Chapter 8 Bonding General Concepts Copyright 2018 Cengage

Chapter 8 Bonding: General Concepts Copyright © 2018 Cengage Learning. All Rights Reserved.

Chapter 8 Table of Contents § § § § (8. 1) (8. 2) (8. 3) (8. 4) (8. 5) (8. 6) (8. 7) (8. 8) Types of chemical bonds Electronegativity Bond polarity and dipole moments Ions: Electron configurations and sizes Energy effects in binary ionic compounds Partial ionic character of covalent bonds The covalent chemical bond: A model Covalent bond energies and chemical reactions Copyright © 2018 Cengage Learning. All Rights Reserved.

Chapter 8 Table of Contents § § § (8. 9) (8. 10) (8. 11) (8. 12) (8. 13) The localized electron bonding model Lewis structures Exceptions to the octet rule Resonance Molecular structure: The VSEPR model Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds An Introduction to Chemical Bonds § Forces that hold groups of atoms together and make them function as a unit § Bond energy: Energy required to break a bond § Ionic bonding: Results from electrostatic attractions of closely packed, oppositely charged ions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds An Introduction to Chemical Bonds (continued) § Ionic compound: Results when a metal reacts with a nonmetal § Example - When Na and Cl react to form Na. Cl, electrons are transferred from the Na atoms to the Cl atoms to form Na+ and Cl– ions § Ions then aggregate to form solid sodium chloride Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Coulomb’s Law § Used to calculate the energy of interaction between a pair of ions § E has units of joules § r - Distance between the ion centers in nanometers § Q 1 and Q 2 - Numerical ion charges Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Coulomb's Law (continued) § Used to calculate the repulsive energy when two like-charged ions are brought together § Calculated value of the energy will have a positive sign Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Development of Bonding Forces between Two Identical Atoms § Bond will form if the system can lower its total energy in the process § System will act to minimize the sum of the positive (repulsive) energy terms and the negative (attractive) energy term § Bond length: Distance between two atoms when potential energy is minimal Copyright © 2018 Cengage Learning. All Rights Reserved.
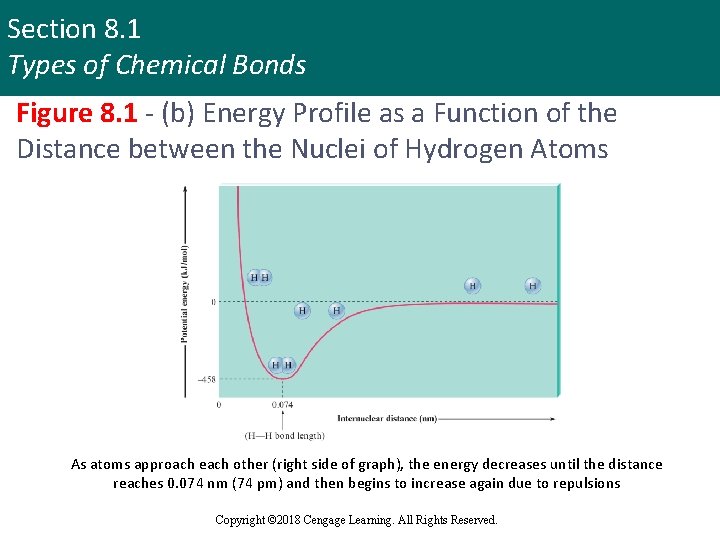
Section 8. 1 Types of Chemical Bonds Figure 8. 1 - (b) Energy Profile as a Function of the Distance between the Nuclei of Hydrogen Atoms As atoms approach each other (right side of graph), the energy decreases until the distance reaches 0. 074 nm (74 pm) and then begins to increase again due to repulsions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Interaction between Two Hydrogen Atoms: Important Features § Energy terms involved are the: § Net potential energy that results from the attractions and repulsions among the charged particles § Kinetic energy due to the motions of the electrons § Zero point of energy is defined with the atoms at infinite separation Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Interaction between Two Hydrogen Atoms: Important Features (continued) § At very short distances the energy rises steeply because of the importance of the repulsive forces when the atoms are very close together § Bond length is the distance at which the system has minimum energy Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Covalent Bonding § Two identical atoms share electrons equally § Bonding results from the mutual attraction of the nuclei for the shared electrons § Polar covalent bond § Characterized by unequal sharing of electrons § Example § Bonding in the hydrogen fluoride (HF) molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Figure 8. 2 - The Effect of an Electric Field on Hydrogen Fluoride Molecules Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 1 Types of Chemical Bonds Join In (1) § What type of bond is formed when electrons are unequally shared by nuclei? a. b. c. d. e. Ionic Covalent Polar ionic Polar covalent None of the above Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity § Ability of an atom in a molecule to attract shared electrons to itself § Can be determined by using Pauling’s model § Consider a hypothetical molecule HX § Relative electronegativities of the H and X atoms are determined by comparing the measured H—X bond energy with the expected H—X bond energy Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity (continued 1) § Difference (Δ) between actual and expected bond energies is given by the following equation: § If H and X have identical electronegativities: § Δ is 0 § (H—X)act and (H—X)exp are the same Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity (continued 2) § If X has a greater electronegativity than H: § Shared electron(s) will tend to be closer to the X atom § Molecule will be polar, with the following charge distribution: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Trends § Attraction between the partially (and oppositely) charged H and X atoms will lead to a greater bond strength § (H—X)act will be larger than (H—X)exp § Greater the difference in the electronegativities of the atoms, the greater is the ionic component of the bond and the greater is the value of Δ Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Figure 8. 3 - The Pauling Electronegativity Values Copyright © 2018 Cengage Learning. All Rights Reserved.
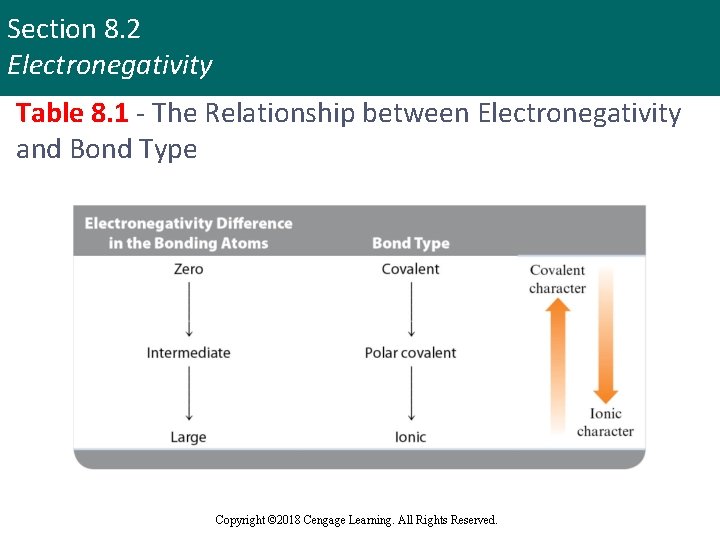
Section 8. 2 Electronegativity Table 8. 1 - The Relationship between Electronegativity and Bond Type Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Interactive Example 8. 1 - Relative Bond Polarities § Order the following bonds according to polarity: § § § H—H O—H Cl—H S—H F—H Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Interactive Example 8. 1 - Solution § Polarity of bonds increases as difference in electronegativity increases § From Pauling’s electronegativity values, the following variation in bond polarity is expected: H—H < S—H < Cl—H < O—H < F—H (2. 1) (2. 5) (2. 1) (3. 0) (2. 1) (3. 5) (2. 1) Copyright © 2018 Cengage Learning. All Rights Reserved. (4. 0) (2. 1)

Section 8. 2 Electronegativity Interactive Example 8. 1 - Solution (continued) § Electronegativity difference Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Exercise § Without using Fig. 8. 3, predict the order of increasing electronegativity in each of the following groups of elements a. b. c. d. C, N, O S, Se, Cl Si, Ge, Sn Tl, S, Ge C<N<O Se < S < Cl Sn < Ge < Si Tl < Ge < S Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Critical Thinking § We use differences in electronegativity to account for certain properties of bonds § What if all atoms had the same electronegativity values? § How would bonding between atoms be affected? § What are some differences we would notice? Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Join In (2) § Which of the following elements forms the most ionic bond with chlorine? a. b. c. d. e. K Al P Kr Br Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 2 Electronegativity Join In (3) § As a general pattern, electronegativity is inversely related to: a. b. c. d. ionization energy atomic size the polarity of the atom the number of neutrons in the nucleus Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Dipole Moment § Molecules that have a center of positive charge and a center of negative charge are called dipolar § Representation of dipolar character § An arrow points to the negative charge center with the tail of the arrow indicating the positive center of charge Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Electrostatic Potential Diagram § Alternative method for representing charge distribution in a molecule § Colors of visible light are used to show the variation in charge distribution § Red indicates the most electron-rich region § Blue indicates the most electron-poor region Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Figure 8. 4 - An Electrostatic Potential Map of HF Red indicates the most electron-rich area (the fluorine atom), and blue indicates the most electron-poor region (the hydrogen atom) Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments of Hydrogen Halides (HX) § Depends on the electronegativity of X Copyright © 2018 Cengage Learning. All Rights Reserved.
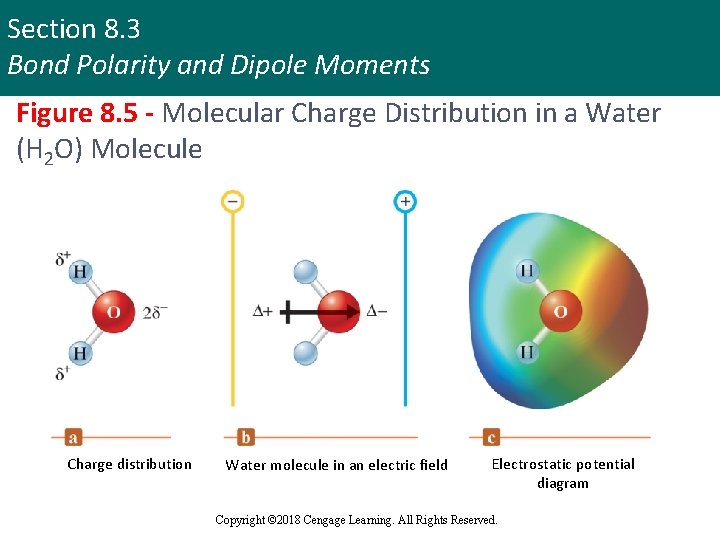
Section 8. 3 Bond Polarity and Dipole Moments Figure 8. 5 - Molecular Charge Distribution in a Water (H 2 O) Molecule Charge distribution Water molecule in an electric field Electrostatic potential diagram Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Dipole Moment in Other Molecules § Some molecules have polar bonds but do not have a dipole moment § Occurs when individual bond polarities are arranged in such a way that they cancel each other out § Example - CO 2 Copyright © 2018 Cengage Learning. All Rights Reserved.
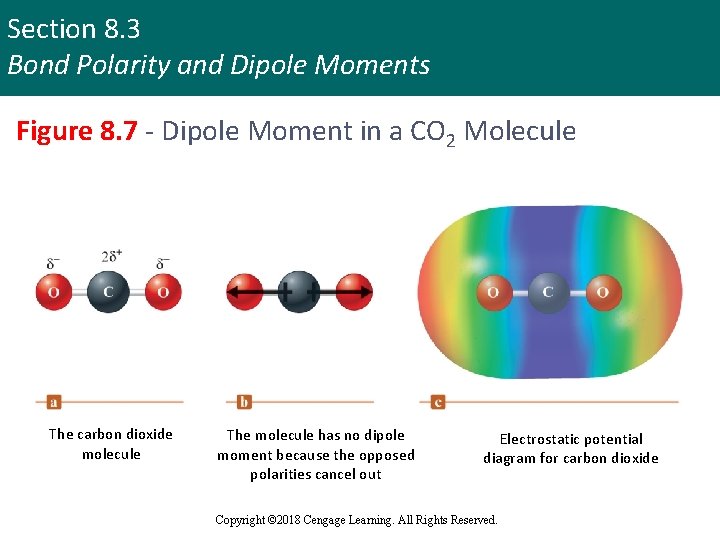
Section 8. 3 Bond Polarity and Dipole Moments Figure 8. 7 - Dipole Moment in a CO 2 Molecule The carbon dioxide molecule The molecule has no dipole moment because the opposed polarities cancel out Electrostatic potential diagram for carbon dioxide Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Table 8. 3 - Types of Molecules with Polar Bonds but No Resulting Dipole Moment Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Bond Polarity and Dipole Moment § For each of the following molecules, show the direction of the bond polarities and indicate which ones have a dipole moment: § HCl, Cl 2, and SO 3 (a planar molecule with the oxygen atoms spaced evenly around the central sulfur atom) § CH 4 (tetrahedral with the carbon atom at the center) § H 2 S (V-shaped with the sulfur atom at the point) Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution § The HCl molecule § Electronegativity of chlorine (3. 0) is greater than that of hydrogen (2. 1) § Thus the chlorine will be partially negative, and the hydrogen will be partially positive § The HCl molecule has a dipole moment Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution (continued 1) § The Cl 2 molecule § The two chlorine atoms share the electrons equally § No bond polarity occurs, and the Cl 2 molecule has no dipole moment § The SO 3 molecule § The electronegativity of oxygen (3. 5) is greater than that of sulfur (2. 5) § This means that each oxygen will have a partial negative charge, and the sulfur will have a partial positive charge Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution (continued 2) § The bond polarities arranged symmetrically as shown cancel, and the molecule has no dipole moment § This molecule is the second type shown in Table 8. 3 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution (continued 3) § The CH 4 molecule § Carbon has a slightly higher electronegativity (2. 5) than does hydrogen (2. 1) § This leads to small partial positive charges on the hydrogen atoms and a small partial negative charge on the carbon Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution (continued 4) § This case is similar to the third type in Table 8. 3, and the bond polarities cancel § The molecule has no dipole moment § The H 2 S molecule § Since the electronegativity of sulfur (2. 5) is slightly greater than that of hydrogen (2. 1), the sulfur will have a partial negative charge, and the hydrogen atoms will have a partial positive charge Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Example 8. 2 - Solution (continued 5) § This case is analogous to the water molecule, and the polar bonds result in a dipole moment oriented as shown: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Join In (4) § Which of the following is the most polar bond without being considered ionic? a. b. c. d. e. C—O Mg—O N—O O—F Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Join In (5) § Which of the following is the least polar bond, yet still considered polar covalent? a. b. c. d. e. C—O Mg—O N—O O—F Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Join In (6) § What is the correct order of the following bonds in terms of decreasing polarity? a. b. c. d. e. N—Cl, P—Cl, As—Cl P—Cl, N—Cl, As—Cl, N—Cl, P—Cl, As—Cl, N—Cl As—Cl, P—Cl, N—Cl Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 3 Bond Polarity and Dipole Moments Join In (7) § In which case is the bond polarity incorrect? a. b. c. d. e. +H—F – +K—O – +Mg—H – +Cl—I – +Si—S – Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Generalizations That Apply to Electron Configurations in Stable Compounds § When two nonmetals react to form a covalent bond, they share electrons in a way that completes the valence electron configurations of both atoms § Both nonmetals attain noble gas electron configurations Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Generalizations That Apply to Electron Configurations in Stable Compounds (continued) § When a nonmetal and a representative-group metal react to form a binary ionic compound, the ions form so that the: § Valence electron configuration of the nonmetal achieves the electron configuration of the next noble gas atom § Valence orbitals of the metal are emptied § Both ions achieve noble gas electron configurations Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Solid State of Ionic Compounds § Solid ionic compounds contain large collection of positive and negative ions packed together in a way that: § Minimizes the –. . – and +. . + repulsions § Maximizes +. . – attractions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Gas Phase of an Ionic Substance § Ions are relatively far apart and would not contain large groups of ions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Predicting Formulas of Ionic Compounds - Example § Consider Ca and O § Valence electron configuration for Ca is [Ar]s 2 and for O is [He]2 s 22 p 4 § Electronegativity of oxygen (3. 5) is much greater than that of calcium (1. 0) § Electrons will be transferred from calcium to oxygen to form oxygen anions and calcium cations in the compound Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Predicting Formulas of Ionic Compounds - Example (continued) § Oxygen needs two electrons to fill its 2 s and 2 p valence orbitals and to achieve the configuration of neon (1 s 22 p 6) § By losing two electrons, calcium can achieve the configuration of argon § Since there are equal numbers of Ca 2+ and O 2–, the empirical formula of the compound is Ca. O Copyright © 2018 Cengage Learning. All Rights Reserved.
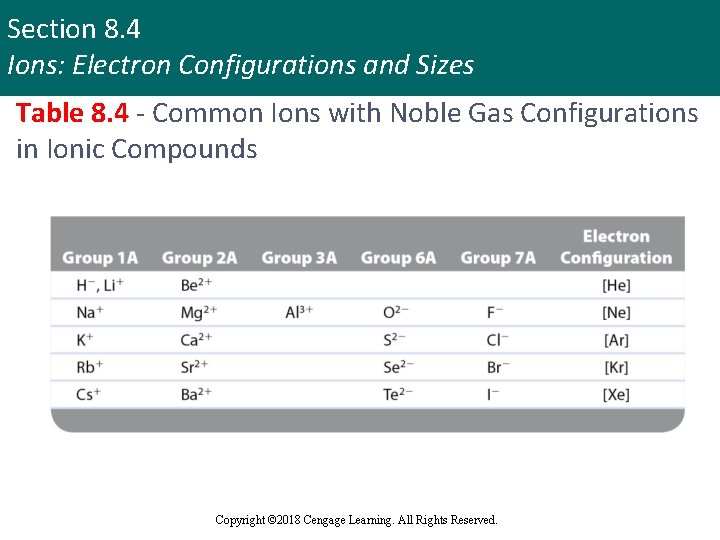
Section 8. 4 Ions: Electron Configurations and Sizes Table 8. 4 - Common Ions with Noble Gas Configurations in Ionic Compounds Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Noble Gas Electron Configurations in Ionic Compounds Trends § In losing electrons to form cations: § Metals in Group 1 A lose one electron § Metals in Group 2 A lose two electrons § Metals in Group 3 A lose three electrons § In gaining electrons to form anions: § Nonmetals in Group 7 A (the halogens) gain one electron § Nonmetals in Group 6 A gain two electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Noble Gas Electron Configurations in Ionic Compounds Exceptions § Representative elements § § Tin forms both Sn 2+ and Sn 4+ ions Lead forms both Pb 2+ and Pb 4+ ions Bismuth forms both Bi 3+ and Bi 5+ ions Thallium forms both Tl+ and Tl 3+ ions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Ion Size § Plays an important role in determining: § Structure and stability of ionic solids § Properties of ions in aqueous solution § Biologic effects of ions § Ionic radii are determined from the measured distance between ion centers in ionic compounds § Involves an assumption about how the distance should be divided up between the two ions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Factors That Influence Ionic Size § Relative sizes of an ion and its parent atom § Positive ion is formed by removing one or more electrons from a neutral atom § Resulting cation is smaller than its parent atom § Negative ions are formed by adding electrons to a neutral atom § Resulting anion is larger than its parent atom Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Factors That Influence Ionic Size (continued) § Position of the parent element in the periodic table § Ion sizes increase down a group § A given period contains both elements that: § Give up electrons to form cations § Accept electrons to form anions Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Isoelectronic Ions § Series of ions containing the same number of electrons § Examples § O 2–, F–, Na+, Mg 2+, and Al 3+ § Trend § Size decreases as Z (nuclear charge) increases Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Figure 8. 8 - Sizes of Ions Related to Positions of the Elements on the Periodic Table Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Critical Thinking § Ions have different radii than their parent atoms § What if ions stayed the same size as their parent atoms? § How would this affect ionic bonding in compounds? Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Interactive Example 8. 3 - Relative Ion Size I § Arrange the following ions in order of decreasing size: § Se 2–, Br–, Rb+, and Sr 2+ Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Interactive Example 8. 3 - Solution § This is an isoelectronic series of ions with the krypton electron configuration § Since these ions all have the same number of electrons, their sizes will depend on the nuclear charge § The Z values are 34 for Se 2–, 35 for Br–, 37 for Rb+, and 38 for Sr 2+ Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Interactive Example 8. 3 - Solution (continued) § Since the nuclear charge is greatest for Sr 2+, it is the smallest of these ions § The Se 2– ion is largest: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 4 Ions: Electron Configurations and Sizes Join In (8) § Which of the following has the largest radius? a. b. c. d. e. S 2– Cl– Ar K+ Ca 2+ Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Lattice Energy § Change in energy that takes place when separated gaseous ions are packed together to form an ionic solid § Defined as the energy released when an ionic solid forms from its ions § Lattice energy has a negative sign Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride 1. Sublimation of solid lithium § Sublimation involves taking a substance from the solid state to the gaseous state § Enthalpy of sublimation for Li(s) is 161 k. J/mol Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride (continued 1) 2. Ionization of lithium atoms to form Li+ ions in the gas phase § Process corresponds to the first ionization energy for lithium, which is 520 k. J/mol 3. Dissociation of fluorine molecules § Form a mole of fluorine atoms by breaking the F—F bonds in a half mole of F 2 molecules Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride (continued 2) § Energy required to break this bond is 154 k. J/mol § Since the bonds are broken in a half mole of fluorine, energy required is (154 k. J)/2 or 77 k. J Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride (continued 3) 4. Formation of F– ions from fluorine atoms in the gas phase § Energy change for this process corresponds to the electron affinity of fluorine, which is – 328 k. J/mol Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride (continued 5) 5. Formation of solid lithium fluoride from the gaseous Li+ and F– ions § This corresponds to the lattice energy for Li. F, which is – 1047 k. J/mol Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Energy Changes in the Formation of Lithium Fluoride (continued 6) § Sum of the five processes yields the desired overall reaction, and the sum of the individual energy changes gives the overall energy change Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Figure 8. 9 - The Energy Changes Involved in the Formation of Solid Lithium Fluoride from Its Elements Numbers in parentheses refer to the reaction steps discussed in the text Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Lattice Energy: Calculations § Represented by a modified form of Coulomb’s law § k - Proportionality constant § Depends on the structure of the solid and the electronic configurations of the ions § Q 1 and Q 2 - Charges on the ions § r - Shortest distance between the centers of the anions and the cations Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Lattice Energy: Calculations (continued) § Lattice energy has a negative sign when Q 1 and Q 2 have opposite signs § Result is expected because bringing cations and anions together is an exothermic process § Process becomes more exothermic as: § Ionic charges increase § Distances between ions in the solid decrease Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Factor Influencing the Composition and Structure of Solid Ionic Compounds § Most important factor involves the balancing of: § Energies required to form highly charged ions § Energy released when highly charged ions combine to form the solid Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 5 Energy Effects in Binary Ionic Compounds Join In (9) § Which of the following ionic compounds has the largest lattice energy (i. e. , the lattice energy most favorable to a stable lattice)? a. b. c. d. e. Cs. I Na. Cl Li. F Cs. F Mg. O Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 6 Partial Ionic Character of Covalent Bonds Figure 8. 12 - The Three Possible Types of Bonds Covalent bond formed between identical F atoms Polar covalent bond of HF, with both ionic and covalent components Copyright © 2018 Cengage Learning. All Rights Reserved. An ionic bond with no electron sharing

Section 8. 6 Partial Ionic Character of Covalent Bonds Formula for Percent Ionic Character of a Bond § Totally ionic bonds between discrete pairs of atoms do not exist § Evident from calculations of percent ionic character for bonds of various binary compounds in the gas phase Copyright © 2018 Cengage Learning. All Rights Reserved.
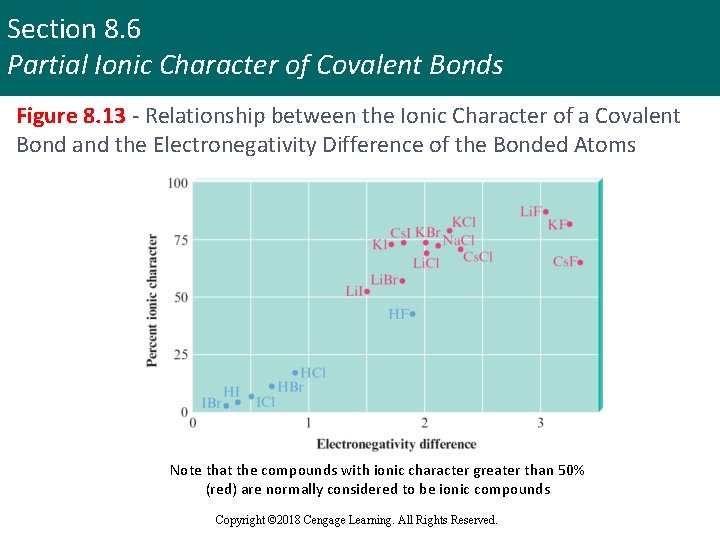
Section 8. 6 Partial Ionic Character of Covalent Bonds Figure 8. 13 - Relationship between the Ionic Character of a Covalent Bond and the Electronegativity Difference of the Bonded Atoms Note that the compounds with ionic character greater than 50% (red) are normally considered to be ionic compounds Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 6 Partial Ionic Character of Covalent Bonds Complications in Identifying Ionic Compounds § Ionic character increases with electronegativity difference, but none of the bonds reaches 100% ionic character § Therefore, no individual bonds are completely ionic § Many substances contain polyatomic ions § Bonds within the ions are covalent Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 6 Partial Ionic Character of Covalent Bonds Complications in Identifying Ionic Compounds (continued) § Avoided by adopting the operational definition of ionic compound § Any compound that conducts an electric current when melted will be classified as ionic Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 7 The Covalent Chemical Bond: A Model Bonds § Forces that cause a group of atoms to behave as a unit § Result from the tendency of a system to seek its lowest possible energy § Bonds occur when collections of atoms are more stable (lower in energy) than the separate atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 7 The Covalent Chemical Bond: A Model Chemical Bonds § Models that depict molecular stability § Example - Methane has a tetrahedron structure (four equal triangular faces) § Bond concept is a human invention § Provides a method for dividing up the energy evolved when a stable molecule is formed from its component atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 7 The Covalent Chemical Bond: A Models § Attempt to explain how nature operates on the microscopic level based on experiences in the macroscopic world § Originate from observations of the properties of nature Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 7 The Covalent Chemical Bond: A Model Bonding Model § Provides a framework to systematize chemical behavior § Molecules are perceived as collections of common fundamental components § Physically sensible § Makes sense that atoms can form stable compounds by sharing electrons § Shared electrons give a lower energy state because they are simultaneously attracted by two nuclei Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Types of Bonds § Single bond § One pair of electrons is shared § Double bond § Two pairs of electrons are shared § Triple bond § Three pairs of electrons are shared § Bond length shortens with the increase in the number of shared electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Table 8. 5 - Average Bond Energies (k. J/mol) Copyright © 2018 Cengage Learning. All Rights Reserved.
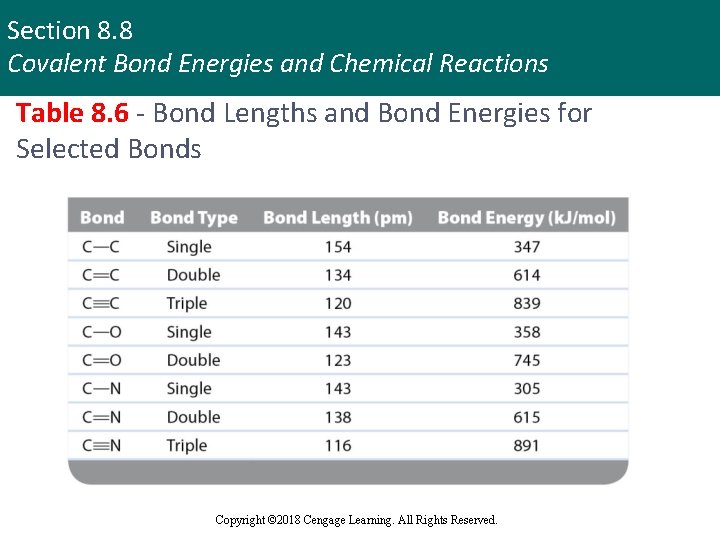
Section 8. 8 Covalent Bond Energies and Chemical Reactions Table 8. 6 - Bond Lengths and Bond Energies for Selected Bonds Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Bond Energy § Energy must be added to the system to break bonds (endothermic process) § Energy terms associated with bond breaking have positive signs § Energy is released when bonds are formed (exothermic process) § Energy terms associated with bond making carry a negative sign Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Enthalpy Change for a Reaction § Can be calculated as follows: § Σ - Sum of terms § D - Bond energy per mole of bonds § Always has a positive sign § n - Moles of a particular type of bond Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Interactive Example 8. 5 - ΔH from Bond Energies § Use the bond energies listed in Table 8. 5, and calculate ΔH for the reaction of methane with chlorine and fluorine to give Freon-12 (CF 2 Cl 2) Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Interactive Example 8. 5 - Solution § Break the bonds in the gaseous reactants to give individual atoms and then assemble these atoms into the gaseous products by forming new bonds § Then combine the energy changes to calculate ΔH: § ΔH = Energy required to break bonds – energy released when bonds form § Minus sign gives the correct sign to the energy terms for the exothermic processes Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Interactive Example 8. 5 - Solution (continued 1) § Reactant bonds broken: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Interactive Example 8. 5 - Solution (continued 2) § Product bonds formed: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Interactive Example 8. 5 - Solution (continued 3) § We now can calculate ΔH: § Since the sign of the value for the enthalpy change is negative, this means that 1194 k. J of energy is released per mole of CF 2 Cl 2 formed Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Join In (10) Bond Average Bond Energy (k. J/mol) H—H 432 F—F 154 H—F 565 § Given the average bond energies above, calculate ∆H for the following reaction: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 8 Covalent Bond Energies and Chemical Reactions Join In (10) (continued) a. b. c. d. e. – 21 k. J 544 k. J -544 k. J 1151 k. J Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 9 The Localized Electron Bonding Model Localized Electron (LE) Model § Assumes that a molecule is composed of atoms that are bound together by sharing pairs of electrons using the atomic orbitals of the bound atoms § Lone pairs: Pairs of electrons localized on an atom § Bonding pairs: Pairs of electrons found in the space between atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 9 The Localized Electron Bonding Model Parts of the LE Model § Description of the valence electron arrangement in the molecule using Lewis structures § Prediction of the geometry of the molecule using the valence shell electron-pair repulsion (VSEPR) model § Description of the type of atomic orbitals used by the atoms to share electrons or hold lone pairs Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Lewis Structure § Depicts the arrangement of valence electrons among atoms in a molecule § Named after G. N. Lewis § Only valence electrons are included § Most important requirement for the formation of a stable compound § Atoms must attain noble gas electron configurations Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Principle of Achieving a Noble Gas Electron Configuration - Hydrogen § Follows a duet rule § Forms stable molecules where it shares two electrons § Example - Two hydrogen atoms, each with one electron, combine to form the H 2 molecule § By sharing electrons, each hydrogen has a filled valence shell Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Principle of Achieving a Noble Gas Electron Configuration - Helium § Does not form bonds because it is a noble gas § Valence orbital is already filled § Electron configuration - 1 s 2 § Represented by the following Lewis structure: He: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Principle of Achieving a Noble Gas Electron Configuration Second-Row Nonmetals (Carbon through Fluorine) § Form stable molecules when surrounded by enough electrons to fill valence orbitals § Obey the octet rule § Octet rule: Elements form stable molecules when surrounded by eight electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Principle of Achieving a Noble Gas Electron Configuration Second-Row Nonmetals (Carbon Through Fluorine) (continued) § Each fluorine atom in F 2 is surrounded by eight electrons § Two electrons that are shared with the other atom constitute a bonding pair § Each fluorine atom also has three pairs of electrons not involved in bonding § These are the lone pairs Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Principle of Achieving a Noble Gas Electron Configuration - Neon § Does not form bonds because it already has an octet of valence electrons § Represented by the following Lewis structure: § Only the valence electrons (2 s 22 p 6) are represented in the Lewis structure Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Problem Solving Strategy - Steps for Writing Lewis Structures 1. Sum the valence electrons from all the atoms 2. Use a pair of electrons to form a bond between each pair of bound atoms 3. Arrange the remaining electrons to satisfy the duet rule for hydrogen and the octet rule for the second-row elements Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Drawing the Lewis Structure of Water § Sum the valence electrons for H 2 O 1 + 6 = 8 valence electrons H H O § Using a pair of electrons per bond, draw the O—H single bonds H—O—H § A line is used to indicate each pair of bonding electrons Copyright © 2018 Cengage Learning. All Rights Reserved.
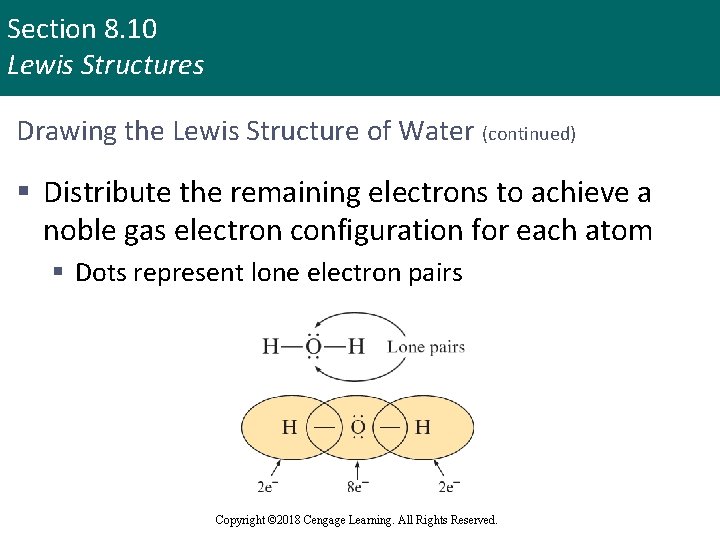
Section 8. 10 Lewis Structures Drawing the Lewis Structure of Water (continued) § Distribute the remaining electrons to achieve a noble gas electron configuration for each atom § Dots represent lone electron pairs Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Interactive Example 8. 6 - Writing Lewis Structures § Give the Lewis structure for each of the following: a. b. c. d. e. f. HF N 2 NH 3 CH 4 CF 4 NO+ Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Interactive Example 8. 6 - Solution § In each case, apply the three steps for writing Lewis structures § Recall that lines are used to indicate shared electron pairs and that dots are used to indicate nonbonding pairs (lone pairs) Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Interactive Example 8. 6 - Solution (continued) Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 10 Lewis Structures Join In (11) § Which of the following does not contain at least one double bond in the Lewis structure? a. b. c. d. H 2 CO C 2 H 4 CO 2 C 3 H 8 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Boron § Tends to form compounds in which the boron atom has fewer than eight electrons around it § Boron trifluoride (BF 3) § Reacts energetically with molecules that have available lone pairs § Boron atom is electron-deficient § Has 24 valence electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Boron (continued) § Octet rule is satisfied by drawing the structure with a double bond § Characteristic of boron to form molecules in which the boron atom is electron-deficient Copyright © 2018 Cengage Learning. All Rights Reserved.
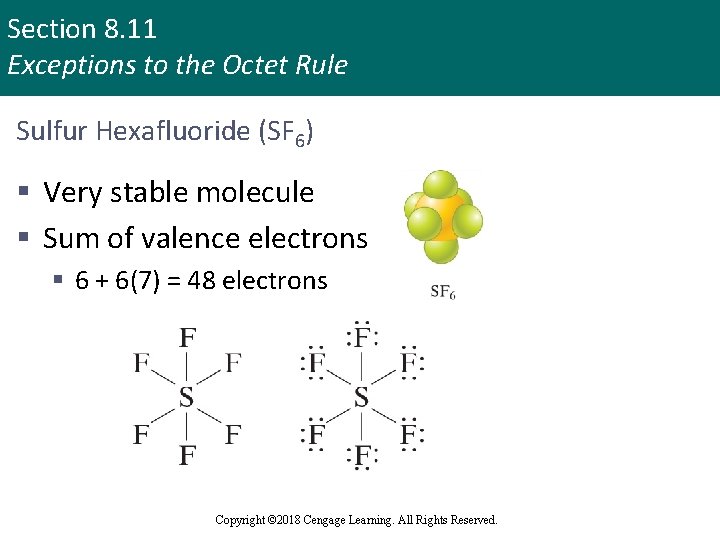
Section 8. 11 Exceptions to the Octet Rule Sulfur Hexafluoride (SF 6) § Very stable molecule § Sum of valence electrons § 6 + 6(7) = 48 electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Sulfur Hexafluoride (SF 6) (continued) § Sulfur exceeds the octet rule § Localized electron model assumes that the empty 3 d orbitals can be used to accommodate extra electrons § Sulfur atom in SF 6 can have 12 electrons around it by using the 3 s and 3 p orbitals to hold 8 electrons, with the extra 4 electrons placed in the formerly empty 3 d orbitals Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 7 - Lewis Structures for Molecules That Violate the Octet Rule I § Write the Lewis structure for PCl 5 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 7 - Solution 1. Sum the valence electrons 5 + 5(7) = 40 electrons P Cl 2. Indicate single bonds between bound atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 7 - Solution (continued) 3. Distribute the remaining electrons § In this case, 30 electrons (40 – 10) remain § These are used to satisfy the octet rule for each chlorine atom § Final Lewis structure is § Phosphorus, a third-row element, has exceeded the octet rule by two electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Lewis Structure of Molecules with More Than One Atom That Exceed the Octet Rule § Assume that the extra electrons should be placed on the central atom § Example - Lewis structure of I 3– (22 valence electrons) § Central iodine exceeds the octet rule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 8 - Lewis Structures for Molecules That Violate the Octet Rule II § Write the Lewis structure for the following: a. CIF 3 b. Rn. Cl 2 c. ICl 4– Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 8 - Solution a. The chlorine atom (3 rd row) accepts the extra electrons b. Radon, a noble gas in Period 6, accepts the extra electrons Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Interactive Example 8. 8 - Solution (continued) c. Iodine exceeds the octet rule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Join In (12) § Which of the following has a Lewis structure most like that of CO 32–? a. b. c. d. e. CO 2 SO 32– NO 3– O 3 NO 2 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 11 Exceptions to the Octet Rule Join In (13) § Which molecule or ion violates the octet rule? a. b. c. d. e. H 2 O NO 3– PF 3 I 3– None of these Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance: An Introduction § Invoked when more than one valid Lewis structure can be written for a particular molecule § Resulting electron structure of the molecule is given by the average of these resonance structures § Represented by double-headed arrows Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance: An Introduction (continued) § Arrangement of nuclei is the same across all structures § Placement of electrons differs § Arrows show that the actual structure is an average of the three resonance structures § Concept is necessary to compensate for the defective assumption of the LE model § LE model postulates that electrons are localized between a given pair of atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 9 - Resonance Structures § Describe the electron arrangement in the nitrite anion (NO 2–) using the localized electron model Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 9 - Solution § NO 2– possesses 18 valence electrons (5 + 2(6) + 1 = 18) § Indicating the single bonds gives the structure O—N—O § Remaining 14 electrons (18 – 4) can be used to produce these structures: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 9 - Solution (continued) § This is a resonance situation, and two equivalent Lewis structures can be drawn § The electronic structure of the molecule is correctly represented not by either resonance structure but by the average of the two § There are two equivalent N—O bonds, each one intermediate between a single and a double bond Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Odd-Electron Molecules § Few molecules formed from nonmetals contain odd numbers of electrons § Example - Nitric oxide (NO) § Emitted into the air where it reacts with oxygen to form NO 2 (g), which is another odd-electron molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Formal Charge § Difference between the number of valence electrons on the free atom and the number of valence electrons assigned to the atom in the molecule § Used to evaluate nonequivalent Lewis structures § Nonequivalent Lewis structures contain different numbers of single and multiple bonds Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Determining Formal Charge of an Atom in a Molecule § Information required § Number of valence electrons on the free neutral atom § Free neutral atom has zero net charge because the number of electrons equals the number of protons § Number of valence electrons belonging to the atom in a molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Determining Formal Charge of an Atom in a Molecule (continued 1) § Assign valence electrons in the molecule to the various atoms, making the following assumptions: § Lone pair electrons belong entirely to the atom in question § Shared electrons are divided equally between the two sharing atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Determining Formal Charge of an Atom in a Molecule (continued 2) § Number of valence electrons assigned to a given atom is calculated as follows: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Fundamental Assumptions about Formal Charges to Evaluate Lewis Structures § Atoms in molecules try to achieve formal charges as close to zero as possible § Any negative formal charges are expected to reside on the most electronegative atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Rules Governing Formal Charge § To calculate the formal charge on an atom: 1. Take the sum of the lone pair electrons and one-half the shared electrons § This is the number of valence electrons assigned to the atom in the molecule 2. Subtract the number of assigned electrons from the number of valence electrons on the free, neutral atom to obtain the formal charge Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Rules Governing Formal Charge (continued) § Sum of the formal charges of all atoms in a given molecule or ion must be equal to the overall charge on that species § Nonequivalent Lewis structures § Species with formal charges closest to zero and with any negative formal charges on the most electronegative atoms are considered to best describe the bonding in the molecule or ion Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 10 - Formal Charges § Give possible Lewis structures for Xe. O 3, an explosive compound of xenon § Which Lewis structure or structures are most appropriate according to the formal charges? Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 10 - Solution § For Xe. O 3 (26 valence electrons) we can draw the following possible Lewis structures: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Example 8. 10 - Solution (continued) § Based on the ideas of formal charge, we would predict that the Lewis structures with the lower values of formal charge would be most appropriate for describing the bonding in Xe. O 3 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Cautions about Formal Charge § Formal charges provide estimates of charge § Should not be considered as actual atomic charges § Evaluation of Lewis structures using formal charge ideas can lead to erroneous predictions § Experimental evidence must be used to make the final decisions on the correct description of the bonding in a molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Join In (14) § How many resonance structures does the molecule SO 2 have? a. b. c. d. e. 0 1 2 3 4 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Join In (15) § Which of the following statements concerning resonance structures is correct? a. The concept of resonance is used because the Lewis structure model is incomplete when describing bonding in a molecule b. For a species having three resonance structures, it is best to think of the species as existing as each of these structures one-third of the time Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 12 Resonance Join In (15) (continued) c. All charged molecules have resonance structures d. The octet rule must not be violated in writing resonance structures Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Valence Shell Electron-Pair Repulsion (VSEPR) Model § Used to predict the molecular structure of molecules formed from nonmetals § Molecular structure: Three-dimensional arrangement of molecules in an atom § Main postulate § Structure around a given atom is determined principally by minimizing electron-pair repulsions § Bonding and nonbonding pairs around a given atom will be placed as far apart as possible Copyright © 2018 Cengage Learning. All Rights Reserved.
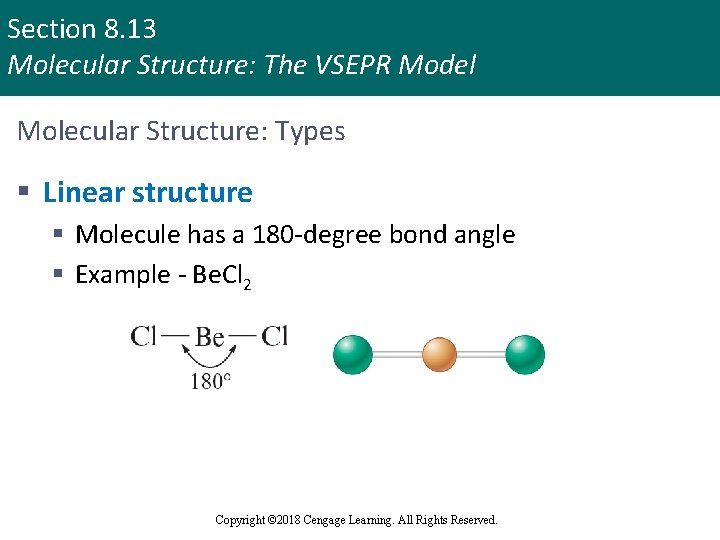
Section 8. 13 Molecular Structure: The VSEPR Model Molecular Structure: Types § Linear structure § Molecule has a 180 -degree bond angle § Example - Be. Cl 2 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Molecular Structure: Types (continued 1) § Trigonal planar structure § Molecule has a planar (flat) and triangular structure with 120° bond angles § Example - BF 3 Copyright © 2018 Cengage Learning. All Rights Reserved.
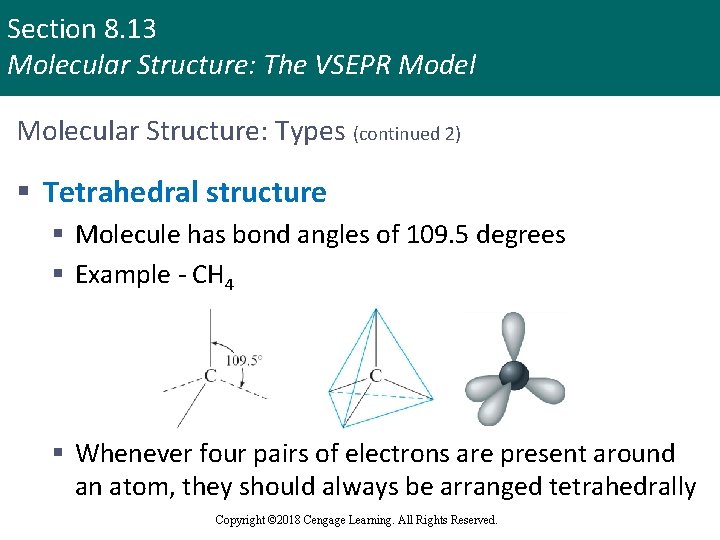
Section 8. 13 Molecular Structure: The VSEPR Model Molecular Structure: Types (continued 2) § Tetrahedral structure § Molecule has bond angles of 109. 5 degrees § Example - CH 4 § Whenever four pairs of electrons are present around an atom, they should always be arranged tetrahedrally Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Problem-Solving Strategy - Steps to Apply the VSEPR Model 1. Draw the Lewis structure for the molecule 2. Count the electron pairs and arrange them in the way that minimizes repulsion 3. Determine the positions of the atoms from the way the electron pairs are shared 4. Determine the name of the molecular structure from the positions of the atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Example 8. 11 - Prediction of Molecular Structure I § Describe the molecular structure of the water molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Example 8. 11 - Solution § The Lewis structure for water is § There are four pairs of electrons: two bonding pairs and two nonbonding pairs § To minimize repulsions, these pairs are best arranged in a tetrahedral array Copyright © 2018 Cengage Learning. All Rights Reserved.
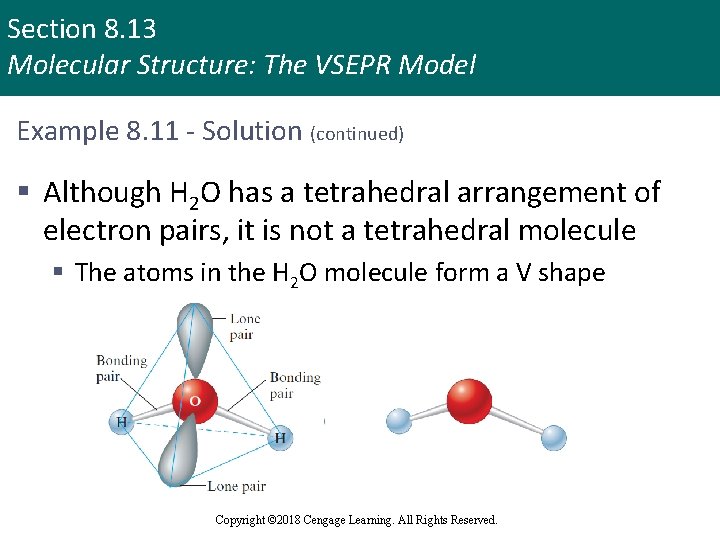
Section 8. 13 Molecular Structure: The VSEPR Model Example 8. 11 - Solution (continued) § Although H 2 O has a tetrahedral arrangement of electron pairs, it is not a tetrahedral molecule § The atoms in the H 2 O molecule form a V shape Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Addition to the Original Postulate of the VSEPR Model § Lone pairs require more room than bonding pairs and tend to compress the angles between the bonding pairs Copyright © 2018 Cengage Learning. All Rights Reserved.
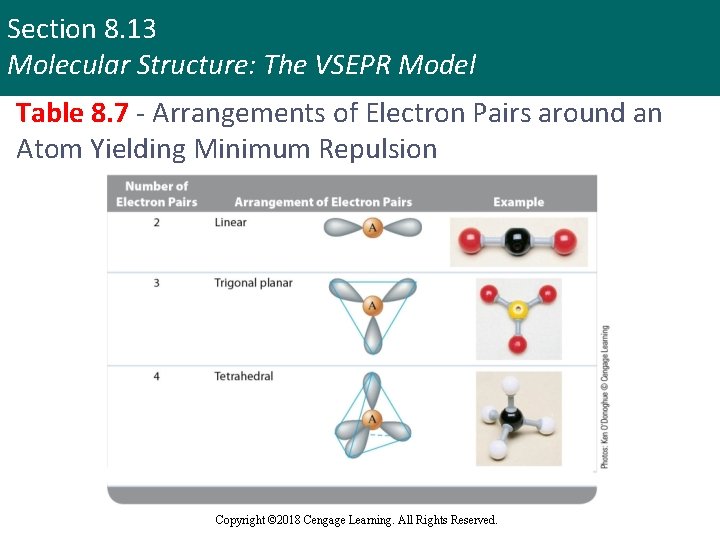
Section 8. 13 Molecular Structure: The VSEPR Model Table 8. 7 - Arrangements of Electron Pairs around an Atom Yielding Minimum Repulsion Copyright © 2018 Cengage Learning. All Rights Reserved.
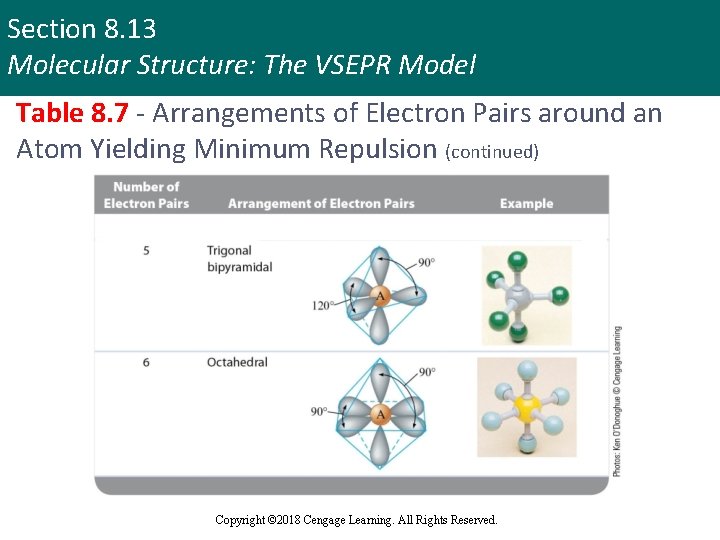
Section 8. 13 Molecular Structure: The VSEPR Model Table 8. 7 - Arrangements of Electron Pairs around an Atom Yielding Minimum Repulsion (continued) Copyright © 2018 Cengage Learning. All Rights Reserved.
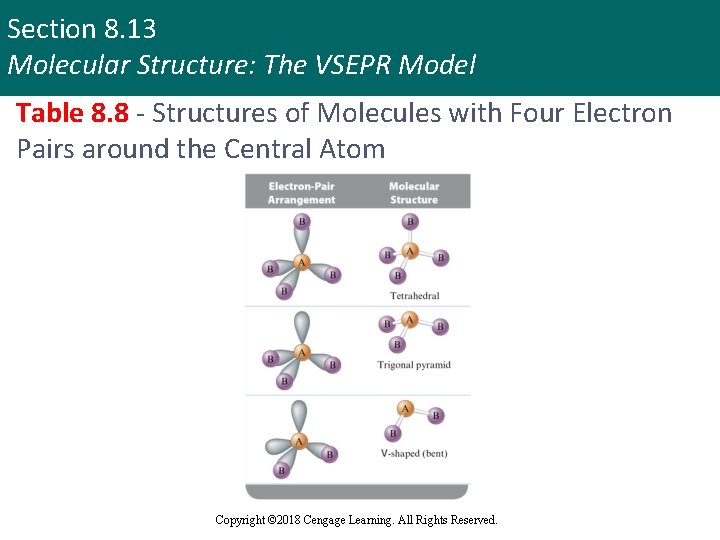
Section 8. 13 Molecular Structure: The VSEPR Model Table 8. 8 - Structures of Molecules with Four Electron Pairs around the Central Atom Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Molecular Structure: Types (continued 3) § Trigonal bipyramidal structure § Produces minimum repulsion for molecules that contain five pairs of electrons around the central atom § Bond angles - 90° and 120° § Octahedral structure § Produces minimum repulsion for molecules that contain six pairs of electrons around the central atom § Bond angle - 90° Copyright © 2018 Cengage Learning. All Rights Reserved.
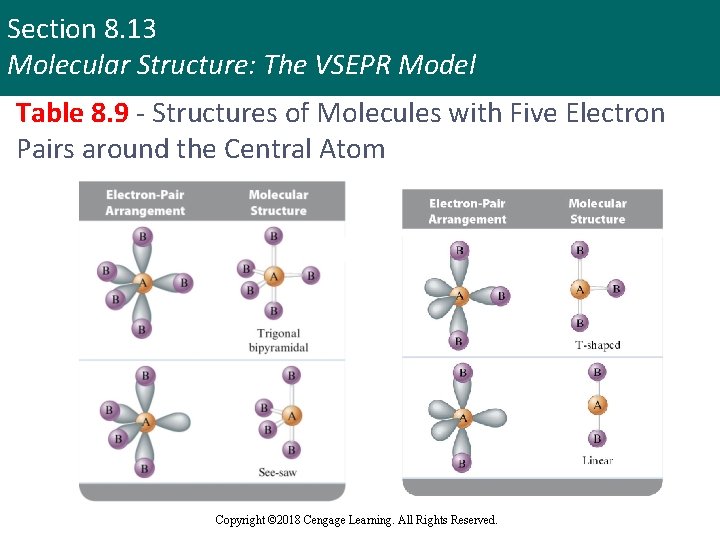
Section 8. 13 Molecular Structure: The VSEPR Model Table 8. 9 - Structures of Molecules with Five Electron Pairs around the Central Atom Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Critical Thinking § You and a friend are studying for a chemistry exam § What if your friend tells you that all molecules with polar bonds are polar molecules? § How would you explain to your friend that this is not correct? § Provide two examples to support your answer Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Prediction of Molecular Structure II § When phosphorus reacts with excess chlorine gas, the compound phosphorus pentachloride (PCl 5) is formed § In the gaseous and liquid states, this substance consists of PCl 5 molecules, but in the solid state it consists of a 1: 1 mixture of PCl 4+ and PCl 6– ions § Predict the geometric structures of PCl 5, PCl 4+, and PCl 6 – Copyright © 2018 Cengage Learning. All Rights Reserved.
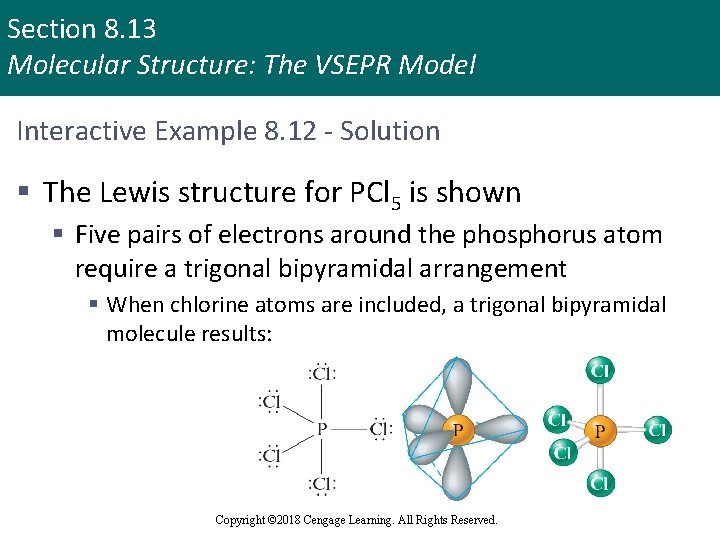
Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Solution § The Lewis structure for PCl 5 is shown § Five pairs of electrons around the phosphorus atom require a trigonal bipyramidal arrangement § When chlorine atoms are included, a trigonal bipyramidal molecule results: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Solution (continued 1) § The Lewis structure for the PCl 4+ ion [5 + 4(7) – 1 = 32 valence electrons] is shown below Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Solution (continued 2) § There are four pairs of electrons surrounding the phosphorus atom in the PCl 4+ ion, which requires a tetrahedral arrangement of the pairs § Since each pair is shared with a chlorine atom, a tetrahedral PCl 4+ cation results Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Solution (continued 3) § The Lewis structure for PCl 6– [5 + 6(7) + 1 = 48 valence electrons] is shown below Copyright © 2018 Cengage Learning. All Rights Reserved.
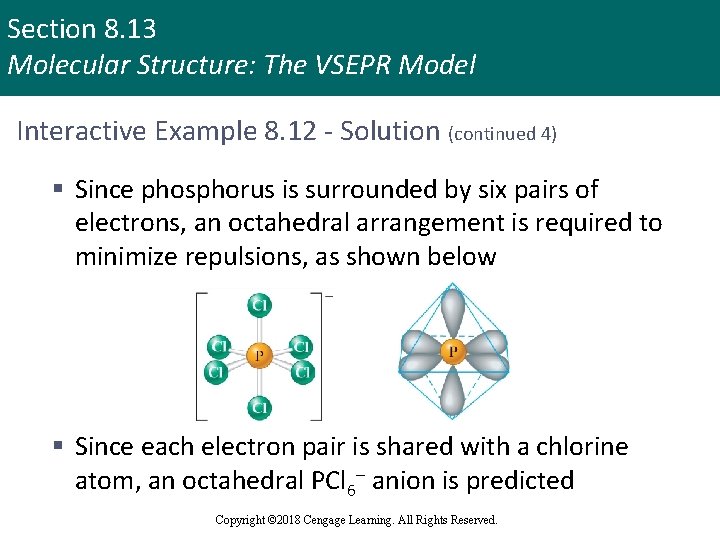
Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 12 - Solution (continued 4) § Since phosphorus is surrounded by six pairs of electrons, an octahedral arrangement is required to minimize repulsions, as shown below § Since each electron pair is shared with a chlorine atom, an octahedral PCl 6– anion is predicted Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Prediction of Molecular Structure III § Because the noble gases have filled s and p valence orbitals, they were not expected to be chemically reactive § In fact, for many years these elements were called inert gases because of this supposed inability to form any compounds § However, in the early 1960 s several compounds of krypton, xenon, and radon were synthesized Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Prediction of Molecular Structure III (continued) § For example, a team at the Argonne National Laboratory produced the stable colorless compound xenon tetrafluoride (Xe. F 4) § Predict its structure and whether it has a dipole moment Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Solution § The Lewis structure for Xe. F 4 is § The xenon atom in this molecule is surrounded by six pairs of electrons, which means an octahedral arrangement Copyright © 2018 Cengage Learning. All Rights Reserved.
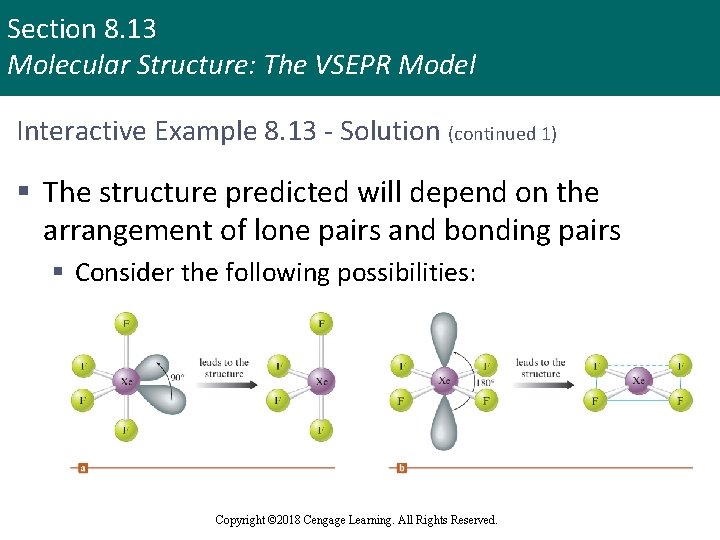
Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Solution (continued 1) § The structure predicted will depend on the arrangement of lone pairs and bonding pairs § Consider the following possibilities: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Solution (continued 2) § The bonding pairs are indicated by the presence of the fluorine atoms § Since the structure predicted differs in the two cases, we must decide which of these arrangements is preferable, and the key is to look at the lone pairs § In the structure in part (a), the lone pair–lone pair angle is 90 degrees § In the structure in part (b), the lone pairs are separated by 180 degrees Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Solution (continued 3) § Since lone pairs require more room than bonding pairs, a structure with two lone pairs at 90 degrees is unfavorable § Thus the arrangement (b) is preferred, and the molecular structure is predicted to be square planar § Note that this molecule is not described as being octahedral § There is an octahedral arrangement of electron pairs, but the atoms form a square planar structure Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 13 - Solution (continued 4) § Although each Xe—F bond is polar (fluorine has a greater electronegativity than xenon), the square planar arrangement of these bonds causes the polarities to cancel § Thus Xe. F 4 has no dipole moment Copyright © 2018 Cengage Learning. All Rights Reserved.
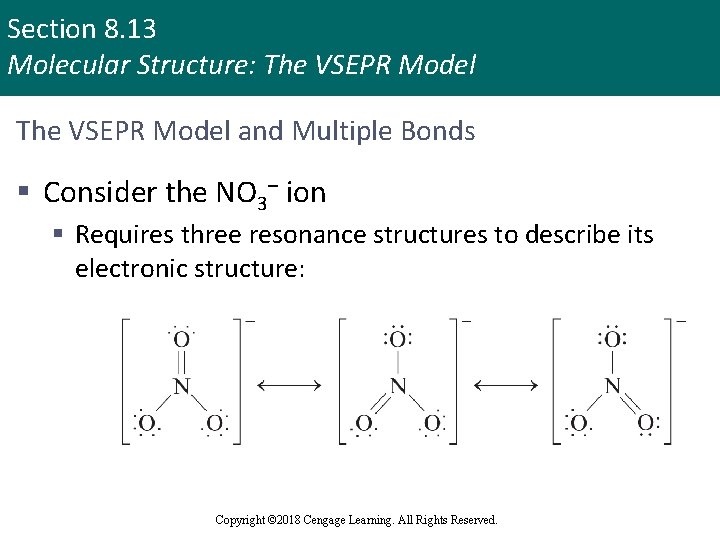
Section 8. 13 Molecular Structure: The VSEPR Model and Multiple Bonds § Consider the NO 3– ion § Requires three resonance structures to describe its electronic structure: Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model and Multiple Bonds (continued 1) § Known to be planar with 120 -degree bond angles § Structure is expected for three pairs of electrons around a central atom, which means that a double bond should be counted as one effective pair in using the VSEPR model Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model and Multiple Bonds (continued 2) § Rules § Multiple bonds count as one effective electron pair § When a molecule exhibits resonance, any one of the resonance structures can be used to predict the molecular structure using the VSEPR model Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 14 - Structures of Molecules with Multiple Bonds § Predict the molecular structure of the sulfur dioxide molecule § Is this molecule expected to have a dipole moment? Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 14 - Solution § First, we must determine the Lewis structure for the SO 2 molecule, which has 18 valence electrons § Expected resonance structures are § To determine the molecular structure, we must count the electron pairs around the sulfur atom Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 14 - Solution (continued 1) § In each resonance structure the sulfur has one lone pair, one pair in a single bond, and one double bond § Counting the double bond as one pair yields three effective pairs around the sulfur § A trigonal planar arrangement is required, which yields a Vshaped molecule Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Interactive Example 8. 14 - Solution (continued 2) § Thus the structure of the SO 2 molecule is expected to be V-shaped, with a 120 -degree bond angle § The molecule has a dipole moment directed as shown § Since the molecule is V-shaped, the polar bonds do not cancel Copyright © 2018 Cengage Learning. All Rights Reserved.
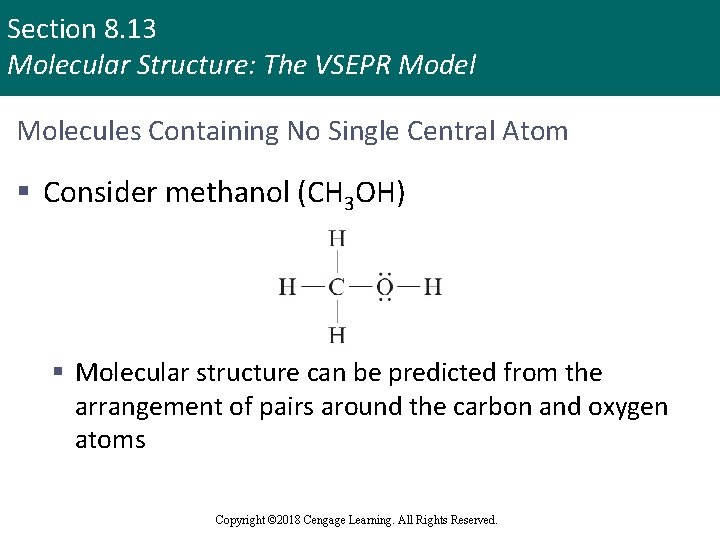
Section 8. 13 Molecular Structure: The VSEPR Model Molecules Containing No Single Central Atom § Consider methanol (CH 3 OH) § Molecular structure can be predicted from the arrangement of pairs around the carbon and oxygen atoms Copyright © 2018 Cengage Learning. All Rights Reserved.
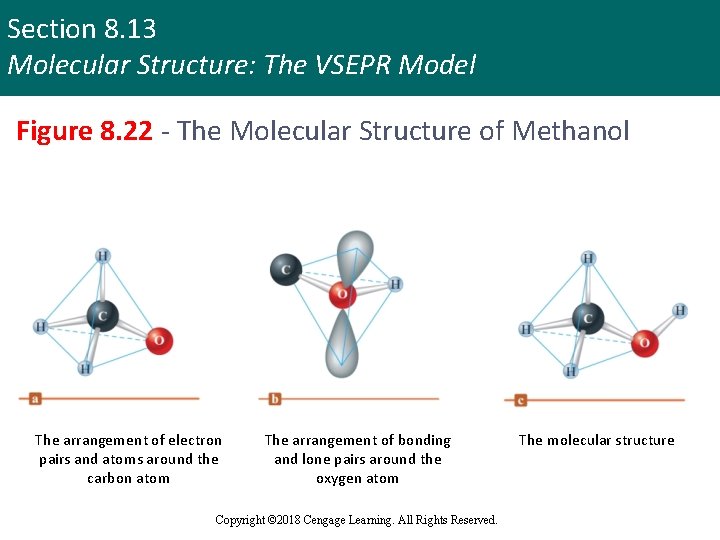
Section 8. 13 Molecular Structure: The VSEPR Model Figure 8. 22 - The Molecular Structure of Methanol The arrangement of electron pairs and atoms around the carbon atom The arrangement of bonding and lone pairs around the oxygen atom Copyright © 2018 Cengage Learning. All Rights Reserved. The molecular structure

Section 8. 13 Molecular Structure: The VSEPR Model: Advantages § Correctly predicts the molecular structures of most molecules formed from nonmetallic elements § Used to predict the structures of molecules with hundreds of atoms Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Limitation of the VSEPR Model § Does not always provide an accurate prediction § Example - Phosphine (PH 3) § Lewis structure of PH 3 is analogous to that of ammonia (NH 3) § Predicted molecular structure of PH 3 is similar to that of NH 3, with bond angles of approximately 107° Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Limitation of the VSEPR Model (continued) § Bond angles of phosphine are 94° Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (16) § Which of the following molecules is polar? a. b. c. d. Xe. F 4 Xe. F 2 BF 3 NF 3 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (17) § Which of the following molecules does not have a dipole moment? a. b. c. d. e. H 2 S H 2 O H 2 Xe All of these have a dipole moment None of these has a dipole moment Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (18) § Which of the following molecules has a dipole moment? a. b. c. d. e. CF 4 SF 4 Xe. F 4 All of these have a dipole moment None of these has a dipole moment Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (19) § Which of the following molecules has a dipole moment? a. b. c. d. BCl 3 Si. Cl 4 PCl 3 Cl 2 Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (20) § Which of the following statements about the species N 2, CO, CN–, and NO+ is false? a. b. c. d. All are isoelectronic Each contains a triple bond All are linear The bond in each species is polar Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (21) § What is the approximate measure of the bond angles about the carbon atom in the formaldehyde molecule, H 2 C O? a. b. c. d. e. 120° 60° 109° 180° 90° Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (22) § What type of structure does the Xe. OF 2 molecule have? a. b. c. d. e. Pyramidal Tetrahedral T-shaped Trigonal planar Octahedral Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (23) § Which of the following statements best describes BF 3 and NF 3? (Note: Geometry refers to the electron pair arrangement, and shape refers to the atom arrangement) a. They have variable geometries and shapes due to their potential resonance structures b. They have the same geometry and different shapes Copyright © 2018 Cengage Learning. All Rights Reserved.

Section 8. 13 Molecular Structure: The VSEPR Model Join In (23) (continued) c. They have the same geometry and the same shape d. They have different geometries and the same shape e. They have different geometries and different shapes Copyright © 2018 Cengage Learning. All Rights Reserved.
- Slides: 195