Chapter 8 Chemical Bonding I Basic Concepts Copyright

Chapter 8 Chemical Bonding I: Basic Concepts Copyright Mc. Graw-Hill 2009 1

8. 1 Lewis Dot Symbols • Valence electrons determine an element’s chemistry. • Lewis dot symbols represent the valence electrons of an atom as dots arranged around the atomic symbol. • Most useful for main-group elements Copyright Mc. Graw-Hill 2009 2
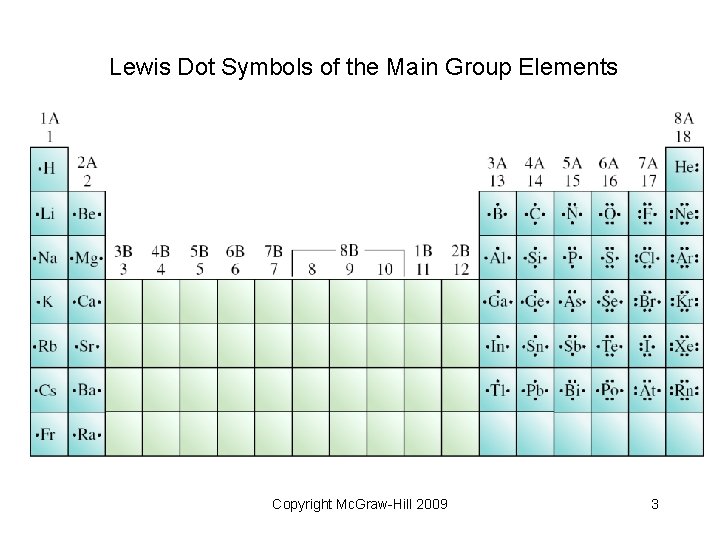
Lewis Dot Symbols of the Main Group Elements Copyright Mc. Graw-Hill 2009 3

Write Lewis dot symbols for the following: (a) N (b) S 2 (c) K+ Copyright Mc. Graw-Hill 2009 4
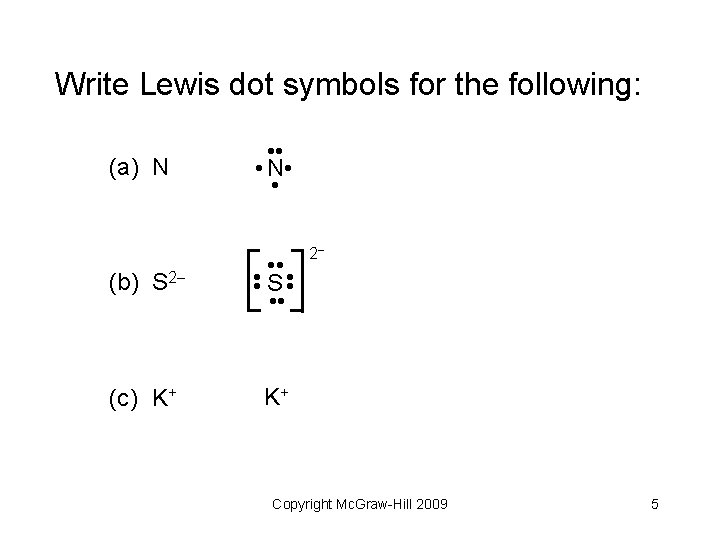
Write Lewis dot symbols for the following: (a) N (c) K+ • • S • • 2 • • (b) S 2 • • • N • • K+ Copyright Mc. Graw-Hill 2009 5

8. 2 Ionic Bonding • Ionic bond: electrostatic force that holds oppositely charge particles together • Formed between cations and anions • Example Na+ + • • Cl • • + • • Na • • • Cl • • • IE 1 + EA 1 = 496 k. J/mol 349 k. J/mol = 147 k. J/mol m. p. = 801 o. C = – 410. 9 k. J/mol Copyright Mc. Graw-Hill 2009 6
Microscopic View of Na. Cl Formation Copyright Mc. Graw-Hill 2009 7

• Lattice energy = the energy required to completely separate one mole of a solid ionic compound into gaseous ions + - - + + - Na. Cl(s) Na+(g) + Cl (g) + - + + - Hlattice = +788 k. J/mol Because they are defined as an amount of energy, lattice energies are always positive. Copyright Mc. Graw-Hill 2009 8

• Coulombic attraction: Q 1 Q = amount of charge d = distance of separation Q 2 d • Lattice energy (like a coulombic force) depends on • Magnitude of charges • Distance between the charges Copyright Mc. Graw-Hill 2009 9
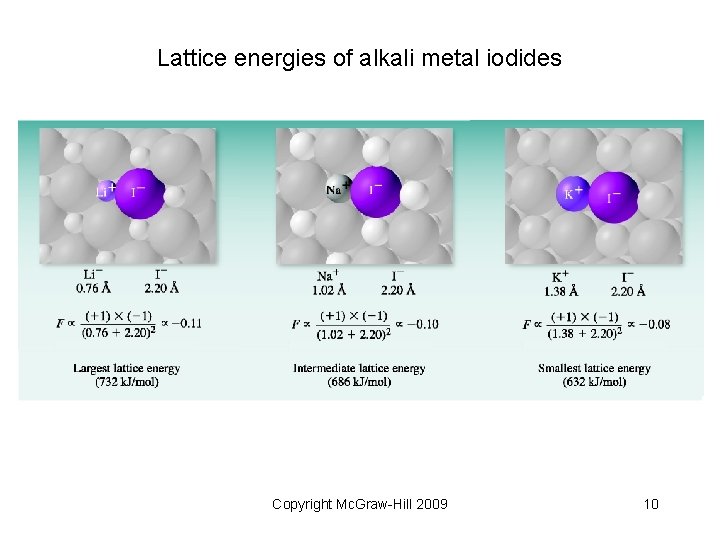
Lattice energies of alkali metal iodides Copyright Mc. Graw-Hill 2009 10
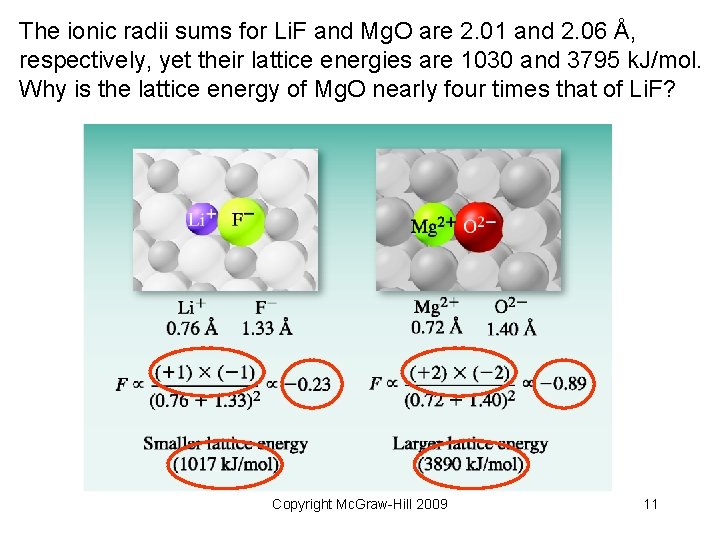
The ionic radii sums for Li. F and Mg. O are 2. 01 and 2. 06 Å, respectively, yet their lattice energies are 1030 and 3795 k. J/mol. Why is the lattice energy of Mg. O nearly four times that of Li. F? Copyright Mc. Graw-Hill 2009 11
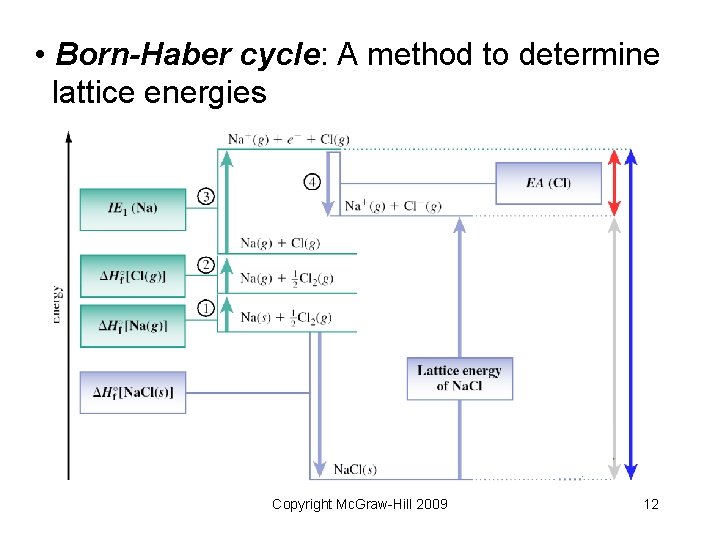
• Born-Haber cycle: A method to determine lattice energies Copyright Mc. Graw-Hill 2009 12

• Born-Haber cycle for Ca. O Ca(s) #1 Ca(g) #2 Ca 2+(g) + + (1/2) O 2(g) #3 O(g) #4 O 2 (g) #6 Ca. O(s) #5 #1 Heat of sublimation = Hf [Ca(g)] = +178 k. J/mol #2 1 st & 2 nd ionization energies = I 1(Ca) + I 2(Ca) = +1734. 5 k. J/mol #3 (1/2) Bond enthalpy = (1/2) D(O=O) = Hf [O(g)] = +247. 5 k. J/mol #4 1 st & 2 nd electron affinities = EA 1(O) + EA 2(O) = +603 k. J/mol #5 (Lattice Energy) = Hlattice[Ca. O(s)] = (the unknown) #6 Standard enthalpy of formation = Hf [Ca. O(s)] = 635 k. J/mol Hlattice = +3398 k. J/mol +178 +1734. 5 +247. 5 +603 Hlatt = 635 Copyright Mc. Graw-Hill 2009 13

8. 3 Covalent Bonding • Atoms share electrons to form covalent bonds. + • H HH • • H • or H–H • In forming the bond the atoms achieve a more stable electron configuration. Copyright Mc. Graw-Hill 2009 14

• Octet: Eight is a “magic” number of electrons. • Octet Rule: Atoms will gain, lose, or share electrons to acquire eight valence electrons Examples: Na+ Copyright Mc. Graw-Hill 2009 • • O H H • • H • + • O • • • + • • Cl • • + • • Na • • • Cl • • • 15
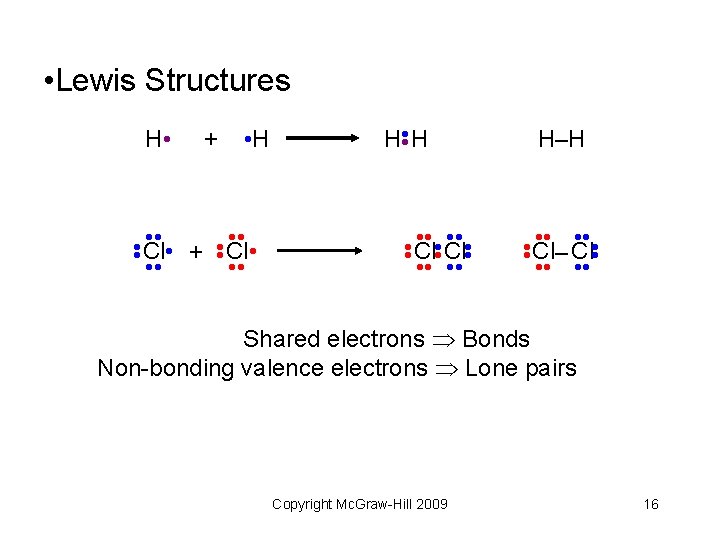
• Lewis Structures + • H H–H • • Cl– Cl • • • • Cl Cl • • • • • • Cl • + Cl • • • HH • • H • Shared electrons Bonds Non-bonding valence electrons Lone pairs Copyright Mc. Graw-Hill 2009 16

• Multiple Bonds - The number of shared electron pairs is the number of bonds. • • Single Bond • • O C O • • O =C=O • • Double Bond N • • • • N • • Cl– Cl • • • • • • Cl Cl • • Triple Bond Copyright Mc. Graw-Hill 2009 17

• Bond strength and bond length bond strength single < double < triple bond length single > double > triple Bond Strength Bond Length N–N N=N N N 163 k. J/mol 418 k. J/mol 941 k. J/mol 1. 47 Å 1. 24 Å 1. 10 Å Copyright Mc. Graw-Hill 2009 18

8. 4 Electronegativity and Polarity • Nonpolar covalent bond = electrons are shared equally by two bonded atoms • Polar covalent bond = electrons are shared unequally by two bonded atoms Copyright Mc. Graw-Hill 2009 19

• Electron density distributions + red high electron density green intermediate electron density blue low electron density - H–F alternate representations Copyright Mc. Graw-Hill 2009 20

• Electronegativity: ability of an atom to draw shared electrons to itself. - More electronegative elements attract electrons more strongly. • relative scale • related to IE and EA • unitless • smallest electronegativity: Cs 0. 7 • largest electronegativity: F 4. 0 Copyright Mc. Graw-Hill 2009 21

Electronegativity: The Pauling Scale Copyright Mc. Graw-Hill 2009 22
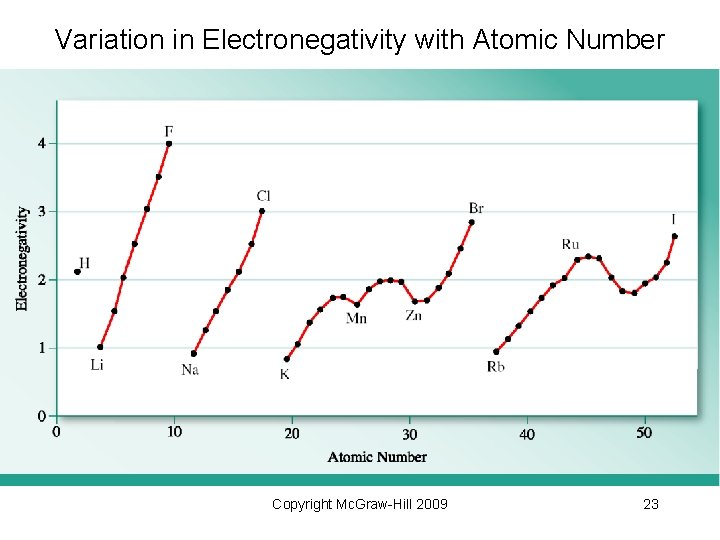
Variation in Electronegativity with Atomic Number Copyright Mc. Graw-Hill 2009 23
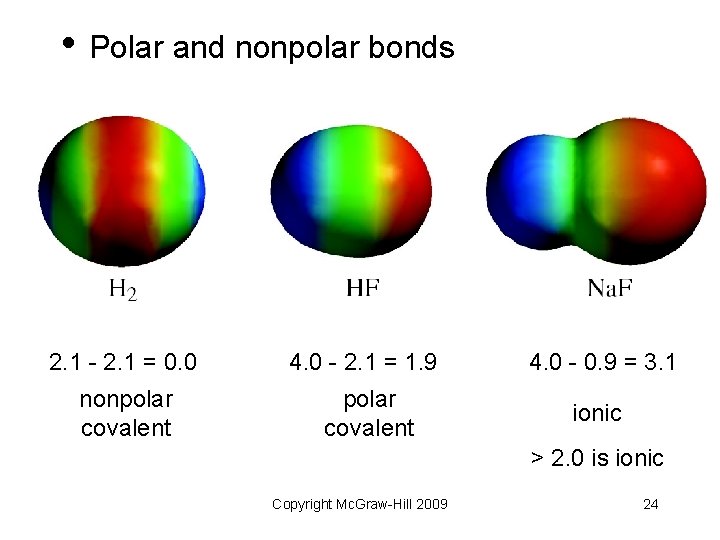
• Polar and nonpolar bonds 2. 1 - 2. 1 = 0. 0 4. 0 - 2. 1 = 1. 9 4. 0 - 0. 9 = 3. 1 nonpolar covalent ionic > 2. 0 is ionic Copyright Mc. Graw-Hill 2009 24
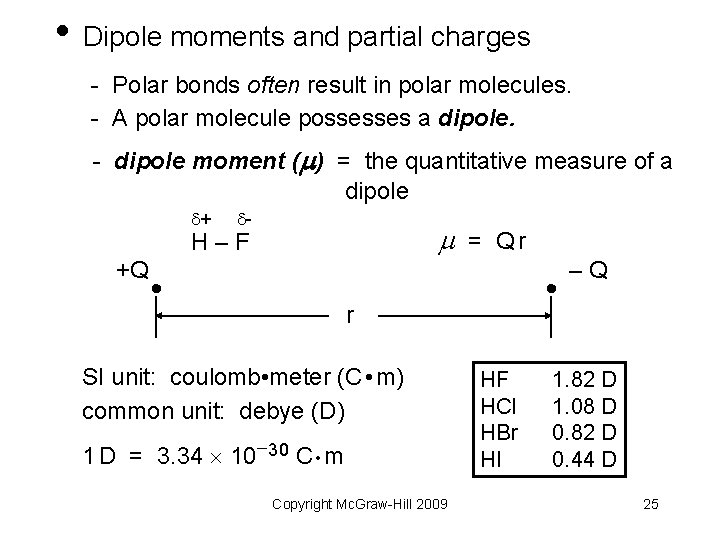
• Dipole moments and partial charges - Polar bonds often result in polar molecules. - A polar molecule possesses a dipole. - dipole moment ( ) = the quantitative measure of a dipole + - = Qr H–F +Q –Q r SI unit: coulomb • meter (C • m) common unit: debye (D) 1 D = 3. 34 10 30 C • m Copyright Mc. Graw-Hill 2009 HF HCl HBr HI 1. 82 D 1. 08 D 0. 82 D 0. 44 D 25

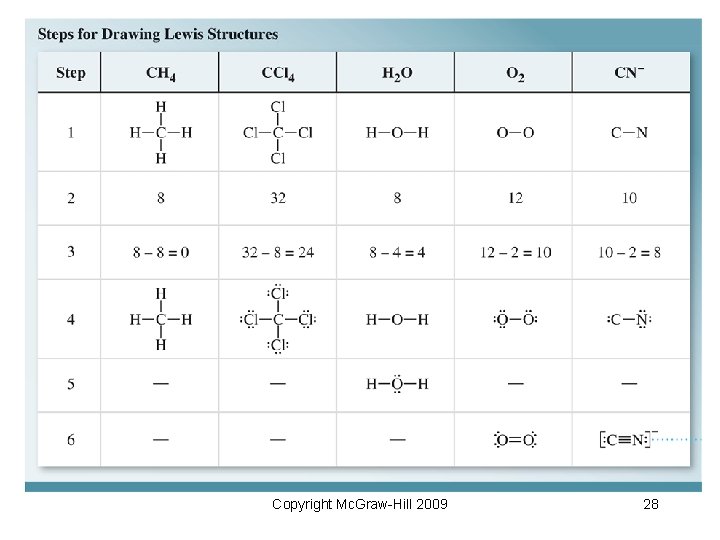
8. 5 Drawing Lewis Structures 1) Draw skeletal structure with the central atom being the least electronegative element. 2) Sum the valence electrons. Add 1 electron for each negative charge and subtract 1 electron for each positive charge. 3) Subtract 2 electrons for each bond in the skeletal structure. 4) Complete electron octets for atoms bonded to the central atom except for hydrogen. 5) Place extra electrons on the central atom. 6) Add multiple bonds if atoms lack an octet. Copyright Mc. Graw-Hill 2009 26
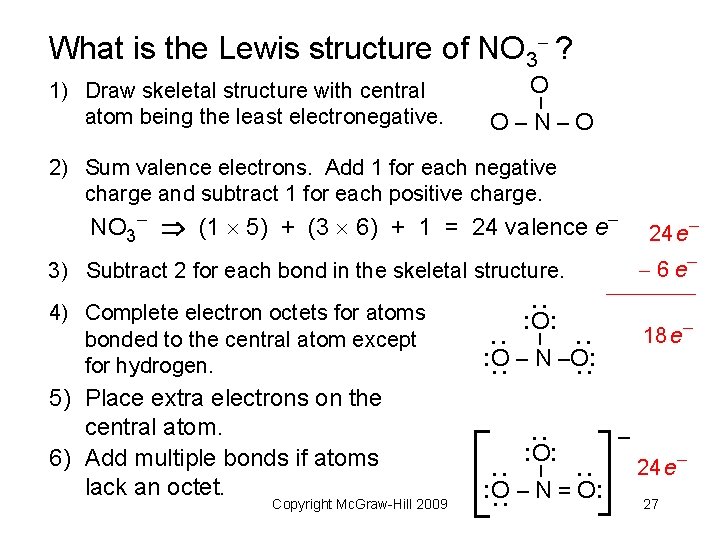
What is the Lewis structure of NO 3 ? O – 1) Draw skeletal structure with central atom being the least electronegative. O–N–O 2) Sum valence electrons. Add 1 for each negative charge and subtract 1 for each positive charge. NO (1 5) + (3 6) + 1 = 24 valence e 3 24 e 6 e 4) Complete electron octets for atoms bonded to the central atom except for hydrogen. 18 e : : : O – N –O: : – – : O: : Copyright Mc. Graw-Hill 2009 : O: : : 5) Place extra electrons on the central atom. 6) Add multiple bonds if atoms lack an octet. : – 3) Subtract 2 for each bond in the skeletal structure. : O – N = O: 24 e 27
Copyright Mc. Graw-Hill 2009 28

8. 6 Lewis Structures and Formal Charge • The electron surplus or deficit, relative to the free atom, that is assigned to an atom in a Lewis structure. Formal = Charge Total nonbonding electrons 11 2 Total bonding electrons : : Example: Total valence electrons H 2 O = H : O : H H: orig. valence e = 1 non-bonding e = 0 1/2 bonding e = 1 formal charge = 0 O: orig. valence e = non-bonding e = 1/2 bonding e = formal charge = 6 4 2 0 Formal charges are not “real” charges. Copyright Mc. Graw-Hill 2009 29

Example: Formal charges on the atoms in ozone Copyright Mc. Graw-Hill 2009 30

Formal charge guidelines − A Lewis structure with no formal charges is generally better than one with formal charges. − Small formal charges are generally better than large formal charges. − Negative formal charges should be on the more electronegative atom(s). H Example: H C O H or H O ? H Answer: • • C C + • • O H H Copyright Mc. Graw-Hill 2009 C H • • O • • 31

Identify the best structure for the isocyanate ion below: : C = N = O: 2 0 : C N – O: 1 +1 – 1 : C – N O: : – : (c) +1 : : (b) : : (a) – 3 +1 +1 Copyright Mc. Graw-Hill 2009 32
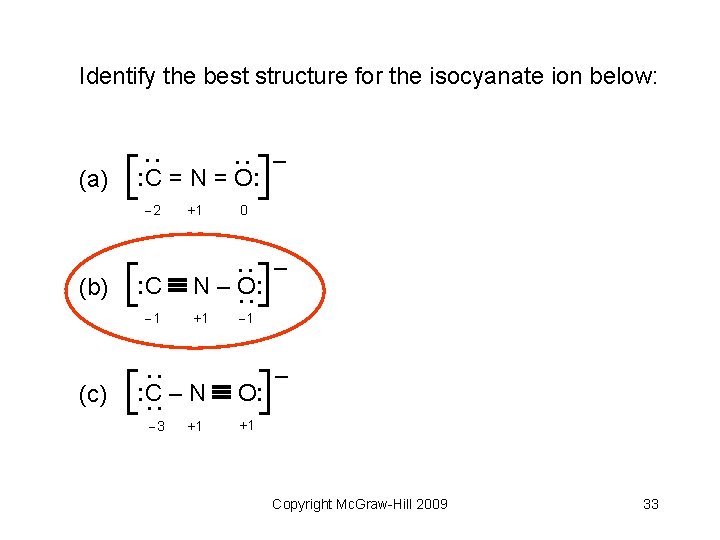
Identify the best structure for the isocyanate ion below: : C = N = O: 2 0 : C N – O: 1 +1 – 1 : C – N O: : – : (c) +1 : : (b) : : (a) – 3 +1 +1 Copyright Mc. Graw-Hill 2009 33
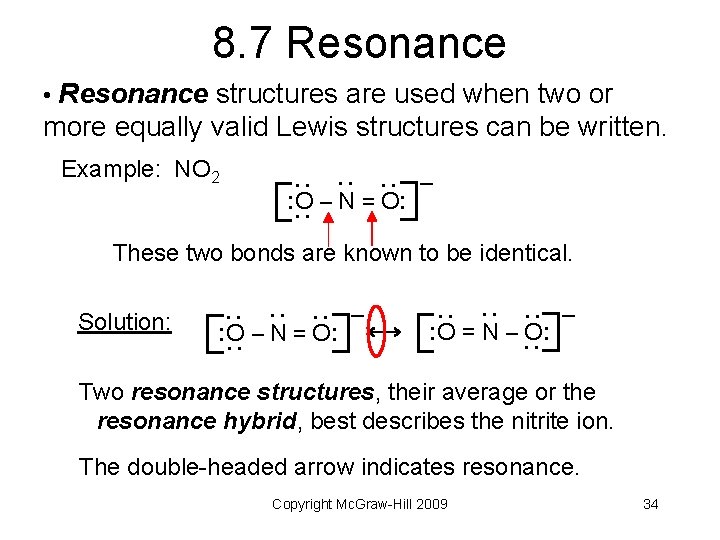
8. 7 Resonance • Resonance structures are used when two or more equally valid Lewis structures can be written. : : : : Example: NO 2 : O – N = O: – These two bonds are known to be identical. : : : : O – N = O: – : : : Solution: : O = N – O: – Two resonance structures, their average or the resonance hybrid, best describes the nitrite ion. The double-headed arrow indicates resonance. Copyright Mc. Graw-Hill 2009 34
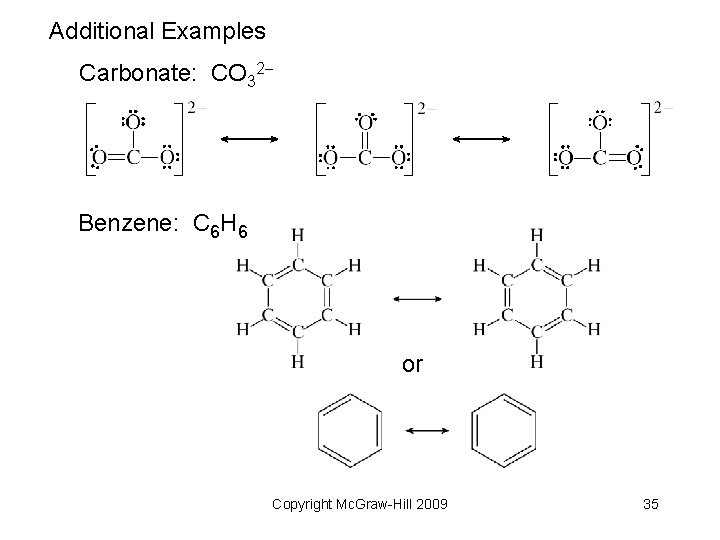
Additional Examples Carbonate: CO 32 Benzene: C 6 H 6 or Copyright Mc. Graw-Hill 2009 35

8. 8 Exceptions to the Octet Rule • Exceptions to the octet rule fall into three categories: − Molecules with an incomplete octet − Molecules with an odd number of electrons − Molecules with an expanded octet Copyright Mc. Graw-Hill 2009 36
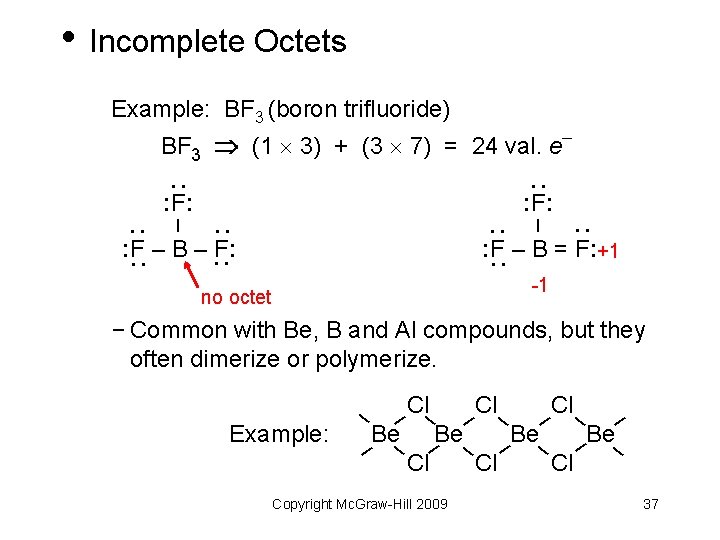
• Incomplete Octets Example: BF 3 (boron trifluoride) BF 3 (1 3) + (3 7) = 24 val. e : : : F: : – : : F – B = F: +1 : : F – B – F: -1 no octet − Common with Be, B and Al compounds, but they often dimerize or polymerize. Example: Cl– Cl– – – Be– Be– – – Cl – Copyright Mc. Graw-Hill 2009 37

• Odd Numbers of Electrons Example: NO (nitrogen monoxide or nitric oxide) : : NO (1 5) + (1 6) = 11 valence e 0. 0 1 +1. Are these both : N = O: equally good? better Example: NO 2 (nitrogen dioxide) NO 2 (1 5) + (2 6) = 17 val. e best +1. 1 : O = N – O: 1 +1. 0 : : O – N = O: 0 : : : O = N – O: 0 : O – N = O: Are these all equally good? Copyright Mc. Graw-Hill 2009 38
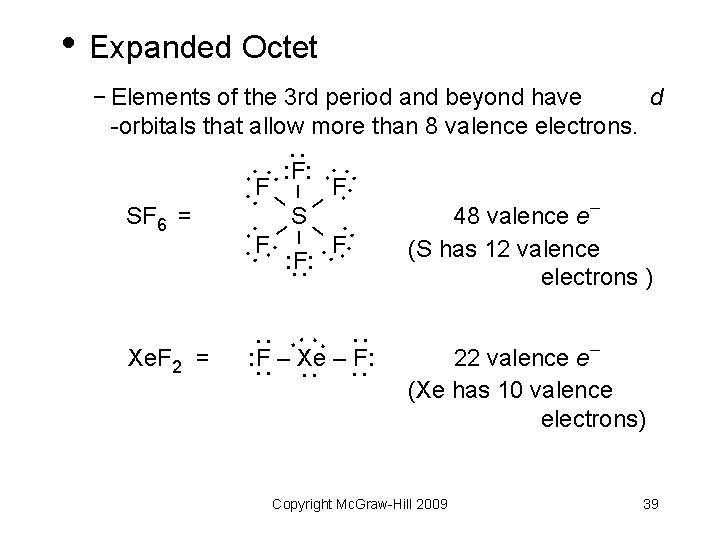
• Expanded Octet − Elements of the 3 rd period and beyond have d -orbitals that allow more than 8 valence electrons. : : F: : F– F: – : –S – F: F: : – SF 6 = : : : : : : F – Xe – F: : Xe. F 2 = 48 valence e (S has 12 valence electrons ) 22 valence e (Xe has 10 valence electrons) Copyright Mc. Graw-Hill 2009 39

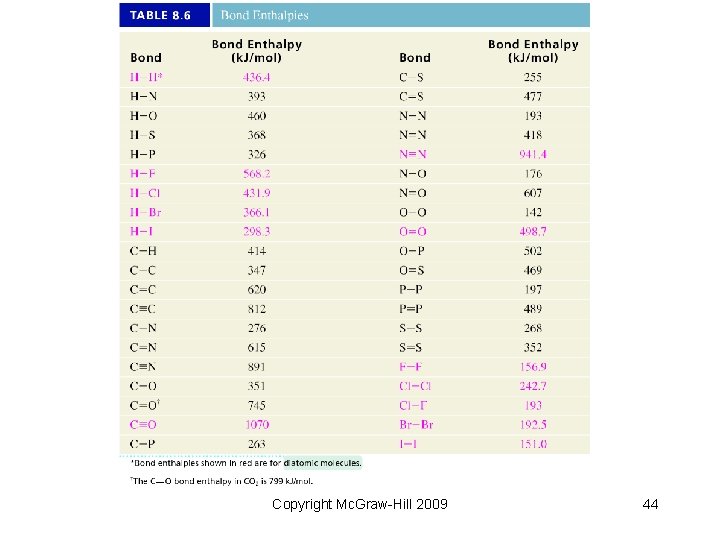
8. 9 Bond Enthalpy • Bond enthalpy is the energy associated with breaking a particular bond in one mole of gaseous molecules. − Bond enthalpy is one measure of molecular stability. − Symbol: Ho − For diatomic molecules these are accurately measured quantities. Cl 2(g) Cl(g) + Cl(g) Ho = 243. 4 k. J HCl(g) H(g) + Cl(g) Ho = 431. 9 k. J single bonds O 2(g) O(g) + O(g) Ho = 495. 0 k. J double bond N 2(g) N(g) + N(g) triple bond Ho = 945. 4 k. J Copyright Mc. Graw-Hill 2009 40
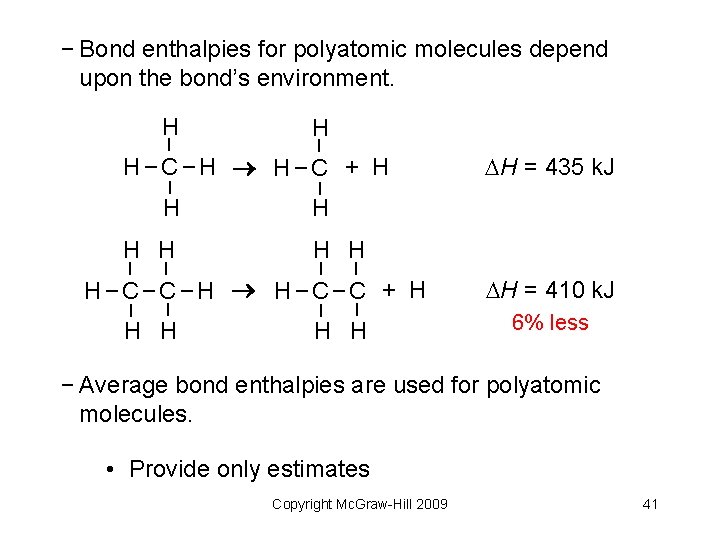
− Bond enthalpies for polyatomic molecules depend upon the bond’s environment. – – – H H–C–H H–C + H H H – – – H H H–C–C–H H–C–C + H – H H H = 435 k. J H H H = 410 k. J 6% less − Average bond enthalpies are used for polyatomic molecules. • Provide only estimates Copyright Mc. Graw-Hill 2009 41
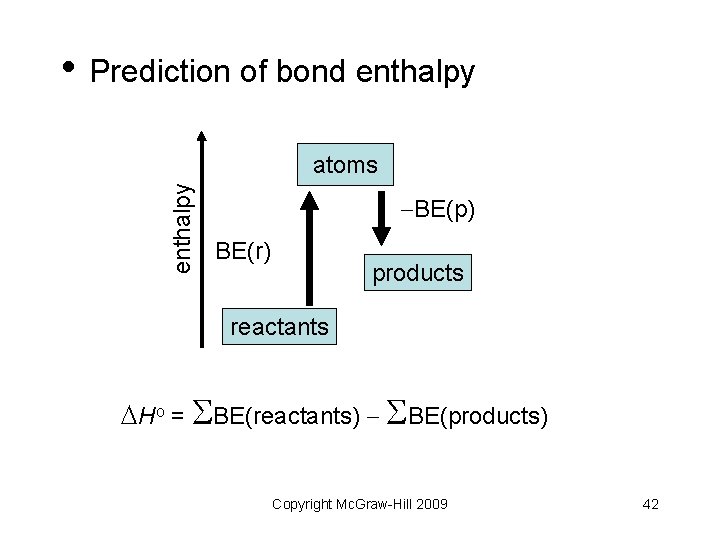
• Prediction of bond enthalpy atoms BE(p) BE(r) products reactants Ho = BE(reactants) BE(products) Copyright Mc. Graw-Hill 2009 42

Example: Calculate the enthalpy of reaction for CH 4(g) + Br 2(g) CH 3 Br(g) + HBr(g) Solution: Consider ONLY bonds broken or formed. H H + Br – – H–C–H H – C – Br – – H H + H – Br Hrxn = [ BE(C–H) + BE(Br–Br) ] – [ BE(C–Br) + BE(H–Br) ] = [ (413) + (193) ] – [ (276) + (366) ] = – 36 k. J/mol Copyright Mc. Graw-Hill 2009 43
Copyright Mc. Graw-Hill 2009 44

Key Points • • • Lewis dot symbols Ionic bonding Lattice energy Born-Haber cycle Covalent bonding Octet rule Lewis structures Bond order Bond polarity Copyright Mc. Graw-Hill 2009 45

Key Points • • • Electronegativity Dipole moment Drawing lewis structures Formal charge Resonance structures Incomplete octets Odd numbers of electrons Expanded octets Bond enthalpy Copyright Mc. Graw-Hill 2009 46
- Slides: 46