Chapter 8 AcidBase Equilibria 8 1 BrnstedLowry Acids

Chapter 8 Acid-Base Equilibria • 8 -1 Brønsted-Lowry Acids and Bases • 8 -2 Water and the p. H Scale • 8 -3 The Strengths of Acids and Bases • 8 -4 Equilibria Involving Weak Acids and Bases • 8 -5 Buffer Solutions • 8 -6 Acid-Base Titration Curves • 8 -7 Polyprotic Acids • 8 -8 Lewis Acids and Bases 9/5/2021 OFB Chapter 8 1

Acid and Base Definitions 1) Arrhenius (Section 4. 3) • • Acids are H+ donors Bases are OH- donors 2) Broadened Definition (Section 4. 3) • • Acids are substances that increase [H+] Bases are substances that increase [OH-] 3) Brønsted-Lowry (Section 8. 1) • • 4) Lewis (Section 8. 8) • • 9/5/2021 OFB Chapter 8 2

Chapter 8 Acid-Base Equilibria • Brønsted-Lowry – Acids – Bases – Conjugate Base – Conjugate Acid 9/5/2021 OFB Chapter 8 3

Acid-Base Equilibria Brønsted-Lowry Acids and Bases A Brønsted-Lowry acid is a substance that can donate a hydrogen ion. A Brønsted-Lowry base is a substance that can accept a hydrogen ion. In the Brønsted-Lowry Acid and Base concept, acids and bases occur as conjugate acid-base pairs. 9/5/2021 OFB Chapter 8 4

Conjugate Base - subtract an H+ from the acid Conjugate Acid add H+ to the base Examples 1. 2. is the conjugate base of H 2 O is the conjugated base of H 3 O+ (called the hydronium ion) 3. 4. is the conjugated acid of OH(or shown as H+) is the conjugate acid of H 2 O 9/5/2021 OFB Chapter 8 5
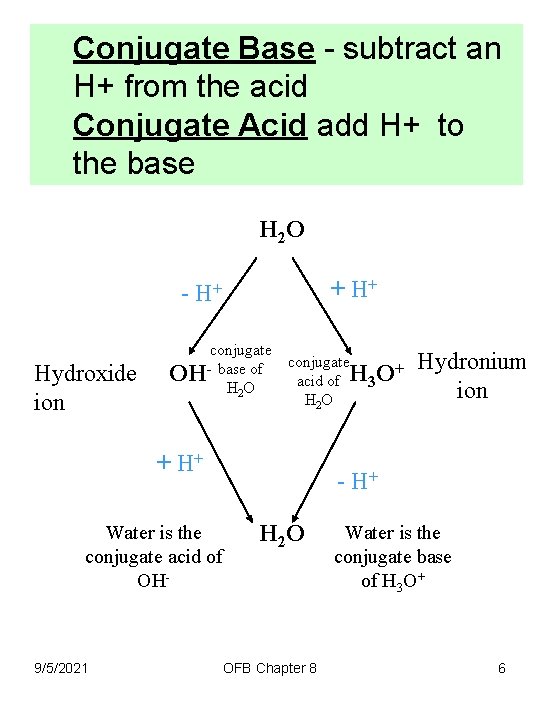
Conjugate Base - subtract an H+ from the acid Conjugate Acid add H+ to the base H 2 O + H+ - H+ Hydroxide ion conjugate base of OH H 3 O + acid of H 2 O + H+ Water is the conjugate acid of OH- 9/5/2021 Hydronium ion - H+ H 2 O OFB Chapter 8 Water is the conjugate base of H 3 O+ 6
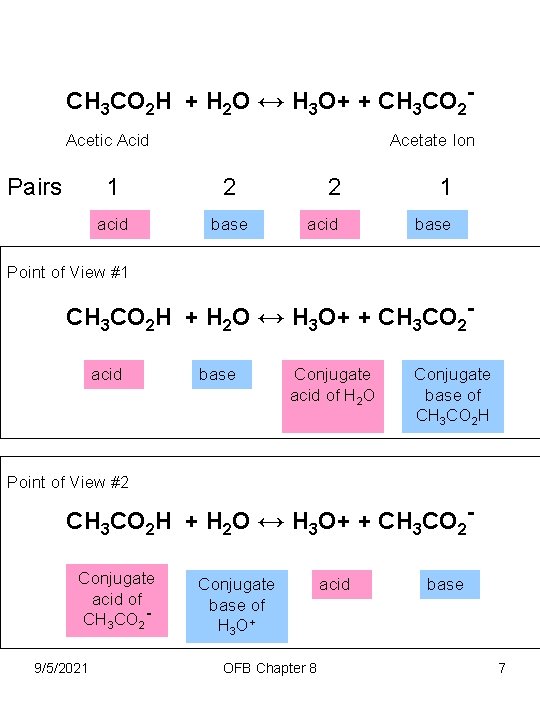
CH 3 CO 2 H + H 2 O ↔ H 3 O+ + CH 3 CO 2 Acetic Acid Pairs Acetate Ion 1 2 acid base 2 1 acid base Point of View #1 CH 3 CO 2 H + H 2 O ↔ H 3 O+ + CH 3 CO 2 acid base Conjugate acid of H 2 O Conjugate base of CH 3 CO 2 H Point of View #2 CH 3 CO 2 H + H 2 O ↔ H 3 O+ + CH 3 CO 2 Conjugate acid of CH 3 CO 29/5/2021 Conjugate base of H 3 O + OFB Chapter 8 acid base 7
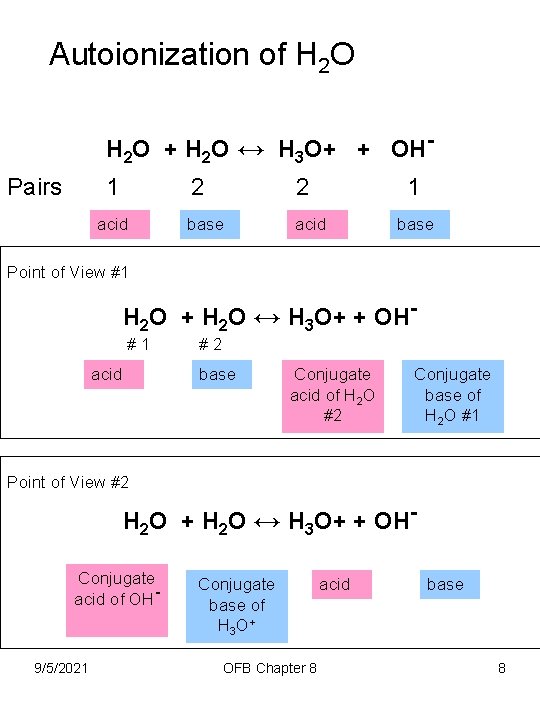
Autoionization of H 2 O + H 2 O ↔ H 3 O+ + OHPairs 1 acid 2 2 1 base acid base Point of View #1 H 2 O + H 2 O ↔ H 3 O+ + OH#1 acid #2 base Conjugate acid of H 2 O #2 Conjugate base of H 2 O #1 Point of View #2 H 2 O + H 2 O ↔ H 3 O+ + OHConjugate acid of OH- 9/5/2021 Conjugate base of H 3 O + OFB Chapter 8 acid base 8

Exercise 8 -1: Trimethylamine (C 3 H 9 N) is a soluble weak base with a foul odor (it contributes to the smell of rotten fish). Write the formula of its conjugate acid. “… the formula of a conjugate acid is obtained by adding H+ to the formula of the base. ” 9/5/2021 OFB Chapter 8 9
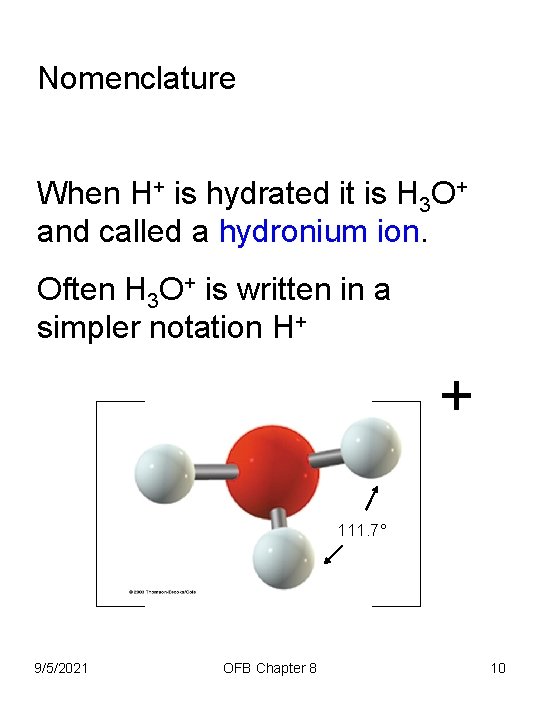
Nomenclature When H+ is hydrated it is H 3 O+ and called a hydronium ion. Often H 3 O+ is written in a simpler notation H+ + 111. 7° 9/5/2021 OFB Chapter 8 10
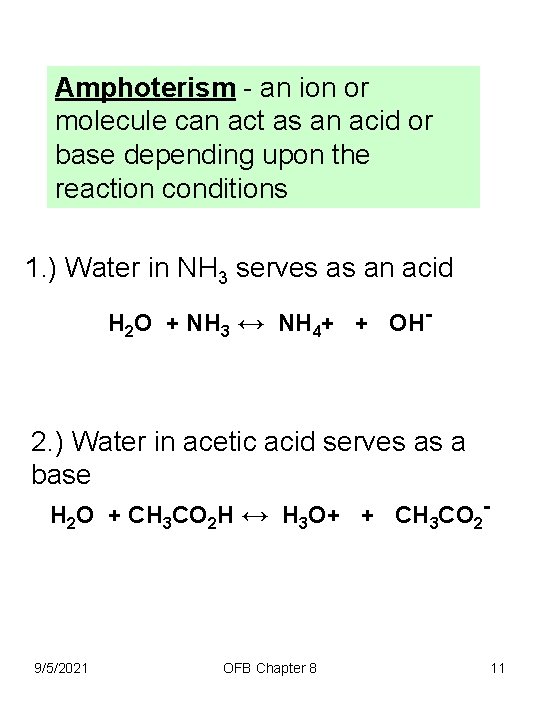
Amphoterism - an ion or molecule can act as an acid or base depending upon the reaction conditions 1. ) Water in NH 3 serves as an acid H 2 O + NH 3 ↔ NH 4+ + OH- 2. ) Water in acetic acid serves as a base H 2 O + CH 3 CO 2 H ↔ H 3 O+ + CH 3 CO 2 - 9/5/2021 OFB Chapter 8 11

3. ) Acetic Acid is also amphoteric, if in the presence of a strong acid serves as a base H 2 SO 4 + CH 3 CO 2 H ↔ CH 3 CO 2 H 2+ + HSO 4 - Amphoterism - an ion or molecule can act as an acid or base depending upon the reaction conditions 9/5/2021 OFB Chapter 8 12
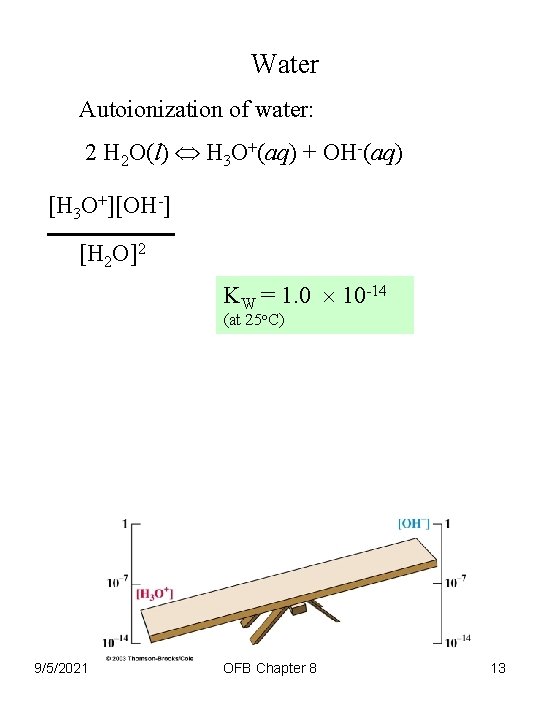
Water Autoionization of water: 2 H 2 O(l) H 3 O+(aq) + OH-(aq) [H 3 O+][OH-] [H 2 O]2 KW = 1. 0 10 -14 (at 25 o. C) 9/5/2021 OFB Chapter 8 13

Strong Acids and Bases A strong acid is one that reacts essentially completely with water to produce H 3 O+(aq). Hydrochloric acid (HCl) is a strong acid: HCl (aq) + H 2 O(l) H 3 O+(aq) + Cl-(aq) (reaction essentially complete) Dissolving 0. 10 mol of HCl in enough water to make 1. 0 L of solution gives a final concentration of 0. 10 M for H 3 O+(aq). [H 3 O+][OH-] = KW 9/5/2021 OFB Chapter 8 14

A strong base is one that reacts Strong Acids and Bases essentially completely with water to produce OH-(aq) ions. Sodium hydroxide (Na. OH) is a strong base: Others are NH 2 - (amide ion) and H- (Hydride ion) Na. OH(s) Na+(aq) + OH-(aq) (reaction essentially complete) Dissolving 0. 10 mol of Na. OH in enough water to make 1. 0 L of solution gives a final concentration of 0. 10 M for OH-(aq). [H 3 O+][OH-] = KW 9/5/2021 OFB Chapter 8 15
![The p. H Function p. H = -log 10[H 3 O+] p. H < The p. H Function p. H = -log 10[H 3 O+] p. H <](http://slidetodoc.com/presentation_image_h2/8a9eae5d0101853dc8bf552dee593268/image-16.jpg)
The p. H Function p. H = -log 10[H 3 O+] p. H < 7 acidic solution [H 3 O+] > [OH-] p. H = 7 neutral solution [H 3 O+] = [OH-] p. H > 7 basic solution [H 3 O+] < [OH-] 9/5/2021 OFB Chapter 8 16

The p. H Function EXAMPLE 8 -3 Calculate the p. H (at 25 o. C) of an aqueous solution that has an OH-(aq) concentration of 1. 2 10 -6 M. 9/5/2021 OFB Chapter 8 17
![p. H = -log 10[H 3 O+] If converting from p. H to [H+] p. H = -log 10[H 3 O+] If converting from p. H to [H+]](http://slidetodoc.com/presentation_image_h2/8a9eae5d0101853dc8bf552dee593268/image-18.jpg)
p. H = -log 10[H 3 O+] If converting from p. H to [H+] = 10 -p. H = 10 -X 9/5/2021 OFB Chapter 8 18
![Recall [H 3 O+][OH-] = KW = 10 -14 p. H + p. OH Recall [H 3 O+][OH-] = KW = 10 -14 p. H + p. OH](http://slidetodoc.com/presentation_image_h2/8a9eae5d0101853dc8bf552dee593268/image-19.jpg)
Recall [H 3 O+][OH-] = KW = 10 -14 p. H + p. OH = p. Kw = 14 9/5/2021 OFB Chapter 8 19
![The p. H Function Exercise page 8 -4: Calculate [H 3 O+] and [OH-] The p. H Function Exercise page 8 -4: Calculate [H 3 O+] and [OH-]](http://slidetodoc.com/presentation_image_h2/8a9eae5d0101853dc8bf552dee593268/image-20.jpg)
The p. H Function Exercise page 8 -4: Calculate [H 3 O+] and [OH-] in saliva that has a p. H of 6. 60 at 25 o. C. 9/5/2021 OFB Chapter 8 20

Strengths of Acids and Bases • Strong acids (completely ionized in water, i. e. , 100%) 1. 2. 3. 4. 5. 6. 7. HI HBr HCl. O 4 (perchloric acid) HCl. O 3 (chloric acid) H 2 SO 4 HNO 3 HA + H 2 O ↔ H 3 O+ + A – HA = generic acid 9/5/2021 OFB Chapter 8 21
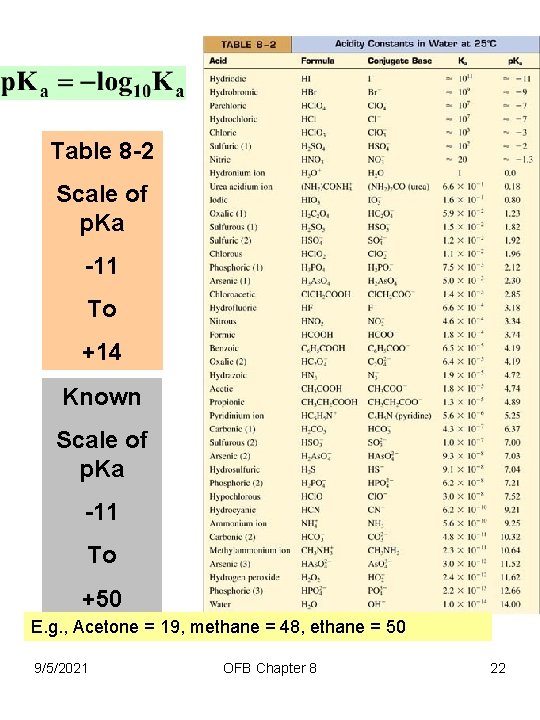
Table 8 -2 Scale of p. Ka -11 To +14 Known Scale of p. Ka -11 To +50 E. g. , Acetone = 19, methane = 48, ethane = 50 9/5/2021 OFB Chapter 8 22
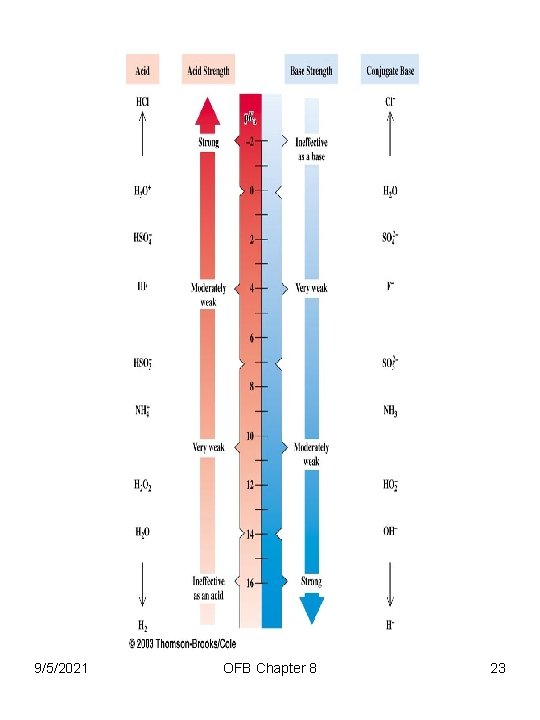
9/5/2021 OFB Chapter 8 23
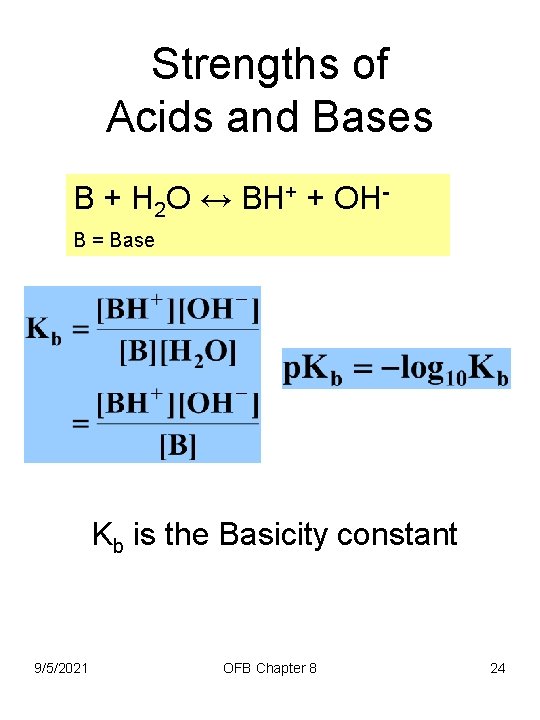
Strengths of Acids and Bases B + H 2 O ↔ BH+ + OHB = Base Kb is the Basicity constant 9/5/2021 OFB Chapter 8 24
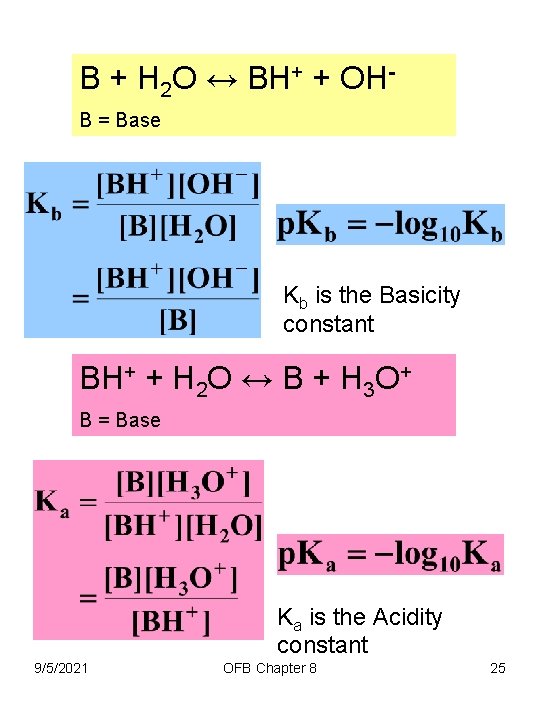
B + H 2 O ↔ BH+ + OHB = Base Kb is the Basicity constant BH+ + H 2 O ↔ B + H 3 O+ B = Base Ka is the Acidity constant 9/5/2021 OFB Chapter 8 25
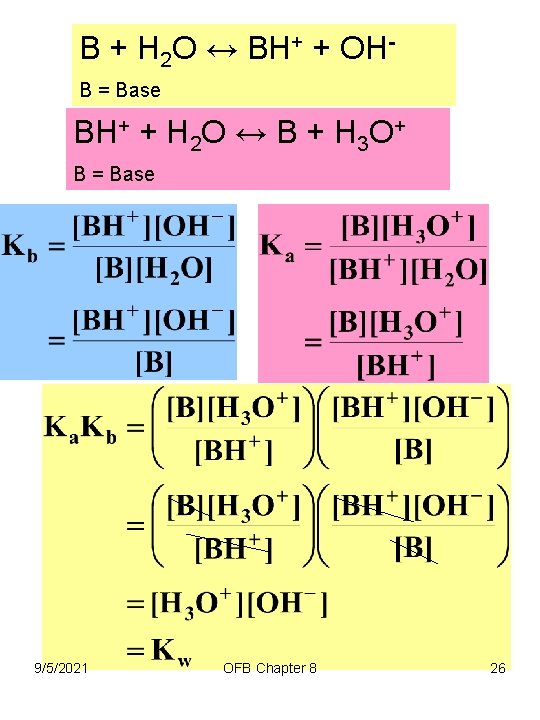
B + H 2 O ↔ BH+ + OHB = Base BH+ + H 2 O ↔ B + H 3 O+ B = Base 9/5/2021 OFB Chapter 8 26

Chapter 8 Acid-Base Equilibria • Base Strength – strong acids have weak conjugate bases – weak acids have strong conjugate bases The strength of a base is inversely related to the strength of its conjugate acid; the weaker the acid, the stronger its conjugate base, and vice versa p. Ka + p. Kb = p. Kw 9/5/2021 OFB Chapter 8 27

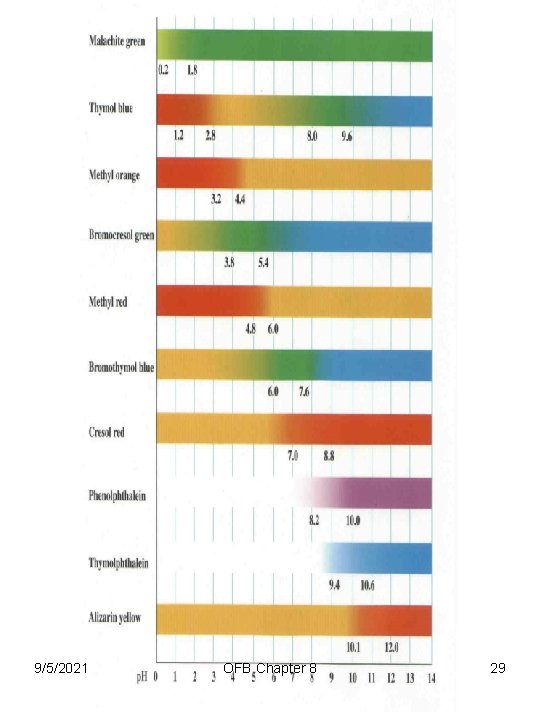
Indicators (denoted by In) A soluble compound, generally an organic dye, that changes its color noticeably over a fairly short range of p. H. Typically, a weak organic acid that has a different color than its conjugate base. HIn(aq) + H 2 O(l) H 3 O+(aq) + In-(aq) [H 3 O+][In-] [HIn] 9/5/2021 = Ka OFB Chapter 8 28
9/5/2021 OFB Chapter 8 29
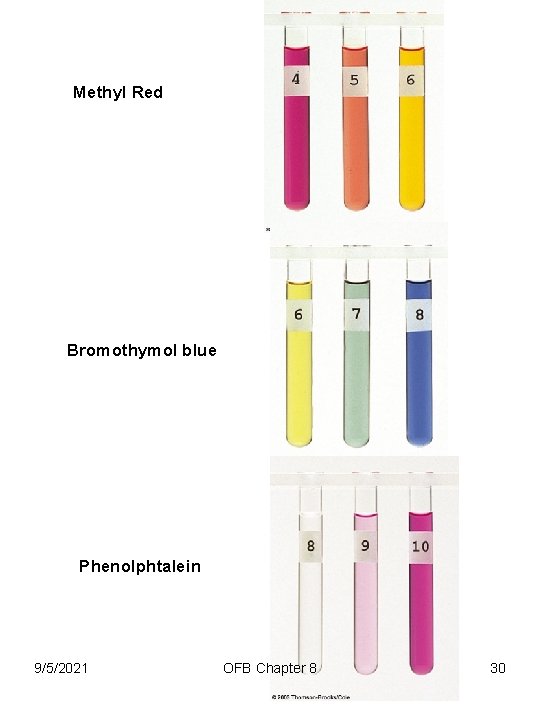
Methyl Red Bromothymol blue Phenolphtalein 9/5/2021 OFB Chapter 8 30

9/5/2021 OFB Chapter 8 31
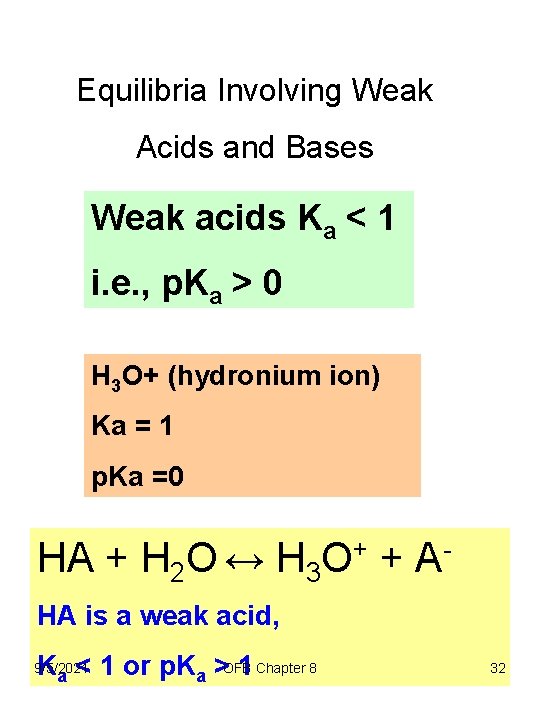
Equilibria Involving Weak Acids and Bases Weak acids Ka < 1 i. e. , p. Ka > 0 H 3 O+ (hydronium ion) Ka = 1 p. Ka =0 HA + H 2 O ↔ H 3 O+ + AHA is a weak acid, Ka < 1 or p. Ka >OFB 1 Chapter 8 9/5/2021 32
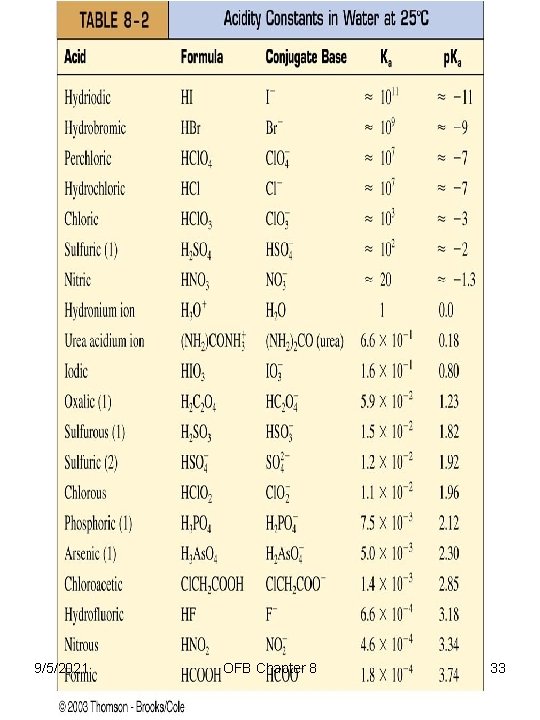
9/5/2021 OFB Chapter 8 33
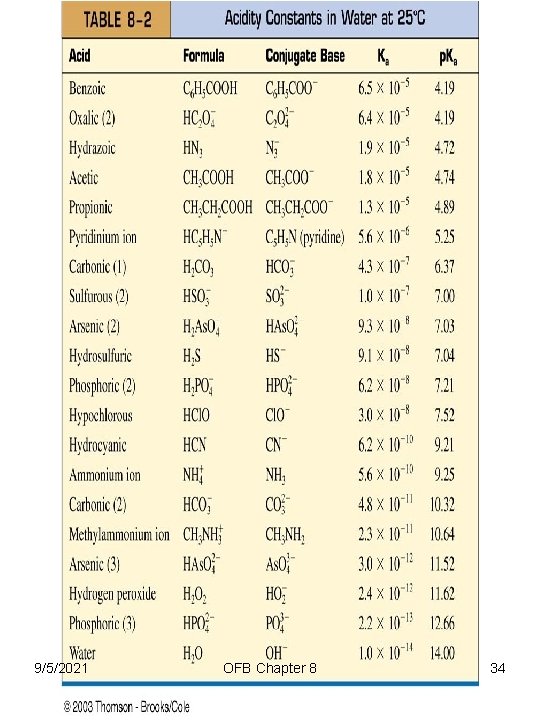
9/5/2021 OFB Chapter 8 34
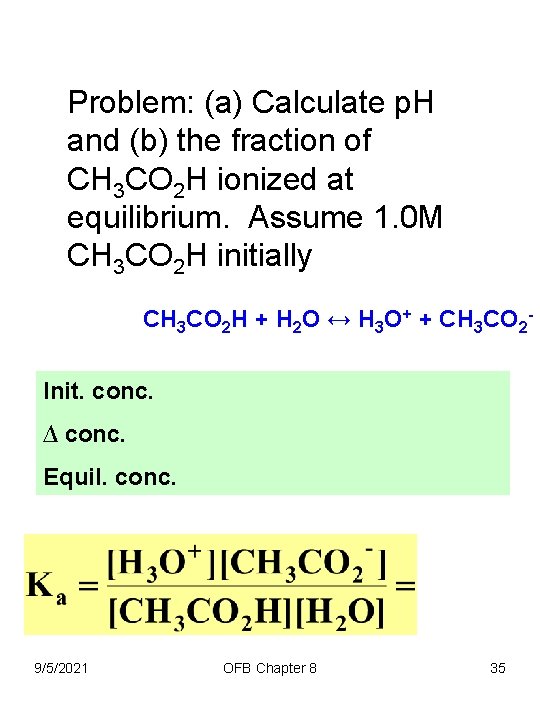
Problem: (a) Calculate p. H and (b) the fraction of CH 3 CO 2 H ionized at equilibrium. Assume 1. 0 M CH 3 CO 2 H initially CH 3 CO 2 H + H 2 O ↔ H 3 O+ + CH 3 CO 2 Init. conc. ∆ conc. Equil. conc. 9/5/2021 OFB Chapter 8 35

A trick to solving: Assume that y is small (less than 5% of the initial conc. ) 9/5/2021 OFB Chapter 8 36

Problem: (a) Calculate p. H and (b) the fraction of CH 3 CO 2 H ionized at equilibrium. Assume 1. 0 M CH 3 CO 2 H initially CH 3 CO 2 H + H 2 O ↔ H 3 O+ + CH 3 CO 2 - (b) Fraction CH 3 CO 2 H ionized at equilibrium 9/5/2021 OFB Chapter 8 37
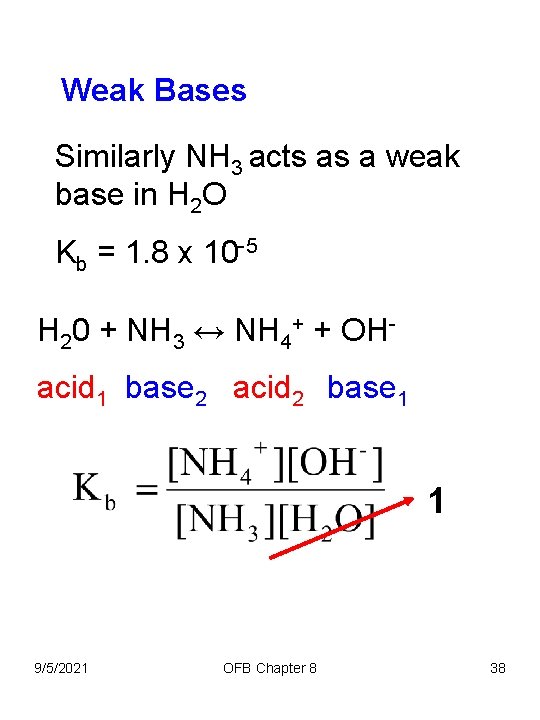
Weak Bases Similarly NH 3 acts as a weak base in H 2 O Kb = 1. 8 x 10 -5 H 20 + NH 3 ↔ NH 4+ + OHacid 1 base 2 acid 2 base 1 1 9/5/2021 OFB Chapter 8 38
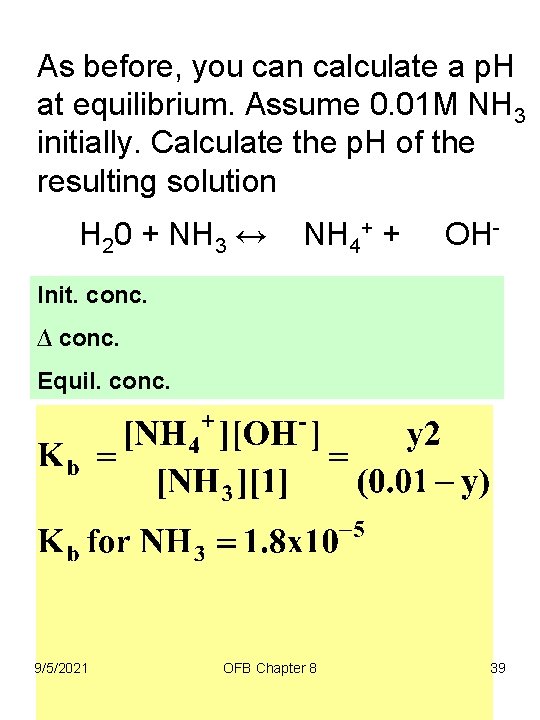
As before, you can calculate a p. H at equilibrium. Assume 0. 01 M NH 3 initially. Calculate the p. H of the resulting solution H 20 + NH 3 ↔ NH 4+ + OH- Init. conc. ∆ conc. Equil. conc. 9/5/2021 OFB Chapter 8 39

As before, you can calculate a p. H at equilibrium. Assume 0. 01 M NH 3 initially. Calculate the p. H of the resulting solution 9/5/2021 OFB Chapter 8 40

• Hydrolysis is a term applied to reactions of aquated ions that change the p. H from 7 • When Na. Cl is placed in water, the resulting solution is observed to be neutral (p. H = 7) • However when sodium acetate (Na. C 2 H 3 O 2) is dissolved in water the resulting solution is basic • Other salts behave similarly, NH 4 Cl and Al. Cl 3 give acid solutions. • These interactions between salts and water are called hydrolysis 9/5/2021 OFB Chapter 8 41

Despite the special term, hydrolysis, there is no reason to treat hydrolysis in a special manner. It is still a Brønsted-Lowry Acid and Base Reaction 9/5/2021 OFB Chapter 8 42

Example problem: Suppose a 0. 1 mole solution sodium acetate is dissolved in 1 liter of water. What is the p. H of the solution? Init. conc. ∆ conc. Equil. conc. 1. Find Kb 2. Find [OH-] 3. Find [H+] 9/5/2021 OFB Chapter 8 4. Find p. H 43
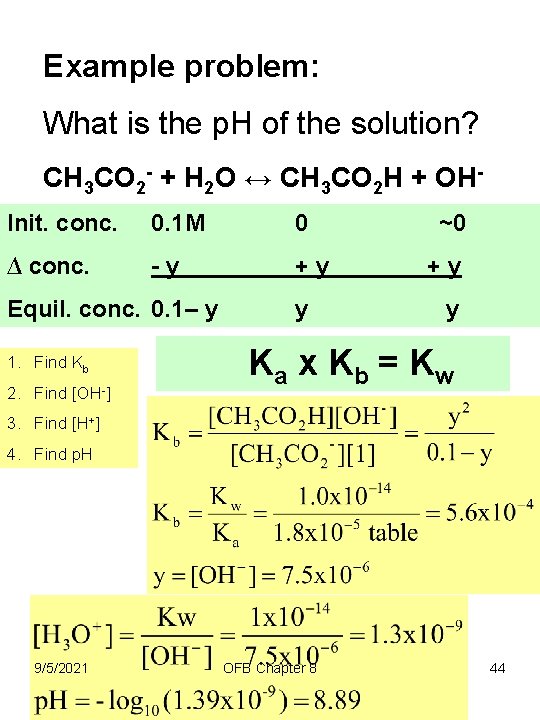
Example problem: What is the p. H of the solution? CH 3 CO 2 - + H 2 O ↔ CH 3 CO 2 H + OHInit. conc. 0. 1 M 0 ∆ conc. -y +y Equil. conc. 0. 1– y 1. Find Kb 2. Find [OH-] y ~0 +y y Ka x K b = K w 3. Find [H+] 4. Find p. H 9/5/2021 OFB Chapter 8 44
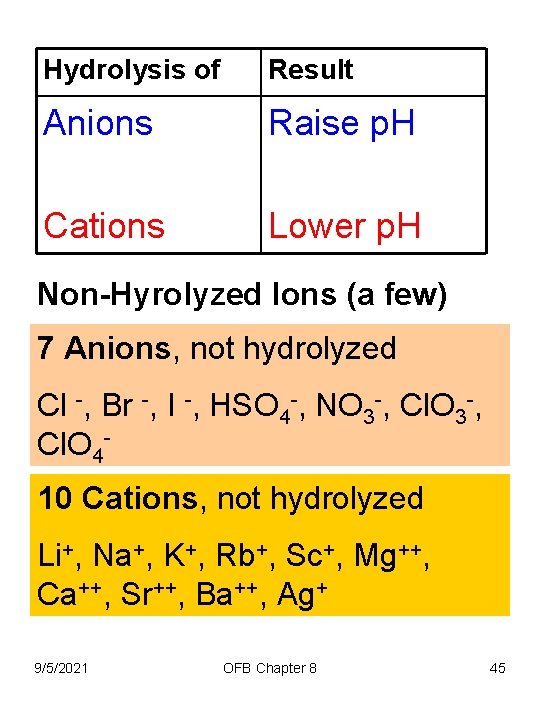
Hydrolysis of Result Anions Raise p. H Cations Lower p. H Non-Hyrolyzed Ions (a few) 7 Anions, not hydrolyzed Cl -, Br -, I -, HSO 4 -, NO 3 -, Cl. O 410 Cations, not hydrolyzed Li+, Na+, K+, Rb+, Sc+, Mg++, Ca++, Sr++, Ba++, Ag+ 9/5/2021 OFB Chapter 8 45

Hydrolysis of Result Anions Raise p. H Cations Lower p. H Can predict p. H of some salts (relative p. H) Na 3 PO 4 is basic (a non hydrolyzed cation and a hydrolyzed anion) Fe. Cl 3 is acidic (a hydrolyzed cation and a 9/5/2021 OFB Chapter 8 non hydrolyzed anion) 46

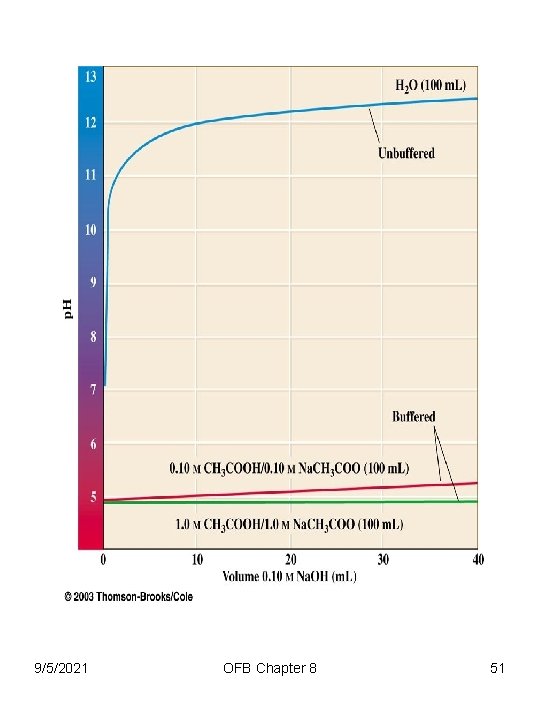
Chapter 8 Acid-Base Equilibria • Buffer Solutions: important in biochemical and physiological processes • Organisms (and humans) have built-in buffers to protect them against large changes in p. H. • Buffers any solutions that maintain an approximately constant p. H despite small additions of acids or bases 9/5/2021 OFB Chapter 8 47

Buffers any solutions that maintain an approximately constant p. H despite small additions of acids or bases Human blood (p. H=7. 4) is maintained by a combination of CO 3 -2, PO 4 -3 and protein buffers, which accept H+ Death = 7. 0 < p. H > 7. 8 = Death How Do Buffers Work? 9/5/2021 OFB Chapter 8 48
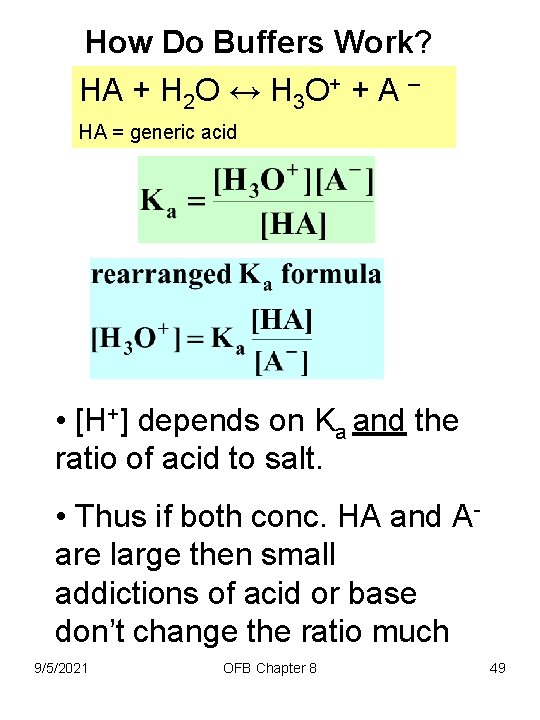
How Do Buffers Work? HA + H 2 O ↔ H 3 O+ + A – HA = generic acid • [H+] depends on Ka and the ratio of acid to salt. • Thus if both conc. HA and Aare large then small addictions of acid or base don’t change the ratio much 9/5/2021 OFB Chapter 8 49
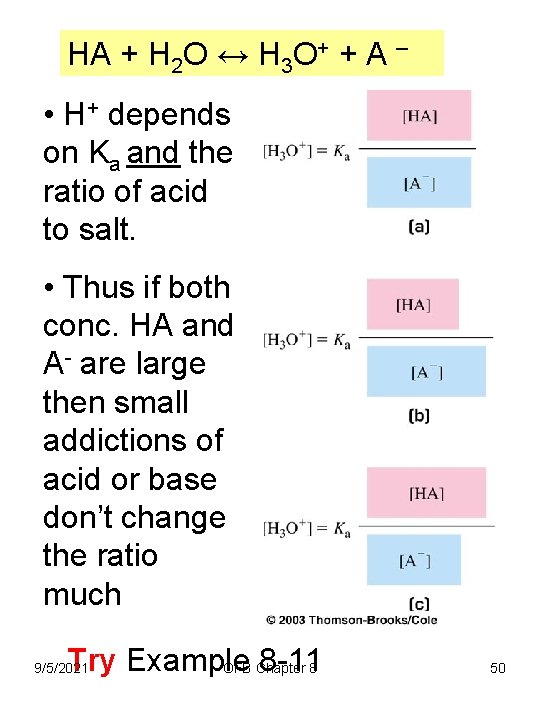
HA + H 2 O ↔ H 3 O+ + A – • H+ depends on Ka and the ratio of acid to salt. • Thus if both conc. HA and A- are large then small addictions of acid or base don’t change the ratio much Try Example 8 -118 OFB Chapter 9/5/2021 50
9/5/2021 OFB Chapter 8 51
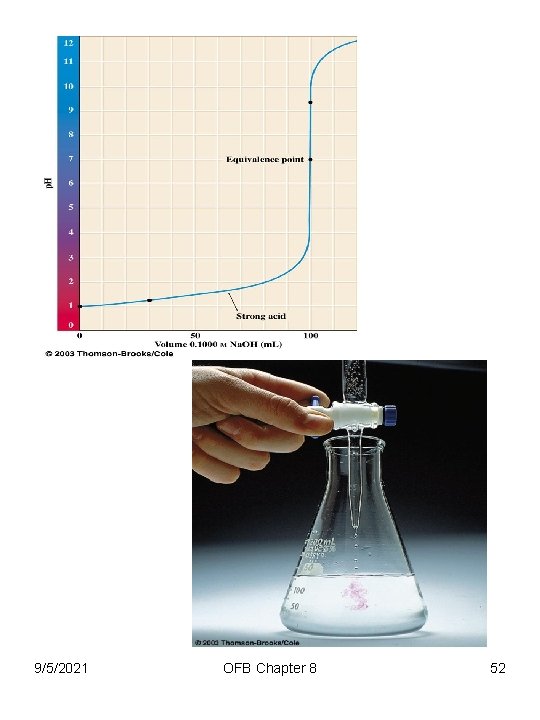
9/5/2021 OFB Chapter 8 52

8 -8 Lewis Acids • Extremely important for Organic Chemistry • Recall from Chapter 3 that main group elements in a molecule are surrounded by 8 electrons (the Octet Rule). • Hydrogen by 2 electrons • Carbon is 6 C or 1 s 2 2 p 2 • Hydrogen is 1 H or 1 s 1 9/5/2021 OFB Chapter 8 53
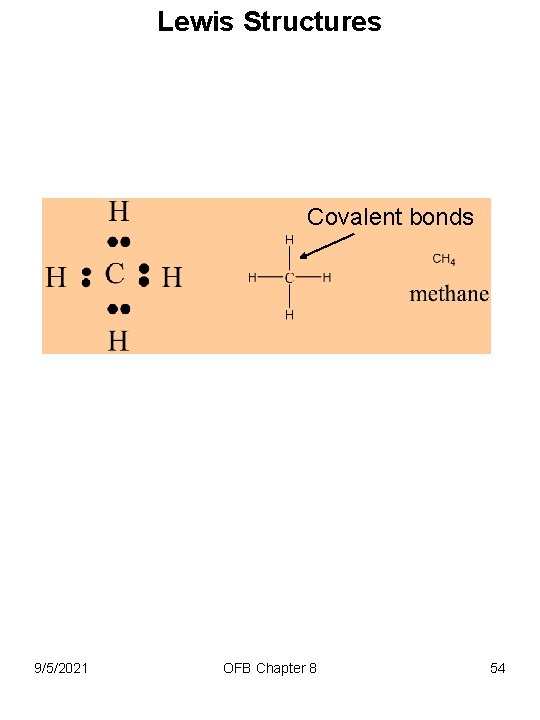
Lewis Structures Covalent bonds 9/5/2021 OFB Chapter 8 54
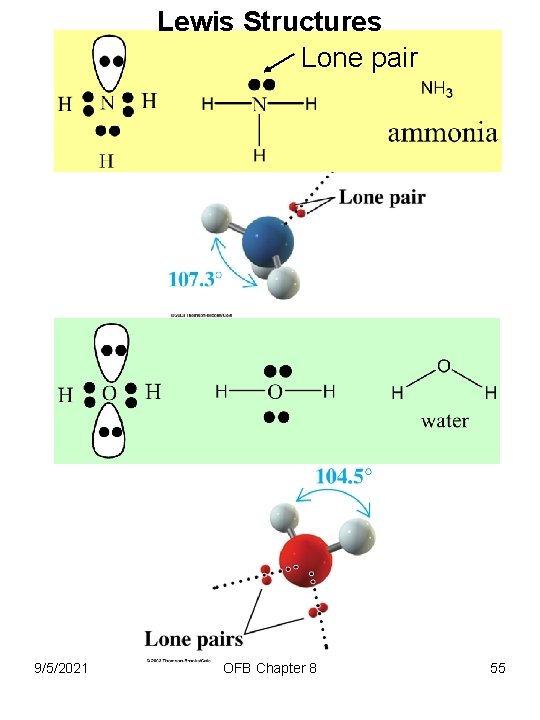
Lewis Structures Lone pair 9/5/2021 OFB Chapter 8 55
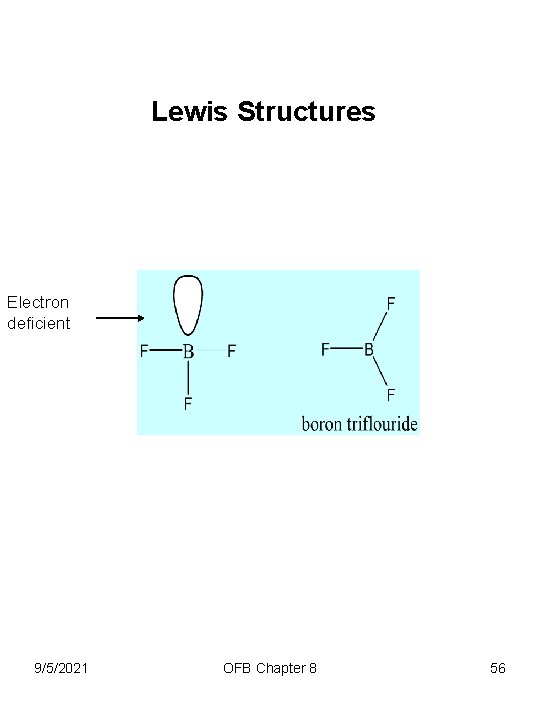
Lewis Structures Electron deficient 9/5/2021 OFB Chapter 8 56

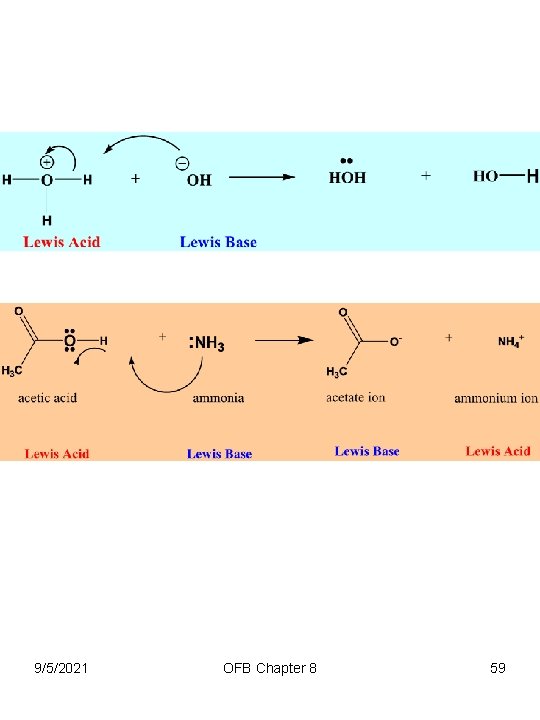
Chapter 8 Acid-Base Equilibria • Lewis Acids-Bases – Lewis Acid is any species that accepts electron pairs • Called electrophiles (electron seeking) • e. g. , Boron, Al, Group III elements • BF 3, Mg 2+, H 3 O+ BH 3, Sn. Cl 4, CH 3+, Al. Cl 3, HCl 9/5/2021 OFB Chapter 8 57
Chapter 8 Acid-Base Equilibria • Lewis Acids-Bases – Lewis Base is any species that donates electrons through coordination to its lone pair • Called Nucleophiles (nucleus seeking) • e. g. , Group V, VII elements • Br -1, Cl -1, F -1, HO -1, : NH 3, H 2 O: : 9/5/2021 OFB Chapter 8 58
9/5/2021 OFB Chapter 8 59
- Slides: 59