Chapter 8 Chemical Bonding and Molecular Structure Chemical

Chapter 8 Chemical Bonding and Molecular Structure Chemical Bonding

WHAT IS A BOND? • Bonds are attractive forces that hold groups of atoms together and make them function as a unit. • Bonding relates to physical properties such as melting point, hardness and electrical and thermal conductivity as well as solubility characteristics. Chemical Bonding

WHAT IS A BOND? • The system is achieving the lowest possible energy state by bonding. • Being bound requires less energy than existing in the elemental form. • It takes energy to break a bond, not make a bond! • Energy is RELEASED when a bond is formed, therefore, it REQUIRES energy Chemical to break a bond. Bonding

CHEMICAL BONDS • Three basic types of bonds: ØIonic • Electrostatic attraction between ions; electrons are transferred ØCovalent • Sharing of electrons ØMetallic • Metal atoms bonded to several other atoms Chemical Bonding

COULOMB’S LAW • used to calculate the energy of an ionic bond. • the energy interaction between a pair of ions. • There is a (-) sign; indicated an attractive force – energy is lower. Chemical Bonding

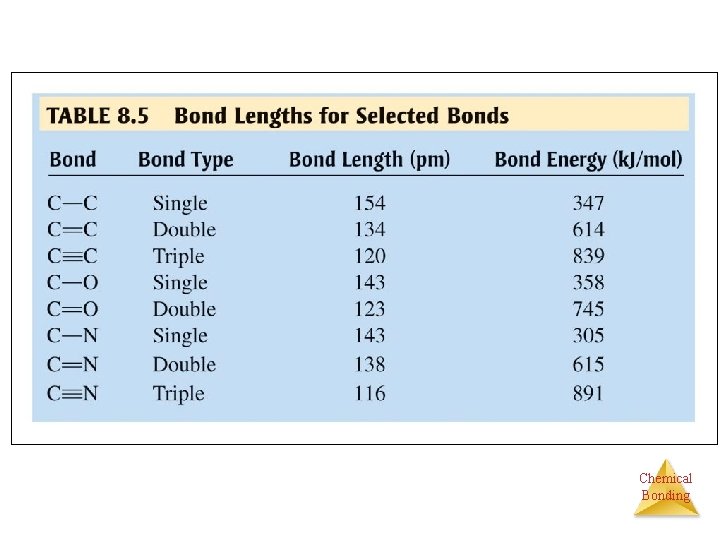
BOND LENGTH Øthe distance between the two nuclei where the system energy is at a minimum between the two nuclei. Øenergy is given off when two atoms achieve greater stability together than apart. § Attractive forces — proton - electron § Repulsive forces — electron - electron § Small energy decrease - van der Waals IMFs § Large energy decrease - chemical bonds Chemical Bonding
Chemical Bonding
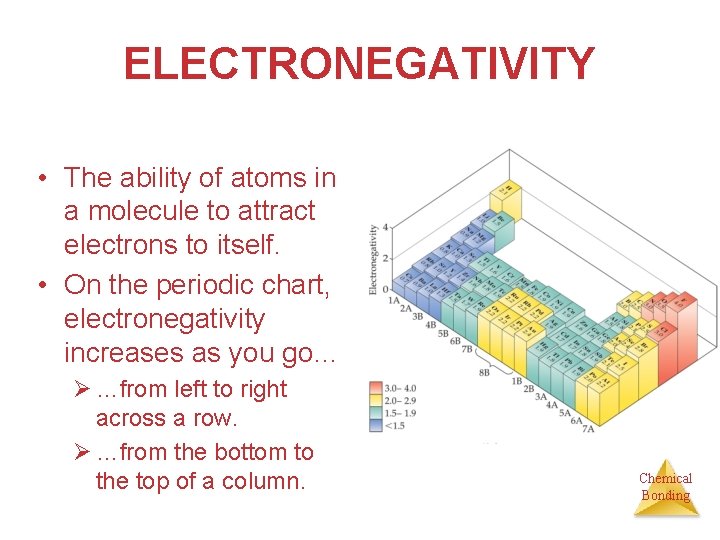
ELECTRONEGATIVITY • The ability of atoms in a molecule to attract electrons to itself. • On the periodic chart, electronegativity increases as you go… Ø …from left to right across a row. Ø …from the bottom to the top of a column. Chemical Bonding

ELECTRONEGATIVITY • Ionic: Eneg difference >1. 7 • Polar Covalent: Eneg difference is ≥ 0. 4 and ≤ 1. 7 • Nonpolar Covalent: Eneg difference <0. 4 Chemical Bonding

PRACTICE ONE • Order the following bonds according to polarity: H—H, O—H, Cl—H, S—H, and F—H. Chemical Bonding

PRACTICE ONE - answer • • • H—H S—H Cl—H O—H F—H 0. 0 difference 0. 4 difference 0. 9 difference 1. 4 difference 1. 9 difference Polarity increases Chemical Bonding

BOND POLARITY AND DIPOLE MOMENTS DIPOLAR MOLECULES § A molecule with a somewhat positive end a somewhat negative end. § a dipole moment. § also molecules with preferential orientation in an electric field; all diatomic molecules with a polar covalent bond are dipolar. Chemical Bonding
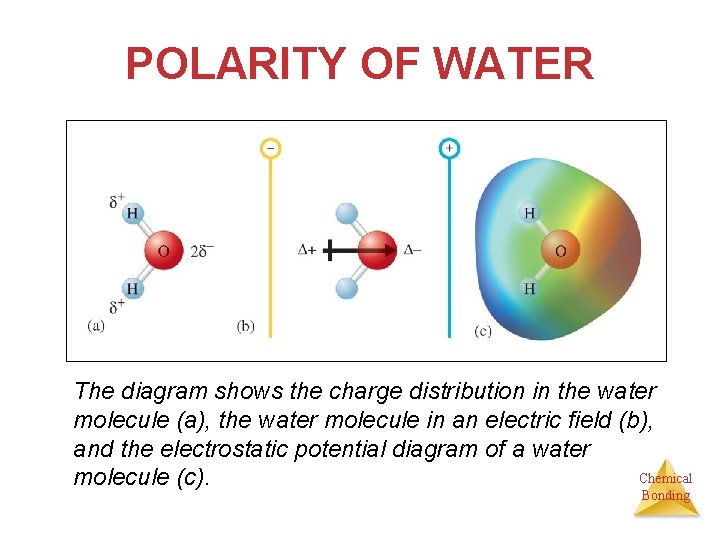
POLARITY OF WATER The diagram shows the charge distribution in the water molecule (a), the water molecule in an electric field (b), and the electrostatic potential diagram of a water Chemical molecule (c). Bonding

IONS: configurations and sizes • Goal is to achieve a noble gas configuration. • COVALENT: two nonmetals share electrons so each has a noble gas configuration. • IONIC: Metal and representative group metal form a binary ionic compound where electrons are transferred so each gets a noble gas configuration Chemical Bonding

IONIC COMPOUNDS • The final result of ionic bonding is a solid, regular array of cations and anions called a crystal lattice; • This configuration limits (-) ion/(-) ion and (+) ion/(+) ion interactions and maximizes (+)ion and (-)ion interactions. • *Ion size plays a role in determining the structure and stability of ionic solids and the properties of ions in aqueous solutions. Chemical Bonding

PRACTICE TWO • Arrange the ions Se 2 -, Br-, Rb+, and Sr+2 in order of decreasing size. Chemical Bonding
![PRACTICE TWO - answer In order of decreasing size: (all have [Kr] electron configuration) PRACTICE TWO - answer In order of decreasing size: (all have [Kr] electron configuration)](http://slidetodoc.com/presentation_image_h2/26cc25b7fbfb15d96821a8a2aa9cbbfa/image-17.jpg)
PRACTICE TWO - answer In order of decreasing size: (all have [Kr] electron configuration) Se 2 - Br- Rb+ Sr+2 Largest smallest Sr+2 has greatest Zeff and thus the strongest attractive force; metal ions are always smaller than their atoms. Chemical Bonding

PRACTICE THREE Choose the largest ion in each of the following groups. a. Li+, Na+, K+, Rb+, Cs+ b. Ba 2+, Cs+, I-, Te 2 - Chemical Bonding

PRACTICE THREE - answer a. Li+, Na+, K+, Rb+, Cs+ - all in the same group; principle quantum number increases. b. Ba 2+, Cs+, I-, Te 2 - Isoelectronic with [Xe] e- configuration; smallest Zeff is the largest. Chemical Bonding

Energies of Ionic Bonding It takes 495 k. J/mol to remove electrons from sodium. Chemical Bonding

Energies of Ionic Bonding We get 349 k. J/mol back by giving electrons to chlorine. Chemical Bonding
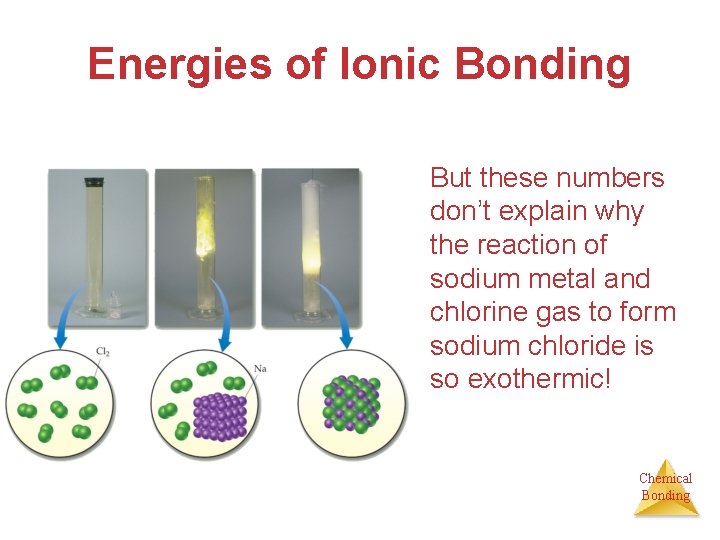
Energies of Ionic Bonding • But these numbers don’t explain why the reaction of sodium metal and chlorine gas to form sodium chloride is so exothermic! Chemical Bonding
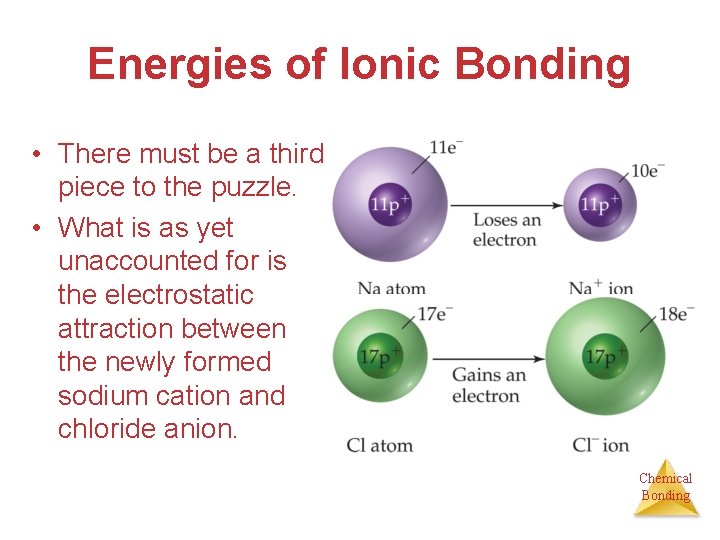
Energies of Ionic Bonding • There must be a third piece to the puzzle. • What is as yet unaccounted for is the electrostatic attraction between the newly formed sodium cation and chloride anion. Chemical Bonding

LATTICE ENERGY • This third piece of the puzzle is the LATTICE ENERGY: The energy required to completely separate a mole of a solid ionic compound into its gaseous ions. • The energy associated with electrostatic interactions is governed by Coulomb’s law: Q 1 Q 2 Eel = r Chemical Bonding

LATTICE ENERGY • Lattice energy increases with the charge on the ions. • It also increases with decreasing size of ions. Chemical Bonding
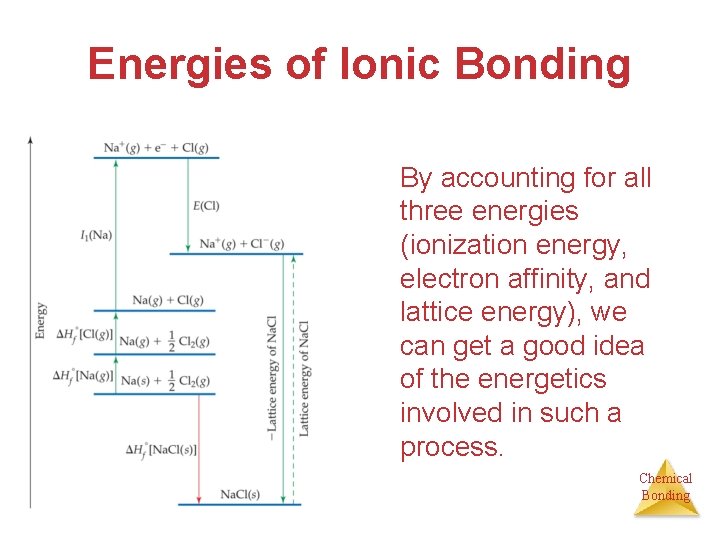
Energies of Ionic Bonding By accounting for all three energies (ionization energy, electron affinity, and lattice energy), we can get a good idea of the energetics involved in such a process. Chemical Bonding

Energies of Ionic Bonding • These phenomena also helps explain the “octet rule. ” • Metals, for instance, tend to stop losing electrons once they attain a noble gas configuration because energy would be expended that cannot be overcome by lattice energies. Chemical Bonding

CALCULATING IONIC CHARACTER Ionic vs. Covalent • Ionic compounds generally have greater than 50% ionic character; Eneg differences greater than 1. 7 • Percent ionic character is difficult to calculate for compounds containing polyatomic ions. Chemical Bonding

COVALENT BONDING • Most compounds are covalently bonded, especially carbon compounds. • Strengths of the Bond Model - associates quantities of energy with the formation of bonds between elements - allows the drawing of structures showing the spatial relationship between atoms in a molecule; provides a visual tool to understanding chemical structure Chemical Bonding

COVALENT BONDING • Weaknesses of the Bond Model - bonds are not actual physical structures; bonds cannot adequately explain some phenomena like resonance. Chemical Bonding

COVALENT BONDING • Atoms form covalent bonds because they seek the lowest possible energy. • We can calculate the ΔH for most chemical reactions by comparing the energy required versus energy released. Chemical Bonding
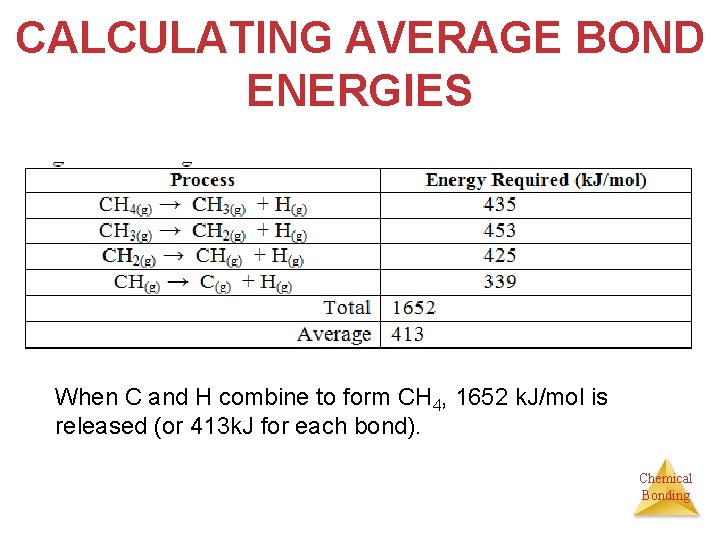
CALCULATING AVERAGE BOND ENERGIES When C and H combine to form CH 4, 1652 k. J/mol is released (or 413 k. J for each bond). Chemical Bonding

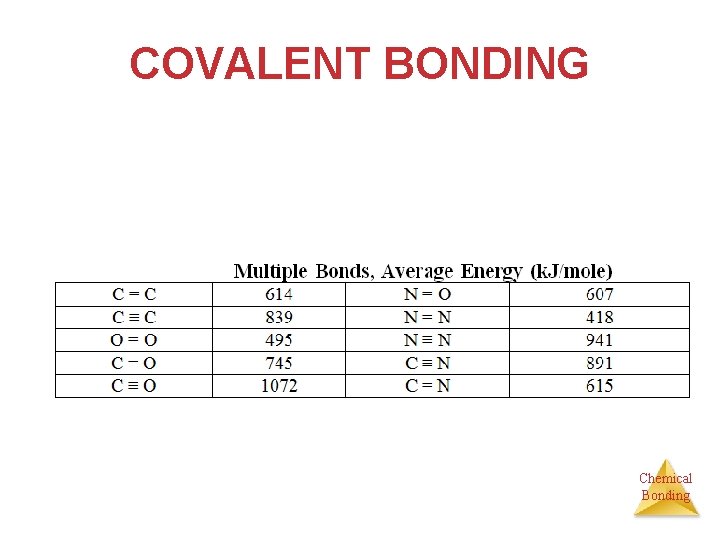
COVALENT BONDING • Single and multiple bonds Øsingle bond - one pair of electrons shared → sigma (σ) bond ØMultiple bonds are most often formed by C, N, O, P and S atoms — C-NOPS Ødouble bond - two pairs of electrons shared → one σ bond and one π bond Øtriple bond - three pairs of electrons shared Chemical → one σ bond and two π bonds Bonding
COVALENT BONDING Chemical Bonding
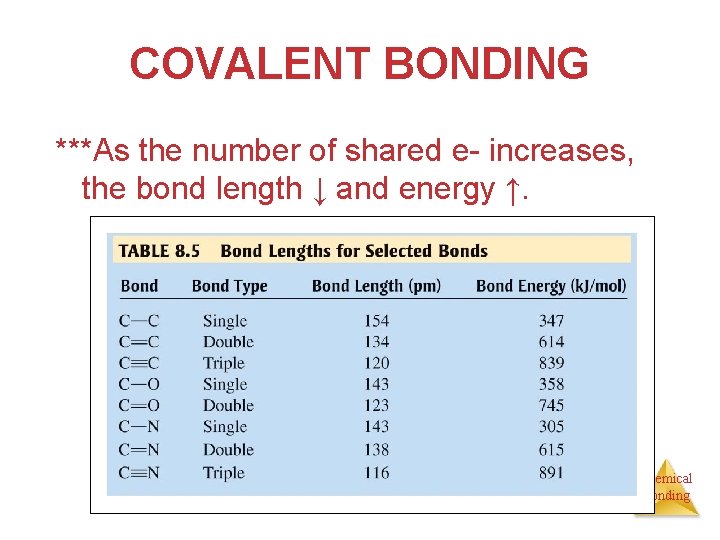
COVALENT BONDING ***As the number of shared e- increases, the bond length ↓ and energy ↑. Chemical Bonding

BOND ENERGY AND ENTHALPY • using bond energy to calculate approximate energies for reactions. • ΔH = sum of the energies required to break old bonds(endothermic) + sum of the energies released in forming new bonds (exothermic). • ΔH =∑D(Bonds broken) -∑D(Bonds formed) • (D represents bond energy per mole of bonds and always has a positive sign) Chemical Bonding

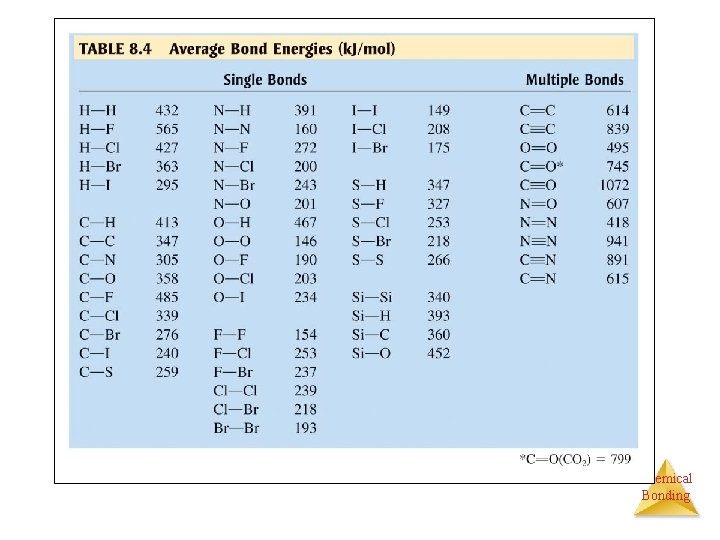
PRACTICE FOUR • Using the bond energies in in the table in your text, calculate Δ H for the reaction of methane with chlorine and fluorine to give Freon, CF 2 Cl 2. CH 4(g) + 2 Cl 2(g) + 2 F 2(g) → CF 2 Cl 2(g) + 2 HF(g) + 2 HCl(g) • Break the bonds and then assemble with new bonds Reactants → atoms → products E required E released Chemical Bonding
Chemical Bonding

PRACTICE FOUR Reactant Bonds Broken: • CH 4: 4 mol C – H 4(413 k. J) • 2 Cl 2: 2 mol Cl – Cl 2(478 k. J) • 2 F 2: 2 mol F – F 2(308 k. J) • Total 2438 k. J Chemical Bonding

PRACTICE FOUR Product Bonds Formed: • CF 2 Cl 2: 2 mol C – F 2(485 k. J) 2 mol C - Cl 2(339 k. J) • HF: 2 mol H – F 2(565 k. J) • HCl: 2 mol H – Cl 2(427 k. J) • Total 3632 k. J Chemical Bonding

PRACTICE FOUR ΔH =∑Dbroken -∑Dformed ΔH =2438 k. J - 3632 k. J = -1194 k. J This energy is released when CF 2 Cl 2 is formed. Chemical Bonding

THE LOCALIZED ELECTRON (LE) BONDING MODEL • Assumes that a molecule is composed of atoms that are bound together by sharing pairs of electrons using the atomic orbitals of the bound atoms. Electron pairs are assumed to be localized on a particular atom - lone pairs - or in the space between two atoms - bonding pairs. • Lone electron pairs - electrons localized on an atom (unshared) • Bonding electron pairs - electrons found in the Chemical space between atoms (shared pairs) Bonding

LE BONDING MODEL Derivations of the Localized Model 1. Lewis Structures describe the valence electron arrangement 2. Geometry of the molecule is predicted with VSEPR 3. Description of the type of atomic orbitals “blended” by the atoms to share electrons or hold lone pairs (hybrids—next chapter). Chemical Bonding

LEWIS STRUCTURES • "the most important requirement for the formation of a stable compound is that the atoms achieve noble gas configurations • Duet rule - hydrogen, lithium, beryllium, and boron form stable molecules when they share two electrons (helium configuration) • Octet Rule - elements carbon and beyond form stable molecules when they are surrounded by eight electrons Chemical Bonding

LEWIS STRUCTURES 1. H is always a terminal atom and connected to only one other atom. 2. Lowest electronegativity element is central atom in molecule. 3. Add the TOTAL number of valence electrons from all atoms. 4. Place one pair of electrons, a σ bond, between each pair of bonded atoms. 5. Arrange the remaining atoms to satisfy the duet rule for hydrogen and the octet rule for the Chemical Bonding second row elements.
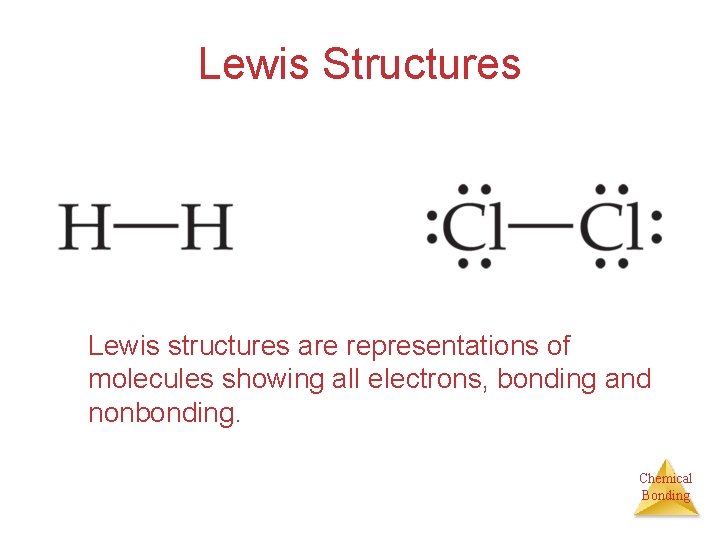
Lewis Structures Lewis structures are representations of molecules showing all electrons, bonding and nonbonding. Chemical Bonding

Writing Lewis Structures PCl 3 5 + 3(7) = 26 1. Find the sum of valence electrons of all atoms in the polyatomic ion or molecule. Ø If it is an anion, add one electron for each negative charge. Ø If it is a cation, subtract one electron for each positive charge. Chemical Bonding

Writing Lewis Structures 2. The central atom is the least electronegative element that isn’t hydrogen. Connect the outer atoms to it by single bonds. Keep track of the electrons: 26 6 = 20 Chemical Bonding
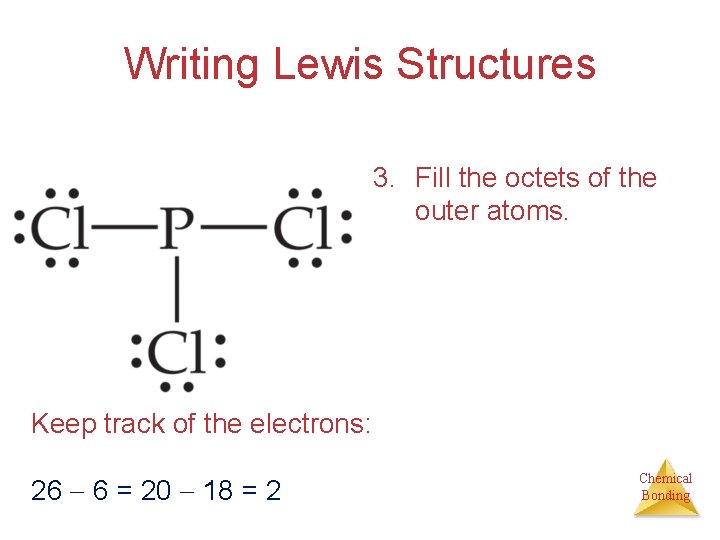
Writing Lewis Structures 3. Fill the octets of the outer atoms. Keep track of the electrons: 26 6 = 20 18 = 2 Chemical Bonding
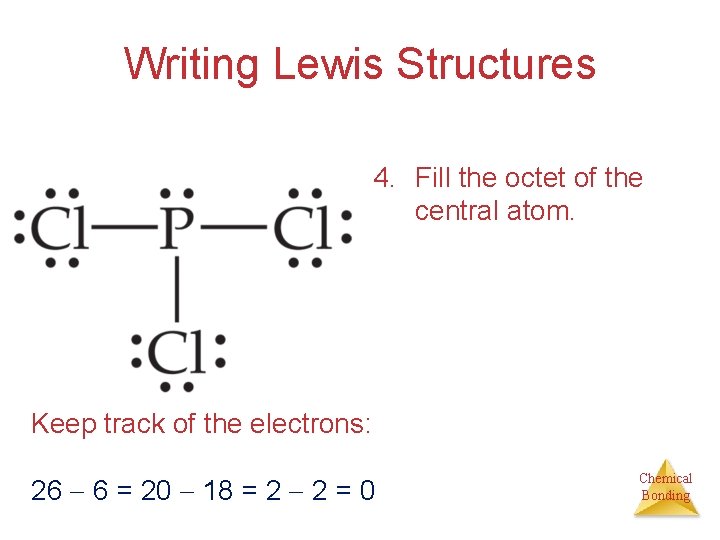
Writing Lewis Structures 4. Fill the octet of the central atom. Keep track of the electrons: 26 6 = 20 18 = 2 2 = 0 Chemical Bonding
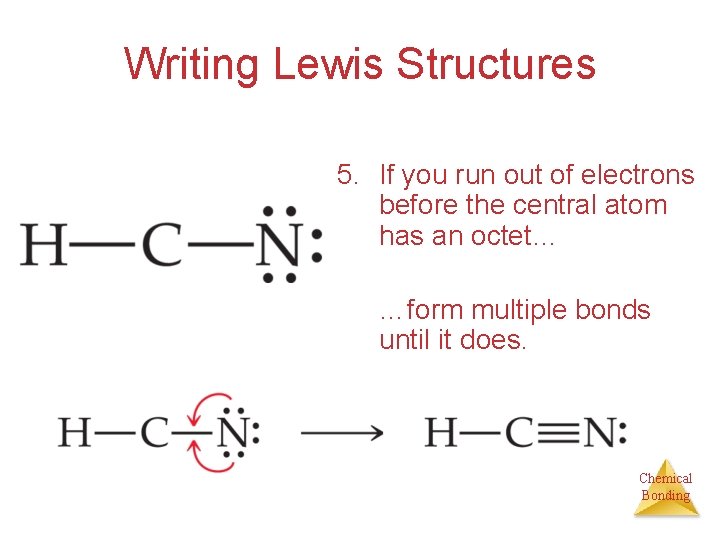
Writing Lewis Structures 5. If you run out of electrons before the central atom has an octet… …form multiple bonds until it does. Chemical Bonding

PRACTICE FIVE – do this now • Give the Lewis structure for each of the following: a. HF b. N 2 c. NH 3 d. CH 4 e. CF 4 f. NO+ Chemical Bonding

PRACTICE FIVE Chemical Bonding

Exceptions to the Octet Rule • There are three types of ions or molecules that do not follow the octet rule: ØIons or molecules with less than an octet. ØIons or molecules with more than eight valence electrons (an expanded octet). ØIons or molecules with an odd number of electrons. Chemical Bonding

LESS THAN OCTET • H at most only two electrons (one bond) • Be. H 2, only 4 valence electrons around Be (only two bonds) • Boron compounds, only 6 valence electrons (three bonds) • ammonia boron trifluoride is a classic Lewis A/B reaction. Chemical Bonding

LESS THAN OCTET Example: Boron Trifluoride 1. Note that boron only has six electrons around it 2. BF 3 is electron deficient and acts as a Lewis acid (electron pair acceptor) 3. Boron can form molecules that obey the octet rule Chemical Bonding

MORE THAN EIGHT ELECTRONS • can only happen if the central element has d-orbitals which means it is from the 3 rd period or greater and can thus be surrounded by more than four valence pairs in certain compounds. • The number of bonds depends on the balance between the ability of the nucleus to attract electrons and the repulsion between the pairs. Chemical Bonding

MORE THAN EIGHT ELECTRONS Example: Sulfur Hexafluoride 1. Note that sulfur has 12 electrons around it, exceeding the octet rule 2. Sulfur hexafluoride is very stable 3. SF 6 fills the 3 s and 3 p orbitals with 8 of the valence electrons, and places the other 4 in the higher energy 3 d orbital Chemical Bonding

Odd Number of Electrons Though relatively rare and usually quite unstable and reactive, there are ions and molecules with an odd number of electrons. NO, NO 2, Cl. O 2 Chemical Bonding

More About the Octet Rule 1. Second row elements C, N, O and F should always obey the octet rule 2. B and Be (second row) often have fewer than eight electrons around them, and form electron deficient, highly reactive molecules 3. Second row elements never exceed the octet rule Chemical Bonding

More About the Octet Rule 4. Third row and heavier elements often satisfy (or exceed) the octet rule 5. Satisfy the octet rule first. If extra electrons remain, place them on elements having available d orbitals. When necessary to exceed the octet rule for one of several third row elements, assume that the extra electrons be placed on the central atom. Chemical Bonding

COORDINATE COVALENT BONDS Some atoms, such as N and P, tend to share a lone pair with another atom that is short of electrons, leading to the formation of a coordinate covalent bond. These bonds are in all coordination compounds and Lewis Acids/Bases. Chemical Bonding

COORDINATE COVALENT BONDS N is sharing the lone PAIR of electrons by drawing an arrow from it to the H+, remember H+ has NO electrons to contribute to the bond. Note that all four bonds are actually identical in length and strength. Chemical Bonding

PRACTICE SIX • Write the Lewis structure for PCl 5. Chemical Bonding
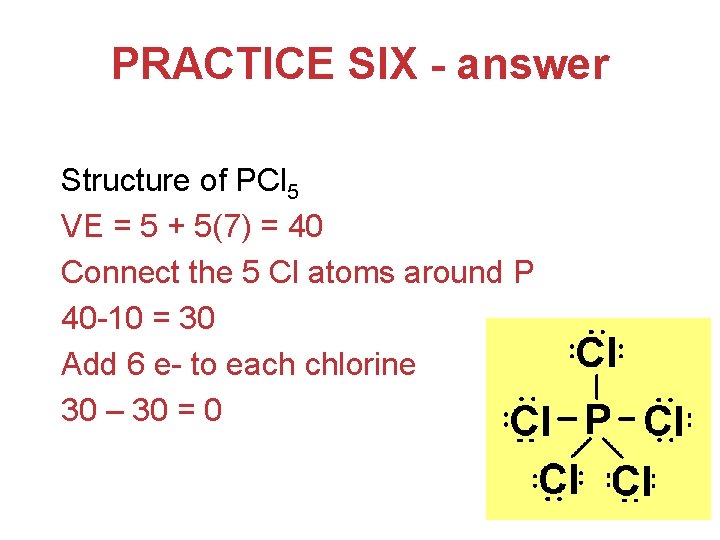
PRACTICE SIX - answer Structure of PCl 5 VE = 5 + 5(7) = 40 Connect the 5 Cl atoms around P 40 -10 = 30 Add 6 e- to each chlorine 30 – 30 = 0 Chemical Bonding
PRACTICE SEVEN Write the Lewis structure for each molecule or ion. a. Cl. F 3 b. Xe. O 3 c. Rn. Cl 2 d. Be. Cl 2 e. ICl 4 - Note that this is a negative one ion. Correct it please Chemical Bonding
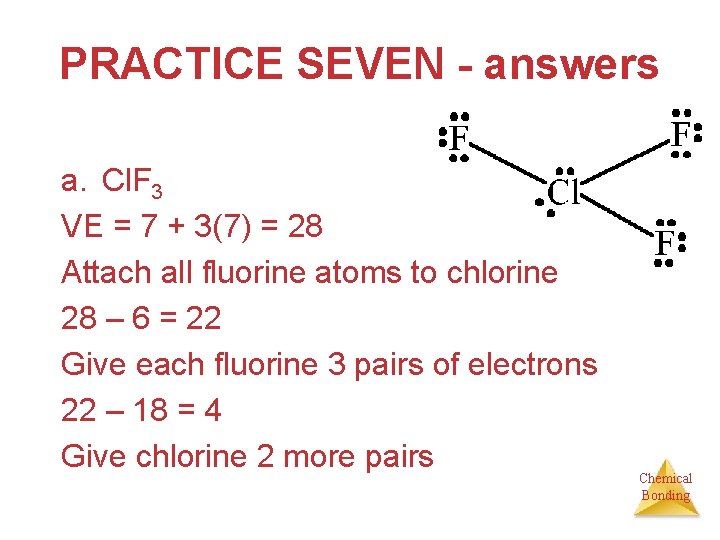
PRACTICE SEVEN - answers a. Cl. F 3 VE = 7 + 3(7) = 28 Attach all fluorine atoms to chlorine 28 – 6 = 22 Give each fluorine 3 pairs of electrons 22 – 18 = 4 Give chlorine 2 more pairs Chemical Bonding

PRACTICE SEVEN - answers b. Xe. O 3 VE = 8 + 3(6) = 26 Attach three oxygen to xenon 26 – 6 = 20 Give each oxygen 3 pairs of electrons 20 – 18 = 2 Xenon gets one more pair of electrons Chemical Bonding

PRACTICE SEVEN - answers c. Rn. Cl 2 VE = 8 + 2(7) = 22 Attach chlorine atoms to radon 22 – 4 = 18 Give each chlorine 3 pairs of electrons 18 – 12 = 6 Radon gets three more pairs of electrons. Chemical Bonding

PRACTICE SEVEN - answers d. Be. Cl 2 VE = 2 + 2(7) = 16 Attach two chlorine to Be 16 – 4 = 12 Give each chlorine 3 pairs of electrons 12 – 12 = 0 Chemical Bonding
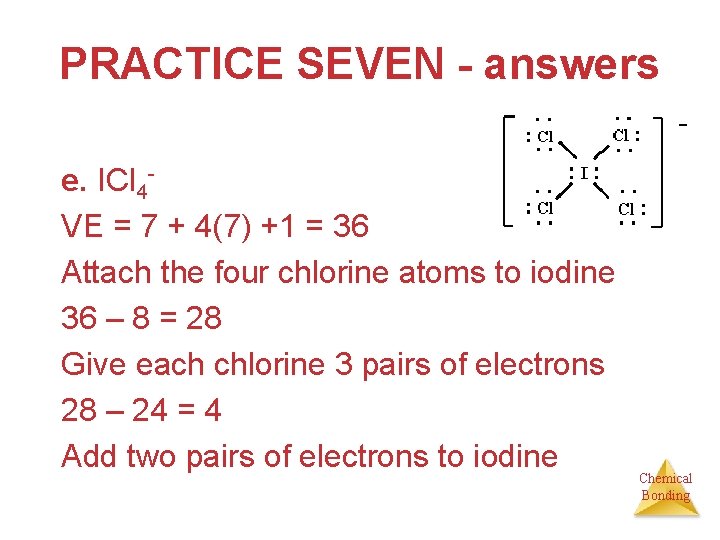
PRACTICE SEVEN - answers e. ICl 4 VE = 7 + 4(7) +1 = 36 Attach the four chlorine atoms to iodine 36 – 8 = 28 Give each chlorine 3 pairs of electrons 28 – 24 = 4 Add two pairs of electrons to iodine Chemical Bonding
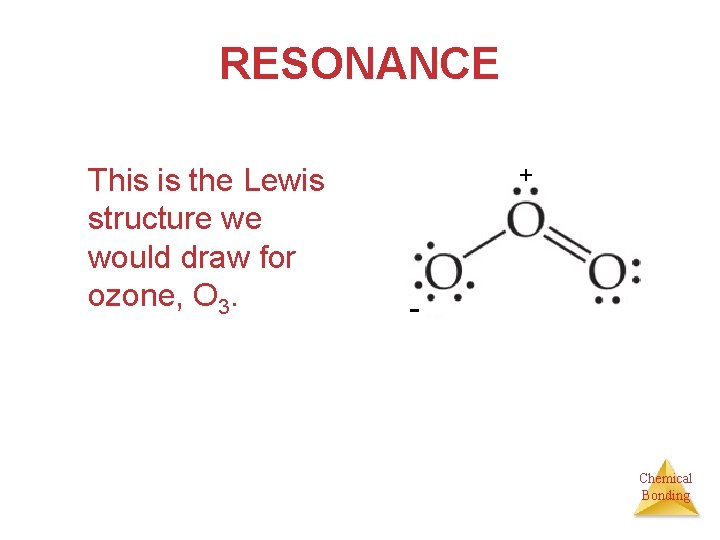
RESONANCE This is the Lewis structure we would draw for ozone, O 3. + - Chemical Bonding
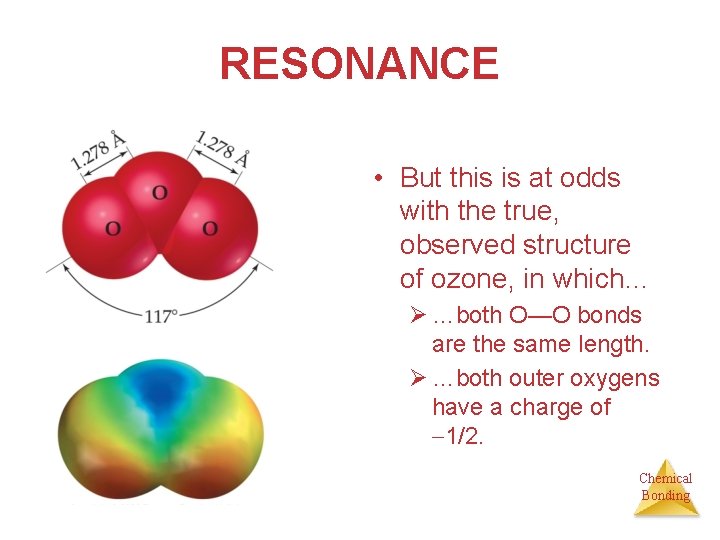
RESONANCE • But this is at odds with the true, observed structure of ozone, in which… Ø …both O—O bonds are the same length. Ø …both outer oxygens have a charge of 1/2. Chemical Bonding
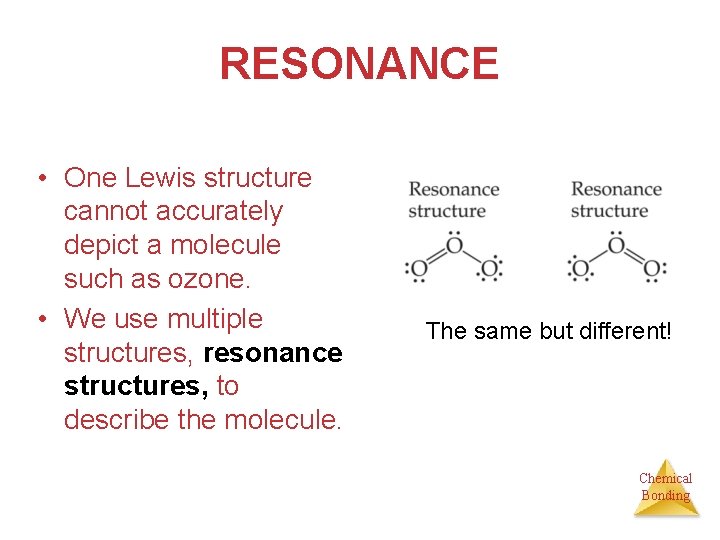
RESONANCE • One Lewis structure cannot accurately depict a molecule such as ozone. • We use multiple structures, resonance structures, to describe the molecule. The same but different! Chemical Bonding
RESONANCE Just as green is a synthesis of blue and yellow… …ozone is a synthesis of these two resonance structures. Chemical Bonding

RESONANCE • In truth, the electrons that form the second C—O bond in the double bonds below do not always sit between that C and that O, but rather can move among the two oxygens and the carbon. • They are not localized, but rather are delocalized. Chemical Bonding
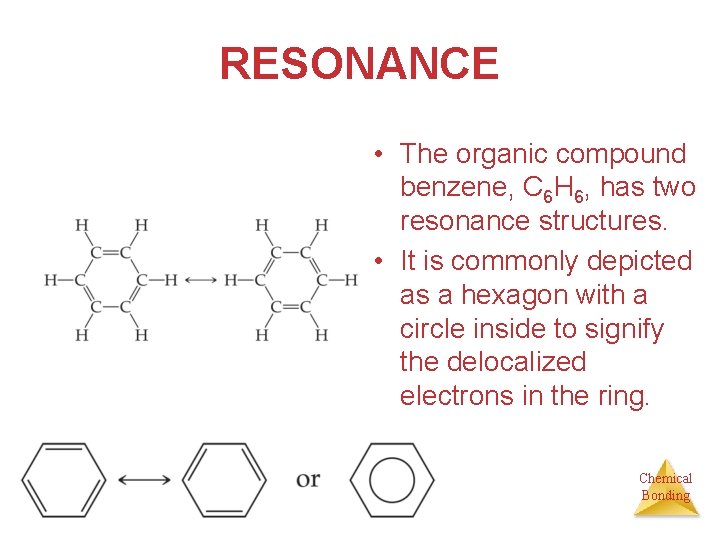
RESONANCE • The organic compound benzene, C 6 H 6, has two resonance structures. • It is commonly depicted as a hexagon with a circle inside to signify the delocalized electrons in the ring. Chemical Bonding

RESONANCE KEY POINTS 1. Resonance structures differ only in the assignment of electron pair positions, NEVER atom positions. 2. Resonance structures differ in the number of bond pairs between a given pair of atoms. 3. The actual structure is an average of Chemical the depicted resonance structures. Bonding

PRACTICE EIGHT • Describe the electron arrangement in the nitrite anion, NO 2 -, using the localized electron model. • Follow what you know to obtain the Lewis Structure for the ion. Chemical Bonding

PRACTICE EIGHT • Describe the electron arrangement in the nitrite anion, NO 2 -, using the localized electron model. • • 5 + 2(6) + 1 = 18 valence e. Single bonded structure: O – N – O 18 valence - 4 = 14 valence e. Make one double bond – now 12 valence Chemical Bonding
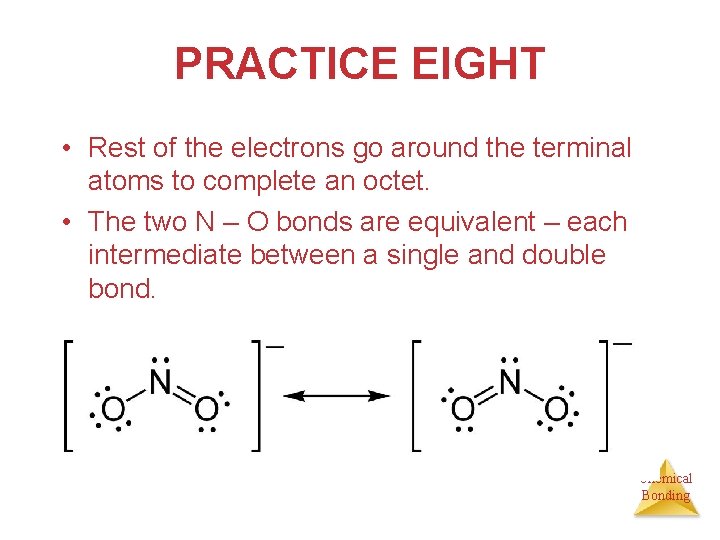
PRACTICE EIGHT • Rest of the electrons go around the terminal atoms to complete an octet. • The two N – O bonds are equivalent – each intermediate between a single and double bond. Chemical Bonding

FORMAL CHARGE • Use formal charge to determine the most favored resonance structure. • Formal Charge - The number of valence electrons on the free element minus the number of electrons assigned to the atom in the molecule. Chemical Bonding

FORMAL CHARGE • lone pair (unshared electrons belong completely to the atom in question. • shared electrons are divided equally between the sharing atoms • Atom’s formal charge = group number − [# of lone electrons − 2 (# of bonding electrons)] • The sum of the formal charges must Chemical Bonding equal an ion’s charge.

FORMAL CHARGE • Use formal charges along with the following to determine resonance structure: • Atoms in molecules (or ions) should have formal charges as small as possible—as close to zero as possible - called principle of electroneutrality. • A molecule (or ion) is most stable when any negative formal charge resides on the most electronegative atom. Chemical Bonding

FORMAL CHARGES Assigning formal charges: Ø For each atom, count the electrons in lone pairs and half the electrons it shares with other atoms. Ø Subtract that from the number of valence electrons for that atom: The difference is its formal charge. Carbon dioxide resonance structures Chemical Bonding

FORMAL CHARGES • So since we want formal charges to be as close to zero as possible, we choose the left structure as the likely structure. Chemical Bonding

FORMAL CHARGES The best Lewis structure… Ø…is the one with the fewest charges. Ø…puts a negative charge on the most electronegative atom. VE - 5 4 6 Assign 7 4 5 -2 0 +1 5 6 -1 4 4 0 6 6 0 5 5 0 4 4 0 6 7 -1 Chemical Bonding
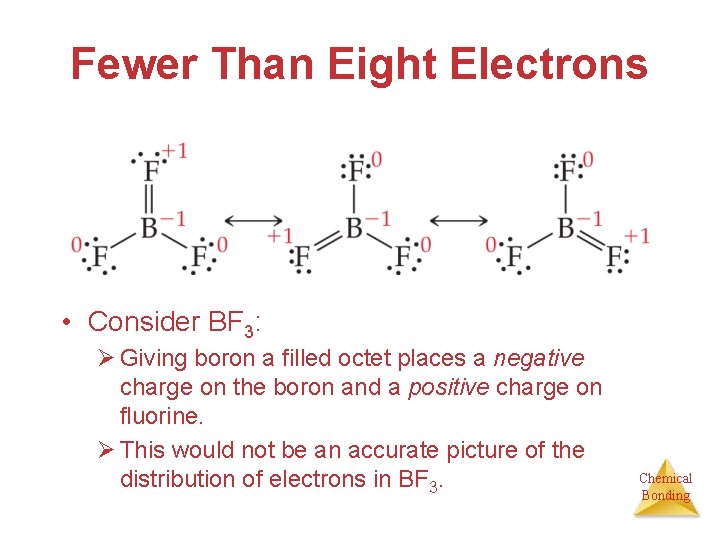
Fewer Than Eight Electrons • Consider BF 3: Ø Giving boron a filled octet places a negative charge on the boron and a positive charge on fluorine. Ø This would not be an accurate picture of the distribution of electrons in BF 3. Chemical Bonding
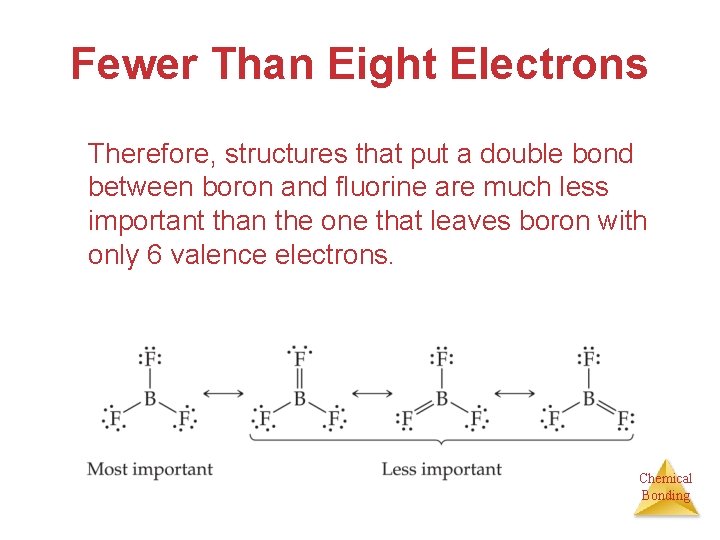
Fewer Than Eight Electrons Therefore, structures that put a double bond between boron and fluorine are much less important than the one that leaves boron with only 6 valence electrons. Chemical Bonding

Fewer Than Eight Electrons The lesson is: If filling the octet of the central atom results in a negative charge on the central atom and a positive charge on the more electronegative outer atom, don’t fill the octet of the central atom. Chemical Bonding
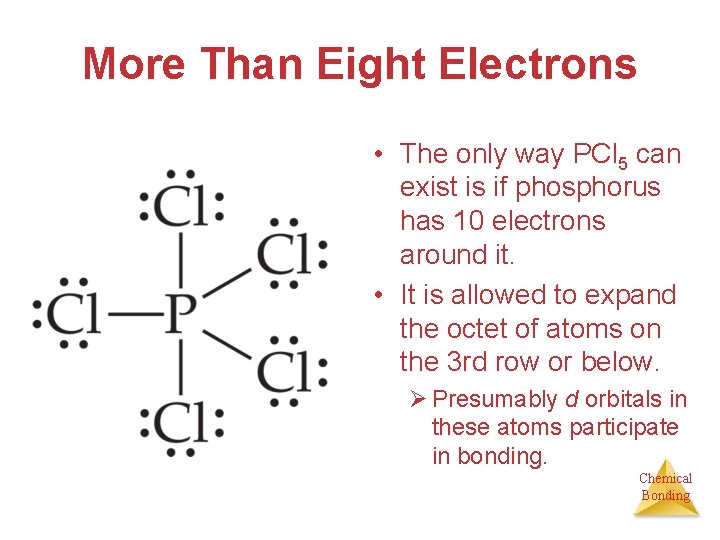
More Than Eight Electrons • The only way PCl 5 can exist is if phosphorus has 10 electrons around it. • It is allowed to expand the octet of atoms on the 3 rd row or below. Ø Presumably d orbitals in these atoms participate in bonding. Chemical Bonding

More Than Eight Electrons • This eliminates the charge on the phosphorus and the charge on one of the oxygens. • The lesson is: When the central atom is on the 3 rd row or below and expanding its octet eliminates some formal charges, do so. Chemical Bonding
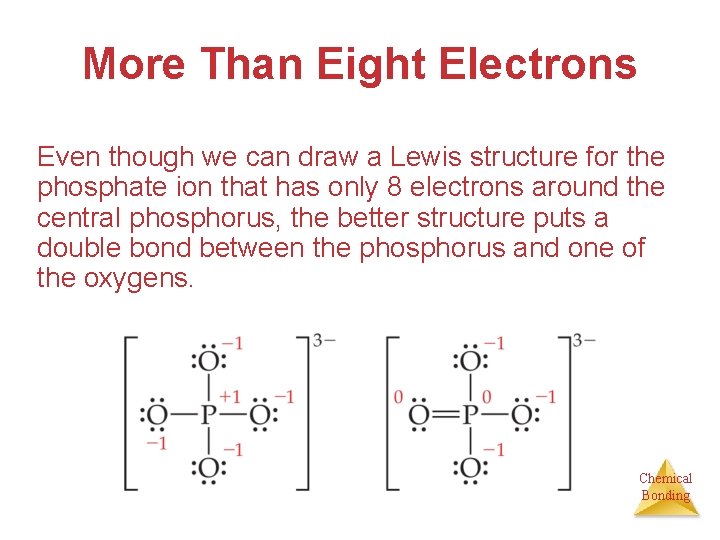
More Than Eight Electrons Even though we can draw a Lewis structure for the phosphate ion that has only 8 electrons around the central phosphorus, the better structure puts a double bond between the phosphorus and one of the oxygens. Chemical Bonding

PRACTICE NINE • Draw all possible structures for the sulfate ion. Decide which is the most plausible using formal charges. Chemical Bonding
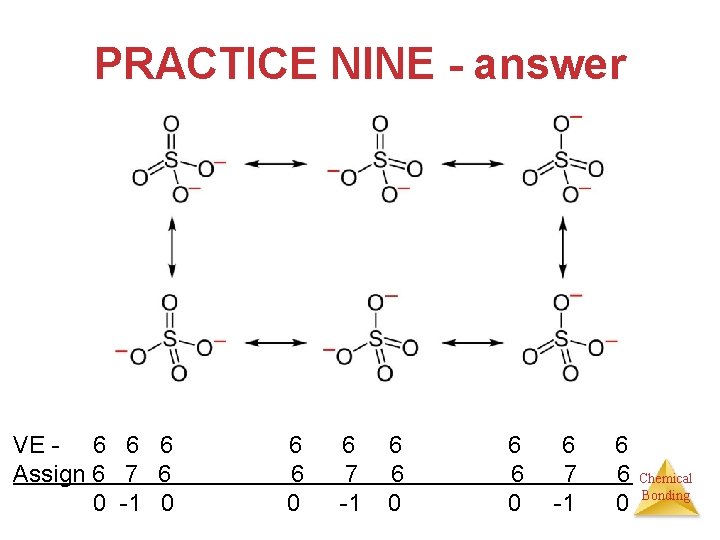
PRACTICE NINE - answer VE - 6 6 6 Assign 6 7 6 0 -1 0 6 6 0 6 7 -1 6 6 0 Chemical Bonding

FORMAL CHARGES REMINDER • Formal charges are only estimates and should not be taken as the actual atomic charges. • Using formal charges can often lead to erroneous structures, so tests based on experiments must be used to make the final decisions on the correct description Chemical of bonding. Bonding

PRACTICE TEN • Give possible Lewis structures for Xe. O 3, an explosive compound of xenon. Which Lewis structure or structures are most appropriate according to the formal charges? Chemical Bonding
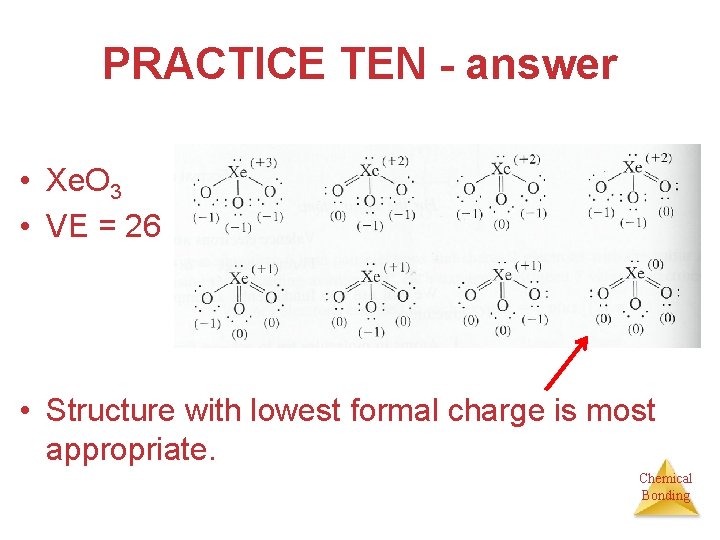
PRACTICE TEN - answer • Xe. O 3 • VE = 26 • Structure with lowest formal charge is most appropriate. Chemical Bonding

VSEPR Chemical Bonding
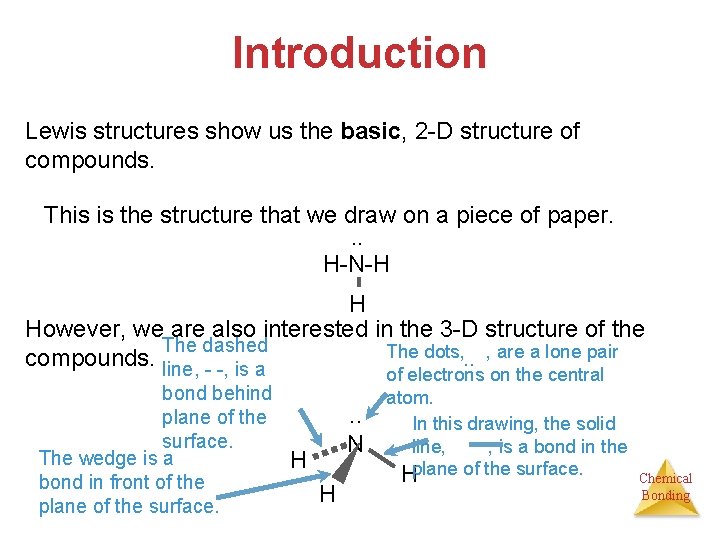
Introduction Lewis structures show us the basic, 2 -D structure of compounds. This is the structure that we draw on a piece of paper. . . H-N-H H However, we are also interested in the 3 -D structure of the The dashed The dots, . . , are a lone pair compounds. line, - -, is a bond behind plane of the surface. The wedge is a bond in front of the plane of the surface. . . N H H of electrons on the central atom. In this drawing, the solid line, , is a bond in the Hplane of the surface. Chemical Bonding

Introduction We use Lewis structures to look at how the atoms of the compound are bound together. Lewis structures will tell us: ü the number of atoms bound to a central atom. ü the number of lone pairs on the central atom. Chemical Bonding

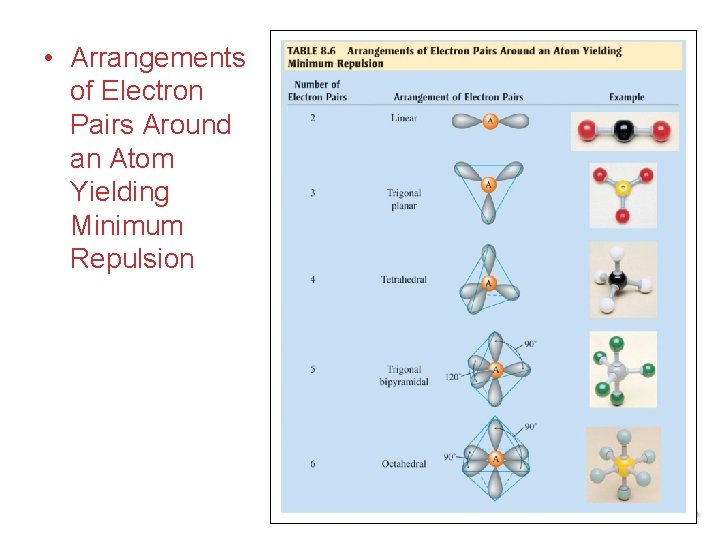
Introduction We will use the VSEPR model to help us determine the 3 -D structure of compounds. VSEPR stands for Valence Shell Electron Pair Repulsion. In this model, we will place the bonding and non-bonding electron pairs as far apart as possible in 3 -D space. Then, we can look at the position of the atoms of the compound and determine the 3 -D structure. Chemical Bonding

Introduction ØElectronic (structural pair)geometry – all the electron pairs surrounding a central atom are considered. ØMolecular geometry - the arrangement in space of the atoms bonded to a central atom and nonbonding electrons become “invisible”. This is not necessarily the same as the electronic geometry. ØANY time lone pairs are present, the electronic and molecular geometries will be Chemical Bonding different.

Introduction Ø Each lone pair or bond pair repels all other lone pairs and bond pairs. Ø Lone pairs have a different repulsion since they are experiencing an attraction or “pull” from only one nucleus as opposed to two nuclei. • works well for elements of the s and p-blocks • VSEPR does not apply to transition element compounds (exceptions). • They do not cause distortion when bond angles are 120º or greater. Chemical Bonding
LONE PAIRS Chemical Bonding

Application - TWO Two orbitals around a central atom: B A B We arrange the orbitals around the atom as far apart as possible - 180° apart. The orbitals are in a linear structure. With AB 2, there are two bonding orbitals and zero lone pairs. The atoms are in a linear structure. Chemical Bonding

Application - TWO Two orbitals around a central atom: B A We arrange the orbitals around the atom as far apart as possible - 180° apart. The orbitals are in a linear structure. With AB, there is one bonding orbital and one lone pair. The atoms are in a linear structure. Chemical Bonding
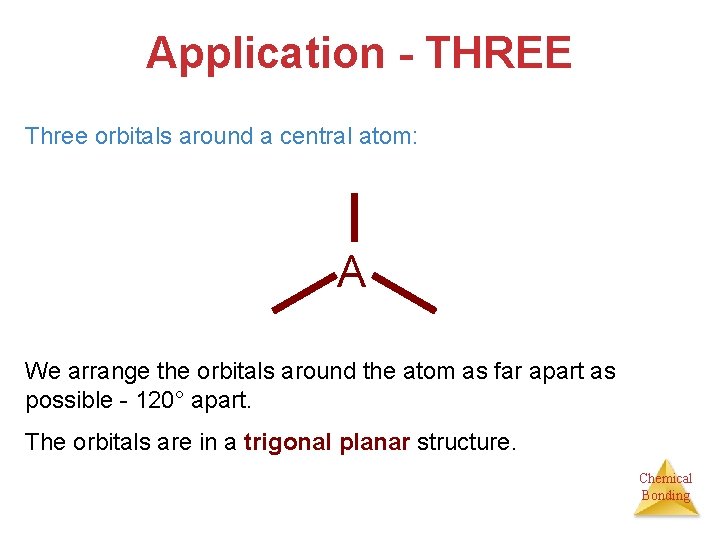
Application - THREE Three orbitals around a central atom: A We arrange the orbitals around the atom as far apart as possible - 120° apart. The orbitals are in a trigonal planar structure. Chemical Bonding
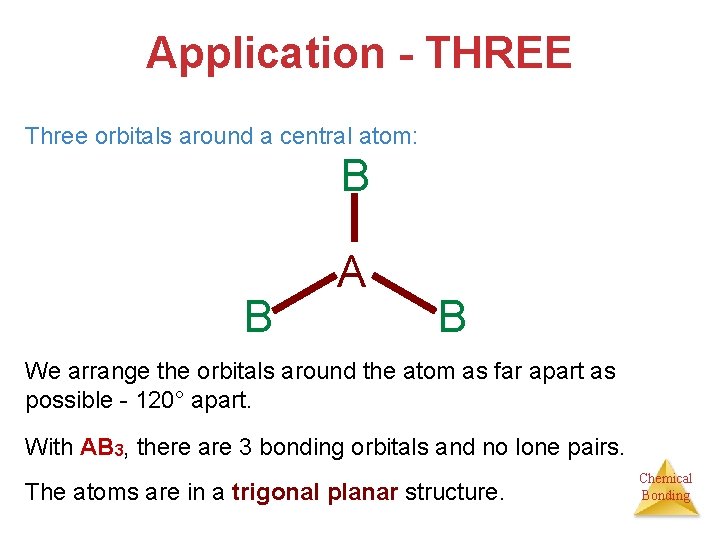
Application - THREE Three orbitals around a central atom: B B A B We arrange the orbitals around the atom as far apart as possible - 120° apart. With AB 3, there are 3 bonding orbitals and no lone pairs. The atoms are in a trigonal planar structure. Chemical Bonding
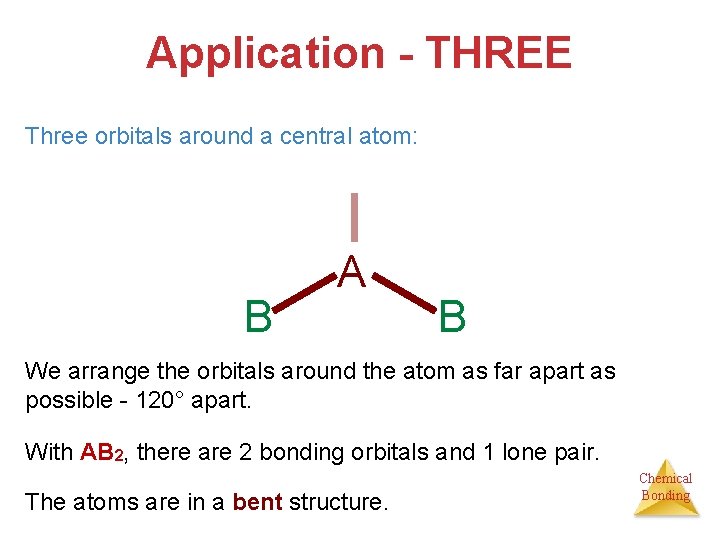
Application - THREE Three orbitals around a central atom: B A B We arrange the orbitals around the atom as far apart as possible - 120° apart. With AB 2, there are 2 bonding orbitals and 1 lone pair. The atoms are in a bent structure. Chemical Bonding
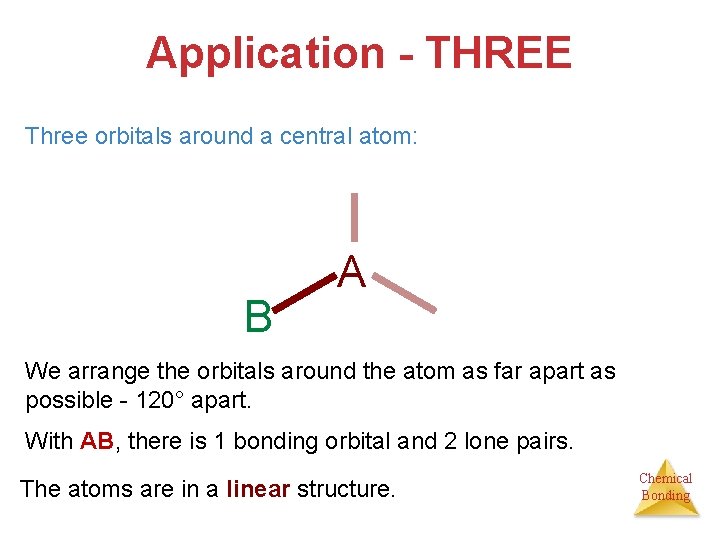
Application - THREE Three orbitals around a central atom: B A We arrange the orbitals around the atom as far apart as possible - 120° apart. With AB, there is 1 bonding orbital and 2 lone pairs. The atoms are in a linear structure. Chemical Bonding

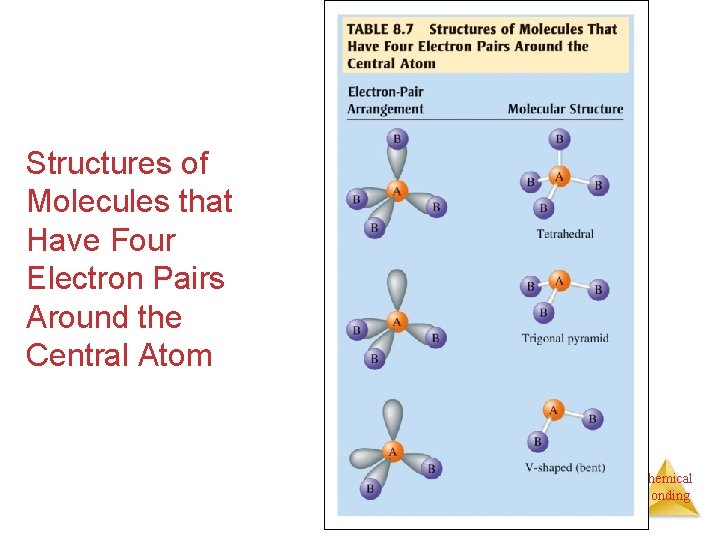
Application - FOUR Four orbitals around a central atom: A We arrange the orbitals around the atom as far apart as possible - into the shape of a tetrahedron – 109. 5°. The orbitals are in a tetrahedral structure. Chemical Bonding
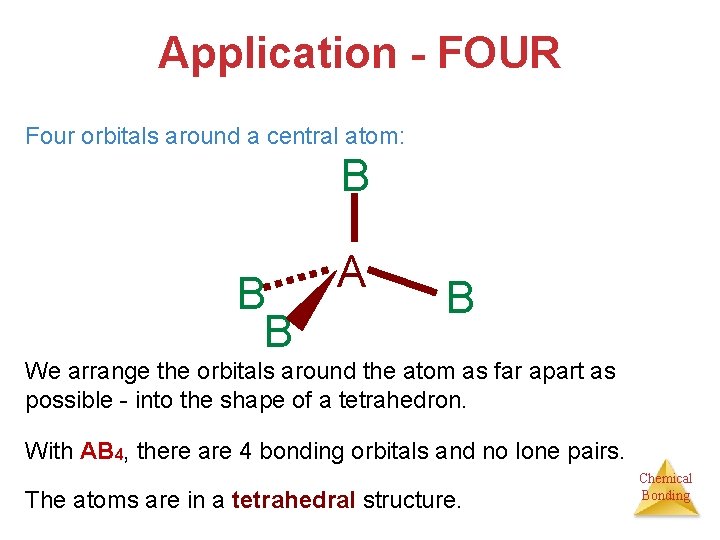
Application - FOUR Four orbitals around a central atom: B B B A B We arrange the orbitals around the atom as far apart as possible - into the shape of a tetrahedron. With AB 4, there are 4 bonding orbitals and no lone pairs. The atoms are in a tetrahedral structure. Chemical Bonding
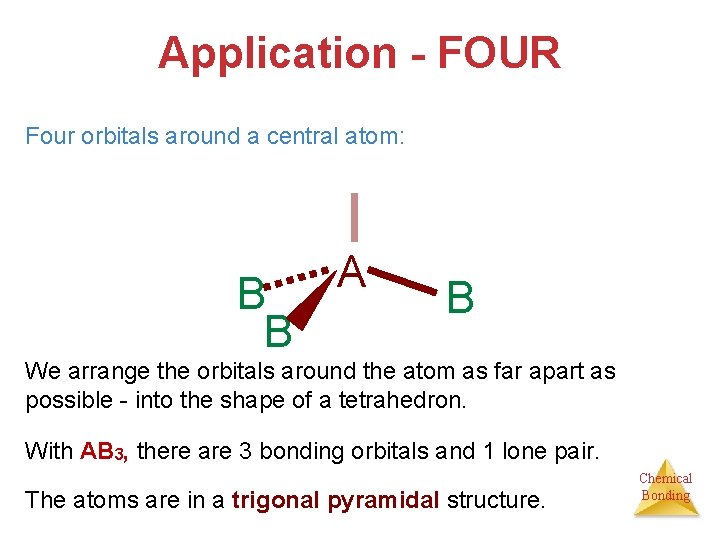
Application - FOUR Four orbitals around a central atom: B B A B We arrange the orbitals around the atom as far apart as possible - into the shape of a tetrahedron. With AB 3, there are 3 bonding orbitals and 1 lone pair. The atoms are in a trigonal pyramidal structure. Chemical Bonding
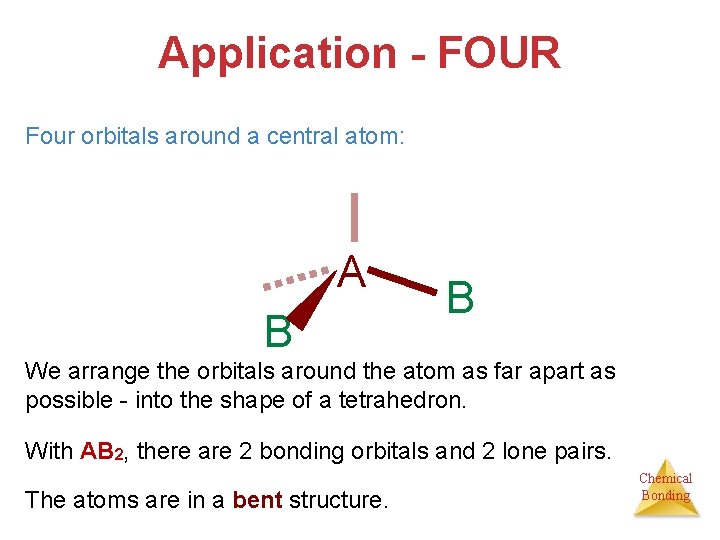
Application - FOUR Four orbitals around a central atom: A B B We arrange the orbitals around the atom as far apart as possible - into the shape of a tetrahedron. With AB 2, there are 2 bonding orbitals and 2 lone pairs. The atoms are in a bent structure. Chemical Bonding
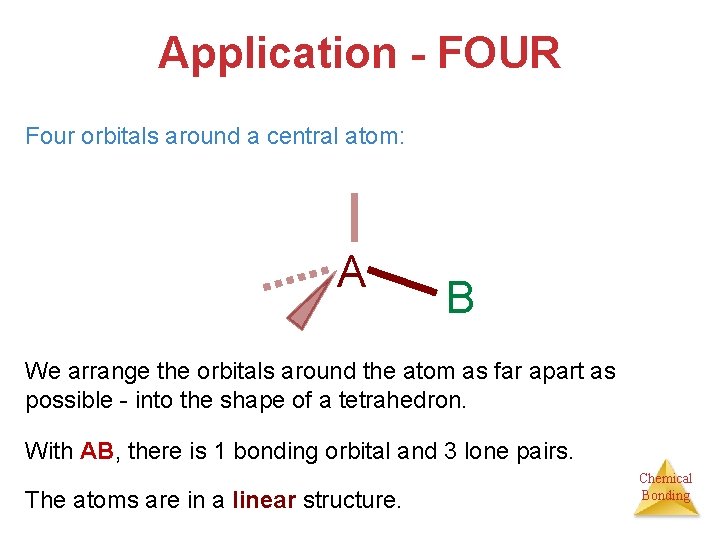
Application - FOUR Four orbitals around a central atom: A B We arrange the orbitals around the atom as far apart as possible - into the shape of a tetrahedron. With AB, there is 1 bonding orbital and 3 lone pairs. The atoms are in a linear structure. Chemical Bonding

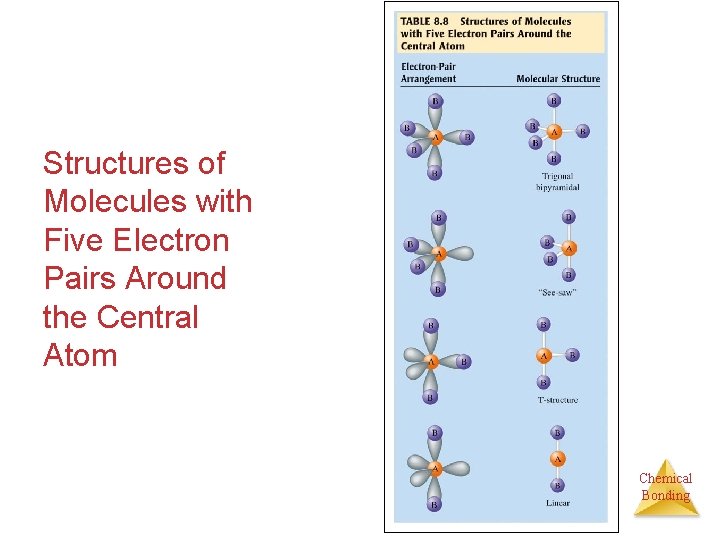
Application - FIVE Five orbitals around a central atom: A We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. The orbitals are in a trigonal bipyramidal structure. Chemical Bonding
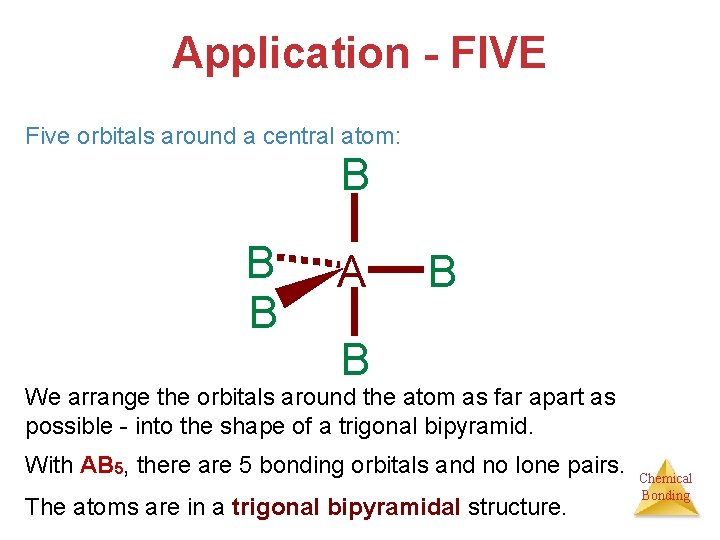
Application - FIVE Five orbitals around a central atom: B B B A B B We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. With AB 5, there are 5 bonding orbitals and no lone pairs. The atoms are in a trigonal bipyramidal structure. Chemical Bonding
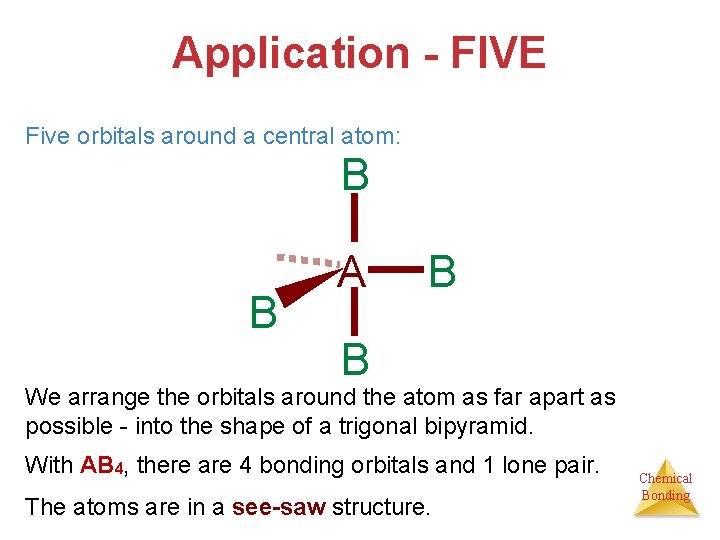
Application - FIVE Five orbitals around a central atom: B B A B B We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. With AB 4, there are 4 bonding orbitals and 1 lone pair. The atoms are in a see-saw structure. Chemical Bonding
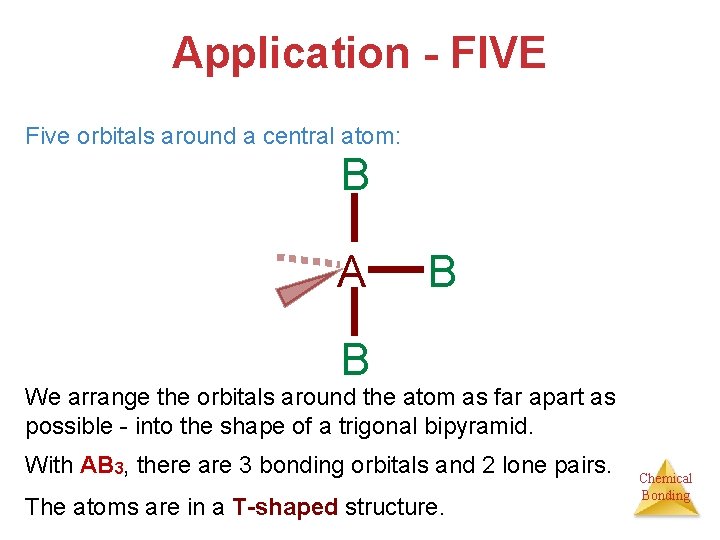
Application - FIVE Five orbitals around a central atom: B A B B We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. With AB 3, there are 3 bonding orbitals and 2 lone pairs. The atoms are in a T-shaped structure. Chemical Bonding
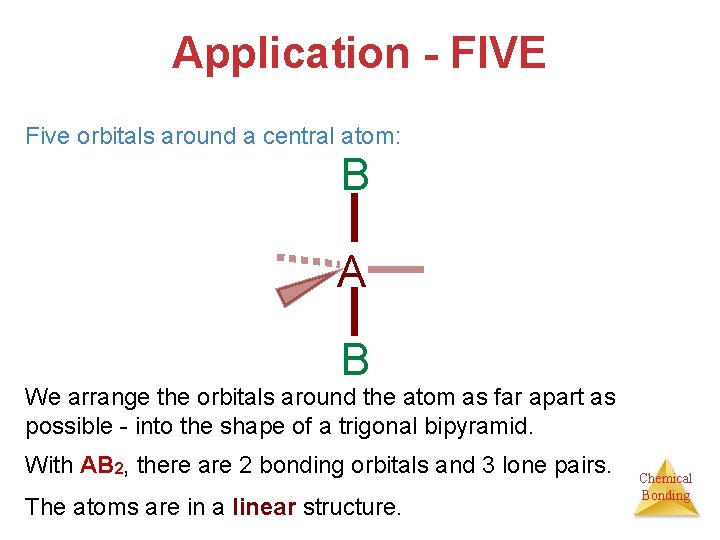
Application - FIVE Five orbitals around a central atom: B A B We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. With AB 2, there are 2 bonding orbitals and 3 lone pairs. The atoms are in a linear structure. Chemical Bonding
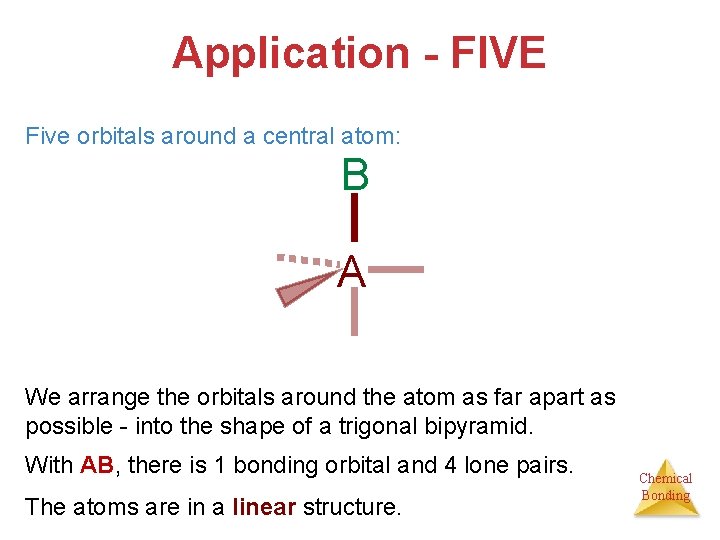
Application - FIVE Five orbitals around a central atom: B A We arrange the orbitals around the atom as far apart as possible - into the shape of a trigonal bipyramid. With AB, there is 1 bonding orbital and 4 lone pairs. The atoms are in a linear structure. Chemical Bonding

Application - SIX six orbitals around a central atom: A We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. The orbitals are in an octahedral structure. Chemical Bonding
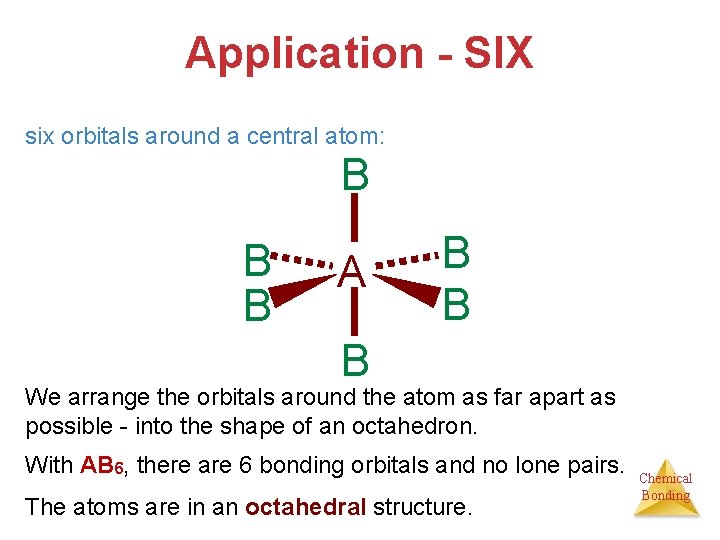
Application - SIX six orbitals around a central atom: B B B A B B B We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB 6, there are 6 bonding orbitals and no lone pairs. The atoms are in an octahedral structure. Chemical Bonding
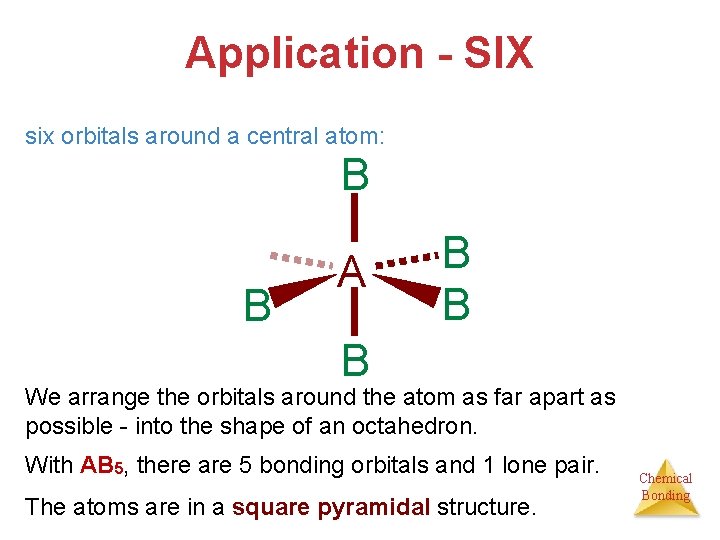
Application - SIX six orbitals around a central atom: B B A B B B We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB 5, there are 5 bonding orbitals and 1 lone pair. The atoms are in a square pyramidal structure. Chemical Bonding
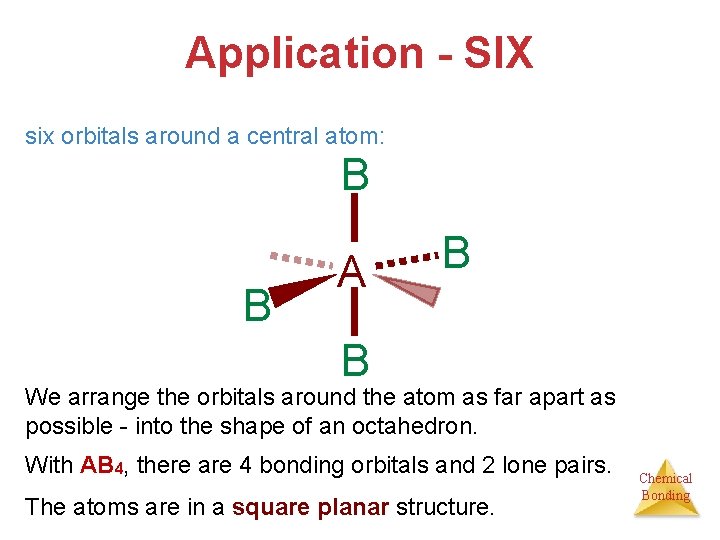
Application - SIX six orbitals around a central atom: B B A B B We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB 4, there are 4 bonding orbitals and 2 lone pairs. The atoms are in a square planar structure. Chemical Bonding
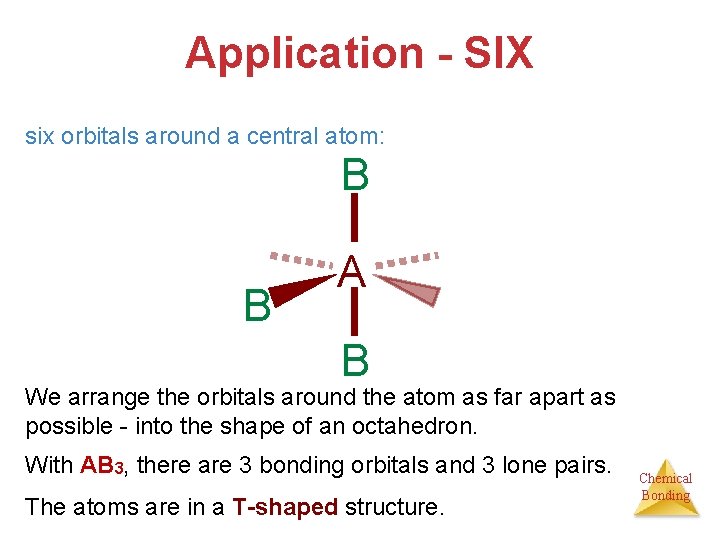
Application - SIX six orbitals around a central atom: B B A B We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB 3, there are 3 bonding orbitals and 3 lone pairs. The atoms are in a T-shaped structure. Chemical Bonding
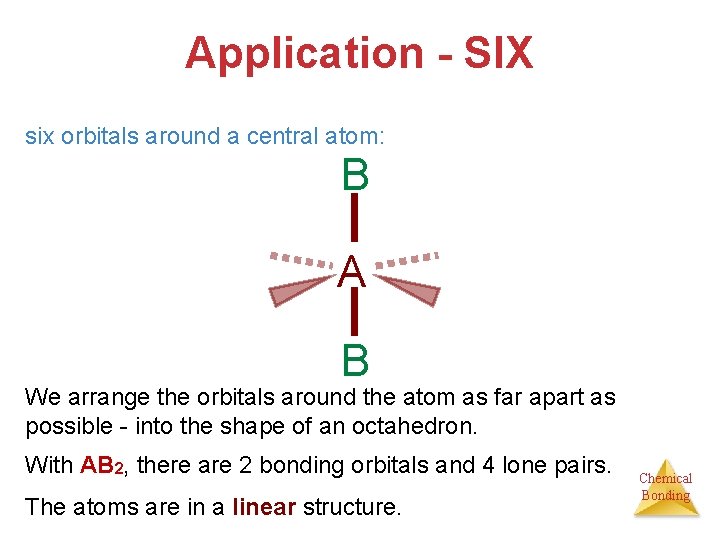
Application - SIX six orbitals around a central atom: B A B We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB 2, there are 2 bonding orbitals and 4 lone pairs. The atoms are in a linear structure. Chemical Bonding
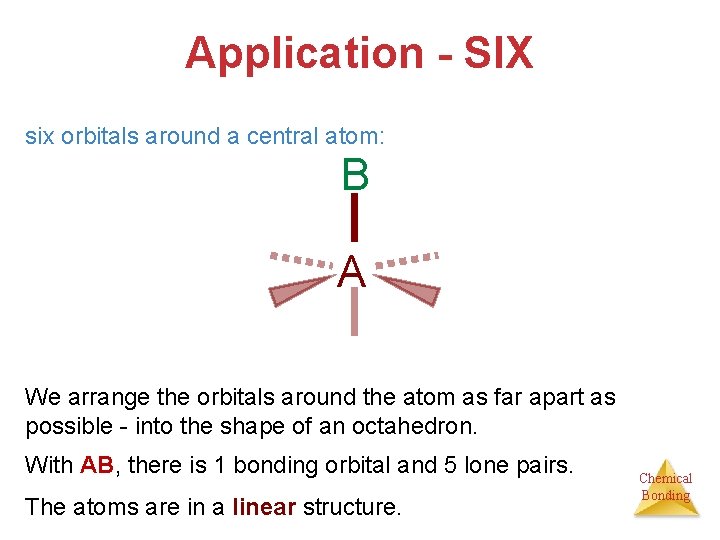
Application - SIX six orbitals around a central atom: B A We arrange the orbitals around the atom as far apart as possible - into the shape of an octahedron. With AB, there is 1 bonding orbital and 5 lone pairs. The atoms are in a linear structure. Chemical Bonding
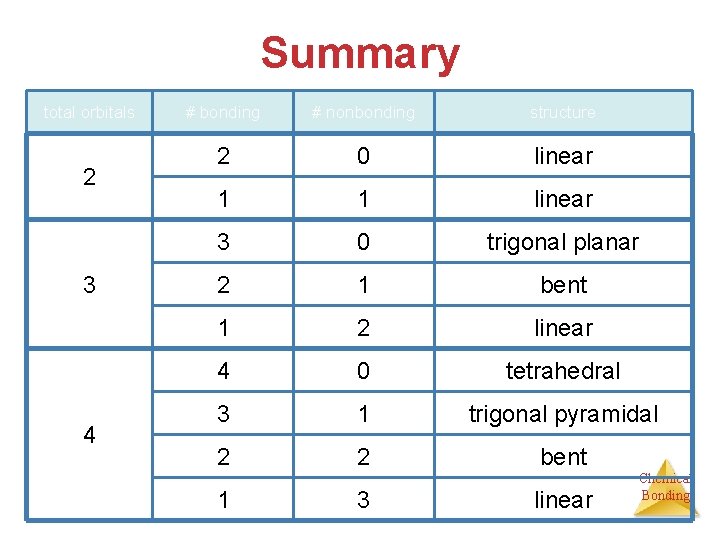
Summary total orbitals 2 3 4 # bonding # nonbonding structure 2 0 linear 1 1 linear 3 0 trigonal planar 2 1 bent 1 2 linear 4 0 tetrahedral 3 1 trigonal pyramidal 2 2 bent 1 3 linear Chemical Bonding
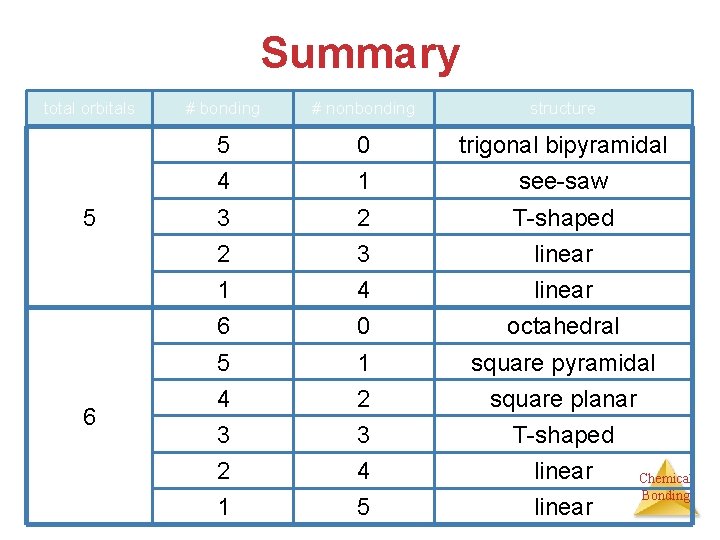
Summary total orbitals 5 6 # bonding # nonbonding structure 5 0 trigonal bipyramidal 4 1 see-saw 3 2 T-shaped 2 3 linear 1 4 linear 6 0 octahedral 5 1 square pyramidal 4 2 square planar 3 3 T-shaped 2 4 linear 1 5 linear Chemical Bonding
• Arrangements of Electron Pairs Around an Atom Yielding Minimum Repulsion Chemical Bonding

REMEMBER 1. structural pairs - σ bond pairs about an atom 2. The presence of lone pairs alters the six basic MOLECULAR geometries, but the electronic or structural pair geometry remains one of the six basic types. Chemical Bonding
Structures of Molecules that Have Four Electron Pairs Around the Central Atom 8– 134 Chemical Bonding
Structures of Molecules with Five Electron Pairs Around the Central Atom Chemical Bonding

DETERMINING GEOMETRY 1. Sketch the Lewis dot structure – do not skip this step! 2. Determine the number of electron pair groups surrounding the central atom(s). Remember that double and triple bonds are treated as a single group. 3. Determine the geometric shape that maximizes the distance between the electron groups. This is the geometry of the electron groups. 4. Mentally allow nonbonding pairs to become invisible. Determine the molecular geometry by looking at the remaining arrangement of atoms (as determined by the Chemical bonding electron groups) around the central atom. Bonding

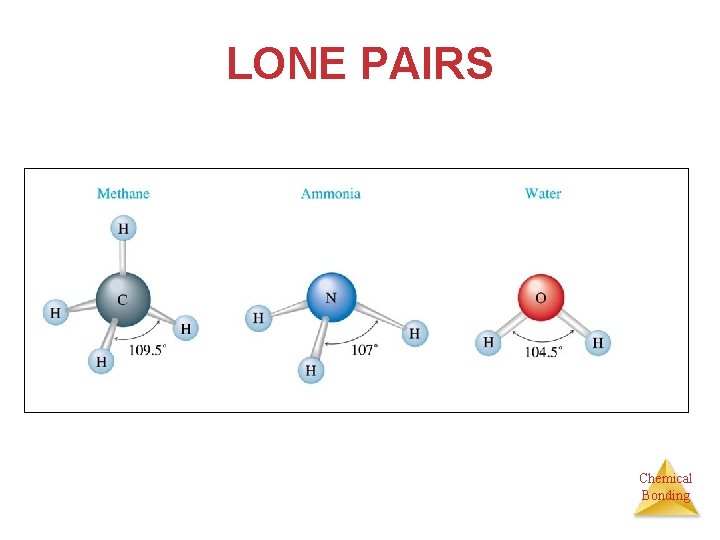
Molecular shapes for central atoms with normal valence • This will be no more than four structural pairs if the atom obeys the octet rule. • Since no lone pairs are present, the molecular and structural pair [or electronic] geometry is the same. 109. 5° bond angle. • Ignore lone pairs AFTER you’ve determined the angles and only the relative positions of the atoms are important in molecular geometry • Examples are ammonia (107. 5°bond angle) and water (104. 5°bond angle). Chemical Bonding

Molecular shapes for central atoms with expanded valence • only elements with a principal energy level of 3 or higher can expand their valence and violate the octet • rule on the high side. • d orbitals are needed for the expansion to a 5 th or 6 th bonding location—the combination of 1 s and 3 p’s provides the four bonding sites that make up the octet rule. • seems to be a limit of three lone pairs about the central atom • Example is Xe. F 4 (equatorial and axial) Chemical Bonding

PRACTICE ELEVEN Prediction of Molecular Structure I • Describe the molecular structure of the water molecule. Chemical Bonding
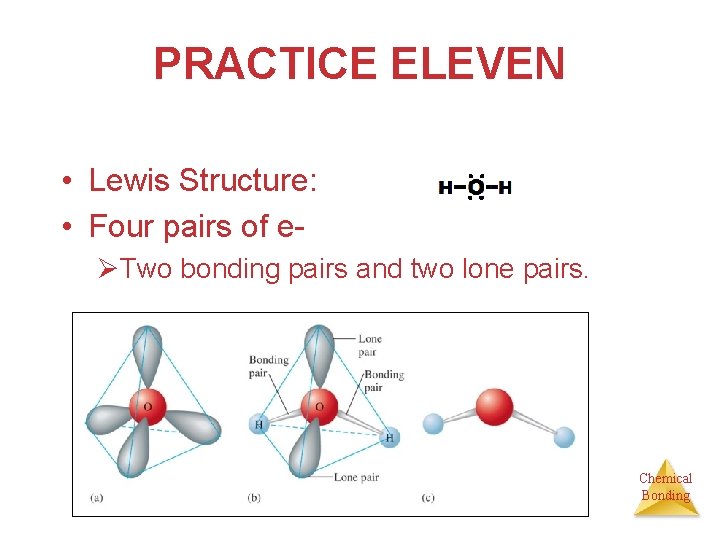
PRACTICE ELEVEN • Lewis Structure: • Four pairs of eØTwo bonding pairs and two lone pairs. Chemical Bonding

PRACTICE TWELVE Prediction of Molecular Structure II • When phosphorus reacts with excess chlorine gas, the compound phosphorus pentachloride (PCl 5) is formed. In the gaseous and liquid states, this substance consists of PCl 5 molecules, but in the solid state it consists of a 1: 1 mixture of PCl 4+ and PCl 6 - ions. Predict the geometric structures of PCl 5, PCl 4+, and PCl 6 -. Chemical Bonding

PRACTICE TWELVE • • PCl 5 VE = 5 + 5(7) = 40 Five bonded etrigonal bipyramidal Chemical Bonding
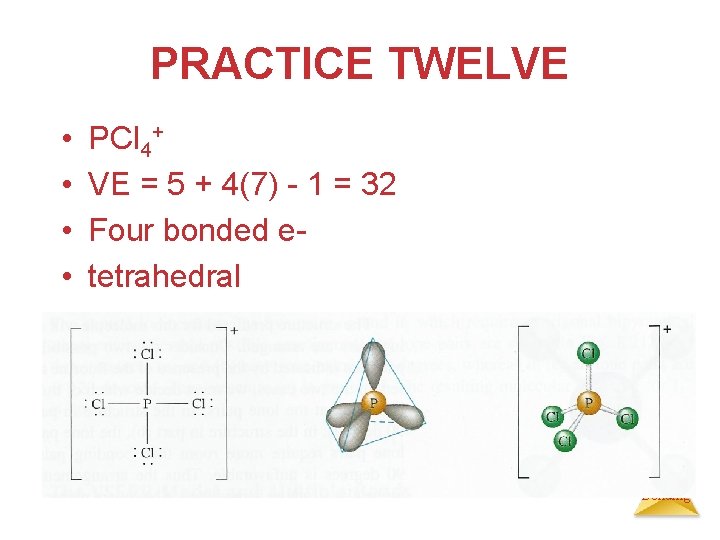
PRACTICE TWELVE • • PCl 4+ VE = 5 + 4(7) - 1 = 32 Four bonded etetrahedral Chemical Bonding
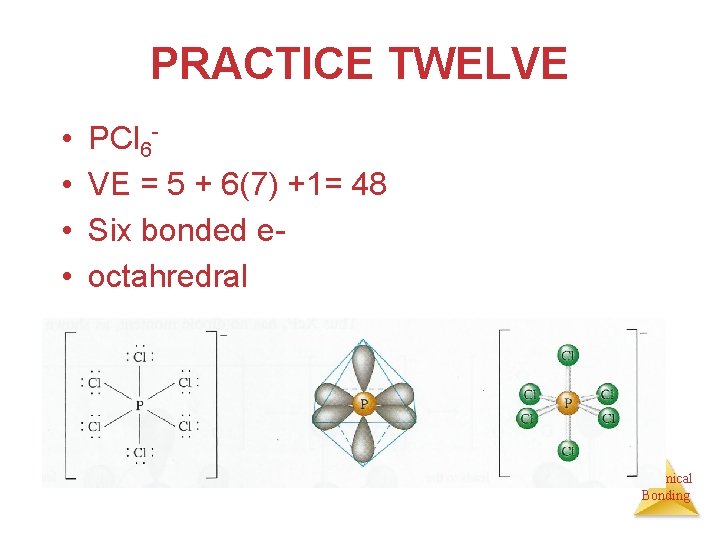
PRACTICE TWELVE • • PCl 6 VE = 5 + 6(7) +1= 48 Six bonded eoctahredral Chemical Bonding

MOLECULAR POLARITY • Polar - bonds can be polar while the entire molecule isn’t and vice versa. • dipole moment - separation of the charge in a molecule; product of the size of the charge and the distance of separation – learned this earlier. Chemical Bonding

• align themselves with an electric field. • align with each other as well in the absence of an electric field • water — two lone pairs establish a strong negative pole • ammonia — one lone pair which establishes a negative pole • note that the direction of the arrow indicating the dipole moment always points to the negative pole Chemical Bonding with the cross hatch on the arrow at the positive pole.

MOLECULAR POLARITY Nonpolar molecule octet rule is obeyed AND all the surrounding bonds are the same; the molecule is nonpolar since all the dipole moments cancel each other out. Example: CO 2 Chemical Bonding

MOLECULAR POLARITY Polar Molecule octet rule is obeyed and all the surrounding bonds are NOT the same; the molecule is polar since all the dipole moments do not cancel each other out. Example: NH 3. Methane is a great example. Replace one H with a halogen and it becomes polar. Replace all and it. Chemical Bonding is nonpolar again!

DO NOW • Pick up handout • Get out your notes and the last two homework sheets Chemical Bonding

PRACTICE THIRTEEN Drawing molecules • Draw CH 4 , CH 3 Cl, CH 2 Cl 2, CHCl 3, CCl 4. Indicate dipole moment(s) where necessary. Chemical Bonding
PRACTICE THIRTEEN CH 4 CH 3 Cl Chemical Bonding
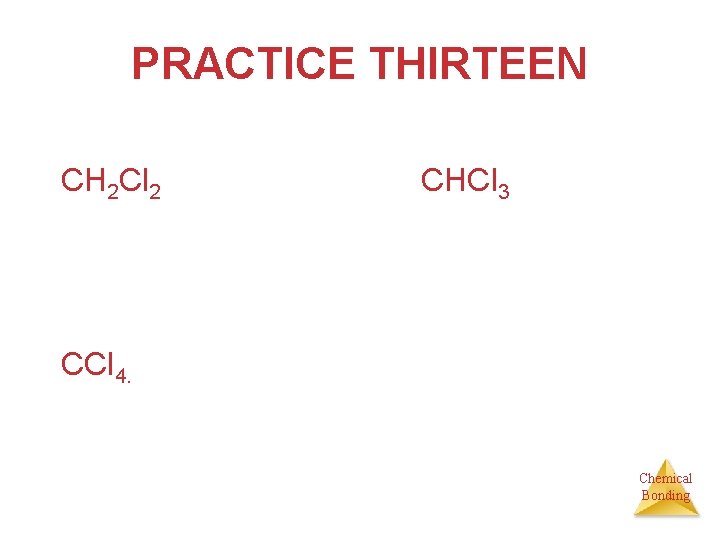
PRACTICE THIRTEEN CH 2 Cl 2 CHCl 3 CCl 4. Chemical Bonding

PRACTICE FOURTEEN A team at the Argonne National Laboratory produced the stable colorless compound xenon tetrafluoride (Xe. F 4). Predict its structure and whether it has a dipole moment. Chemical Bonding
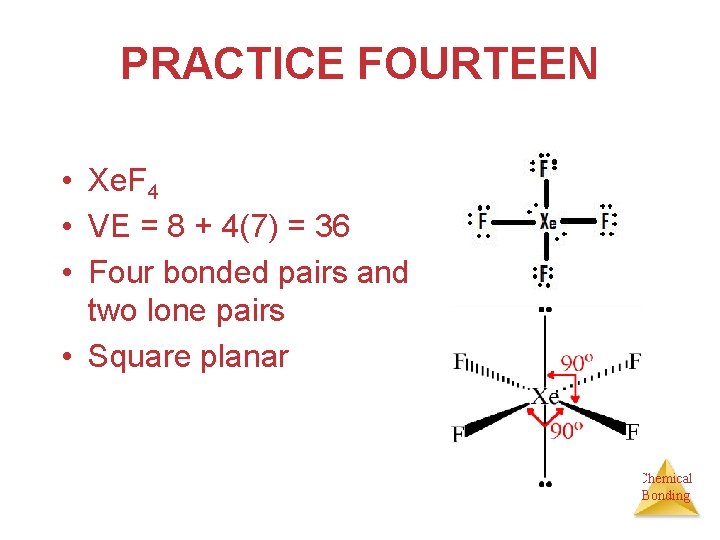
PRACTICE FOURTEEN • Xe. F 4 • VE = 8 + 4(7) = 36 • Four bonded pairs and two lone pairs • Square planar Chemical Bonding

PRACTICE FIFTEEN Structures of Molecules with Multiple Bonds • Predict the molecular structure of the sulfur dioxide molecule. Is this molecule expected to have a dipole moment? Chemical Bonding
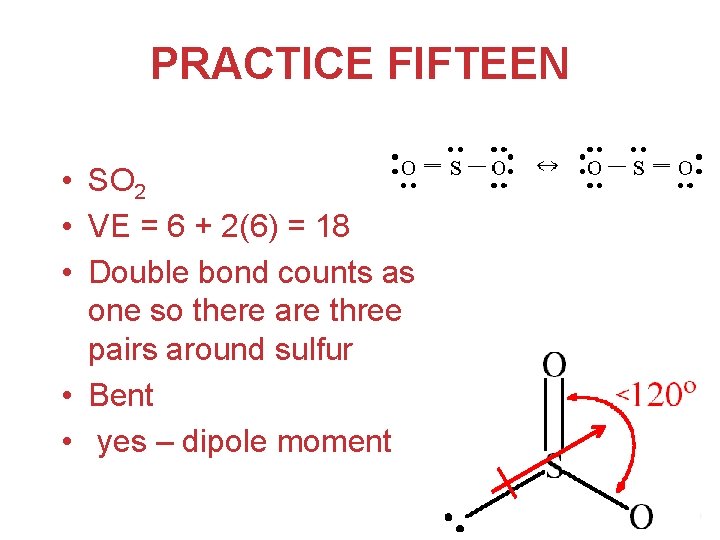
PRACTICE FIFTEEN • SO 2 • VE = 6 + 2(6) = 18 • Double bond counts as one so there are three pairs around sulfur • Bent • yes – dipole moment Chemical Bonding

BONDING SUMMARY • Read on your own. Chemical Bonding
- Slides: 157