Chapter 7 Solvent Cycle Methods for Solute Precipitation






















- Slides: 39

Chapter 7 Solvent Cycle, Methods for Solute Precipitation Heat and Mass Transfer: High Pressure chemical Engineering I (WS)

Flow Scheme of a Solvent Cycle

Solvent Cycle Steps: separate the extract from the solvent (1), clean the solvent for reuse (2), remove the solvent from raffinate (3), adjust composition of solvent mixture (if applicable) (4).

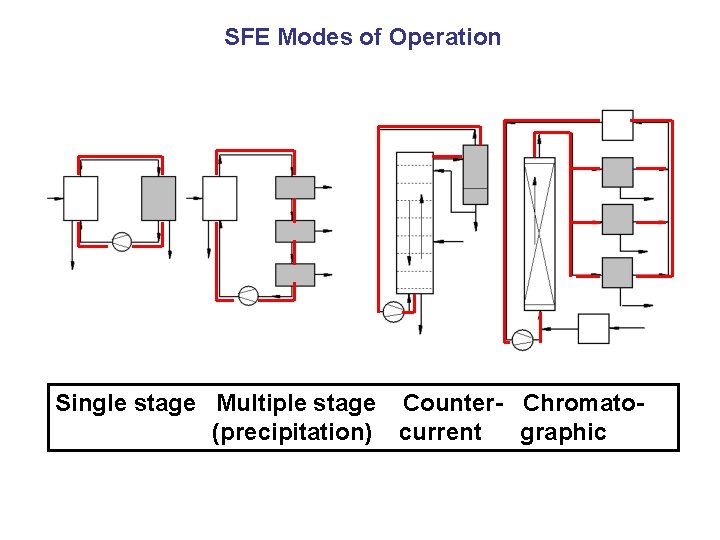
SFE Modes of Operation Single stage Multiple stage Counter- Chromato(precipitation) current graphic

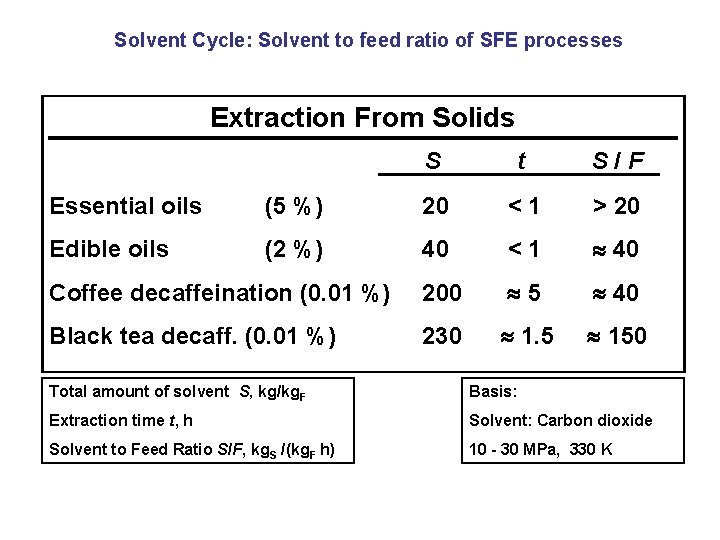
Solvent Cycle: Solvent to feed ratio of SFE processes Extraction From Solids S t S/F Essential oils (5 %) 20 <1 > 20 Edible oils (2 %) 40 <1 40 Coffee decaffeination (0. 01 %) 200 5 40 Black tea decaff. (0. 01 %) 230 1. 5 150 Total amount of solvent S, kg/kg. F Basis: Extraction time t, h Solvent: Carbon dioxide Solvent to Feed Ratio S/F, kg. S /(kg. F h) 10 - 30 MPa, 330 K

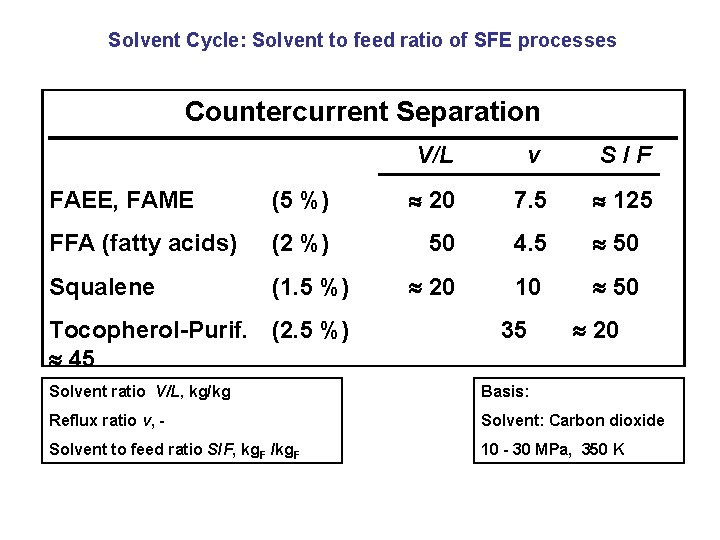
Solvent Cycle: Solvent to feed ratio of SFE processes Countercurrent Separation V/L v S/F FAEE, FAME (5 %) 20 7. 5 125 FFA (fatty acids) (2 %) 50 4. 5 50 Squalene (1. 5 %) 20 10 50 Tocopherol-Purif. (2. 5 %) 45 35 20 Solvent ratio V/L, kg/kg Basis: Reflux ratio v, - Solvent: Carbon dioxide Solvent to feed ratio S/F, kg. F /kg. F 10 - 30 MPa, 350 K

Solvent Cycle: Solvent to feed ratio of SFE processes Chromatographic Separation Pr tr S/F DHA / DPA 1. 5 15 900 x 103 EM Phytol-isomers 10 - 30 6 900 EM 200 SMB Productivity Pr, g. P /(kg. St. Ph h) Basis: Retention time, min Solvent: Carbon dioxide Solvent to feed ratio S/F, kg. F /kg. F 10 - 30 MPa, 310 K

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

Solubility of Caffeine in CO 2 Birtigh, Brunner, Johannsen

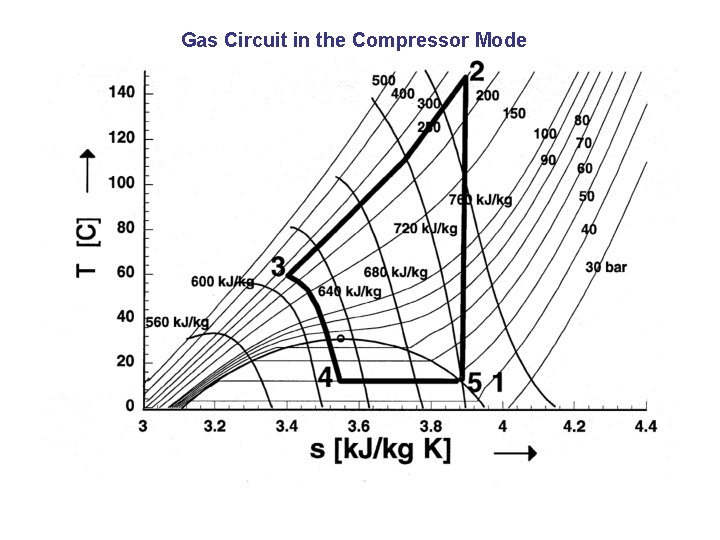
Gas Circuit in the Compressor Mode

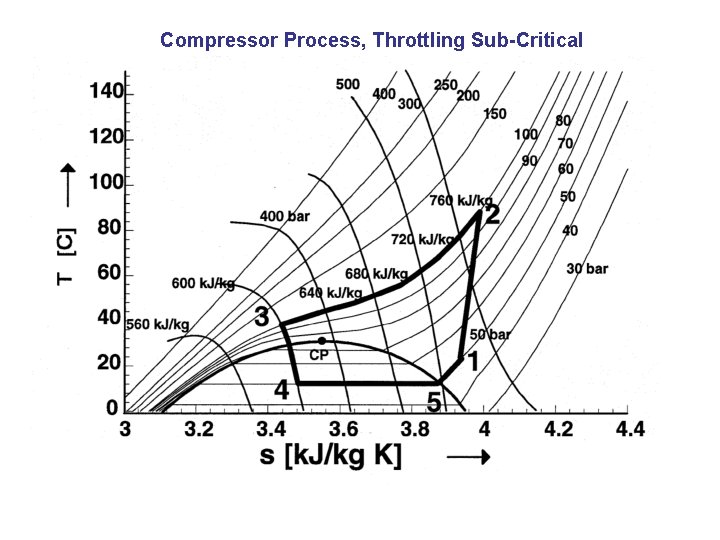
Compressor Process, Throttling Sub-Critical

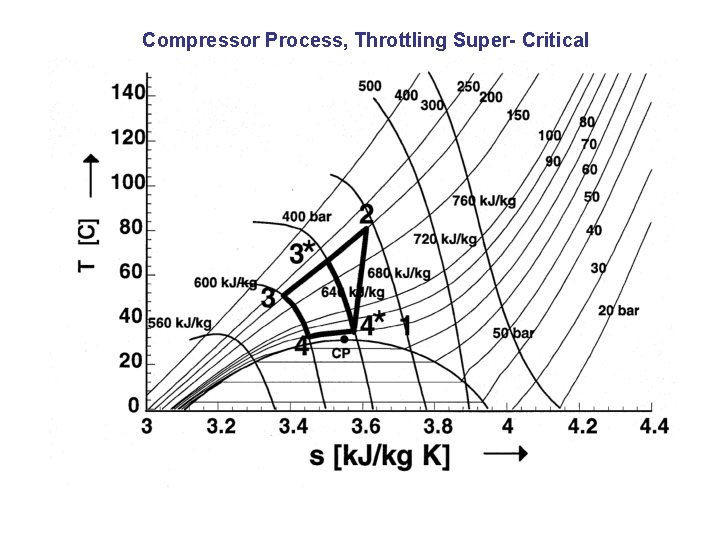
Compressor Process, Throttling Super- Critical

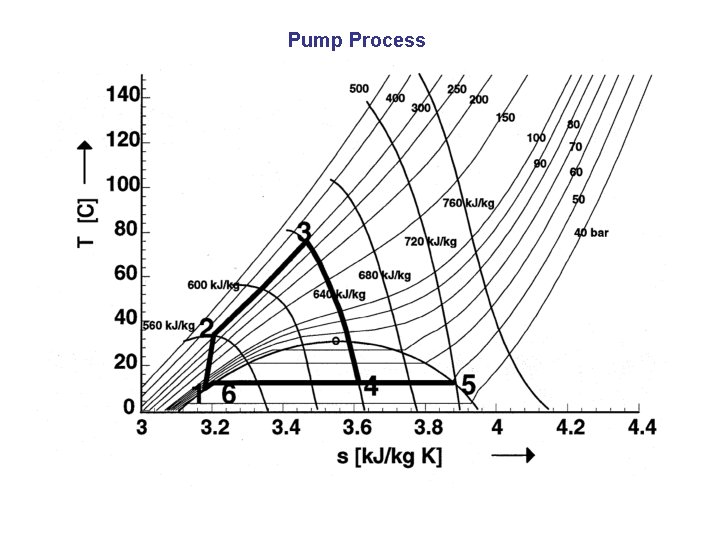
Pump Process

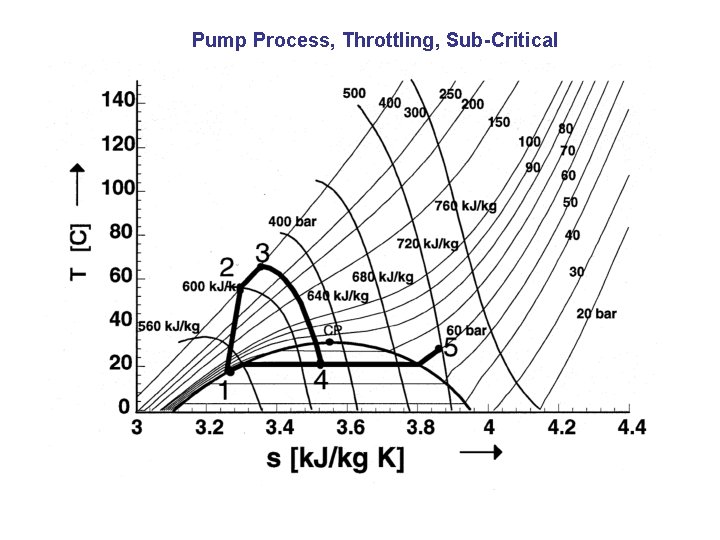
Pump Process, Throttling, Sub-Critical

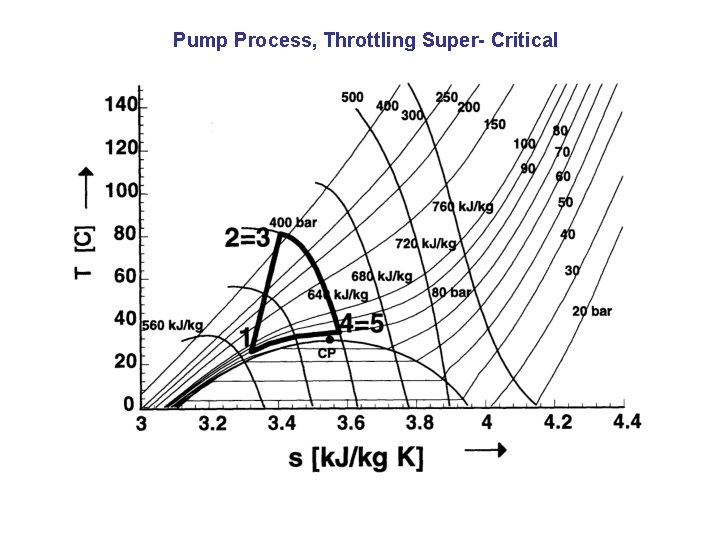
Pump Process, Throttling Super- Critical

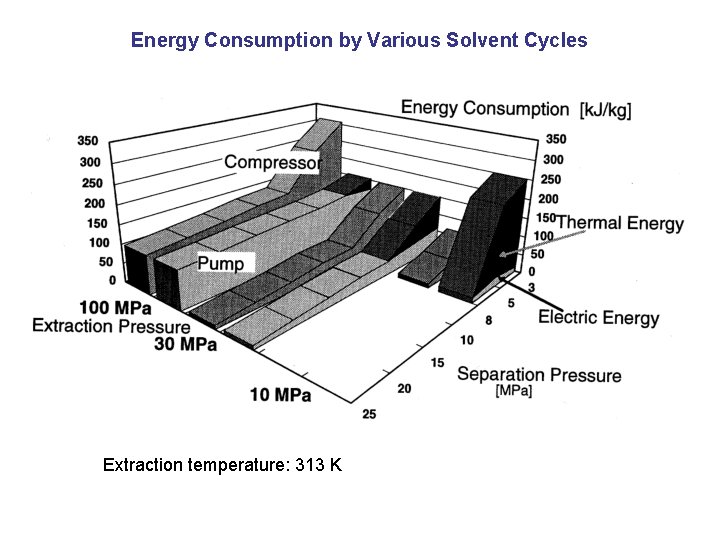
Energy Consumption by Various Solvent Cycles Extraction temperature: 313 K

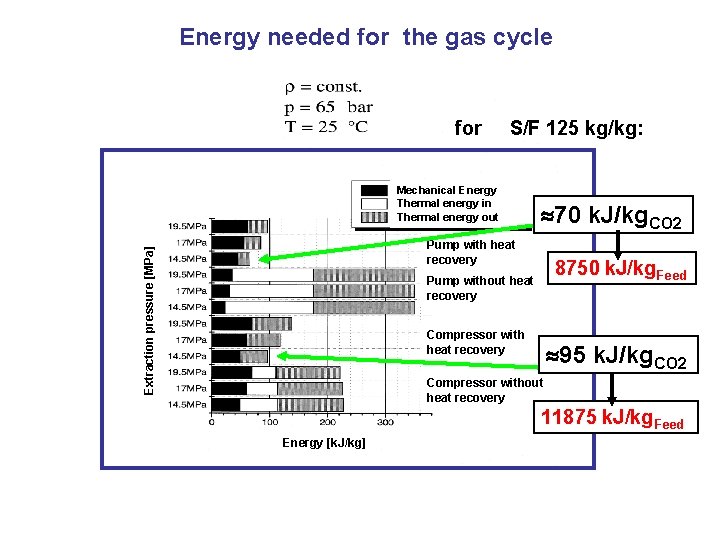
Energy needed for the gas cycle for S/F 125 kg/kg: Mechanical Energy Thermal energy in Thermal energy out 70 k. J/kg. CO 2 Extraction pressure [MPa] Pump with heat recovery 8750 k. J/kg. Feed Pump without heat recovery Compressor with heat recovery 95 k. J/kg. CO 2 Compressor without heat recovery 11875 k. J/kg. Feed Energy [k. J/kg]

Modes For Product Recovery Reduction of pressure or density (temperature) Anti solvent Membrane separation Adsorption Absorption De-Entrainment

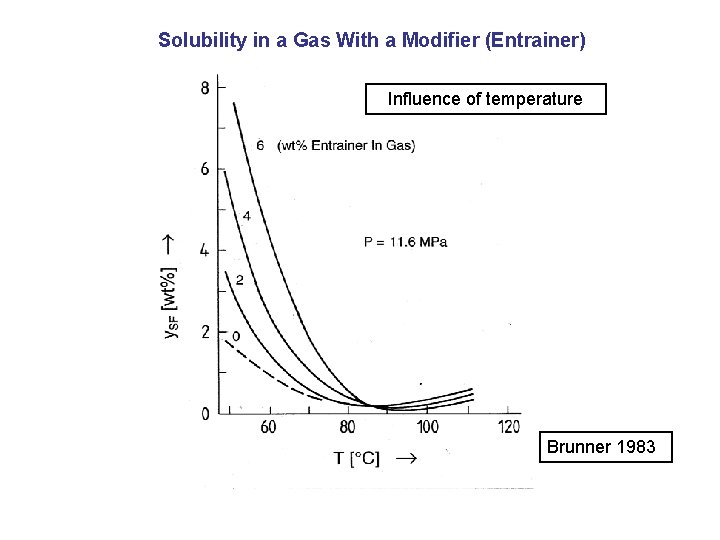
Solubility in a Gas With a Modifier (Entrainer) Influence of temperature Brunner 1983

Solubility of Caffeine in CO 2 Data by: Gährs 1984 Ebeling, Franck 1984 Johannsen, Brunner 1994

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

Anti-Solvent: Solubility of Caffeine in CO 2 Influence of nitrogen Gährs 1984

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

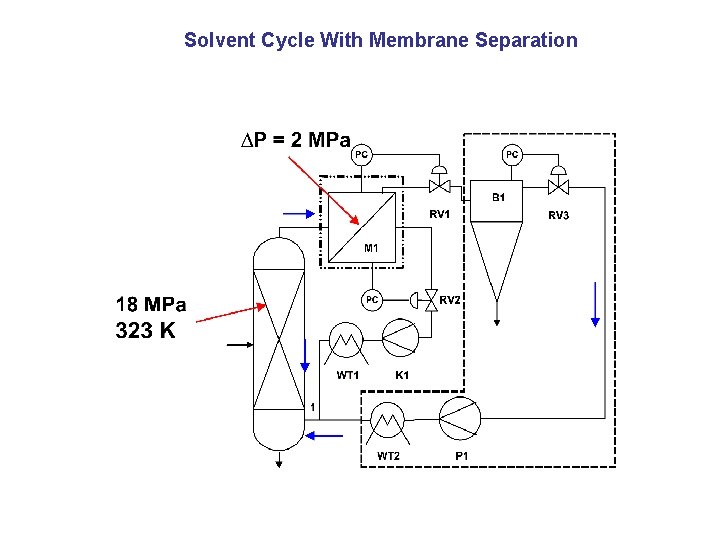
Solvent Cycle With Membrane Separation

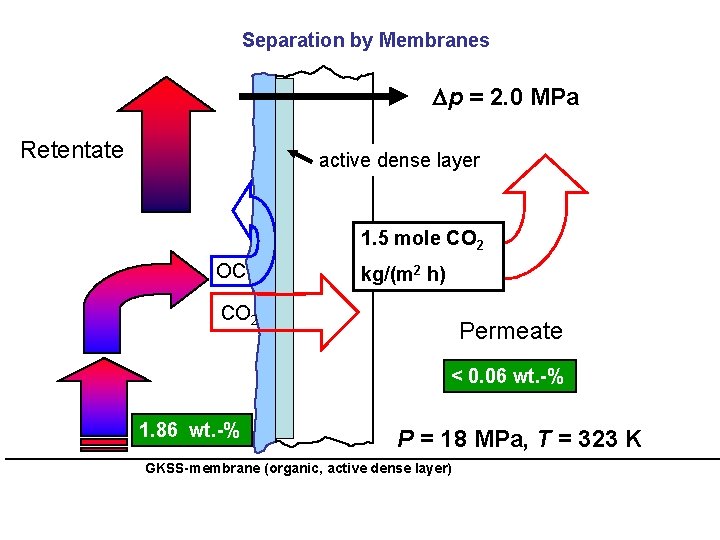
Separation by Membranes p = 2. 0 MPa Retentate active dense layer 1. 5 mole CO 2 OC kg/(m 2 h) CO 2 Permeate < 0. 06 wt. -% 1. 86 wt. -% P = 18 MPa, T = 323 K GKSS-membrane (organic, active dense layer)

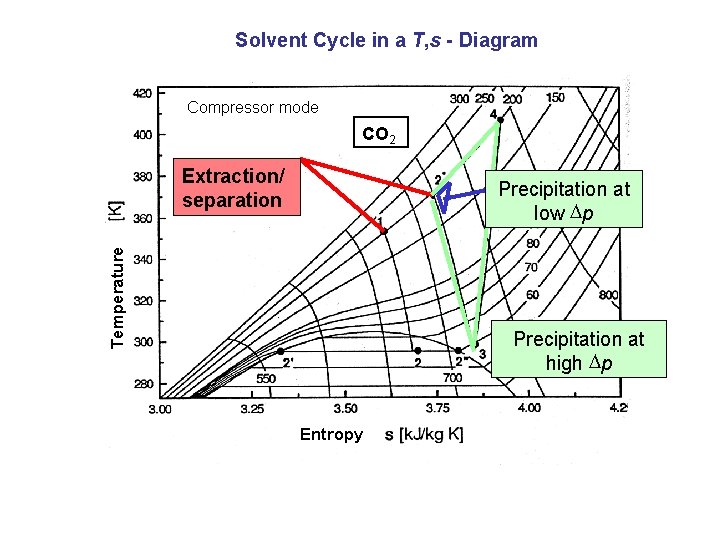
Solvent Cycle in a T, s - Diagram Compressor mode CO 2 Extraction/ separation Temperature Precipitation at low Dp Precipitation at high Dp Entropy

Energy For Different Solvent Cycles Pump-Cycle 53 k. J/ kg. CO 2 Compressor-Cycle 21 k. J/ kg. CO 2 Membrane-Cycle Like in 2 7. 6 k. J/ kg. CO 2 Sartorelli 2001

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

Adsorption of Caffeine on Activated Carbon

Recovery of Tocopherolacetate by Adsorption Silica with 52% loading, loaded by high pressure adsorption Silica with 50% loading, loaded by mixing, conventional process

Recovery of Tocopherolacetate by Adsorption

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

Phase Equilibrium Caffeine - Water - CO 2

Modes For Product Recovery Reduction of pressure or density Anti solvent Membrane separation Adsorption Absorption De-Entrainment. . .

Solubility in a Gas With a Modifier (Entrainer) Influence of temperature Brunner 1983

De-Entrainment Birtigh

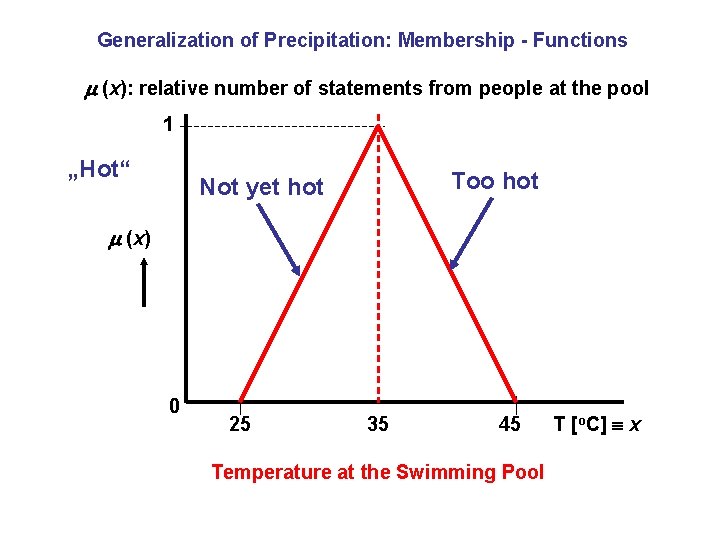
Generalization of Precipitation: Membership - Functions (x): relative number of statements from people at the pool 1 „Hot“ Too hot Not yet hot (x) 0 25 35 45 Temperature at the Swimming Pool T [o. C] x

Membership Functions P Adsorption Membrane Birtigh

Absorption De-Entrain T T Membership Functions Birtigh