CHAPTER 7 REACTIONS IN WATER SOLUTIONS Reactions in















- Slides: 23

CHAPTER 7 – REACTIONS IN WATER SOLUTIONS Reactions in water solution involve dissolved ionic compounds and acids DISSOLVED IONIC COMPOUNDS When an ionic compound dissolves in water, the ions separate from each other DISSOCIATION – The separation of ions from an ionic compound during the dissolving process 7 A-1 (of 23)

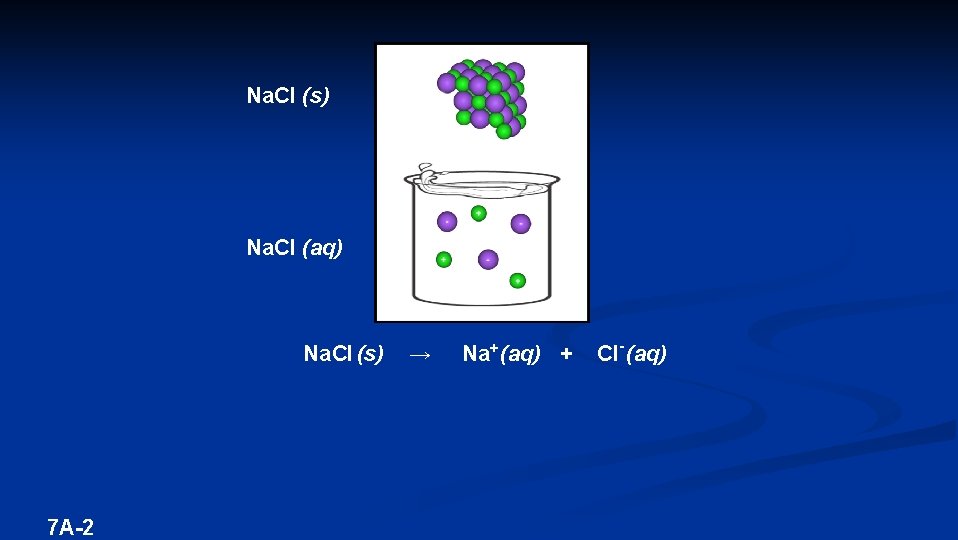
Na. Cl (s) Na. Cl (aq) Na. Cl (s) 7 A-2 → Na+ (aq) + Cl- (aq)

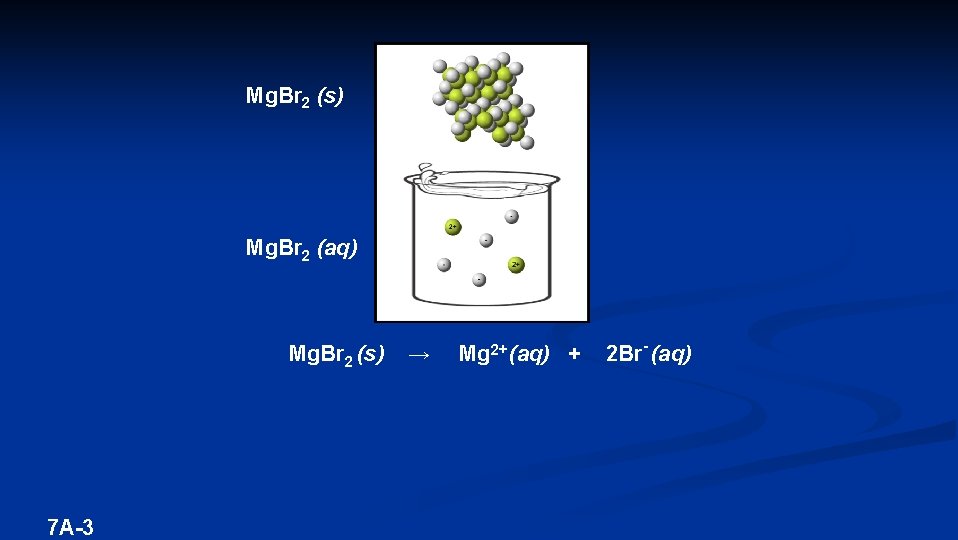
Mg. Br 2 (s) Mg. Br 2 (aq) Mg. Br 2 (s) 7 A-3 → Mg 2+ (aq) + 2 Br- (aq)

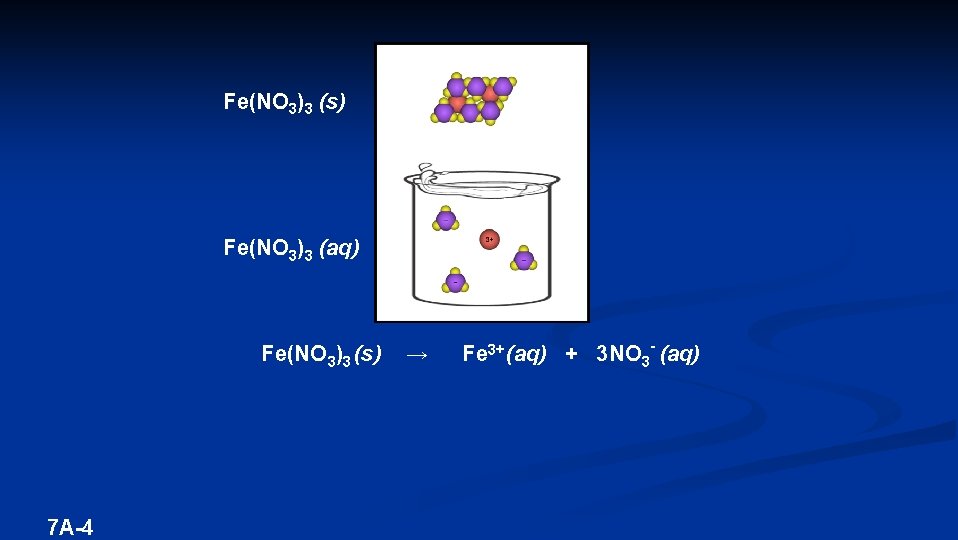
Fe(NO 3)3 (s) Fe(NO 3)3 (aq) Fe(NO 3)3 (s) 7 A-4 → Fe 3+ (aq) + 3 NO 3 - (aq)

PRECIPITATION REACTIONS PRECIPITATE – A solid formed when two solutions are mixed Dissolved ions that have a strong attraction for each other will precipitate out of a solution 7 A-5

5 SOLUBILITY RULES FOR DISSOLVED IONS IN DILUTE SOLUTIONS 1 – Group I and NH 4+ ions never precipitate out of solution with anions 2 – NO 3 - and C 2 H 3 O 2 - ions never precipitate out of solution with cations 3 – Cl-, Br-, and I- ions never precipitate out of solution with cations except with Ag+, Hg 22+, or Pb 2+ ions 4 – SO 42 - ions never precipitate out of solution with cations except with Hg 22+, Pb 2+, Ca 2+, Sr 2+, or Ba 2+ ions 5 – CO 32 -, OH-, PO 43 -, and S 2 - ions do precipitate out of solution with cations except not with Group I and NH 4+ ions 7 A-6

5 SOLUBILITY RULES FOR DISSOLVED IONS IN DILUTE SOLUTIONS 1 – Group I and NH 4+ ions never precipitate out of solution with anions 2 – NO 3 - and C 2 H 3 O 2 - ions never precipitate out of solution with cations 3 – Cl-, Br-, and I- ions never precipitate out of solution with cations except with Ag+, Hg 22+, or Pb 2+ ions 4 – SO 42 - ions never precipitate out of solution with cations except with Hg 22+, Pb 2+, Ca 2+, Sr 2+, or Ba 2+ ions 5 – CO 32 -, OH-, PO 43 -, and S 2 - ions do precipitate out of solution with cations except not with Group I and NH 4+ ions 7 A-7

5 – CO 32 -, OH-, PO 43 -, and S 2 - ions do precipitate out of solution with cations except not with Group I and NH 4+ ions 7 A-8

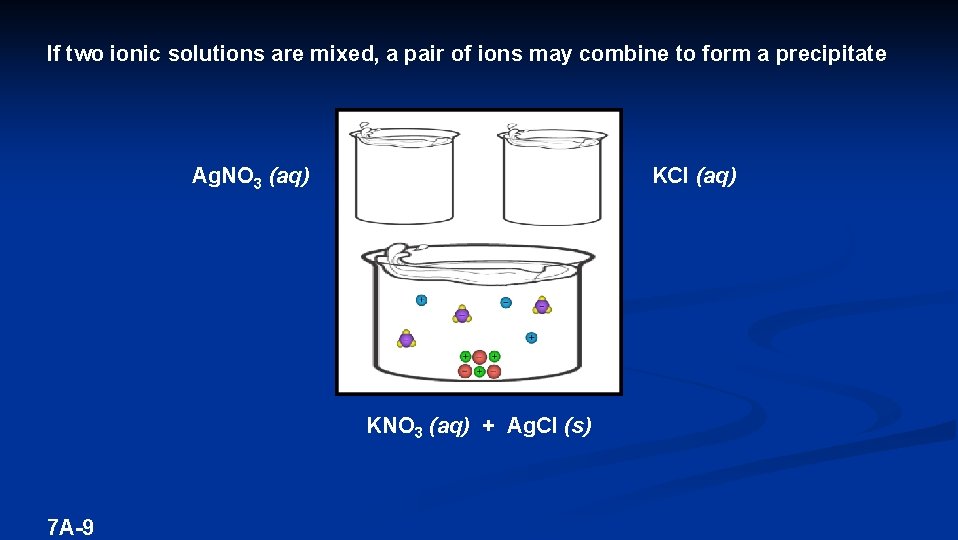
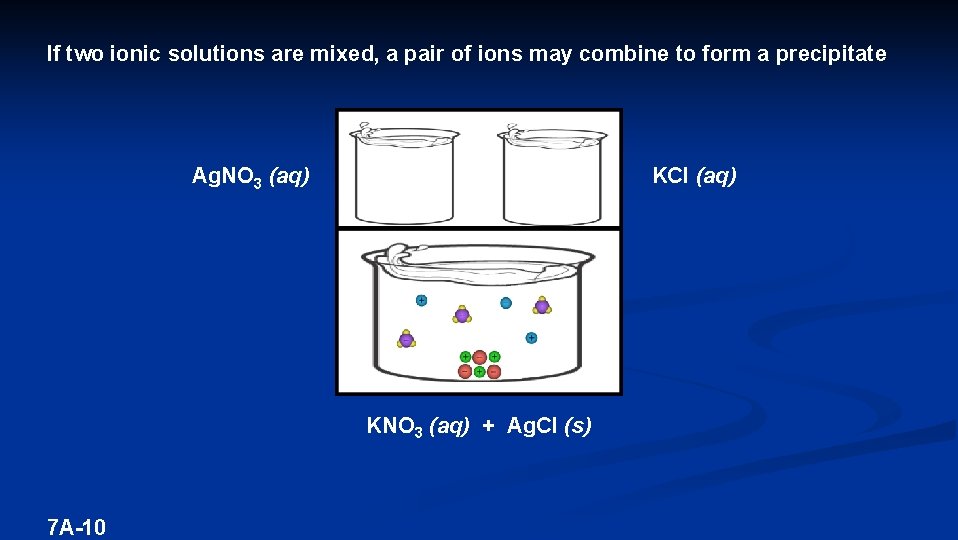
If two ionic solutions are mixed, a pair of ions may combine to form a precipitate Ag. NO 3 (aq) KCl (aq) KNO 3 (aq) + Ag. Cl (s) 7 A-9

If two ionic solutions are mixed, a pair of ions may combine to form a precipitate Ag. NO 3 (aq) KCl (aq) KNO 3 (aq) + Ag. Cl (s) 7 A-10

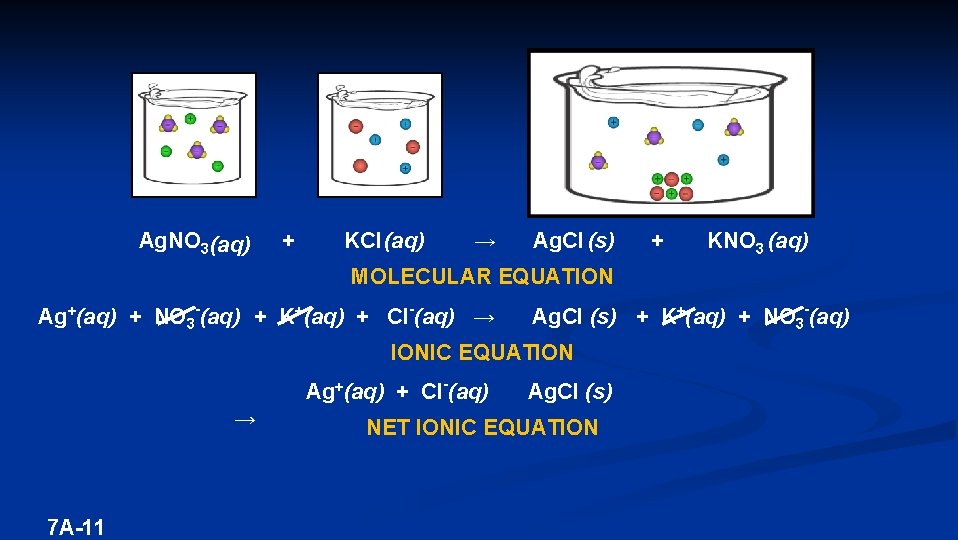
Ag. NO 3 (aq) + KCl (aq) → Ag. Cl (s) + KNO 3 (aq) MOLECULAR EQUATION Ag+(aq) + NO 3 -(aq) + K+(aq) + Cl-(aq) → Ag. Cl (s) + K+(aq) + NO 3 -(aq) IONIC EQUATION Ag+(aq) + Cl-(aq) → 7 A-11 Ag. Cl (s) NET IONIC EQUATION

Solutions of iron (II) chloride and sodium hydroxide are mixed Write in molecular form: (aq) Fe. Cl +2 2 → Write in ionic form: Na. OH (aq) Fe(OH)2 (s) + 2 Na. Cl (aq) Fe 2+(aq) + 2 Cl-(aq) + 2 Na+(aq) + 2 OH-(aq) → Fe(OH)2 (s) + 2 Na+(aq) + 2 Cl-(aq) Write in net ionic form: Fe 2+(aq) + 2 OH-(aq) → Fe(OH)2 (s) 7 A-12

Solutions of potassium sulfate and barium nitrate are mixed Write in molecular form: (aq) K 2 SO 4 + → Write in ionic form: (aq) Ba(NO(aq) 3 )2 7 A-13 Ba. SO 4 (s) 2 K+(aq) + SO 42 - + Ba 2+(aq) + 2 NO 3 -(aq) → 2 K+(aq) + 2 NO 3 -(aq) + Ba. SO 4 (s) Write in net ionic form: → 2 KNO 3 (aq) + SO 42 -(aq) + Ba 2+(aq) Ba. SO 4 (s)

Solutions of aluminum chloride and sodium carbonate are mixed Write in molecular form: 2 Al. Cl (aq) 3 3 + Na 2 CO (aq) 3 →Al 2(CO 3)3 (s) + 6 Na. Cl (aq) Write in ionic form: 2 Al 3+(aq) + 6 Cl(aq) Write in net ionic form: + 6 Na+(aq) + 3 CO 32 -(aq) → (s) Al 2(CO 3)3 + 6 Na+(aq) + 6 Cl-(aq) 2 Al 3+(aq) + 2 CO 32 -(aq) → Al 2(CO 3)3 (s) 7 A-14

STRONG ELECTROLYTE – A compound that dissolves in water and produces many ions Water soluble ionic compounds are strong electrolytes Electrolyte solutions conduct electricity because the dissolved ions are free to move throughout the solution Strong electrolyte solutions conduct electricity well 7 A-15

ACIDS When dissolved in water, acid molecules break apart into hydrogen ions and their corresponding anions IONIZATION – The formation of ions from a molecular compound during the dissolving process 7 A-16

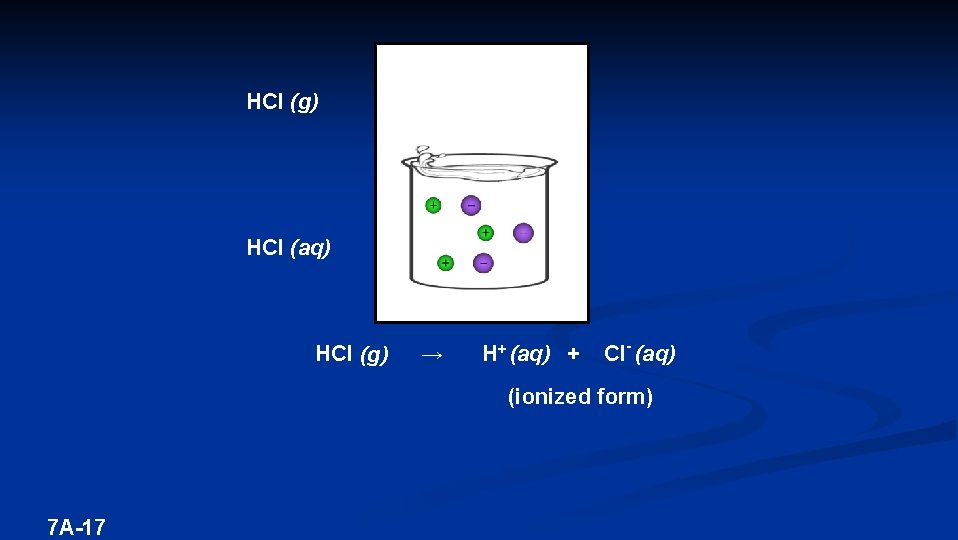
HCl (g) HCl (aq) HCl (g) → H+ (aq) + Cl- (aq) (ionized form) 7 A-17

STRONG ACID – An acid in which all of the molecules release hydrogen ions in water solution HCl, HBr, HI, and oxyacids with at least 2 more O’s than H’s HNO 3 H 2 SO 4 HCl. O 4 H 2 CO 3 Strong acids are strong electrolytes like soluble ionic compounds because their solutions contain many ions and conduct electricity well Strong acids are written in ionized form in ionic equations 7 A-18

Solutions of hydroiodic acid and barium hydroxide are mixed Write in molecular form: 2 (aq) HI + → Write in ionic form: 2 H+(aq) + 2 I- Ba(OH) (aq) 2 2 H(OH) (l) Write in net ionic form: (l) 2 H+(aq) + 2 OH-(aq) → 2 H 2 O (l) 7 A-19 Ba. I 2 (aq) + Ba 2+(aq) + 2 OH-(aq) → (aq) H+(aq) + + OH-(aq) →H 2 O (l) 2 H 2 O + Ba 2+(aq) + 2 I-(aq)

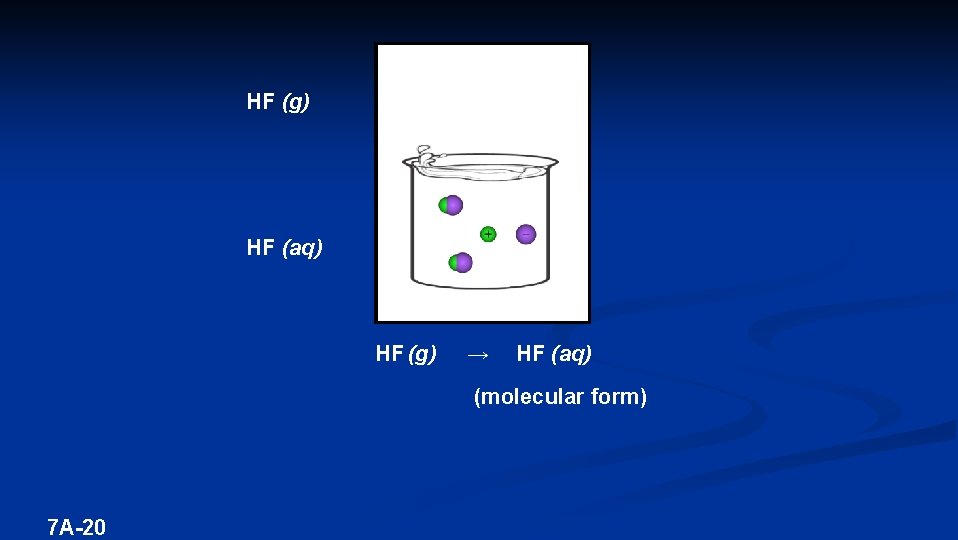
HF (g) HF (aq) HF (g) → HF (aq) (molecular form) 7 A-20

WEAK ACID – An acid in which all of the molecules do not release hydrogen ions in water solution All acids except HCl, HBr, HI, and oxyacids with at least 2 more O’s than H’s Weak acids are weak electrolytes because their solutions contain small numbers of ions and barely conduct electricity Weak acids are written in molecular form in ionic equations 7 A-21

Solutions of sulfurous acid and potassium hydroxide are mixed Write in molecular form: (aq) H 2 SO 3 2 + (aq) KOH → 2 H(OH) (l) + K 2 SO 3 (aq) Write in ionic form: H 2 SO 3 (aq) + 2 K+(aq) + 2 OH-(aq) → Write in net ionic form: → 7 A-22 H 2 SO 3 (aq) + 2 OH-(aq) (l) 2 H 2 O + 2 K+(aq) + SO 32 - (aq) 2 H 2 O (l) + SO 32 - (aq)

Solid copper (II) oxide is added to a solution of hydrochloric acid Write in molecular form: Cu. O (s) +2 HCl (aq) Cu. Cl 2 (aq) → Write in ionic form: Cu. O (s) + 2 H+(aq) + 2 Cl-(aq) → → 7 A-23 H 2 O (l) Cu 2+(aq) + 2 Cl-(aq) + H 2 O (l) Write in net ionic form: Cu. O (s) + 2 H+(aq) + Cu 2+(aq) + 2 H 2 O (l)