Chapter 8 An Introduction To Metabolism Metabolism Catabolic

Chapter 8 An Introduction To Metabolism

Metabolism

Catabolic Pathways

Anabolic Pathways

Energy

Kinetic Energy

Potential Energy
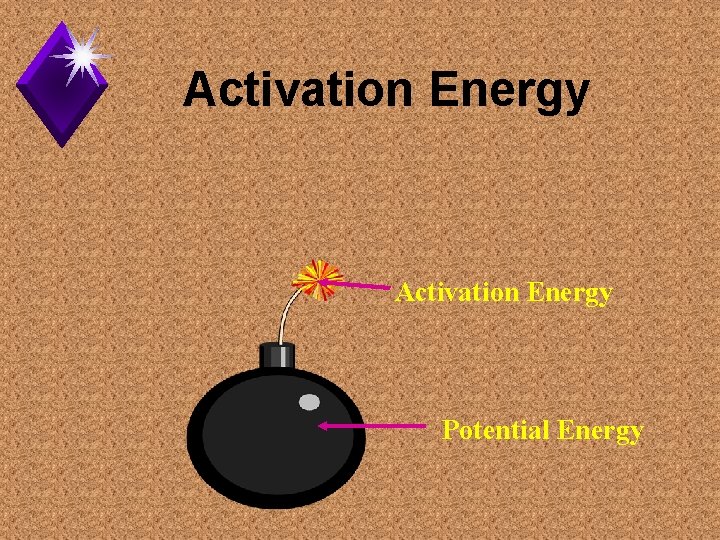
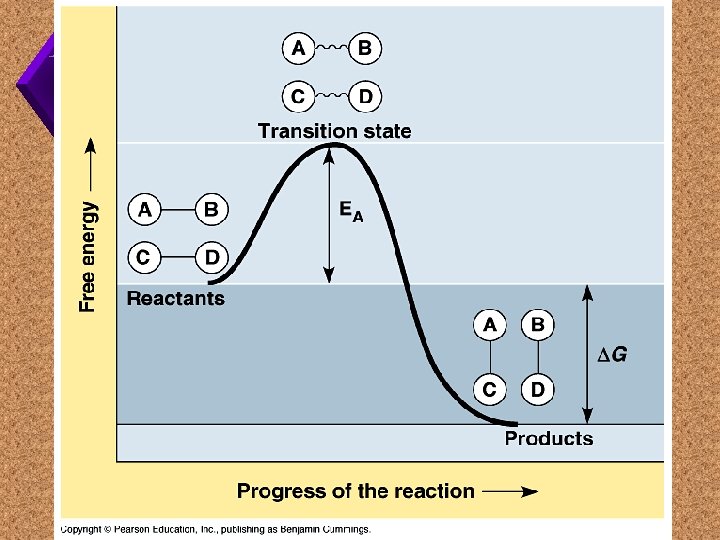
Activation Energy Potential Energy

Energy Transformation u.

1 st Law of Thermodynamics

2 nd Law of Thermodynamics

Entropy

Summary u. The quantity of energy in the universe is constant, but its quality is not.

Free Energy

Free Energy G = H - TS G = free energy of a system H = total energy of a system T = temperature in o. K S = entropy of a system

Free Energy of a System
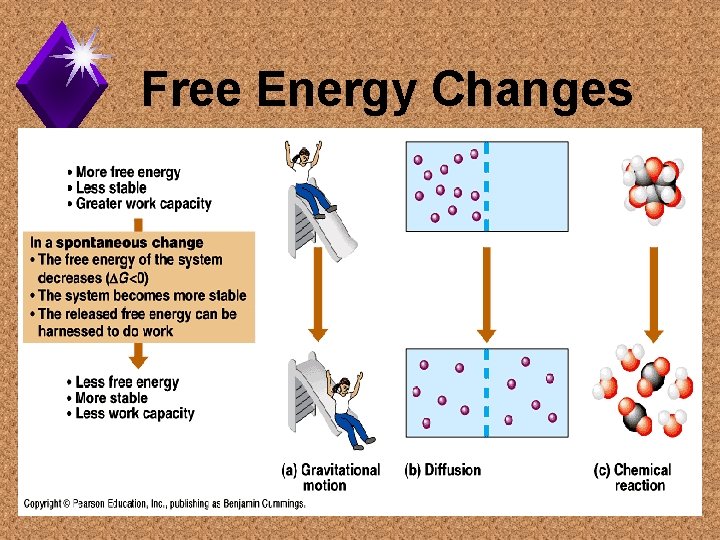
Free Energy Changes

Spontaneous Process

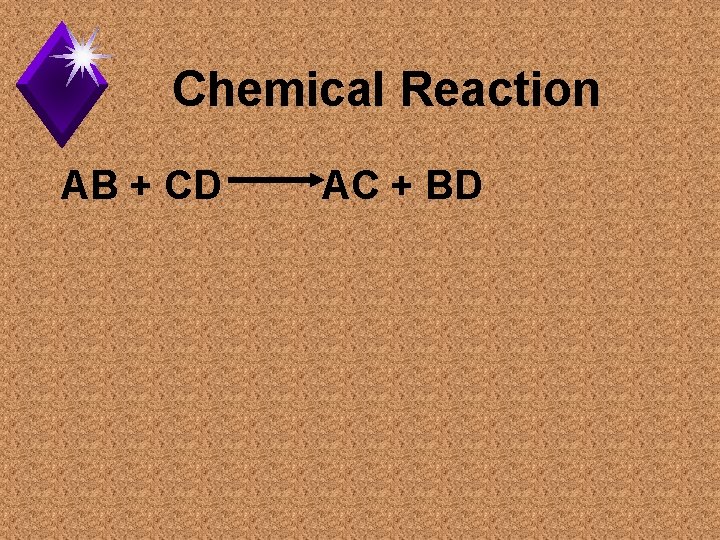
Chemical Reactions

Reaction Types u. Exergonic: u. Endergonic:
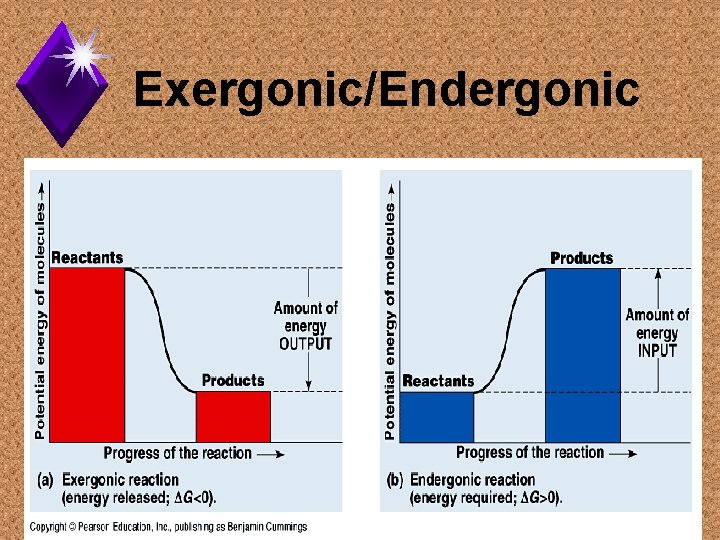
Exergonic/Endergonic

Biological Examples u. Exergonic – u. Endergonic -

Cell - Types of Work

Energy Coupling

ATP
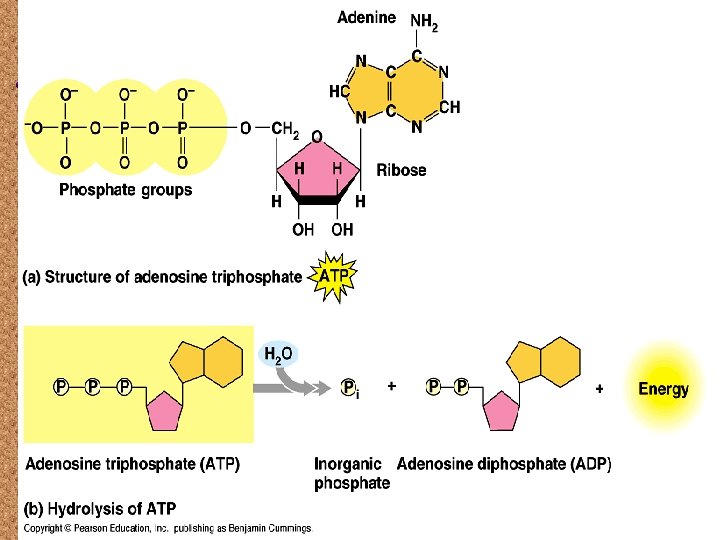
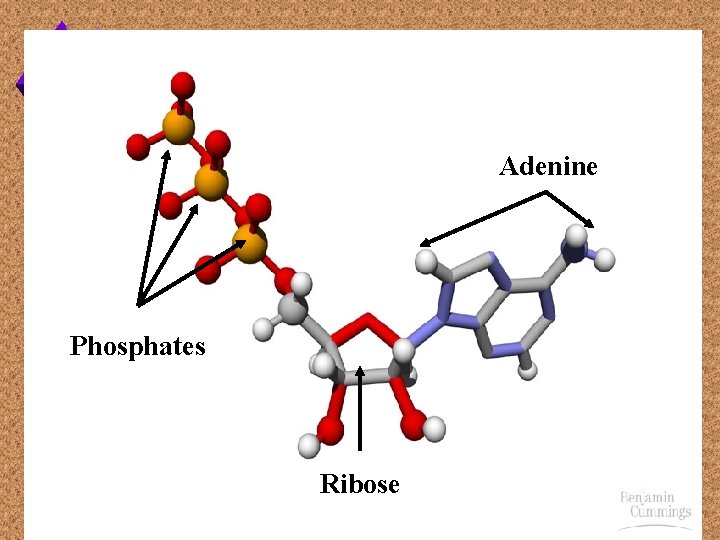
Adenine Phosphates Ribose

Key to ATP

ATP
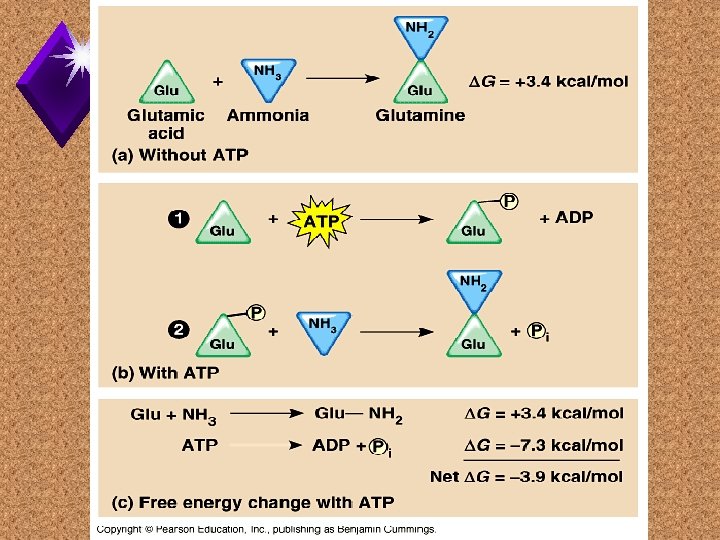

ATP vs Food u. ATP: u. Food:

ATP Cycles
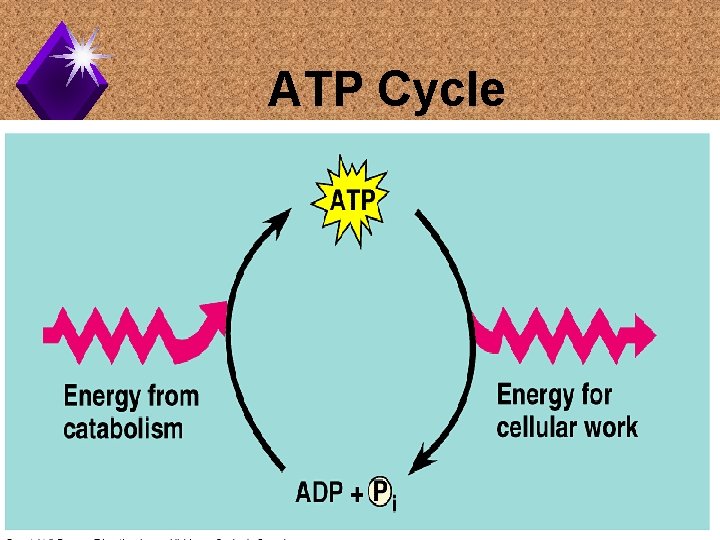
ATP Cycle

ATP in Cells u. A cell's ATP content is recycled every minute. u. Humans use close to their body weight in ATP daily. u. No ATP production equals quick death.

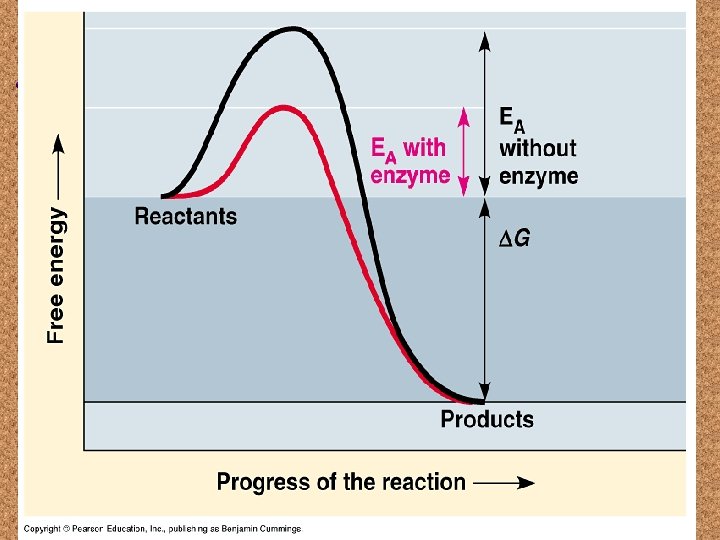
Enzymes
Chemical Reaction AB + CD AC + BD

Enzymes

Enzyme Terms u. Substrate - ase –

Enzyme Name

Active Site
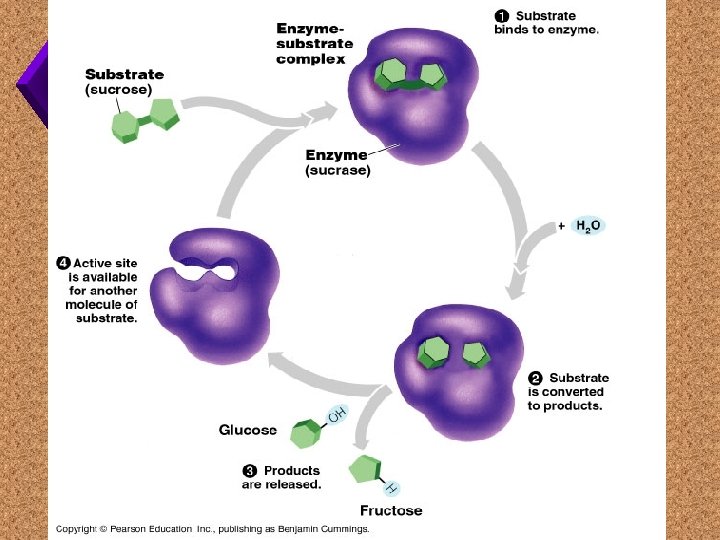

Models of How Enzymes Work 1. 2.

Lock and Key Model

Induced Fit Model
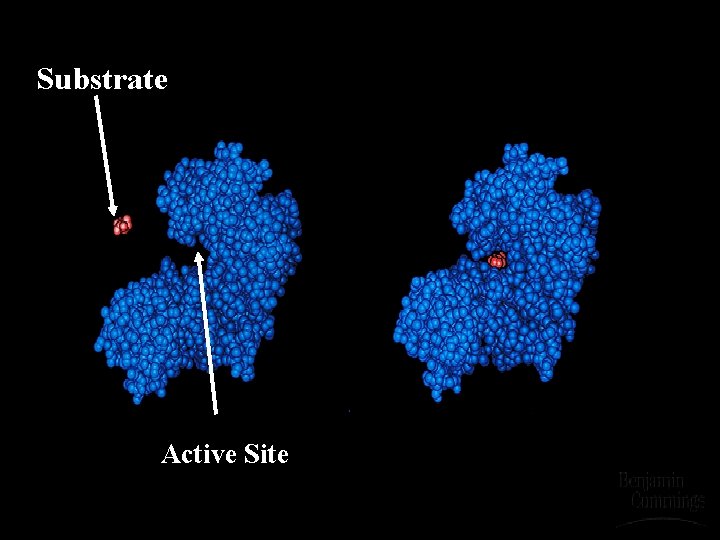
Substrate Active Site

Enzymes

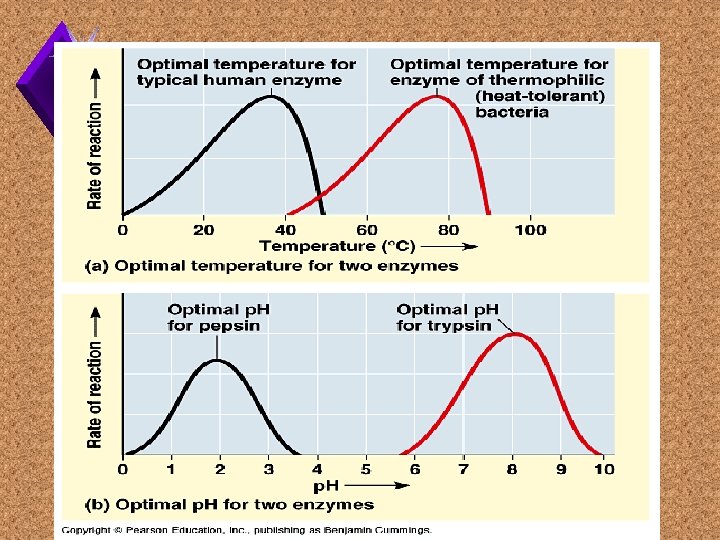
Factors that Affect Enzymes
Environment

Cofactors

Coenzymes

Enzyme Inhibitors u. Competitive u Noncompetitive -
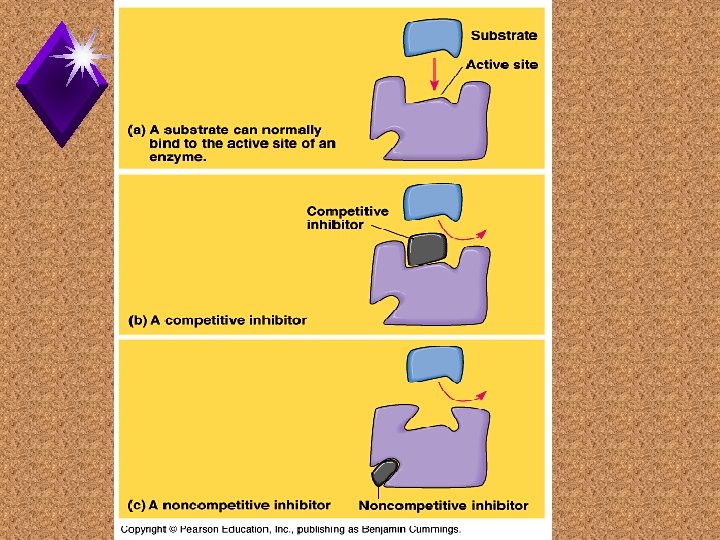

Allosteric Regulation
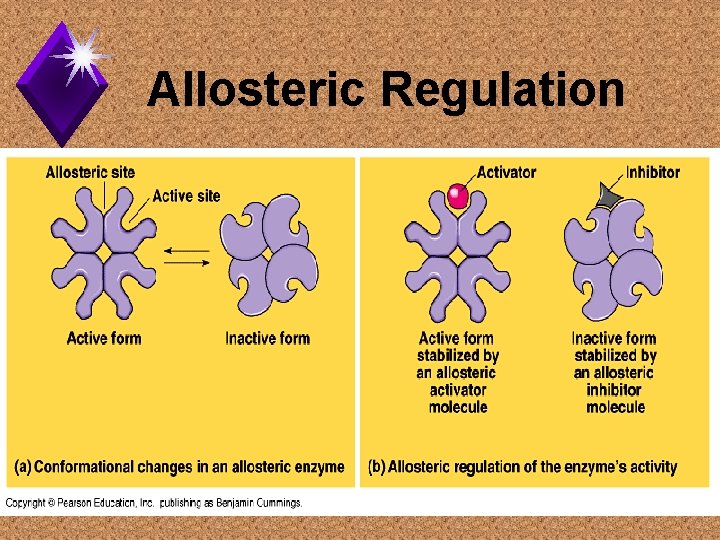
Allosteric Regulation

Control of Metabolism

Types of Control

Feedback Inhibition u.
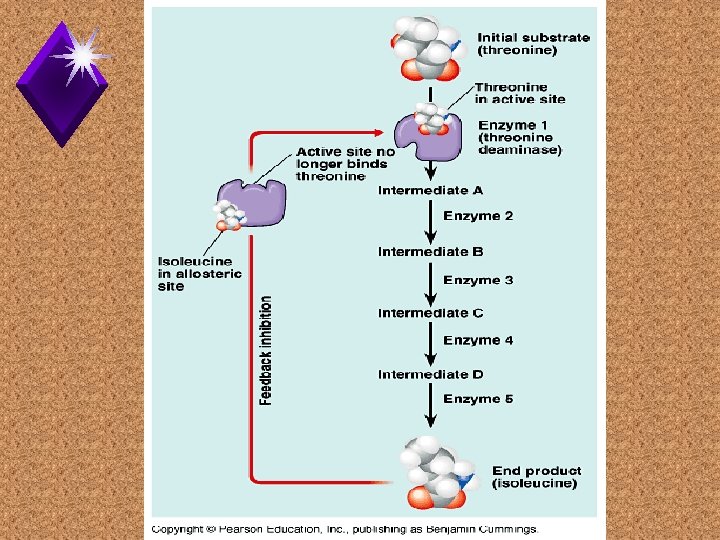

Structural Order

Summary u. Recognize that Life must follow the Laws of Thermodynamics. u. The role of ATP in cell energy. u. How enzymes work.
- Slides: 65