Chapter 8 Chemical Reactions 8 3 Balancing a
Chapter 8 Chemical Reactions 8. 3 Balancing a Chemical Equation 1
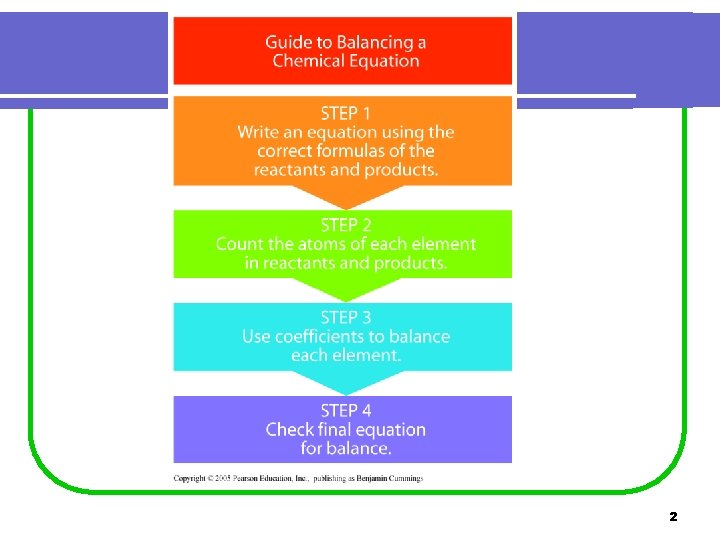
2

Balancing Chemical Equations 1. Write the equation with the correct formulas. NH 3(g) + O 2(g) NO(g) + H 2 O(g) 2. Determine if the equation is balanced. No, not all atoms are balanced. 3. Balance with coefficients in front of formulas. 4 NH 3 + 5 O 2 4 NO + 6 H 2 O 4. Check that atoms of each element are equal in reactants and products. 4 N (4 x 1 N) = 12 H (4 x 3 H) = 10 O (5 x 2 O) = 4 N (4 x 1 N) 12 H (6 x 2 H) 10 O (4 O + 6 O) 3

Steps in Balancing an Equation To balance the following equation, Fe 3 O 4(s) + H 2(g) Fe(s) + H 2 O(l) • Work on one element at a time. • Use only coefficients in front of formulas. • Do not change any subscripts. Fe: Fe 3 O 4(s) + H 2(g) 3 Fe(s) + H 2 O(l) O: Fe 3 O 4(s) + H 2(g) 3 Fe(s) + 4 H 2 O(l) H: Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) 4

Learning Check the balance of atoms in the following: Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) A. Number of H atoms in products. 1) 2 2) 4 3) 8 B. Number of O atoms in reactants. 1) 2 2) 4 3) 8 C. Number of Fe atoms in reactants. 1) 1 2) 3 3) 4 5

Solution Fe 3 O 4(s) + 4 H 2(g) 3 Fe(s) + 4 H 2 O(l) A. Number of H atoms in products. 3) 8 (4 H 2 O) B. Number of O atoms in reactants. 2) 4 (Fe 3 O 4) C. Number of Fe atoms in reactants. 2) 3 (Fe 3 O 4) 6
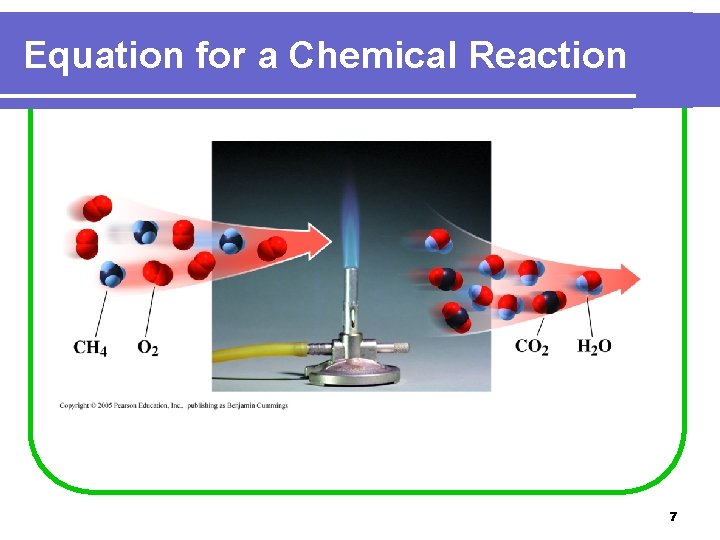
Equation for a Chemical Reaction 7
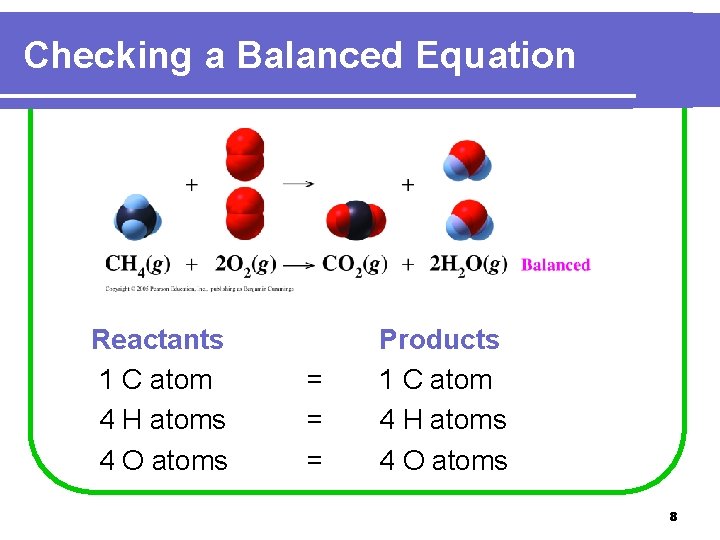
Checking a Balanced Equation Reactants 1 C atom 4 H atoms 4 O atoms = = = Products 1 C atom 4 H atoms 4 O atoms 8

Learning Check Balance each equation and list the coefficients in the balanced equation going from reactants to products: A. __Mg(s) + __N 2(g) 1) 1, 3, 2 B. __Al(s) 1) 3, 3, 2 __Mg 3 N 2(s) 2) 3, 1, 2 + __Cl 2(g) 3) 3, 1, 1 __Al. Cl 3(s) 2) 1, 3, 1 3) 2, 3, 2 9

Solution A. 3) 3, 1, 1 3 Mg(s) + 1 N 2(g) 1 Mg 3 N 2(s) B. 3) 2, 3, 2 2 Al(s) + 3 Cl 2(g) 2 Al. Cl 3(s) 10
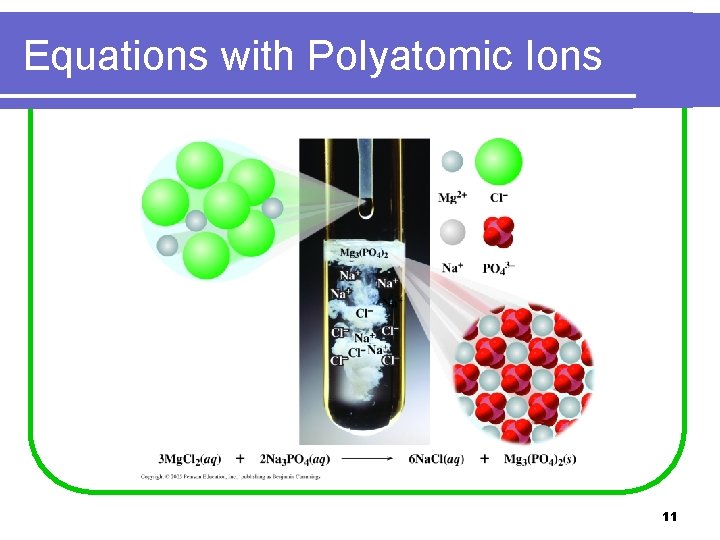
Equations with Polyatomic Ions 11
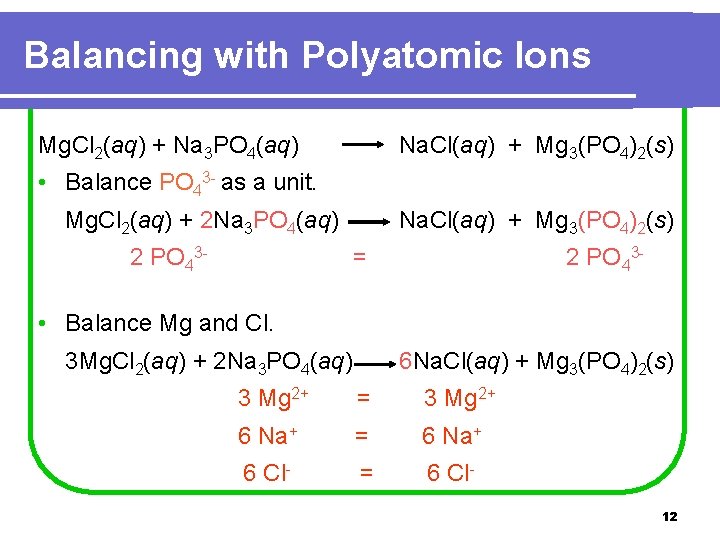
Balancing with Polyatomic Ions Mg. Cl 2(aq) + Na 3 PO 4(aq) Na. Cl(aq) + Mg 3(PO 4)2(s) • Balance PO 43 - as a unit. Mg. Cl 2(aq) + 2 Na 3 PO 4(aq) 2 PO 43 - Na. Cl(aq) + Mg 3(PO 4)2(s) = 2 PO 43 - • Balance Mg and Cl. 3 Mg. Cl 2(aq) + 2 Na 3 PO 4(aq) 6 Na. Cl(aq) + Mg 3(PO 4)2(s) 3 Mg 2+ = 3 Mg 2+ 6 Na+ = 6 Na+ 6 Cl- = 6 Cl 12

Learning Check Balance and list the coefficients from reactants to products: A. __Fe 2 O 3(s) + __C(s) 1) 2, 3, 2, 3 __Fe(s) 2) 2, 3, 4, 3 3) 1, 1, 2, 3 B. __Al(s) + __Fe. O(s) 1) 2, 3, 3, 1 1) 3, 2, 1, 2 __Fe(s) + 2) 2, 1, 1, 1 C. __Al(s) + __H 2 SO 4(aq) + __CO 2(g) __Al 2 O 3(s) 3) 3, 3, 3, 1 __Al 2(SO 4)3(aq) + __H 2(g) 2) 2, 3, 1, 3 3) 2, 3, 2, 3 13

Solution A. 2) 2, 3, 4, 3 2 Fe 2 O 3(s) + 3 C(s) 4 Fe(s) + 3 CO 2(g) B. 1) 2, 3, 3, 1 2 Al(s) + 3 Fe. O(s) C. 2) 2, 3, 1, 3 2 Al(s) + 3 H 2 SO 4(aq) 3 Fe(s) + 1 Al 2 O 3(s) 1 Al 2(SO 4)3(aq) + 3 H 2(g) 14
- Slides: 14