CHAPTER 8 BOND POLARITY Nonpolar Covalent Electrons pulled

CHAPTER 8

BOND POLARITY Nonpolar Covalent: Electrons pulled relatively equally EN difference 0 0. 4 Ex. H-H

BOND POLARITY Polar Covalent Electrons pulled unequally EN difference 0. 5 1. 9 More EN atom will attract electrons and have a slight negative charge Ex. H-Cl

BOND POLARITY Ionic Bond EN difference will be greater than 1. 9 ALL ionic compounds will always have ionic bonds, no matter what the EN difference is Ex. Na. Cl

POLAR BONDS BUT NOT A POLAR MOLECULE? Sometimes, a molecule can have polar bonds but be a nonpolar molecule Ex. Water and Carbon Dioxide

INTERMOLECULAR FORCES Intermolecular attractions are weak attractions between groups. They are weaker than ionic or covalent bonds Inter- among or between groups Intra- inside or within

VAN DER WAALS FORCES Dipole Polar molecules are attracted to each other Slightly negative region is attracted to slightly positive region of another molecule Ex. HCl

VAN DER WAALS FORCES Dispersion (London Dispersion) Weakest of all forces All polar and non polar molecules Non polar molecules can bond to one another Strength increases as the number of electrons increases

HYDROGEN BONDING Strongest of all forces Hydrogen is covalently bonded to fluorine, oxygen, or nitrogen (FON) May be in the same molecule or nearby molecule

BOND LENGTH Distance between centers of bonded atoms Determined by X-ray diffraction of solids Bond # of electrons Bond Order Bond Strength Bond Length Single 2 1 Weakest Longest Double 4 2 Triple 6 3 Strongest Shortest
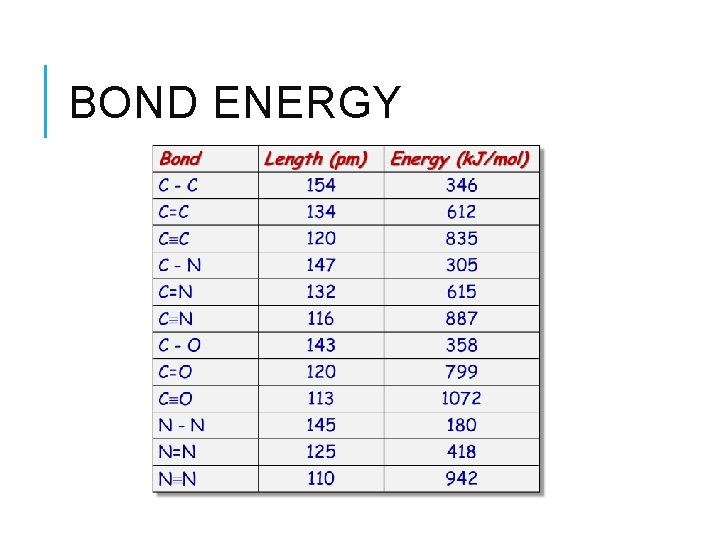
BOND ENERGY

CALCULATE THE BOND ENERGY CO 2 C 2 H 4 PH 3
- Slides: 12