Chapter 8 An Introduction To Metabolism Metabolism u

Chapter 8 An Introduction To Metabolism

Metabolism u. The totality of an organism’s chemical processes. u. Concerned with managing the material and energy resources of the cell.

Catabolic Pathways u. Pathways that break down complex molecules into smaller ones, releasing energy. u. Example: Respiration

Anabolic Pathways u. Pathways that consume energy, building complex molecules from smaller ones. u. Example: Photosynthesis

Energy u. Ability to do work. u. The ability to rearrange a collection of matter. u. Forms of energy: u. Kinetic u. Potential u. Activation


Kinetic Energy u. Energy of action or motion.

Potential Energy u. Stored energy or the capacity to do work.

Activation Energy u. Energy needed to convert potential energy into kinetic energy. Activation Energy Potential Energy

Energy Transformation u. Governed by the Laws of Thermodynamics.

1 st Law of Thermodynamics u. Energy can be transferred and transformed, but it cannot be created or destroyed. u. Also known as the law of “Conservation of Energy”

2 nd Law of Thermodynamics u. Each energy transfer or transformation increases the entropy of the universe.

Entropy u. Measure of disorder.

Summary u. The quantity of energy in the universe is constant, but its quality is not.

Question? u. How does Life go against Entropy? u. By using energy from the environment or external sources (e. g. food, light).

Free Energy u. The portion of a system's energy that can perform work.

Free Energy G = H - TS G = free energy of a system H = total energy of a system T = temperature in o. K S = entropy of a system

Free Energy of a System u. If the system has: umore free energy uit is less stable u. It has greater work capacity

Spontaneous Process u. If the system is unstable, it has a greater tendency to change spontaneously to a more stable state. u. This change provides free energy for work.

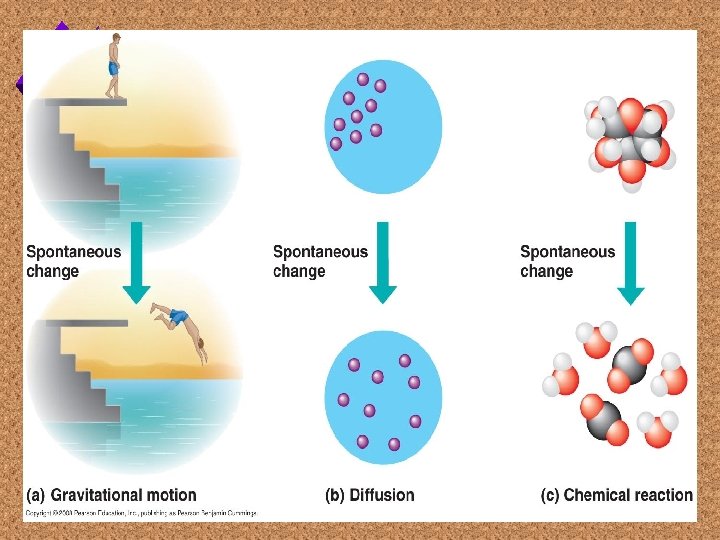
Free Energy Changes

Chemical Reactions u. Are the source of energy for living systems. u. Are based on free energy changes.

Reaction Types u. Exergonic: chemical reactions with a net release of free energy. u. Endergonic: chemical reactions that absorb free energy from the surroundings.
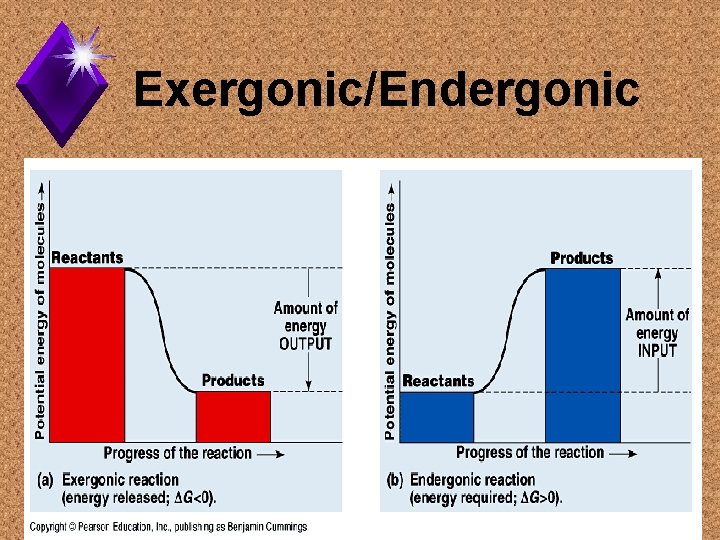
Exergonic/Endergonic

Biological Examples u. Exergonic - respiration u. Endergonic - photosynthesis

Cell - Types of Work u. Mechanical - muscle contractions u. Transport - pumping across membranes u. Chemical - making polymers

Cell Energy u. Couples an exergonic process to drive an endergonic one. u. ATP is used to couple the reactions together.


ATP u. Adenosine u. Made Triphosphate of: - Adenine (nitrogenous base) - Ribose (pentose sugar) - 3 phosphate groups

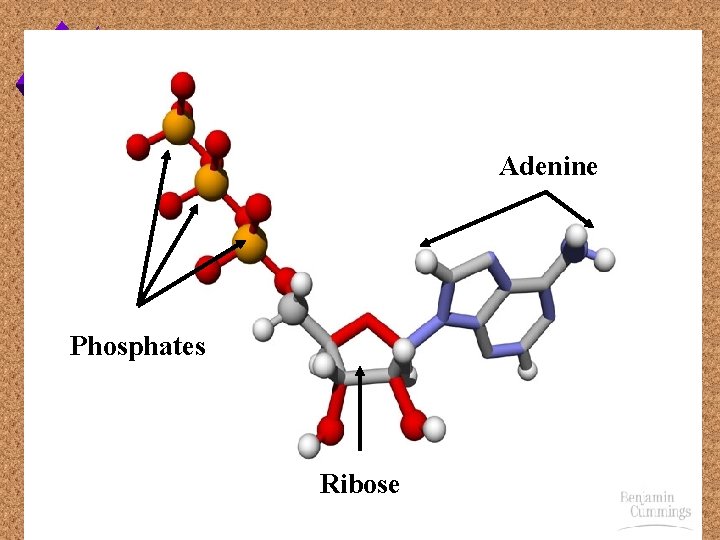
Adenine Phosphates Ribose

Key to ATP u. Is in the three phosphate groups. u. Negative charges repel each other and makes the phosphates unstable.

ATP u. Works by energizing other molecules by transferring phosphate groups.


ATP vs Food u. ATP: u. Renewable energy resource. u. Unstable bonds u. Food: u. Long term energy storage u Stable bonds

ATP Cycles u. Energy released from ATP drives anabolic reactions. u. Energy from catabolic reactions “recharges” ATP.
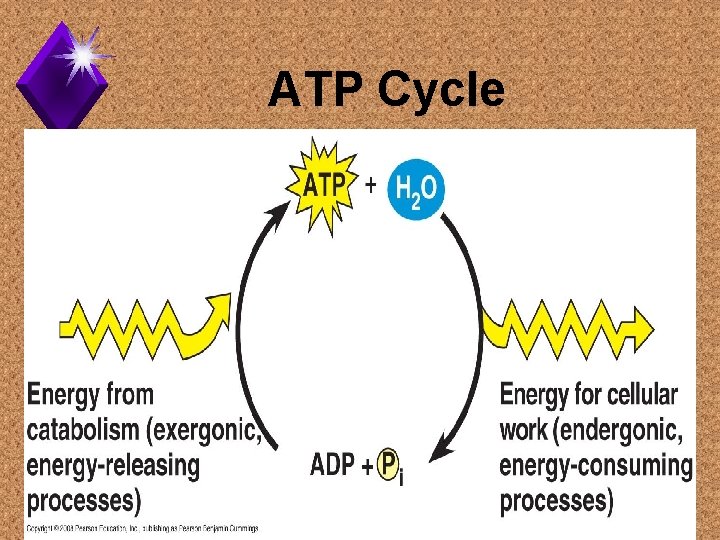
ATP Cycle

ATP in Cells u. A cell's ATP content is recycled every minute. u. Humans use close to their body weight in ATP daily. u. No ATP production equals quick death.

Enzymes u. Biological catalysts made of protein. u. Cause the rate of a chemical reaction to increase.
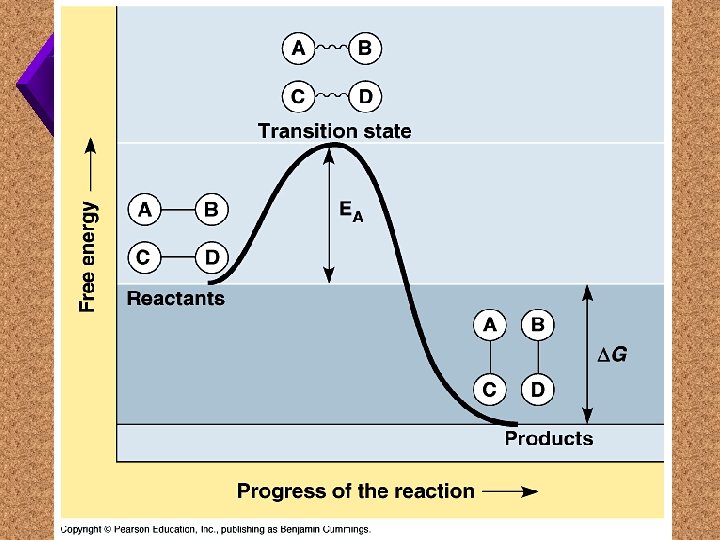

Enzymes u. Lower the activation energy for a chemical reaction to take place.


Enzyme Terms u. Substrate - the material the enzyme works on. u. Enzyme names: Ex. Sucrase - ase name of an enzyme 1 st part tells what the substrate is. (Sucrose)

Enzyme Name u. Some older known enzymes don't fit this naming pattern. u. Examples: pepsin, trypsin

Active Site u. The area of an enzyme that binds to the substrate. u. Structure is designed to fit the molecular shape of the substrate. u. Therefore, each enzyme is substrate specific.



Homework u. Read chapter 8, 41 u. Exam 2 – this week, no broadcast on Thursday u. Chapter 8 – Fri. 10/10 u. Chapter 41 – Mon. 10/20

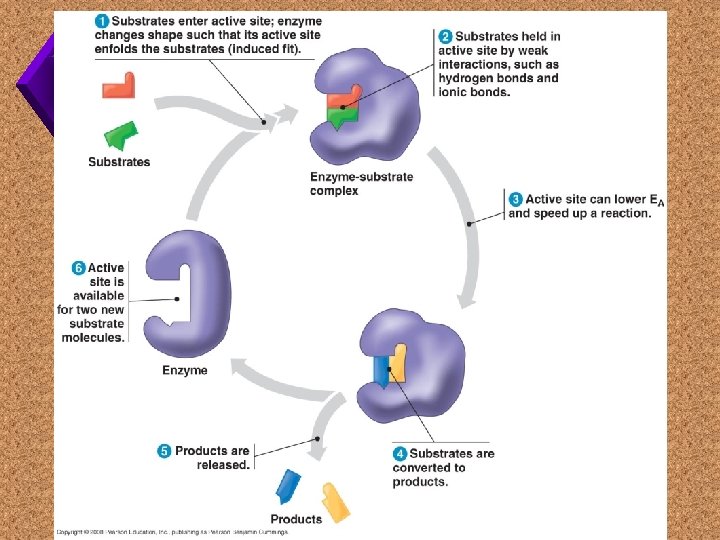
Models of How Enzymes Work 1. Lock and Key model 2. Induced Fit model

Lock and Key Model u. Substrate (key) fits to the active site (lock) which provides a microenvironment for the specific reaction.

Induced Fit Model u. Substrate “almost” fits into the active site, causing a strain on the chemical bonds, allowing the reaction.
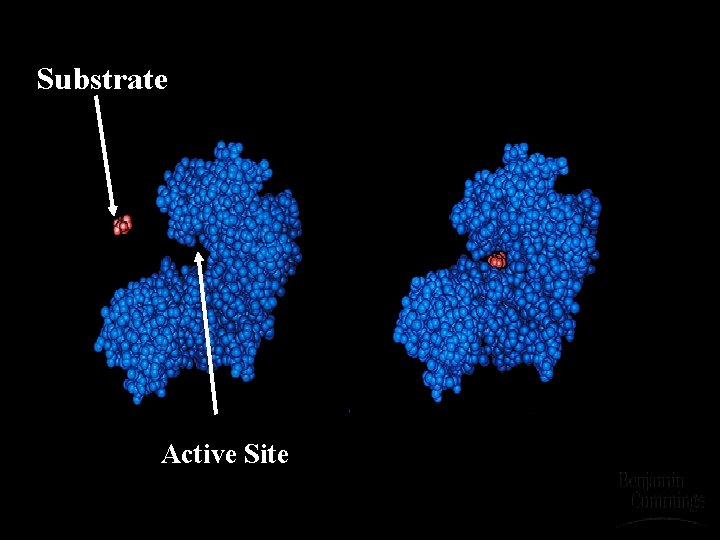
Substrate Active Site

Enzymes u. Usually specific to one substrate. u. Each chemical reaction in a cell requires its own enzyme.

Factors that Affect Enzymes u. Environment u. Cofactors u. Coenzymes u. Inhibitors u. Allosteric Sites

Environment u. Factors that change protein structure will affect an enzyme. u. Examples: up. H shifts utemperature usalt concentrations

u. Cofactors: non-organic helpers required for the proper function of enzymes. u. Nonprotein ions or molecules u. Ex. Fe, Zn, Cu u. NADH

u. Coenzymes: organic helpers to enzymes. Ex. vitamins

Enzyme Inhibitors u. Competitive - mimic the substrate and bind to the active site. u. Noncompetitive - bind to some other part of the enzyme.


Allosteric Regulation u. The control of an enzyme complex by the binding of a regulatory molecule. u. Regulatory molecule may stimulate or inhibit the enzyme complex.
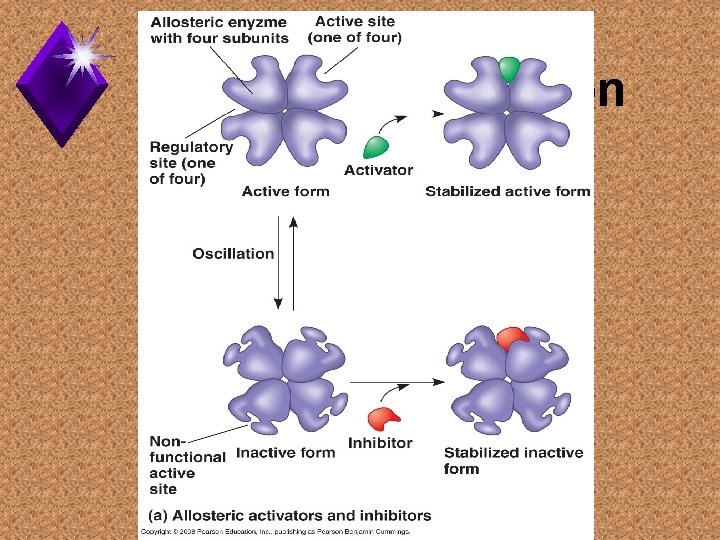
Allosteric Regulation

Control of Metabolism u. Is necessary if life is to function. u. Controlled by switching enzyme activity "off" or "on” or separating the enzymes in time or space.

Types of Control u. Feedback Inhibition u. Structural Order

Feedback Inhibition u. When a metabolic pathway is switched off by its endproduct. u. End-product usually inhibits an enzyme earlier in the pathway.


Structural Order u. Separation of enzymes and metabolic pathways in time or space by the cell's organization. u. Example: enzymes of respiration


Summary u. Recognize that Life must follow the Laws of Thermodynamics. u. The role of ATP in cell energy. u. How enzymes work.
- Slides: 70