Chapter 8 Applications of Aqueous Equilibria 1 Solutions

Chapter 8 Applications of Aqueous Equilibria 1

Solutions of Acids and Bases Containing a Common Ion n e. g. A solution containing a weak acid HA but also its salt Na. A ¨ Major species: HA, Na+, A-, H 2 O. n Common Ion Effect: the shift in an equilibrium position caused by the addition or presence of an ion involved in the equilibrium reaction. n e. g. A solution containing a 1. 0 M HF and 1. 0 M Na. F. ¨ Major species: HF, Na+, F-, H 2 O. HF/Na. F solution less acidic than HF solution 2

n Example: We did an example where we found that the equilibrium concentration of H+ in a 1. 0 M HF solution is 2. 7 x 10 -2 M and the percent dissociation of HF is 2. 7%. Calculate [H+] and the percent dissociation of HF in a solution containing both 1. 0 M HF (Ka = 7. 2 x 10 -4) and 1. 0 M Na. F n Major species: HF, F-, Na+, H 2 O [H+] = x = 7. 2 x 10 -4 n Percent dissociation of HF 3

Buffered Solutions n A buffered solution resists a change in p. H when either hydroxide ions or protons are added. n Usually a weak acid and its conjugate base (CH 3 COOH/CH 3 COONa) or a weak base and its conjugate acid (NH 3/NH 4 Cl) n Example: A buffered solution contains 0. 50 M acetic acid (CH 3 COOH, Ka = 1. 8 x 10 -5) and 0. 50 M sodium acetate (CH 3 COONa). Calculate the p. H of this solution. n Major species: CH 3 COOH, CH 3 COO-, Na+, H 2 O Initial Change 0. 50 M -x Equilibrium 0. 50 - x 0 0. 50 M +x +x +x 0. 50 + x 4
![x ≈ 1. 8 x 10 -5 [H+] = x = 1. 8 x x ≈ 1. 8 x 10 -5 [H+] = x = 1. 8 x](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-5.jpg)
x ≈ 1. 8 x 10 -5 [H+] = x = 1. 8 x 10 -5 M ; p. H = -log[H+] = 4. 74 n Example cont’d: Calculate the change in p. H that occurs when 0. 010 mol of solid Na. OH is added to 1. 0 L of the buffered solution. Compare this p. H change with the change that occurs when 0. 010 mol of solid Na. OH is added to 1. 0 L of water. n Major species: CH 3 COOH, CH 3 COO-, Na+, OH-, H 2 O n Solve in two distinct steps 1) Assume the reaction goes to completion and do stoichiometric calculations ¨ 2) Carry out equilibrium calculations ¨ 5
![Stoichiometry problem Equilibrium problem +x x ≈ 1. 7 x 10 -5 [H+] = Stoichiometry problem Equilibrium problem +x x ≈ 1. 7 x 10 -5 [H+] =](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-6.jpg)
Stoichiometry problem Equilibrium problem +x x ≈ 1. 7 x 10 -5 [H+] = x = 1. 7 x 10 -5 M ; p. H = -log[H+] = 4. 76 6

n The change in p. H produced by the addition of 0. 01 mol of OH- to this buffered solution is then 4. 76 - 4. 74 = +0. 02 n What happens when 0. 01 mol of solid Na. OH is added to 1. 0 L of water to give 0. 01 M Na. OH, thus [OH-] = 0. 01 M p. H = 12. 00 n Thus the change in p. H is 12. 00 - 7. 00 = +5. 00 7
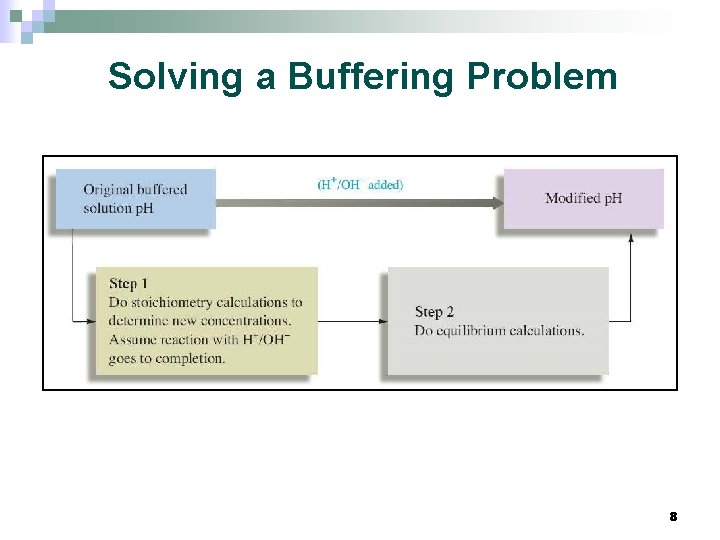
Solving a Buffering Problem 8
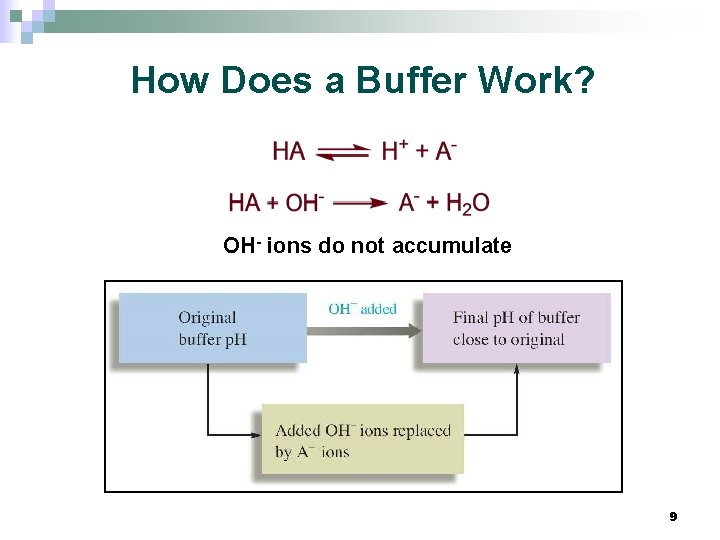
How Does a Buffer Work? OH- ions do not accumulate 9
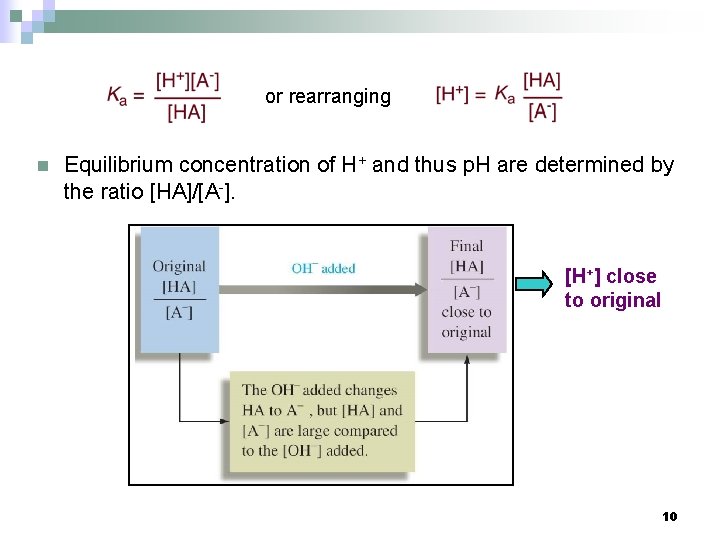
or rearranging n Equilibrium concentration of H+ and thus p. H are determined by the ratio [HA]/[A-]. [H+] close to original 10

Buffered Solutions n When an acid (proton-source) is added to a buffered solution - it will react completely with the base present (same reasoning) H+ ions do not accumulate Henderson-Hasselbach equation n Useful for determining p. H of a buffer solution when [A-]/[HA] ratio is known 11

n n Example: A buffered solution contains 0. 25 M NH 3 (Kb = 1. 8 x 10 -5) and 0. 40 M NH Cl. Calculate the p. H of this solution 4 Major species: NH 3, NH 4+, Cl-, H 2 O x ≈ 1. 1 x 10 -5 [OH-] = x = 1. 1 x 10 -5 M and p. OH = -log(1. 1 x 10 -5) = 4. 95 p. H = 14. 00 -4. 95 = 9. 05 12

n Calculate the p. H of the solution that results when 0. 10 mol of gaseous HCl is added to 1. 0 L of the buffered solution. = 9. 25 - 0. 52 p. H = 8. 73 13

Characteristics of Buffered Solutions 1. Buffered solutions contain relatively large concentrations of a weak acid and its corresponding weak base. 2. When H+ is added to a buffered solution, it reacts completely with the weak base present: H+ + A- HA or H+ + B BH+ 3. When OH- is added to a buffered solution, it reacts completely with the weak acid present: 1. OH- + HA A- + H 2 O or OH+ BH+ B + H 2 O 2. 4. The p. H of the buffered solution is determined by the ratio of the concentration of the weak base to weak acid. If this ratio remains virtually constant, the p. H will remain virtually constant. This will be the case if the concentrations of the buffering materials are large 14 compared with the amounts of H+ or OH- added.

Buffer Capacity n Buffer capacity of a buffered solution describes the amount of protons or hydroxide ions it can absorb without a significant change in p. H. n The p. H of a buffered solution is determined by the ratio [A]/[HA] whereas the capacity of a buffered solution is determined by the magnitudes of HA and A-. n Example: Calculate the change in p. H that occurs when 0. 010 mol of gaseous HCl is added to 1. 0 L of the following solutions ¨Solution A: 5. 00 M CH 3 COOH and 5. 00 M CH 3 COONa ¨Solution B: 0. 050 M CH 3 COOH and 0. 050 M CH 3 COONa For acetic acid, Ka = 1. 8 x 10 -5. 15
![n Solution A: p. H = p. Ka + log[A-]/[HA] = 4. 74 n n Solution A: p. H = p. Ka + log[A-]/[HA] = 4. 74 n](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-16.jpg)
n Solution A: p. H = p. Ka + log[A-]/[HA] = 4. 74 n Solution B: p. H = p. Ka + log[A-]/[HA] = 4. 56 16
![n Optimum buffering occurs when the ratio of [A-]/[HA] is most resistant to change n Optimum buffering occurs when the ratio of [A-]/[HA] is most resistant to change](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-17.jpg)
n Optimum buffering occurs when the ratio of [A-]/[HA] is most resistant to change when H+ or OH- ions are added ¨ when [HA] = [A-] 17

n The p. Ka of the weak acid should be as close as possible to the desired p. H. n e. g. Which weak acid-conjugate base system would you use to prepare a buffer solution of p. H 4. 30? NH 4+, p. Ka = 9. 24 ¨ CH 3 COOH, p. Ka = 4. 74 ¨ C 6 H 5 COOH, p. Ka = 4. 19 ¨ H 2 CO 3, p. Ka = 6. 36 ¨ 18

n n n Example: A chemist needs to prepare a solution buffered at p. H 4. 3 using one of the following acids (and its sodium salt): Chloroacetic acid (Ka = 5 x 10 -5) ; Propanoic acid (Ka = 1. 3 x 10 -5) Benzoic acid (Ka = 6. 4 x 10 -5) ; Hypochlorous acid (Ka = 3. 5 x 10 -8) Calculate the ratio of [HA]/[A- ] required for each system to yield a solution with a p. H of 4. 3. Which system will work best? Solution: A p. H of 4. 3 corresponds to [H+ ] = Ka [HA]/[A- ] Acid [H+ ] = Ka [HA]/[A- ] Chloroacetic acid 5 x 10 -5 = 1. 35 x 10 -3 [HA]/[A- ] 3. 7 x 10 -2 Propanoic acid 5 x 10 -5 = 1. 3 x 10 -5 [HA]/[A- ] 3. 8 Benzoic acid 5 x 10 -5 = 6. 4 x 10 -5 [HA]/[A- ] 0. 78 Hypochlorous acid 5 x 10 -5 = 3. 5 x 10 -8 [HA]/[A- ] 1. 4 x 10 -3 n Since [HA]/[A- ] for benzoic acid is closest to 1, the system of benzoic acid and its sodium salt is the best choice among those given for buffering a solution at p. H 4. 3. n 19

Titration n Titration: used to analyze amount of acid or base in a solution n Process involves delivering a solution of known concentration (the titrant) from a buret into an unknown solution until the substance being analyzed is just consumed n The stoichiometric or equivalence point is signalled by the color change of an indicator. n Titration curve (p. H curve) is a plot of the progress of an acidbase titration showing the p. H of the solution being analyzed as a function of the amount of titrant added 20

Strong Acid - Strong Base Titrations n Millimole (abbreviated mmol) = 10 -3 mol n To compute [H+] at a given point in the titration, we must determine the moles of H+ remaining at that point and divide by the total volume of the solution. 21

n Example: Consider the titration of 50. 0 m. L of 0. 200 M HNO 3 with 0. 100 M Na. OH. n We will calculate the p. H of the solution at selected points during the titration where specific volumes of 0. 100 M Na. OH have been added. n A. No Na. OH has been added ¨ p. H is determined by the H+ from the nitric acid. p. H = -log(0. 200) = 0. 699 n B. 10. 0 m. L of 0. 100 M Na. OH has been added ¨ 1. 0 mmol of added Na. OH will react with 1. 0 mmol of H+ to form water. The p. H will be determined by the remaining H+. 9. 0 mmol H+ left over; total volume = 60. 0 m. L; [H+] = 0. 15 M p. H = -log(0. 15) = 0. 82 22

n C. 20. 0 m. L of 0. 100 M Na. OH has been added ¨ 2. 0 mmol of added Na. OH will react with 2. 0 mmol of H+ to form water. The p. H will be determined by the remaining H+. p. H = -log(0. 11) = 0. 96 n D. 50. 0 m. L of 0. 100 M Na. OH has been added p. H = 1. 30 n E. 100. 0 m. L of 0. 100 M Na. OH has been added 10. 0 mmol of Na. OH has been added; original amount of nitric acid is 10. 0 mmol ¨ This is the stoichiometric point or equivalence point of the titration. ¨ ¨ Solution is neutral, p. H = 7. 00 23
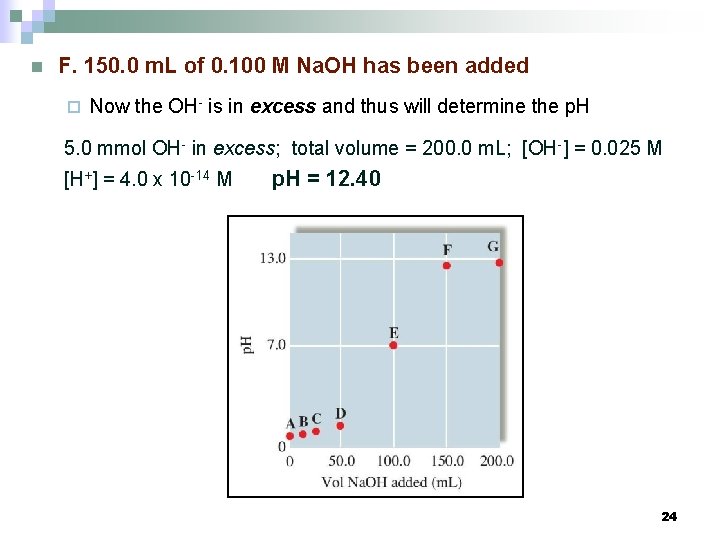
n F. 150. 0 m. L of 0. 100 M Na. OH has been added ¨ Now the OH- is in excess and thus will determine the p. H 5. 0 mmol OH- in excess; total volume = 200. 0 m. L; [OH-] = 0. 025 M [H+] = 4. 0 x 10 -14 M p. H = 12. 40 24
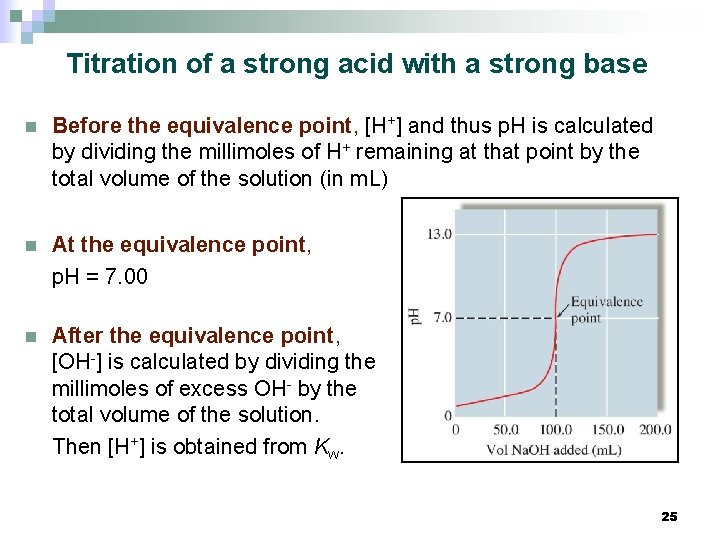
Titration of a strong acid with a strong base n Before the equivalence point, [H+] and thus p. H is calculated by dividing the millimoles of H+ remaining at that point by the total volume of the solution (in m. L) n At the equivalence point, p. H = 7. 00 n After the equivalence point, [OH-] is calculated by dividing the millimoles of excess OH- by the total volume of the solution. Then [H+] is obtained from Kw. 25
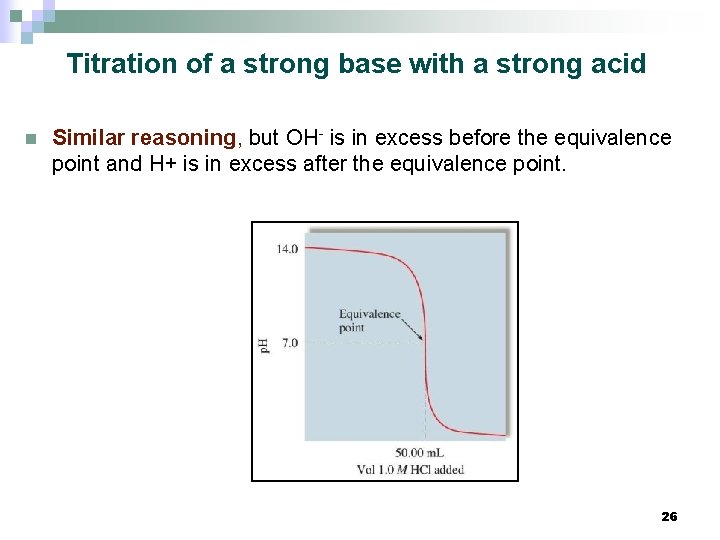
Titration of a strong base with a strong acid n Similar reasoning, but OH- is in excess before the equivalence point and H+ is in excess after the equivalence point. 26
![Weak Acid - Strong Base Titrations n n n To calculate [H+] after a Weak Acid - Strong Base Titrations n n n To calculate [H+] after a](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-27.jpg)
Weak Acid - Strong Base Titrations n n n To calculate [H+] after a certain amount of base has been added, we must deal with the weak acid equilibrium Weak acid reacts essentially to completion with OH- ion. Calculating the p. H curve for a weak acid-strong base titration involves a two-step procedure. 27

n Example: Consider the titration of 50. 0 m. L of 0. 10 M acetic acid (CH 3 COOH, Ka = 1. 8 x 10 -5) with 0. 10 M Na. OH. n We will calculate the p. H of the solution at selected points during the titration where specific volumes of 0. 100 M Na. OH have been added. n A. No Na. OH has been added ¨ This is a typical weak acid equilbrium calculation p. H = 2. 87 n B. 10. 0 m. L of 0. 10 M Na. OH has been added 28
![+x x ≈ 7. 2 x 10 -5 [H+] = x = 7. 2 +x x ≈ 7. 2 x 10 -5 [H+] = x = 7. 2](http://slidetodoc.com/presentation_image_h/1948386416540bda0586c513b731ff44/image-29.jpg)
+x x ≈ 7. 2 x 10 -5 [H+] = x = 7. 2 x 10 -5 M ; p. H = -log[H+] = 4. 14 n C. 25. 0 m. L of 0. 10 M Na. OH has been added 29

n At this point, half of the acid has been used up - halfway to the equivalence point. n Equilibrium concentrations: [CH 3 COOH] = [CH 3 COOH]0 - x ≈ [CH 3 COOH]0 [CH 3 COONa] = [CH 3 COONa]0 + x ≈ [CH 3 COONa]0 p. H = 4. 74 n Thus, at the halfway point in the titration [H+] = Ka n and p. H = p. Ka. D. 40. 0 m. L of 0. 10 M Na. OH has been added p. H = 5. 35 30

n E. 50. 0 m. L of 0. 10 M Na. OH has been added n 5. 0 mmol of Na. OH has been added; original amount of acetic acid is 5. 0 mmol - this is the stoichiometric point or equivalence point n Major species: Na+, CH 3 COO-, H 2 O x = 5. 3 x 10 -6 x = [OH-] = 5. 3 x 10 -6 M ; [H+] = Kw/[OH-] = 1. 9 x 10 -9 M ; p. H = 8. 72 31
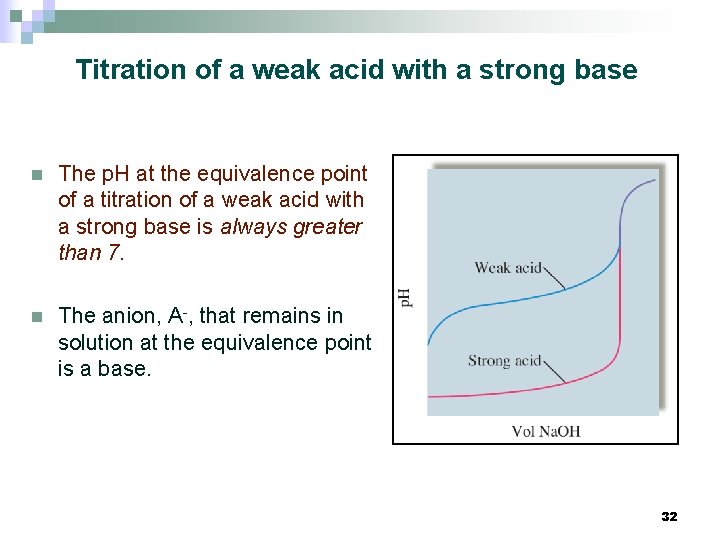
Titration of a weak acid with a strong base n The p. H at the equivalence point of a titration of a weak acid with a strong base is always greater than 7. n The anion, A-, that remains in solution at the equivalence point is a base. 32
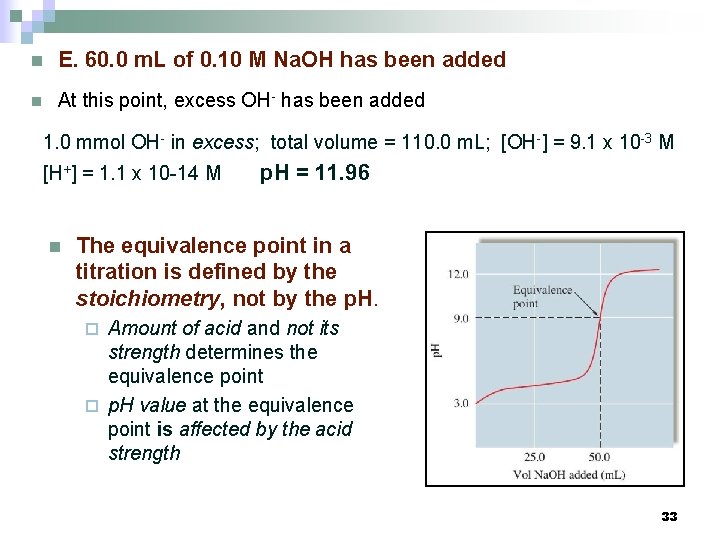
n E. 60. 0 m. L of 0. 10 M Na. OH has been added n At this point, excess OH- has been added 1. 0 mmol OH- in excess; total volume = 110. 0 m. L; [OH-] = 9. 1 x 10 -3 M [H+] = 1. 1 x 10 -14 M n p. H = 11. 96 The equivalence point in a titration is defined by the stoichiometry, not by the p. H. Amount of acid and not its strength determines the equivalence point ¨ p. H value at the equivalence point is affected by the acid strength ¨ 33
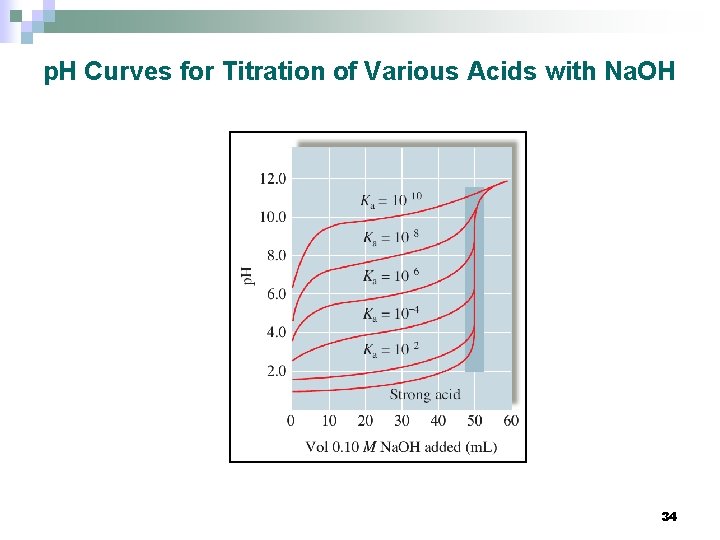
p. H Curves for Titration of Various Acids with Na. OH 34

Example n The Ka of a monoprotic weak acid can be determined using titration. A chemist dissolves 2. 00 mmol of the solid acid in 100. 0 m. L of water and titrates the resulting solution with 0. 0500 M Na. OH. After 20. 00 m. L of Na. OH has been added, the p. H is 6. 00. What is the Ka for the acid? 35

Weak Base - Strong Acid Titrations n Example: Titration of 100. 0 m. L of 0. 050 M NH 3 with 0. 10 M HCl. n Before the addition of HCl n Before the equivalence point Stoichiometry problem Equilibrium problem or (Kb) (Ka) 36
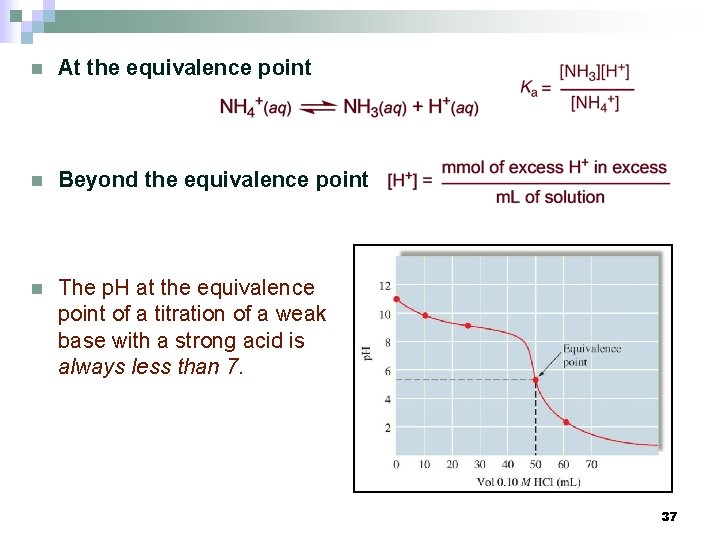
n At the equivalence point n Beyond the equivalence point n The p. H at the equivalence point of a titration of a weak base with a strong acid is always less than 7. 37

38

Acid-Base Indicators n Two main methods for determining the equivalence point of an acid-base titration A p. H meter: is used to monitor the p. H and then plot a titration curve ¨ An acid-base indicator: marks the endpoint of a titration by changing color. ¨ 39

n Acid-base indicators are organic molecules that are themselves weak acids, represented by Hin. ¨ Hin and In- have different colors. ¨ E. g. Phenolphthalein is colorless in its acidic Hin form and pink in its deprotonated In- (basic) form. Rearranging Ka, Color change (from Hin to In-) is apparent at a p. H where 40

Titration of Polyprotic Acids n n n In a polyprotic acid, the acidic protons are titrated in succession. Example: H 3 PO 4 titrated with Na. OH First equivalence point: major species are Na+, H 2 PO 4 -, H 2 O n Second equivalence point: major species are Na+, HPO 42 -, H 2 O n Third equivalence point: major species are Na+, PO 43 -, H 2 O 41

Summary for a Triprotic Acid, H 3 A 42
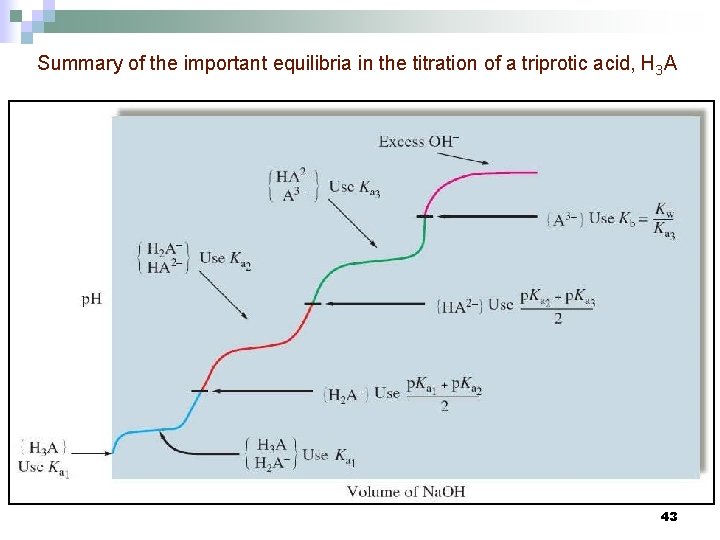
Summary of the important equilibria in the titration of a triprotic acid, H 3 A 43
- Slides: 43