Chapter 7 Whiteboard Review What are ions formed

Chapter 7 Whiteboard Review

» What are ions formed from a single atom called? » Monatomic ions

» Name the following: + » Lithium cation

» Name the following: » Br » bromide anion

» Name the following: 2» O » oxide anion

» Name the following: 2+ » Cobalt (II) cation

» Name the following: 3+ » Aluminum cation
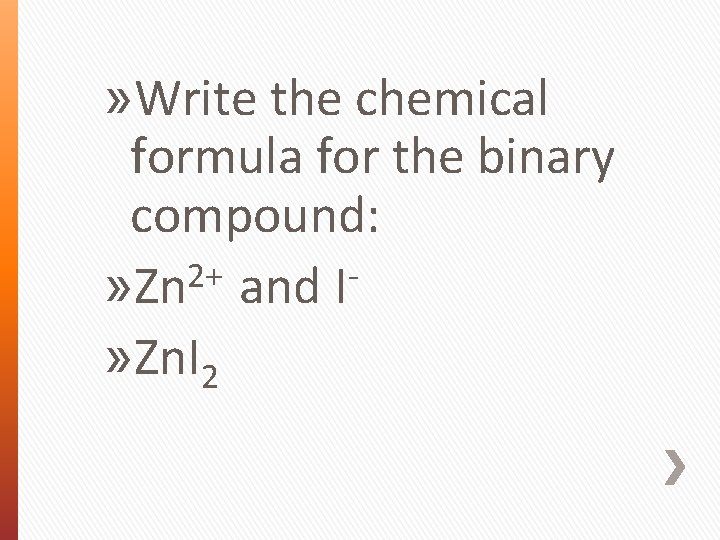
» Write the chemical formula for the binary compound: 2+ » Zn and I » Zn. I 2

» Write the chemical formula for the binary compound formed between the following elements: » Magnesium and chlorine » Mg. Cl 2

» Write the chemical formula for the binary compound formed between the following elements: » Aluminum and sulfur » Al 2 S 3

» Write the formula and give the name for the compounds formed between the following ions: » Fe 2+ and O 2» Fe. O; iron(II) oxide

» Write the formula and give the name for the compounds formed between the following ions: » Fe 3+ and O 2» Fe 2 O 3; iron(III) oxide

» Name the following: 2» CO 3 » Carbonate ion

» Name the following: 3» PO 4 » phosphate ion

» Name the following: + » NH 4 » ammonium ion

» Write the formula for: » Tin(IV) sulfate » Sn(SO 4)2

» Write the formula for: » Calcium nitrite » Ca(NO 2)2

» Give the name of the following compound: » Ca(OH)2 » Calcium hydroxide

» Give the name of the following compound: » KCl. O » Potassium hypochlorite

» Give the name of the following compound: » As 2 O 5 » Diarsenic pentoxide

» Give the name of the following compound: » SO 3 » Sulfur trioxide

» Write the formula for: » Dinitrogen trioxide » N 2 O 3

» Name the following acid: » H 2 SO 4 » Sulfuric acid

» Name the following acid: » HI » Hydriodic acid

» Name the following acid: » HNO 2 » Nitrous acid

» Write the formula for the following acid: » Hypochlorous acid » HCl. O

» Write the formula for the following acid: » carbonic acid » H 2 CO 3

» Assign oxidation numbers to each atom in the following compounds or ions: » HCl » H +1; Cl -1

» Assign oxidation numbers to each atom in the following compounds or ions: » CF 4 » C +4; F -1

» Assign oxidation numbers to each atom in the following compounds or ions: » SO 2 » S +4; O -2

» Assign oxidation numbers to each atom in the following compounds or ions: » HCl. O 3 » H +1; Cl +5; O -2

» Assign oxidation numbers to each atom in the following compounds or ions: 2+ » Sn » +2

» Assign oxidation numbers to each atom in the following compounds or ions: » O 2 » 0

» Name the following compounds first using the prefix system, then using the stock system: » CO 2 » Carbon dioxide; » carbon(IV) oxide

» Name the following compounds first using the prefix system, then using the stock system: » PCl 3 » Phosphorus trichloride; » phosphorus(III) chloride

» Name the following compounds first using the prefix system, then using the stock system: » N 2 O » Dinitrogen monoxide; » nitrogen(I) oxide

» Find the formula mass of the following compound: » H 2 SO 4 » 98. 08 amu

» Find the formula mass of the following compound: » Ca(NO 3)2 » 164. 10 amu
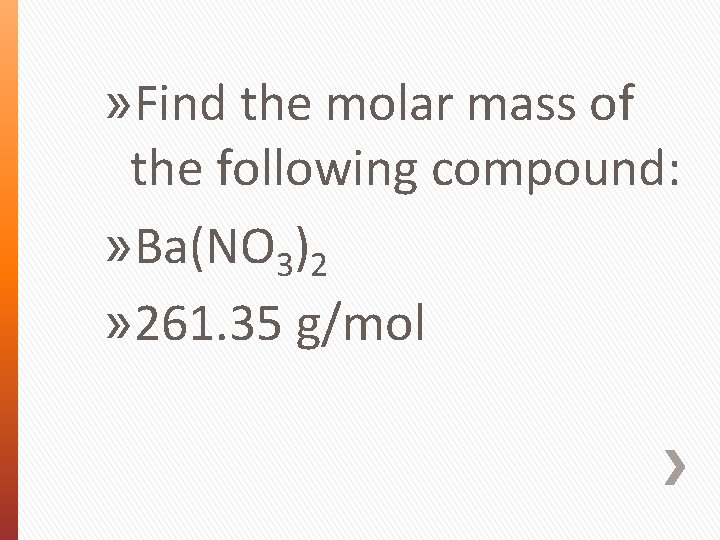
» Find the molar mass of the following compound: » Ba(NO 3)2 » 261. 35 g/mol

» How many moles of atoms of each element are there in one mole of the following compound: » Ba(NO 3)2 » 1 mol Ba; 2 mol N; 6 mol O

» How many moles of atoms of each element are there in one mole of the following compound: » Na. NO 3 » 1 mol Na; 1 mol N; 3 mol O

» What is the mass in grams of 0. 257 mol of calcium nitrate, Ca(NO 3)2: » 42. 2 g

» How many moles of compound are there in 6. 60 g (NH 4)2 SO 4: » 0. 0500 mol

» Find the percentage of water in sodium carbonate decahydrate, . Na 2 CO 3 10 H 20: » 62. 98% H 20

» What is the empirical formula for a compound that contains 32. 38% Na, 22. 65% S, and 44. 99% O? » Na 2 SO 4

» Determine the molecular formula of the compound with an empirical formula of CH and a formula mass of 78. 110 amu. » C 6 H 6

» A sample of a compound with a formula mass of 34. 00 amu is found to consist of 0. 44 g H and 6. 92 g O. Find its molecular formula. » H 2 O 2

» The empirical formula for trichloroisocyanuric acid, the active ingredient in many household bleaches is OCNCl. The molar mass of this compound is 232. 41 g/mol. What is the molecular formula? » O 3 C 3 N 3 Cl 3
- Slides: 48