Chapter 7 Section 3 4 Describing Acids and

Chapter 7, Section 3 & 4 Describing Acids and Bases Acids & Bases in Solution

Properties of Acids • Taste sour • React with metals and carbonates • Turn blue litmus paper red • Some common acids: Table Talk: Can you think of some other foods that are acidic based on their sour taste? – – – Hydrochloric acid Nitric acid Sulfuric acid Carbonic acid Acetic acid DO NOT RANDOMLY TASTE CHEMICALS!

Acids React With Metals • Acids react with magnesium, zinc, and iron to produce hydrogen gas • Corrosive = they wear away other materials Acids React With Carbonates • Carbonate ion = CO 32 • CO 2 gas is formed Hydrochloric acid on limestone

Acids React With Indicators Litmus paper, made from a dye found in lichens, turns red in an acid, blue in a base How Acids are Commonly Used • Many of the vitamins in foods you eat are acids – Ascorbic acid = vitamin C – Folic acid is in leafy green vegetables • Nitric acid and phosphoric acid are used to make fertilizers • Sulfuric acid is in batteries • Muriatic acid helps clean swimming pools

Table Talk: summarize the properties and uses of acids

Properties of Bases • Taste bitter • Feel slippery • Turn red litmus paper blue • Some common bases: – Sodium hydroxide – Calcium hydroxide – Ammonia Common Uses of Bases • In cement • Cleaning solutions • In baking (baking soda)
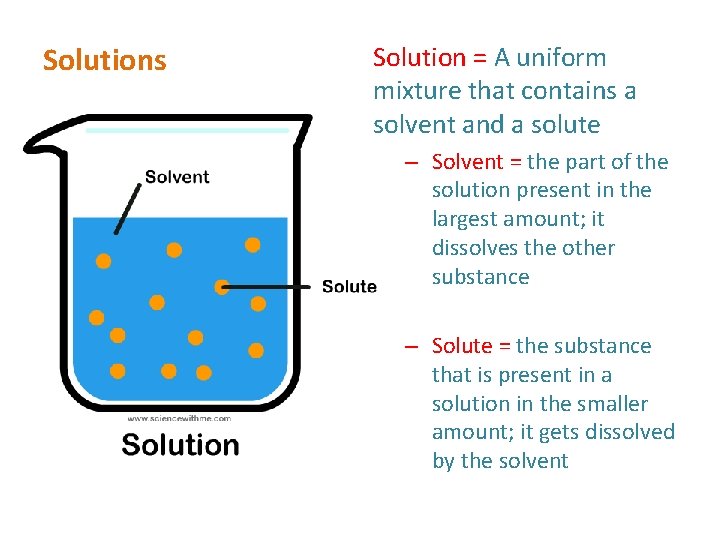
Solutions Solution = A uniform mixture that contains a solvent and a solute – Solvent = the part of the solution present in the largest amount; it dissolves the other substance – Solute = the substance that is present in a solution in the smaller amount; it gets dissolved by the solvent

Acids in Solution • Acids in water solution separate into hydrogen ions (H+) and negative ions • Example: HCl H+ + Cl- Bases in Solution • Most bases are made of positive ions combined with hydroxide ions • When bases dissolve in water, the positive ions and the hyroxide ions separate • Example: NH 3 + H 2 O NH 4+ + OH-

Strength of Acids & Bases “Strength” refers to how well an acid or base produces ions in water Knowing the concentration of hydrogen ions is the key to knowing how acidic or basic a solution is This is measured with the p. H scale, a range of values from 0 -14, with 7 being “neutral” (neither acidic or basic) The lower the p. H, the more concentrated the ions are and the more acidic the solution is. See Brain. Pop “p. H Scale”

Table Talk: If you found that certain foods upset your stomach or gave you heartburn, what could you say about them? How could you ease that uncomfortable feeling?

Acid-Base Reactions • Neutralization = a reaction between an acid and a base • Example: HCl + Na. OH H 2 O + Na+ + Cl- This do-it-yourself volcano is actually an acid-base reaction between vinegar (an acid) and baking soda (a base). The foaming is the formation of CO 2 gas. • After neutralization, a acid-base mixture is less acidic or basic than either of the individual starting solutions

Products of Acid-Base To a chemist, salt = an ionic compound that can be made Reactions from the neutralization of an acid and a base Table Talk: Potassium nitrate, KNO 3, is a salt. Why is it split apart into ions in this equation? Answer: because potassium nitrate is soluble in water. See Brain. Pop “Acids & Bases” & “p. H Scale” In a neutralization reaction, an acid reacts with a base to produce a salt and water: HNO 3 + KOH H 2 O + K+ + NO 3 What do you think would form if a salt was insoluble in water? A ! e t a t i p i c e pr end
- Slides: 12