Chapter 8 Classifying Chemical Compounds Key Ideas All
















- Slides: 20

Chapter 8 Classifying Chemical Compounds

Key Ideas • All chemical compounds are either organic or inorganic. • Inorganic compounds can be molecular or ionic (acids, bases, or salts). • Lewis diagrams (electron dot) can explain how molecular compounds form as a result of bonding pairs of electrons.

Key Ideas • Organic compounds are molecular and contain carbon and hydrogen • All chemical compounds can be classified as either organic or inorganic.

Classifying Inorganic Compounds • Chemical compounds can be classified as either organic or inorganic. • Compounds that have a high percentage of carbon by mass are classified as organic compounds; otherwise they are considered to be inorganic. (Usually have hydrogen as well)

Inorganic compounds • Inorganic compounds do not have a high percentage of carbon by mass and can be either ionic or molecular.

Inorganic Molecules Ø Compounds in this class are • Inorganic, so they contain little or no carbon (most fit into this category) • Molecular, so they have a non-metal bonded to a non-metal. (eg. CO 2 , H 2 O, N 2 O)

Inorganic Ionic Compounds • By the 1500’s scientists had recognized two groups of substances which became known as acids and bases. • When acids react with bases they create a new substance which tasted salty. When water was evaporated from this solution, a crystalline substance remained

Inorganic Ionic Compounds • Svante Arrhenius developed definitions of acids and bases based on the type of ions a substance releases in an aqueous solution. Bases release OH ions (OH-1) and acids release H ions (H+1).

General Properties of Acids, Bases, and Salts. Acidity Ø A solution can be acidic, basic, or neutral based on the relative number of H or OH ions it contains. – A neutral solution has equal numbers of H and OH ions. – An acidic solution has more H ions than OH ions. – A basic (alkaline) solution has more OH ions than H ions

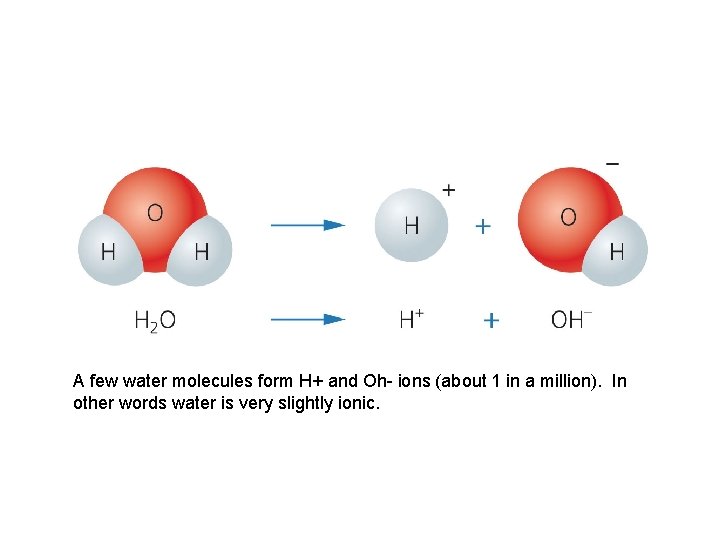
A few water molecules form H+ and Oh- ions (about 1 in a million). In other words water is very slightly ionic.

Measuring Acidity (The p. H Scale) • The p. H of a solution indicates how acidic or basic the solution is. • The normal range of p. H is from 0 to 14, with a p. H of 7 being neutral. • Solutions with a p. H less than 7 are acidic • Solutions with a p. H greater than 7 are basic (alkaline)

p. H Values of some common items

p. H Scale • Every 1 point on the p. H scale represents 10 times more or less H+ ions present. – Eg. A solution with a p. H of 4 has 10 times more H+ ions (10 times more acidic) than one with a p. H of 5. See table 5 page 206. • p. H indicators are chemicals that change colour depending on the p. H of a solution.

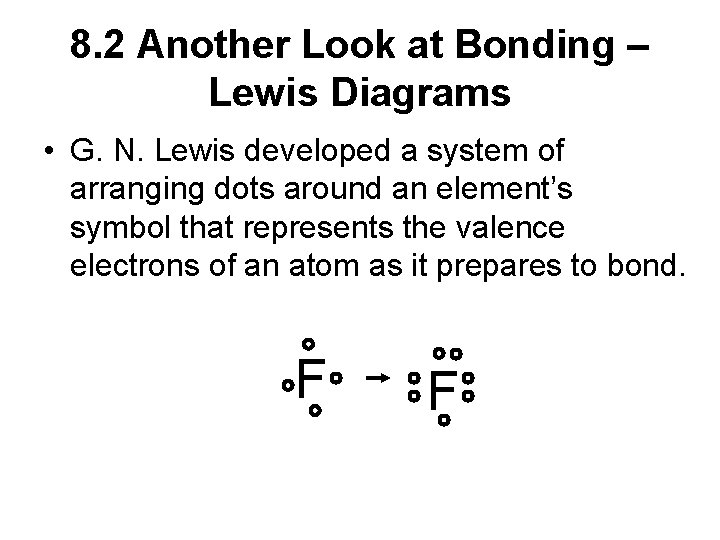
8. 2 Another Look at Bonding – Lewis Diagrams • G. N. Lewis developed a system of arranging dots around an element’s symbol that represents the valence electrons of an atom as it prepares to bond. F F

Lewis Diagrams and Covalent Bonds • A Lewis diagram is a simpler version of a Bohr diagram that has only valence electrons illustrated. • The single valence electrons (unpaired) pair up with another single electron from another atom to form a shared pair, or bonding pair, of electrons. Lewis diagrams are also called electron dot diagrams.

Lewis Diagrams for Atoms • A Lewis diagram can be drawn for any atom by following a few simple rules. 1. Determine the number of valence electrons a. group 1 and 2 have 1 and 2 valence electrons. b. group 13– 18 have 3 -8 valence electrons.

Lewis diagrams 2. Arrange the valence electrons as dots around the atom’s symbol, just like you would in a bohr diagram. One dot per side (up to four) and then start pairing them up

Lewis Diagrams for Molecules • Lewis diagrams can be drawn for molecules. These diagrams illustrate how the atoms come together to form covalent bonds. • To draw a Lewis diagram for a molecule, you must first be given the molecular formula. Atoms are then connected to one another by pairs of shared electrons, or bonding pairs.

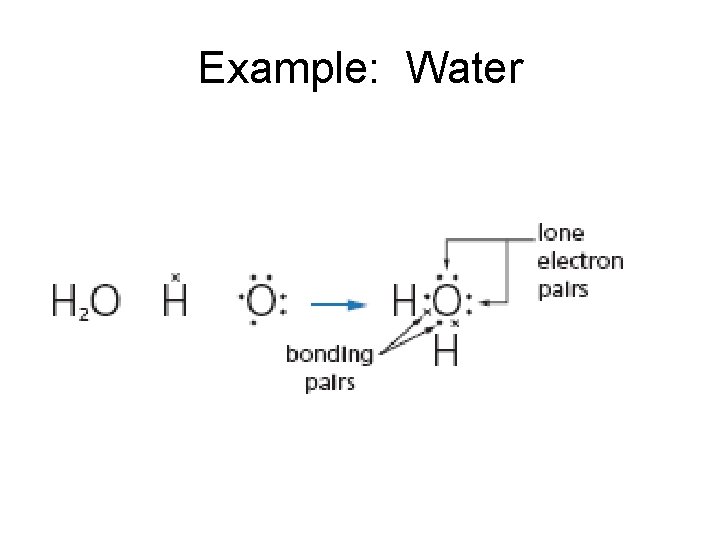
Example: Water

Lewis Diagrams • The purpose of a Lewis diagram is to illustrate that the formation of these covalent molecules is possible. • Lewis diagrams can also be drawn for ions and ionic compounds, but we will not be doing this.