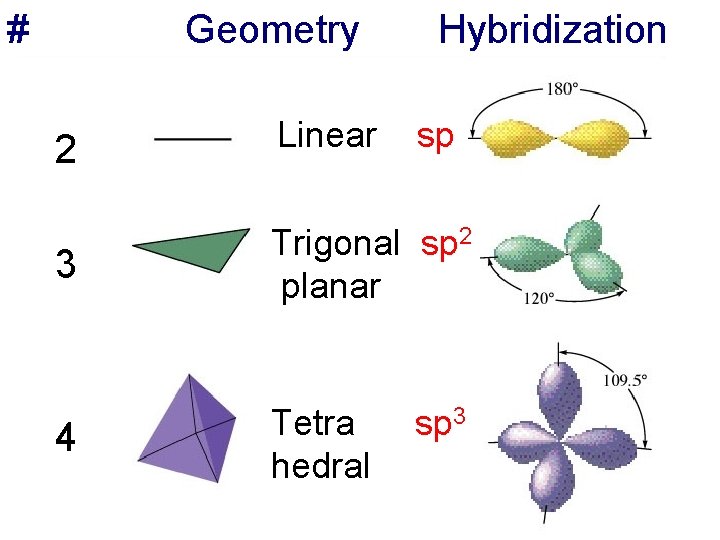
# Geometry Hybridization 2 Linear sp 3 Trigonal sp 2 planar 4 Tetra hedral sp 3
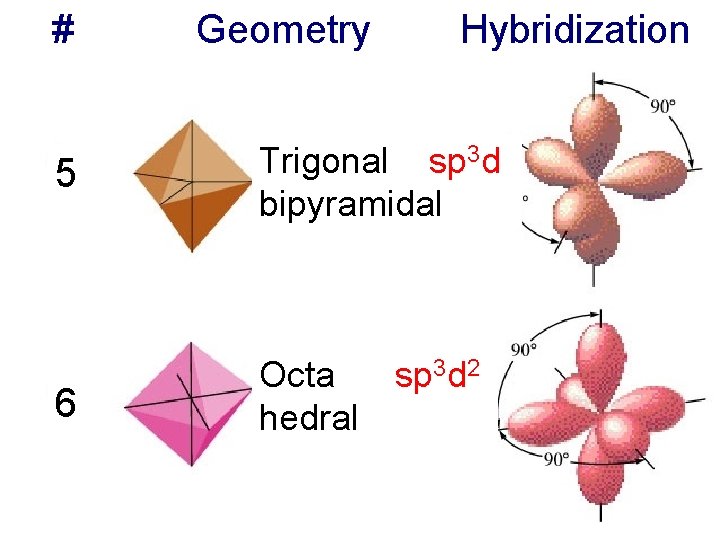
# Geometry Hybridization 5 Trigonal sp 3 d bipyramidal 6 Octa sp 3 d 2 hedral

Geometry & Hybridization - Steps 1. Draw Lewis structure 2. Determine # of effective electron pairs (count double & triple bonds as one pair) 3. Determine basic geometry from number of pairs (e. g. , 5 pairs = trigonal bipyramid) 4. Determine hybridization type from number of pairs (e. g. , 5 pairs = sp 3 d) 5. Form single (sigma) bonds from hybrid orbitals; lone pairs also go in hybrid orbitals 6. Form pi bonds using unhybridized orbitals

Geometry & Hybridization - Steps Following slides give examples of using the steps listed on previous slide for these molecules: 1. Ammonia 2. Ethylene 3. Diatomic nitrogen 4. Acetylene 5. Carbon dioxide 6. Phosphorus pentachloride
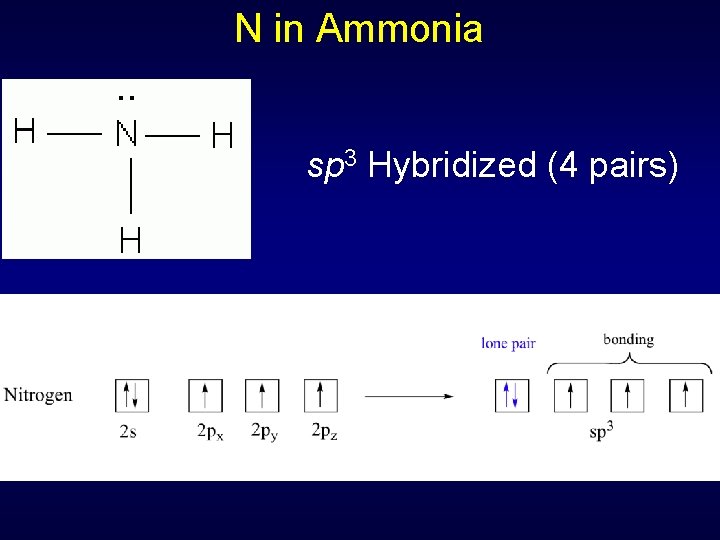
N in Ammonia sp 3 Hybridized (4 pairs)
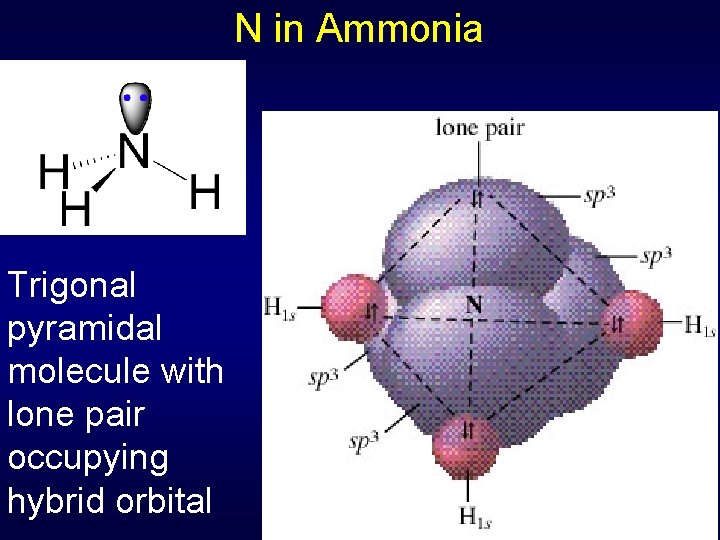
N in Ammonia Trigonal pyramidal molecule with lone pair occupying hybrid orbital
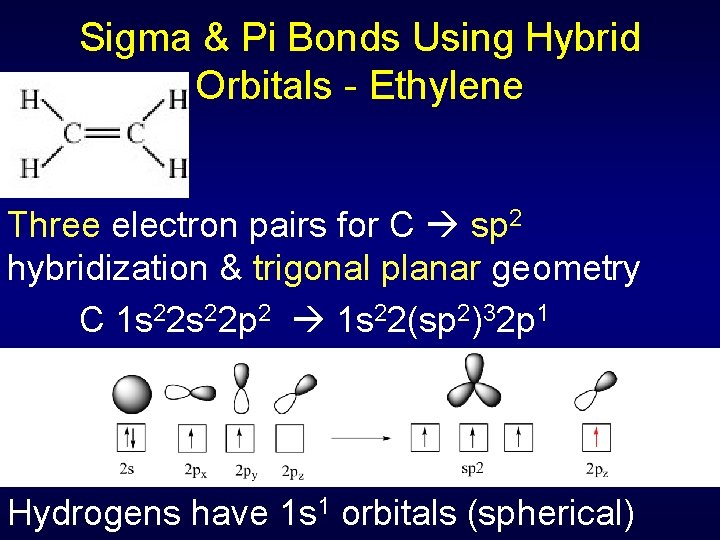
Sigma & Pi Bonds Using Hybrid Orbitals - Ethylene Three electron pairs for C sp 2 hybridization & trigonal planar geometry C 1 s 22 p 2 1 s 22(sp 2)32 p 1 Hydrogens have 1 s 1 orbitals (spherical)
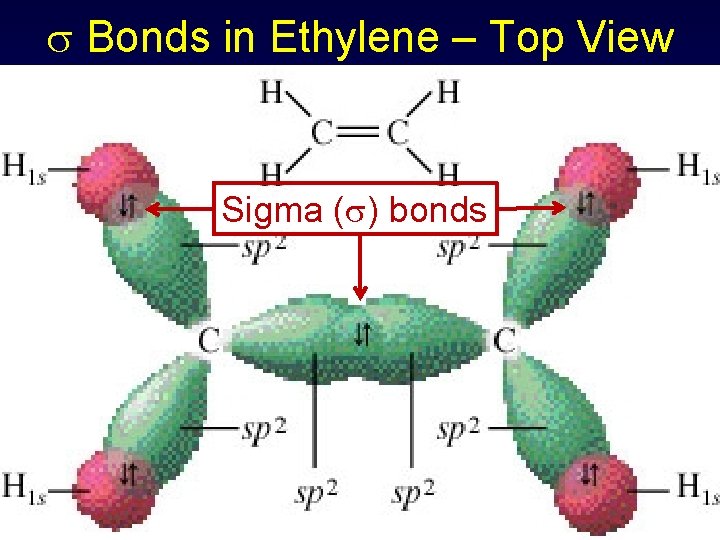
Bonds in Ethylene – Top View Sigma ( ) bonds
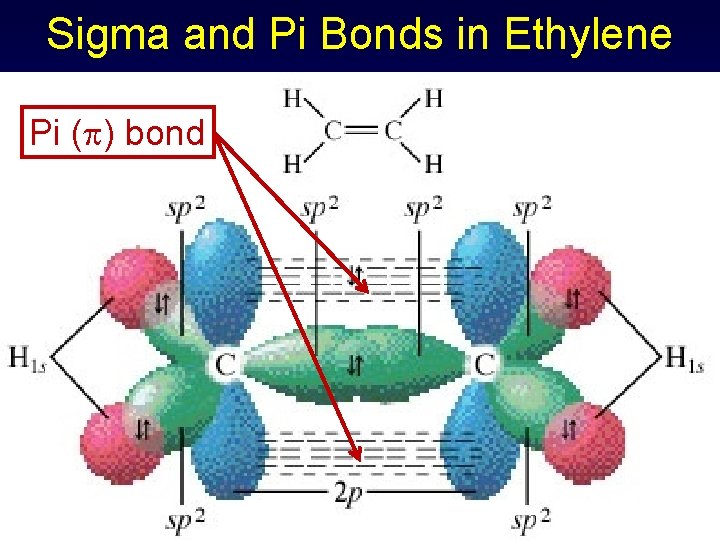
Sigma and Pi Bonds in Ethylene Pi ( ) bond
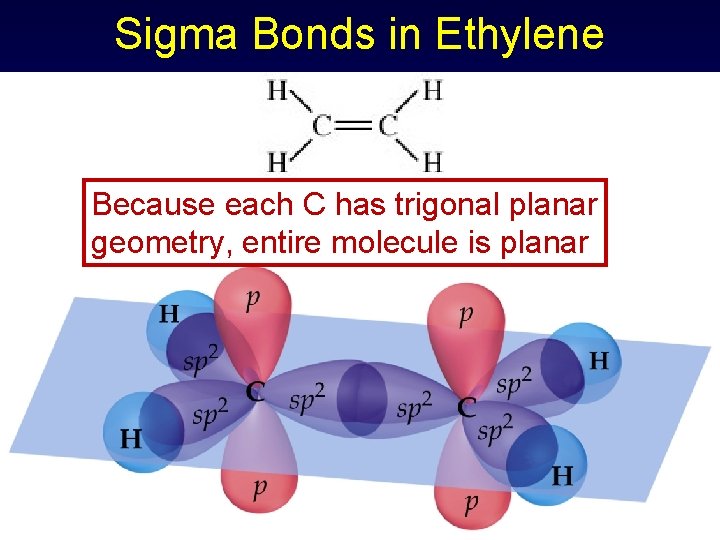
Sigma Bonds in Ethylene Because each C has trigonal planar geometry, entire molecule is planar
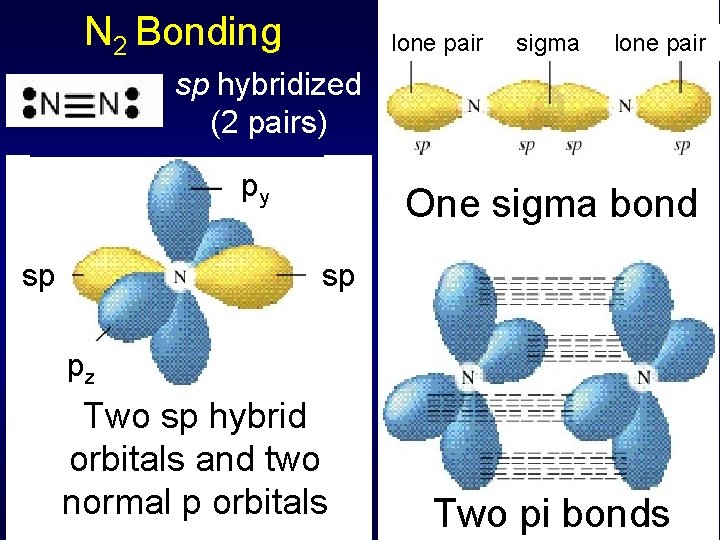
N 2 Bonding lone pair sigma lone pair sp hybridized (2 pairs) py sp One sigma bond sp pz Two sp hybrid orbitals and two normal p orbitals Two pi bonds

Sigma and Pi Bonds in Acetylene Two electron pairs for C sp hybridization, linear geometry (triple bond = single pair) C 1 s 22 p 2 1 s 22(sp)22 p 2 Hydrogens have 1 s 1 orbitals (spherical)
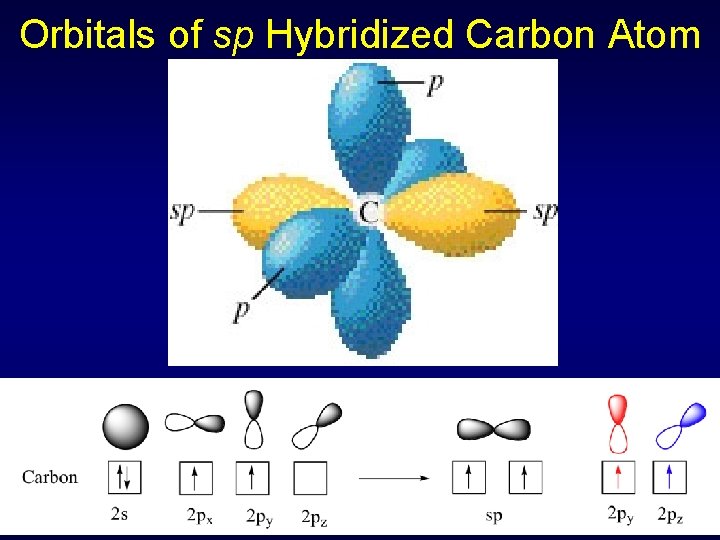
Orbitals of sp Hybridized Carbon Atom

Sigma and Pi Bonds in Acetylene sp hybrid orbitals on C form single (sigma) bond with H and other C Remaining two unhybridized p orbitals overlap to form two pi bonds

Sigma and Pi Bonds in Acetylene

Sigma & Pi Bonds Using Hybrid Orbitals in CO 2 Two electron pairs for C sp hybridization, linear geometry (double bond = single pair) C 1 s 22 p 2 1 s 22(sp)22 p 2 Three electron pairs for O sp 2 hybridization & trigonal planar geometry O 1 s 22 p 4 1 s 22(sp 2)5 p 1
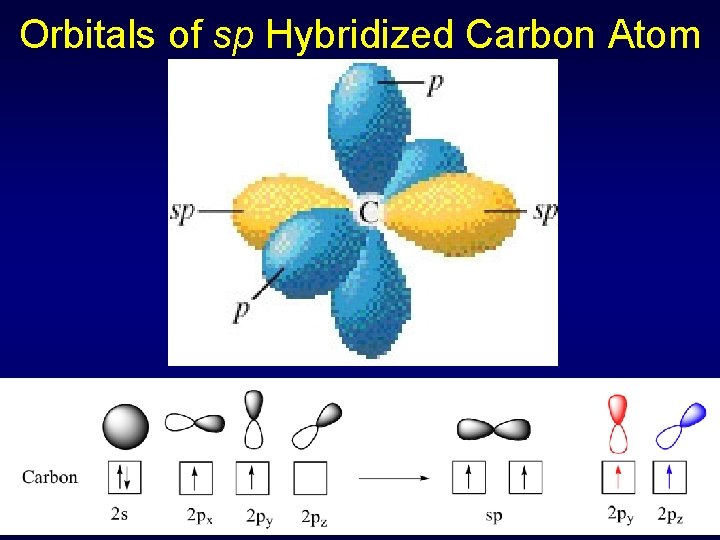
Orbitals of sp Hybridized Carbon Atom
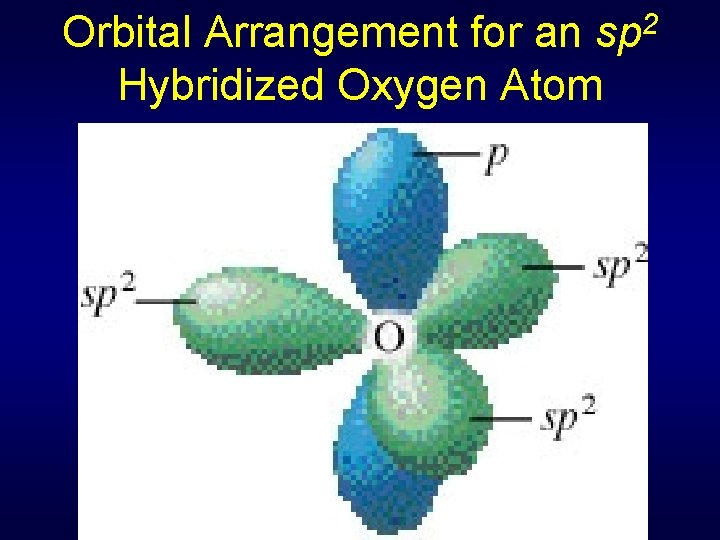
Orbital Arrangement for an sp 2 Hybridized Oxygen Atom
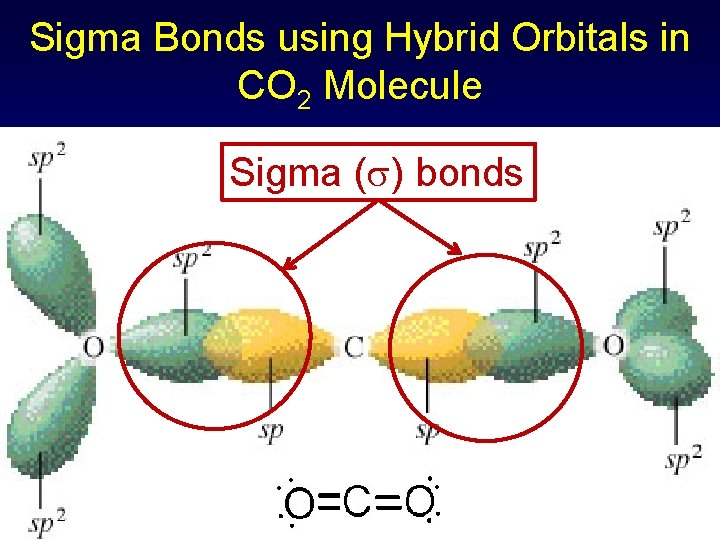
Sigma Bonds using Hybrid Orbitals in CO 2 Molecule Sigma ( ) bonds
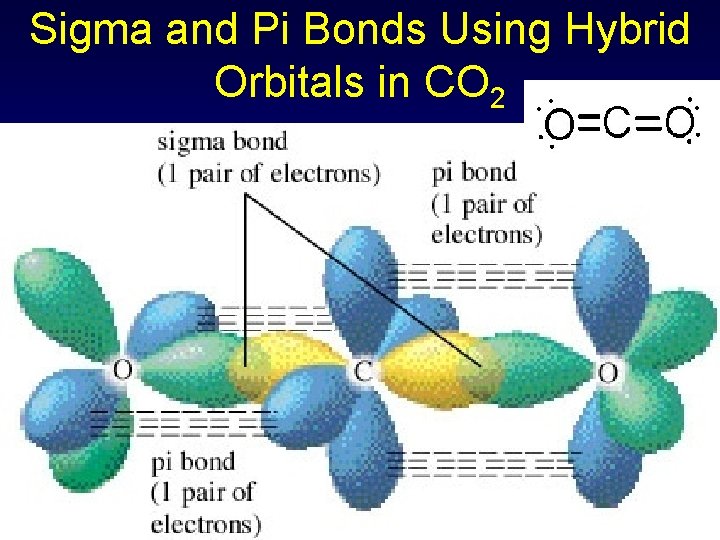
Sigma and Pi Bonds Using Hybrid Orbitals in CO 2

Sigma Bonds Using Hybrid Orbitals in PCl 5 Five electron pairs for P sp 3 d hybridization & trigonal bipyramidal geometry P [Ne]3 s 23 p 3 [Ne](sp 3 d)5 Four electron pairs for Cl sp 3 hybridization & tetrahedral geometry Cl [Ne]3 s 23 p 5 [Ne]3(sp 3)7
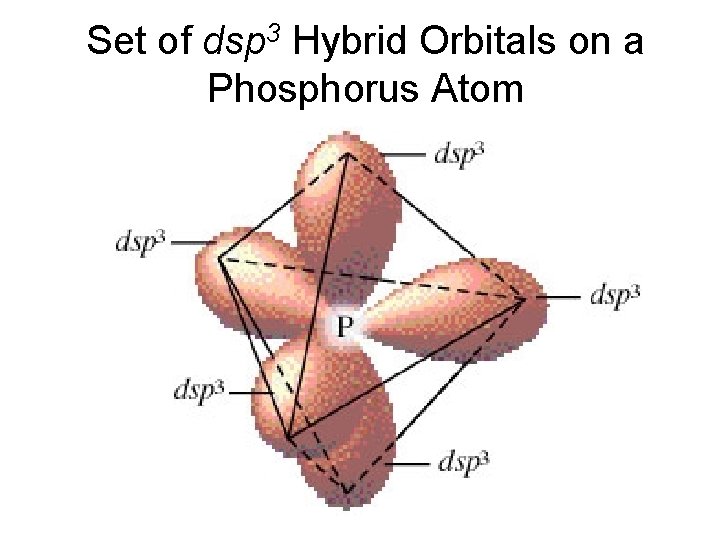
Set of dsp 3 Hybrid Orbitals on a Phosphorus Atom

Structure of PCI 5 and Orbitals Used to Form Sigma Bonds Sigma ( ) bond Lone pairs on Cl in sp 3 orbitals
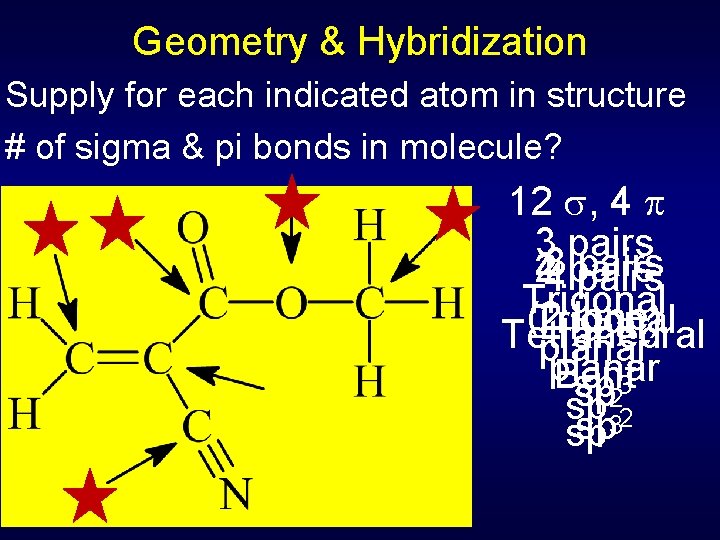
Geometry & Hybridization Supply for each indicated atom in structure # of sigma & pi bonds in molecule? 12 , 4 33 pairs 24 pairs Trigonal (2 lone) Trigonal Linear Tetrahedral planar Bent 3 sp sp 2 3 sp sp

Practice (Shape, Angles, Hybridization) Problems 56 – 60, 65 - 67 page 264 Problems 108, 110 - 112 page 275 Problem 8 page 980

Quantum Mechanics & Molecules

Quantum Mechanics & Molecules Y (wave function) exists for entire molecule and can be obtained from solution to Schrodinger wave equation written for the molecule Y 2 - Square of Y gives probability of finding electron at particular position around molecule – defines what is called a molecular orbital (MO)

Quantum Mechanics & Molecules Using certain types of approximations and today’s computers, wave functions for molecules (not individual atoms) can be obtained and molecular properties calculated from this information Energy, absorption spectrum, dipole moment, etc Molecular orbital theory is most advanced way of describing covalent bonding

Chapter 8 – Covalent Bonding 8. 1 8. 2 8. 3 8. 4 8. 5 The Covalent Bond Naming Molecules Molecular Structures Molecular Shape Electronegativity and Polarity

Section 8. 5 Electronegativity and Polarity A chemical bond’s character is related to each atom’s attraction for the electrons in the bond. • Describe how electronegativity is used to determine bond type and characterize bonds between given pairs of atoms as being polar or nonpolar. • Compare and contrast polar and nonpolar covalent bonds and polar and nonpolar molecules. • Describe the term “dipole moment” and relate it to the terms polar and nonpolar.

Section 8. 5 Electronegativity and Polarity • Identify molecules as being polar or nonpolar. • Describe how polarity affects the solubility of one substance in another substance. • Describe how polarity can give rise to intermolecular forces.

Polar Covalent Bonds Polarity of bond determined by electronegativity difference Difference = 0 Nonpolar Difference > 0 Polar Very large differences • No longer covalent compound
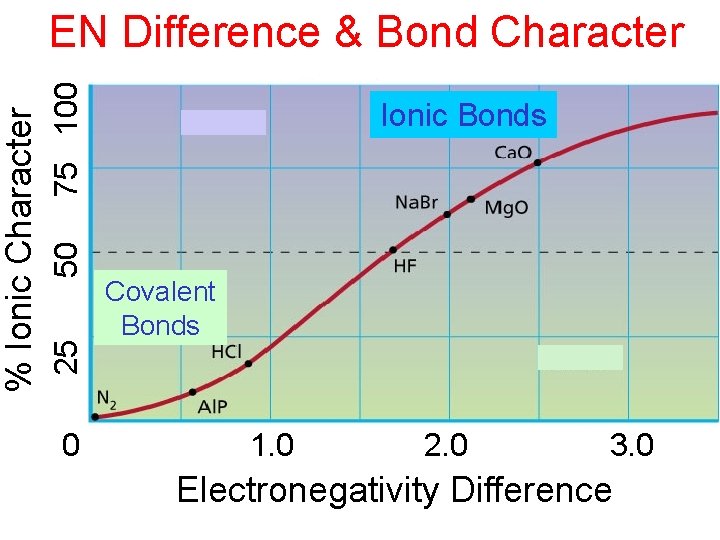
75 100 50 25 % Ionic Character EN Difference & Bond Character 0 Ionic Bonds Covalent Bonds 1. 0 2. 0 3. 0 Electronegativity Difference

Relationship Between EN Difference and Bond Type
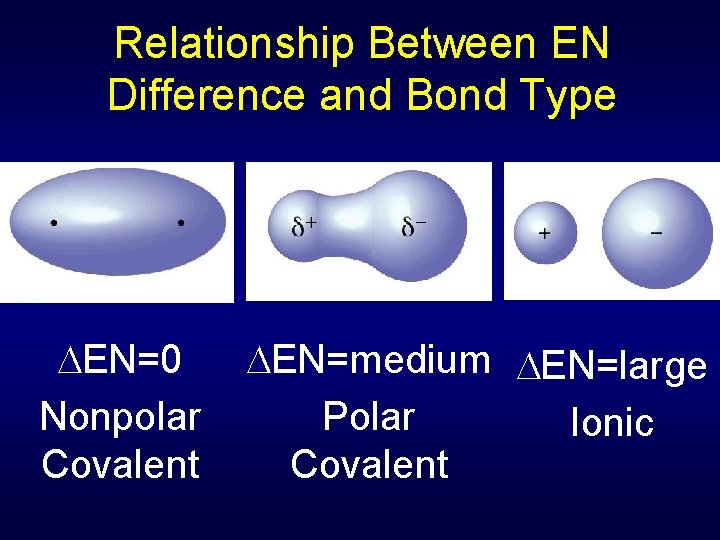
Relationship Between EN Difference and Bond Type EN=0 Nonpolar Covalent EN=medium EN=large Polar Ionic Covalent

Scalars & Vectors Scalar • Completely specified by magnitude and units Vector • Has magnitude, direction, and units v = 3. 5 m/s (scalar) v = 3. 5 m/s to northeast (vector)

Trigonometric Functions

Pythagorean Theorem
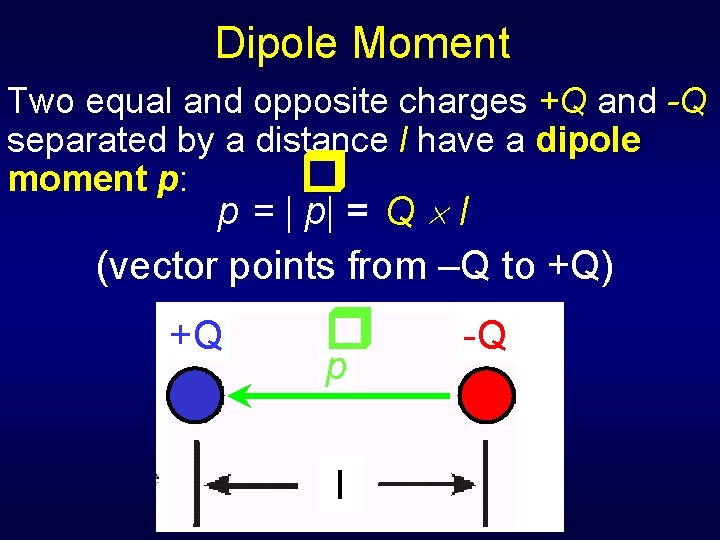
Dipole Moment Two equal and opposite charges +Q and -Q separated by a distance l have a dipole r moment p: p = Q l (vector points from –Q to +Q) +Q r p l -Q
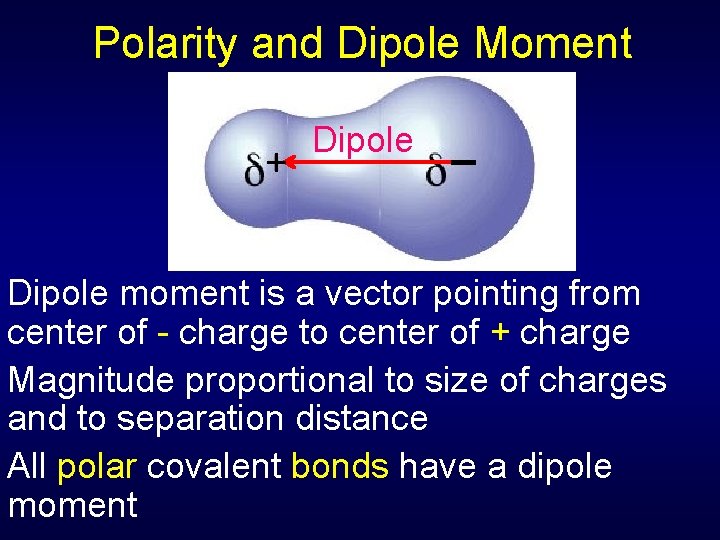
Polarity and Dipole Moment + Dipole _ Dipole moment is a vector pointing from center of - charge to center of + charge Magnitude proportional to size of charges and to separation distance All polar covalent bonds have a dipole moment

Polarity and Dipole Moment p = Q l +Q r -Q p l Units of p are Debye units (D) % ionic character of bond determined by size of measured dipole moment relative to value calculated from using full (ionic) charges as Q
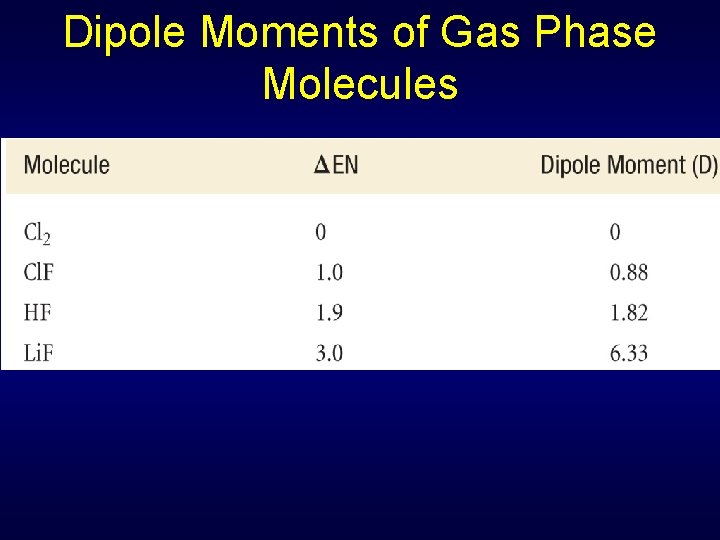
Dipole Moments of Gas Phase Molecules
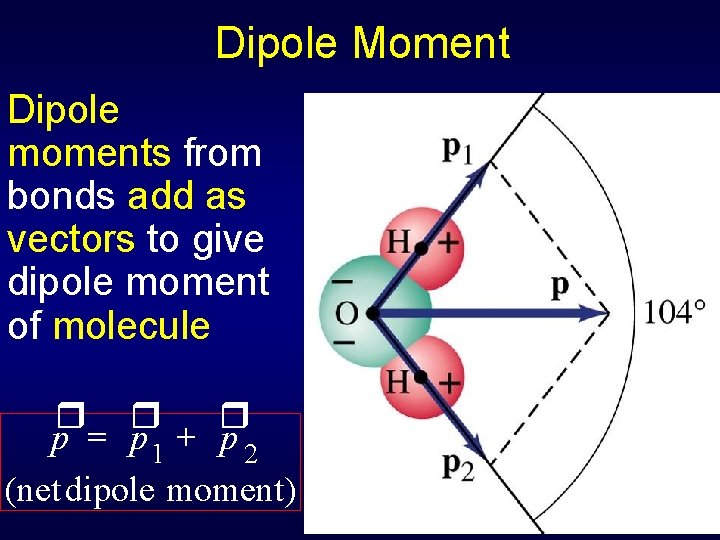
Dipole Moment Dipole moments from bonds add as vectors to give dipole moment of molecule r r r p p 1 + p 2 (net dipole moment)
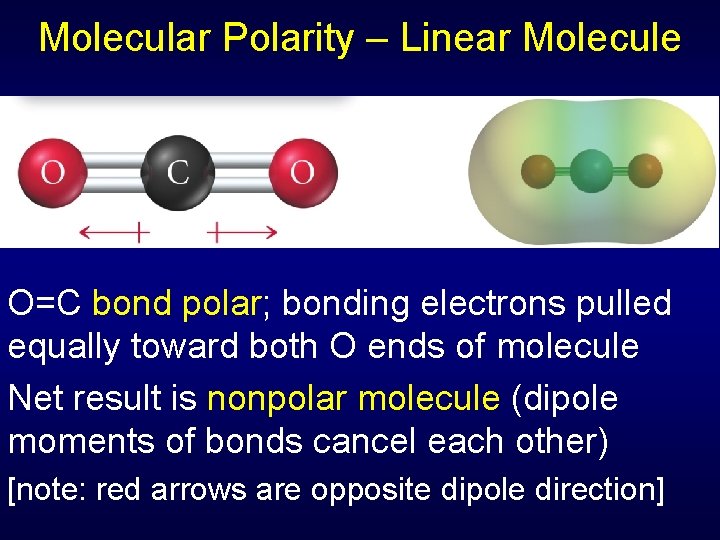
Molecular Polarity – Linear Molecule O=C bond polar; bonding electrons pulled equally toward both O ends of molecule Net result is nonpolar molecule (dipole moments of bonds cancel each other) [note: red arrows are opposite dipole direction]
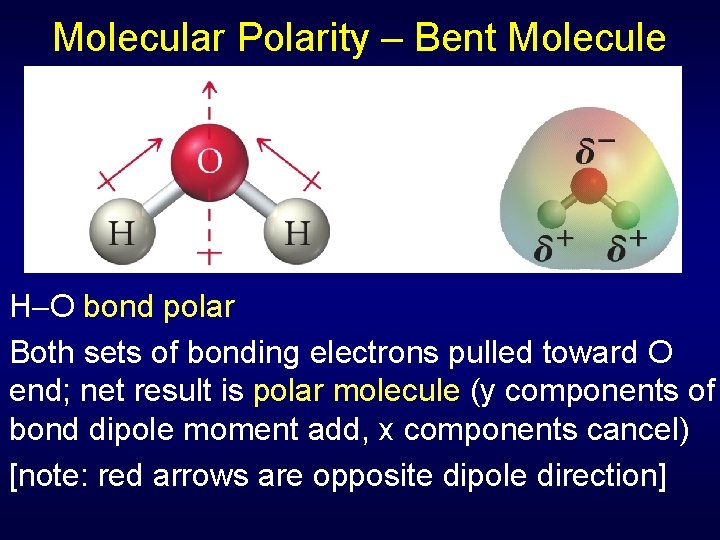
Molecular Polarity – Bent Molecule H O bond polar Both sets of bonding electrons pulled toward O end; net result is polar molecule (y components of bond dipole moment add, x components cancel) [note: red arrows are opposite dipole direction]
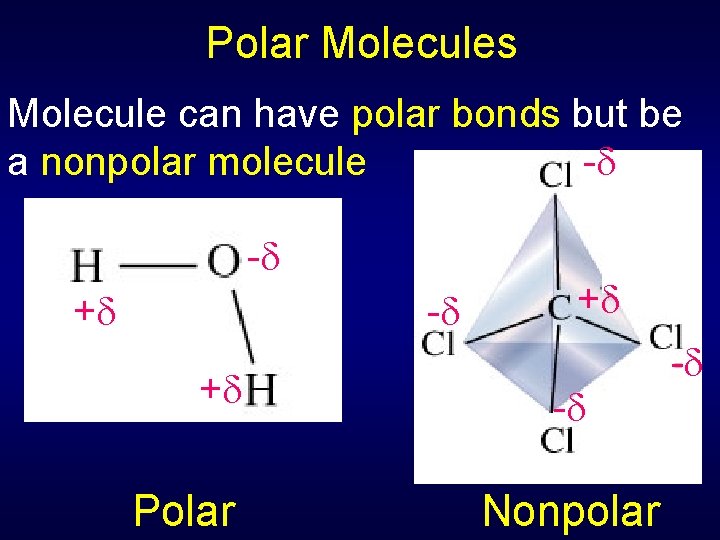
Polar Molecules Molecule can have polar bonds but be - a nonpolar molecule - + Polar + - - Nonpolar
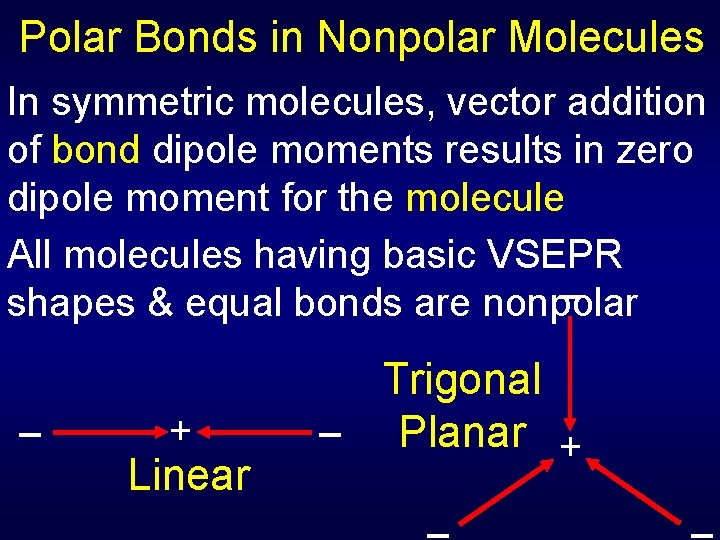
Polar Bonds in Nonpolar Molecules In symmetric molecules, vector addition of bond dipole moments results in zero dipole moment for the molecule All molecules having basic VSEPR shapes & equal bonds are nonpolar + Linear Trigonal Planar +

Practice (Polar Bonds & Polar Molecules) Problems 74 – 77 page 270 Problems 117 – 123 page 275 Problem 9 page 980

Polarity Effects Polarity of molecule determines solubility characteristics – “like dissolves like” Oil (nonpolar) and water (polar) don’t mix

Dipole in an Electric Field The + and – charges in an electric dipole are pulled in opposite directions in an electric field, producing a net torque on the dipole, and orienting it.

Dipole in Electric Field – HF Molecule -δ Field Off +δ F H + Field On

Polar Molecule & Electric Field Polar molecules affected by electric field in an EM wave Oscillating field twists water molecule and energy transferred (heats up) Basis for microwave oven operation

Properties of Covalent Compounds Bonding types affect properties Many properties controlled by intermolecular forces • Forces between molecules • Also known as van der Waals forces Intermolecular forces are weaker than chemical bonds [Note: intermolecular forces treated in more depth in section 12. 2 – Forces of Attraction]

Intermolecular Forces between nonpolar molecules relatively weak • Tend to be gases or volatile liquids • O 2, N 2, small hydrocarbons Forces between polar molecules are stronger due to dipole-dipole forces • Hydrogen bonding a particular strong version - H and F, O, or N
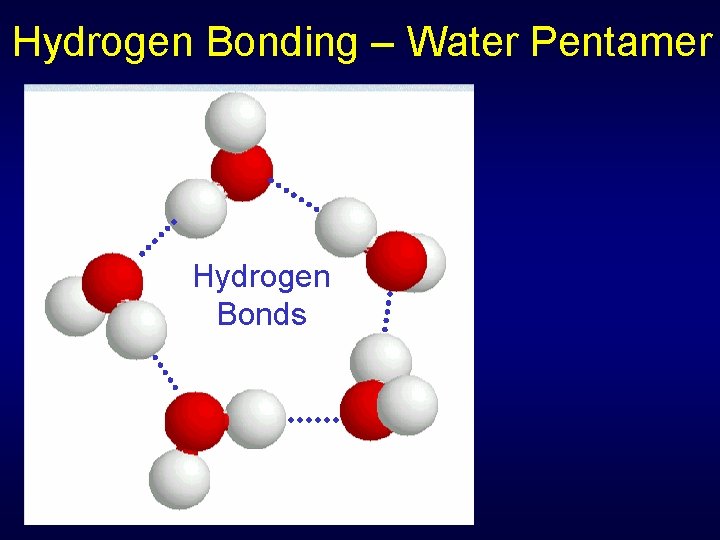
Hydrogen Bonding – Water Pentamer Hydrogen Bonds

Hydrogen Bonding in Nylon Hydrogen bonding helps make nylon strong

End of Chapter 8
- Slides: 166