Chapter 7 Solutions and Other Mixtures Mixtures Variable

Chapter 7 - Solutions and Other Mixtures

Mixtures • Variable combination of 2 or more pure substances. Heterogeneous Homogeneous (solutions)

Heterogeneous Mixtures • Composed of dissimilar components; not uniform structure or composition throughout • Examples: Fruit salad, orange juice, • Types of Heterogeneous Mixtures • Suspensions • Colloids • Emulsions

Suspension • heterogeneous • large particles • The particles are more or less evenly dispersed throughout a liquid • particles settle when mixture is allowed to stand • EX: fresh-squeezed lemonade, italian salad dressing, things that say shake well before using!

Suspension made of two liquids • Example- oil and vinegar • Two layers separate because liquids are immiscible • The heavier (more dense) liquid settles to bottom • The lighter liquid can be decanted (poured) off

Colloid • • Heterogeneous medium-sized particles don’t settle EX: milk, whipped cream, egg whites,

Colloids and the Tyndall Effect • Because the particles in colloids are so small, they pass through ordinary filters and stay dispersed throughout the mixture • The particles ARE large enough to scatter light • This scattering of light is called the Tyndall Effect Mixture on right is a colloid

Emulsions • A colloid in which two or more immiscible liquids in which one liquid is dispersed in the other • Example: Mayonnaise • Small droplets of oil suspended in vinegar

Heterogeneous Mixtures • Examples: • mayonnaise Colloid/emulsion • muddy water suspension • fog colloid • marshmallows colloid • Italian salad dressing suspension

Homogeneous Mixtures • Also called Solutions because they have a uniform composition • Plain water is homogeneous because it is a single substance • Saltwater is also homogeneous because it is a uniform mixture of water molecules, sodium ions, and chloride ions

Solutions • Solutions are a homogeneous mixture made up of tiny particles that do not settle. • Solutions are made up of different substances that are dissolved. • Ex: Kool-Aid, Tea, Sprite • Miscible liquids mix to form solutions • Ex: Sprite, rubbing alcohol • Can be separated by distillation (based on different boiling points)

Solutions • There are two parts of a solution: • Solute • Solvent

Solute • In a solution, the solute is the substance that is dissolved. • For example, in Kool-Aid, the solute would be the sugar.

Solvents • In a solution, the substance that does the dissolving is the solvent. • In Kool-Aid, the solvent is the water.

Solutes and Solvents • Solutes are not always solid, and solvents are not always liquid. • Fuels like gasoline are solutions of petroleum • Other states of matter can form solutions • Ex: the air you breathe is a mixture of oxygen, nitrogen, argon, and other gases • Two solids can be dissolved together to make a solution called an alloy. • Ex: Bronze is a solution of tin and copper like in a trumpet

Solutes and Solvents • Furthermore, two gases can be dissolved together. • Ex: Carbon dioxide is a solution of oxygen and carbon

Examples • In the following examples, tell which substance is the solute, and which is the solvent: • Tea • Carbonated Water • Salt Water

HOW SUBSTANCES DISSOLVE

Water: The Universal Solvent • • Liquid you drink are mostly water Your body is 75% water The earth is 67% water Many things can dissolve in water, and for this reason it is called the Universal solvent

Polarity of Water • Remember from chapter 5, a polar molecule has unequal sharing of electrons • Water is a polar molecule • Polar molecules have a positively and negatively charged sides • Water can dissolve ionic compounds because it can attract the positive and negative ions of ionic compounds

Polarity of Molecules • This attraction between water molecules and ionic compounds results in the ions pulling away from the crystal • In Na. Cl, the sodium ions are attracted to the negative oxygen atoms of the water molecules and the chlorine ions are attracted to the positive hydrogen atoms of the water molecules • Eventually, all ions in the Na. Cl crystal are pulled into solution and completely dissolved

Ionic Compounds and Water • Not all ionic compounds dissolve in water • To dissolve an ionic substance: • Water molecules must exert a force on the ions that is more attractive than the force between ions in the crystal • This applies to all solvent and solute molecules (solvent molecules must exert more force on the particles of the substance than the particles exert on each other
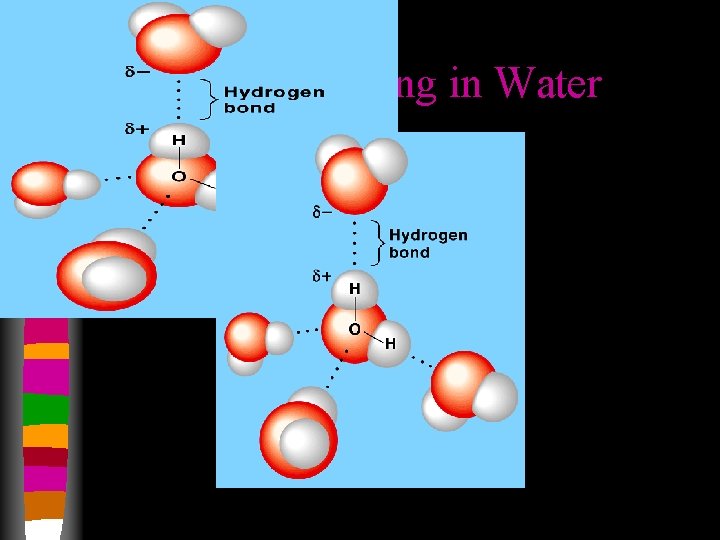
Hydrogen Bonding in Water

Hydrogen Bonding in Water • Hydrogen bonding pulls water molecules close together • The following properties of water can be attributed to hydrogen bonding: • Low molecular mass • Fairly dense • High boiling point • Polar molecules dissolve polar molecules • In ethanol, the –OH group makes it polar, the positive hydrogen atom of a water molecule attracts the negative oxygen atom of an ethanol molecule

Hydrogen Bonding Animation • See how ionic bonds are dissolved in water • http: //www. northland. cc. mn. us/biology/b iology 1111/animations/dissolve. html

What if it’s not polar? • Remember polar molecules dissolve polar molecules • Non-polar molecules dissolve non-polar molecules • Non-polar compounds’ electrons are distributed equally among the atoms
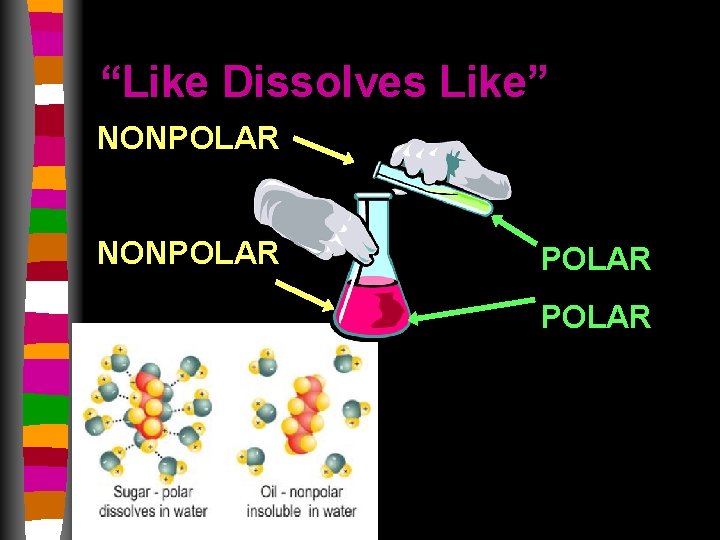
“Like Dissolves Like” NONPOLAR

Factors Affecting Dissolving • Stirring or shaking a solution helps the solute dissolve faster • Solutes with a larger surface area dissolve faster • Stirring moves dissolved solute molecules away from undissolved solute, and more solvent can reach the undissolved solute • Smaller pieces dissolve faster than larger pieces • Solutes dissolve faster when the solvent is hot • KMT-particles are moving faster solvent molecules collide more with solute molecules

Solutes effect the physical properties of solutions • Solutes can lower the freezing point of a solution and raise its boiling point • Ex: salting the road in the winter

SOLUBILITY AND CONCENTRATION

Solubility • Insoluble: solute not able to be dissolved (oil and water) • Soluble: solute able to be dissolve (sugar and water) • Solubility: The maximum amount of a solute that can dissolve in a given quantity of solvent at a given temperature and pressure • Why you can’t dissolve all of a solute

Solubility • Different substances have different solubilities; see Table 1 page 240 • The solubility of Silver Nitrate (216 g/100 g of water)is greater in water than Calcium Chloride (75 g/100 g of water) • Solubility depends on the strength of forces acting between the water and solute molecules compared to forces between the solute molecules

Concentration • Weak solution: only a small amount dissolves in solvent • Strong solution: a large amount dissolves in solvent • These don’t specify concentration • The amount of a particular substance in a given quantity of a mixture, solution, or ore • Concentrated solution: contains a large amount of solute • Dilute solution: contains a small amount of solute

Saturation • In a solution, there’s only so much solute that a solvent can dissolve. • When a solvent can no longer dissolve any solute, it is saturated.

Saturation • Saturation is the point at which no more solute can be dissolved. • Ex: Adding sugar to tea • Solutions can be: • Unsaturated • Supersaturated

Unsaturated Solutions • Unsaturated solutions are capable of dissolving more solute. • Ex: Adding sugar to hot tea. All the sugar is dissolved, the tea is unsaturated.

Saturated Solutions • Saturated solutions can not dissolve any more solute. • Ex: You add more sugar to your hot tea. Grains of sugar sink to the bottom and are not dissolved.

Supersaturated Solutions • Supersaturated solutions have dissolved more solute than is normal. • Usually, supersaturation occurs if the temperature of the solution has been altered. • Ex: Reheating your tea, then adding more sugar.

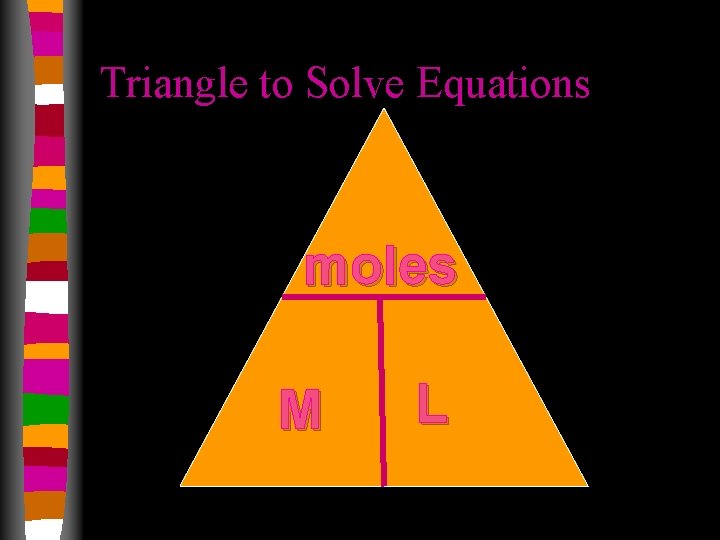
Molarity • Concentration of a solution. substance being dissolved total combined volume
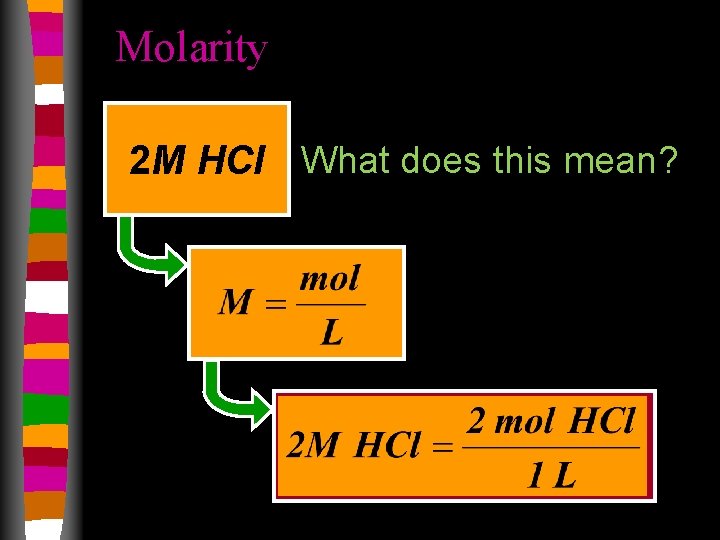
Molarity 2 M HCl What does this mean?

Molarity Calculations molar mass (g/mol) 6. 02 1023 (particles/mol) MASS IN NUMBER MOLES GRAMS OF PARTICLES Molarity (mol/L) LITERS OF SOLUTION

Molarity Calculations • How many grams of Na. Cl are required to make 0. 500 L of 0. 25 M Na. Cl? 0. 500 L 0. 25 mol 58. 44 g 1 L 1 mol = 7. 3 g Na. Cl
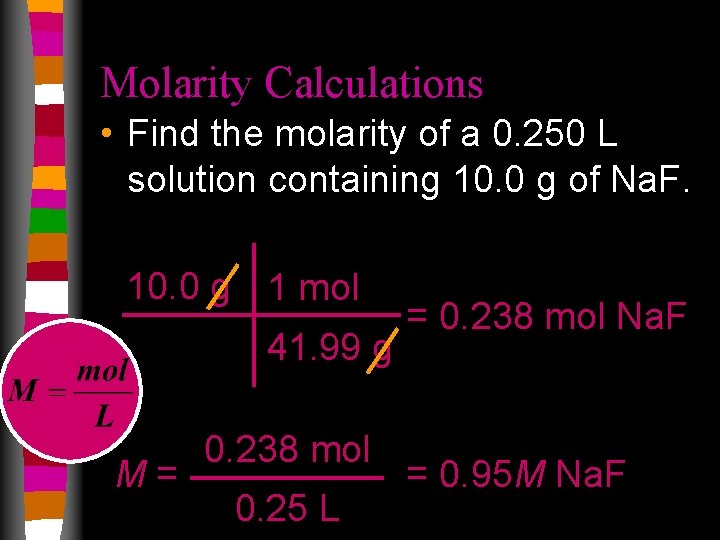
Molarity Calculations • Find the molarity of a 0. 250 L solution containing 10. 0 g of Na. F. 10. 0 g 1 mol 41. 99 g M= 0. 238 mol 0. 25 L = 0. 238 mol Na. F = 0. 95 M Na. F

Other measures of concentration • Mass percent: like in the drugstore, can find 3% hydrogen peroxide • ppm: parts per million • ppb: parts per billion • ppm and ppb are used for solutions that contain very small amounts of solute • USEPA set level of lead in drinking water at 15 ppb
Triangle to Solve Equations moles M L
- Slides: 45