Chapter 7 Properties of Ionic Covalent and Metal













- Slides: 21

Chapter 7: Properties of Ionic Covalent and Metal Materials

Types of Atoms Ionic Compounds: Ions (Cations & Anions) Covalent (Molecular Compounds): Two or more nonmetal atoms Metallic Solids: Only metal atoms

Bond formation Ionic Compounds: Transfer of electrons Form Ionic bonds Covalent (Molecular Compounds): Sharing of electrons Form Covalent bonds Metallic Solids: Free moving valence electrons

Properties Ionic Compounds: • At room temperature they are a crystalline solid. • Hard & Brittle, High melting point Covalent (Molecular Compounds): • At room temperature they can be a low -melting pt solid, liquid or gas. • Usually soft & low melting point

Properties Metallic Solids: • Soft to very hard solids Why? Strength increases with an increase in # of e- available for bonding. • Low to very high melting points • Ductile: Metal can be drawn into thin wires • Malleable: Metal can be hammered into different shapes

Compound Arrangement Ionic Compound • Three dimensional pattern How is the 3 D pattern created? • Opposite charged ions form close-packed layers. • Layers stack together to make a regular repeating 3 D pattern called a: Unit cell • Closely packed layers will keep stacking to form a: Crystal

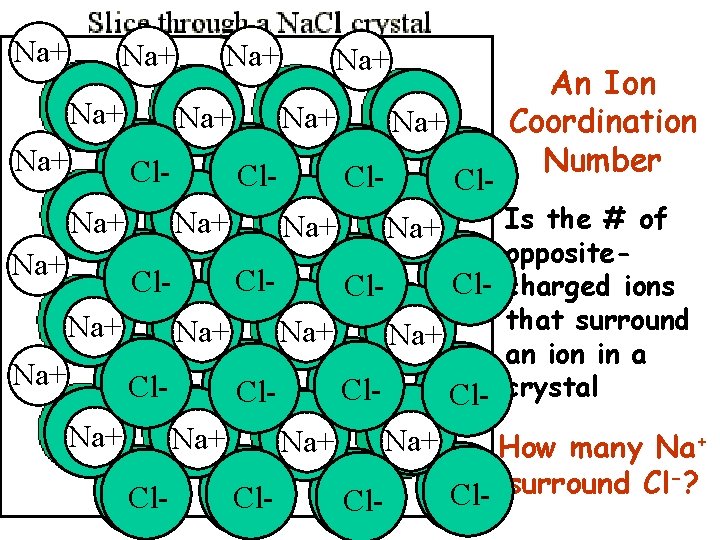
Na+ Na+ An Ion Cl. Na+ Na+ Coordination Number Na+ Na+ Cl. Cl. Is the # of Cl. Na+ Na+ Cl. Na+ Na+ opposite. Na+ Na+ Cl. Cl- charged ions Cl- that surround Cl. Na+ Na+ Na+ Cl. Na+ an ion in a Na+ Na+ Cl. Cl. Cl- crystal Cl. Na+ Na+ Cl. Na+ How many Na+ Cl. Cl- -? surround Cl Cl. Cl-

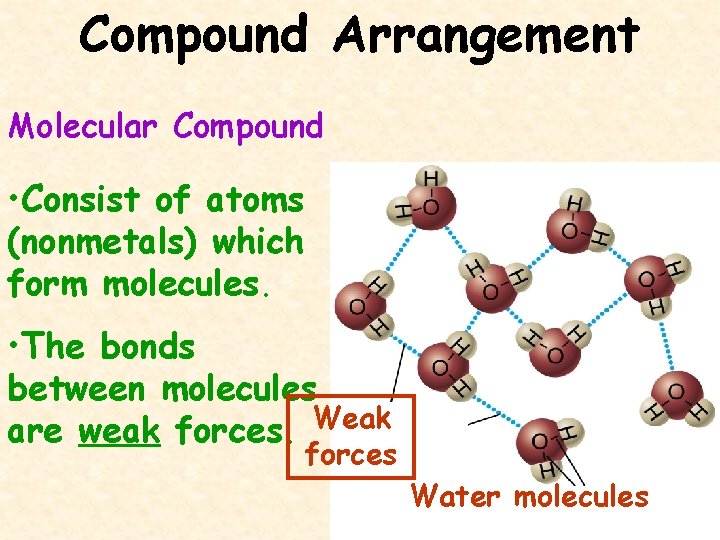
Compound Arrangement Molecular Compound • Consist of atoms (nonmetals) which form molecules. • The bonds between molecules are weak forces. Weak forces Water molecules

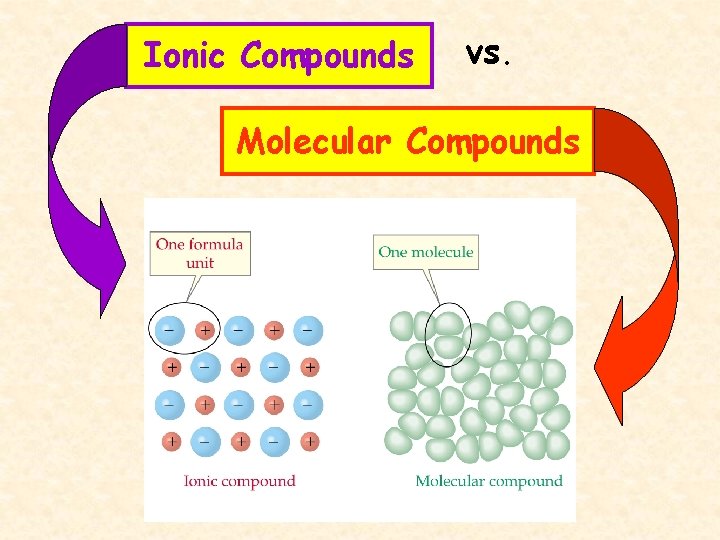
Ionic Compounds vs. Molecular Compounds

Compound Arrangement Metallic Solid • Consist of closely packed cations. How are metallic bonds formed? • They are formed from the attraction of free-moving valence electrons for the cations • These electrons bind the cations together to create a metallic solid.

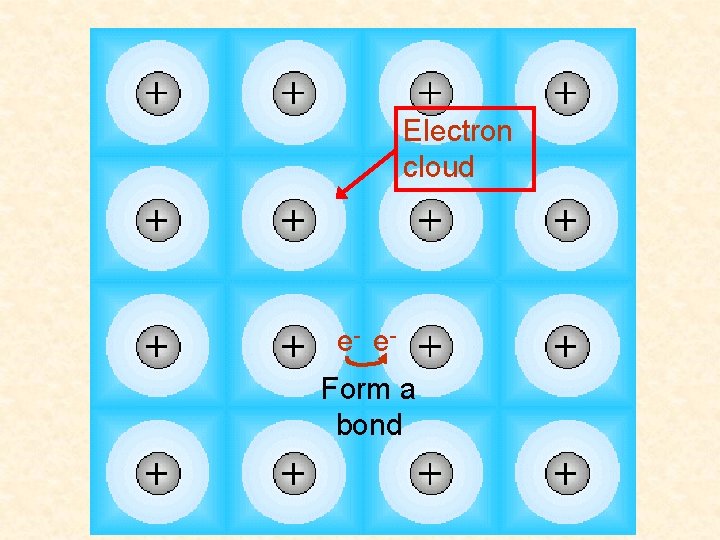
Electron cloud e- e. Form a bond

Type of Bond Force & Strength Ionic Compounds: Electrostatic forces Very Strong Covalent (Molecular Compounds): Intermolecular forces Weak Dispersion forces, Dipole-dipole, H-bonding Metallic Solids: A “sea” of Valence Electrons Strong

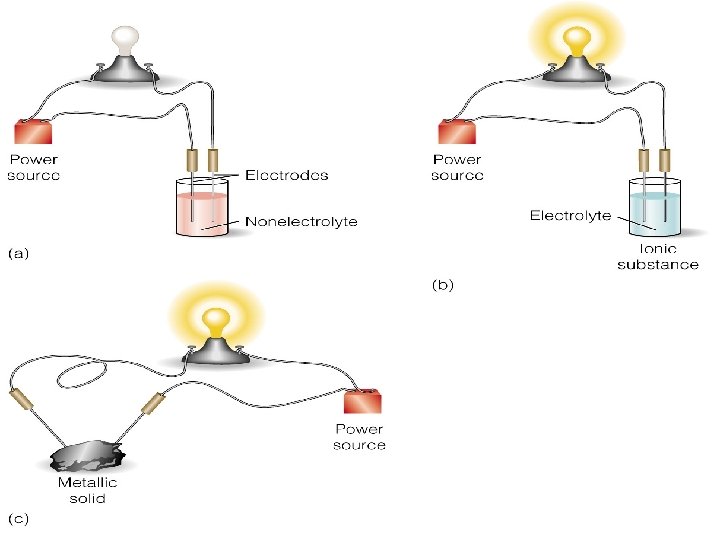
Conductors Ionic Compounds: Good conductors of electricity (dissolved ionic compounds are electrolytes) Two ways ionic compounds conduct electricity: § Conduct an electric current in the melted state. § Conduct electricity when the compound is dissolved in water.

Conductors Ionic Compounds cont… How do ionic compounds conduct electricity? 1. Bonds break (melt) or dissociate (dissolve). 2. The ions separate and are free to move around. The ion 3. Apply voltage allowing movement the cations move to one produces a electrode & the anions to flow of the other. electricity!


Conductors Molecular Compounds: Why? Poor conductors • Intermolecular bonds will break easily, but not form ions. Covalent bonds (holding the molecule together) do not break easily. • Molecular compounds do not readily disassociate in water. • Molecular compounds do not contain ions that can conduct electricity.

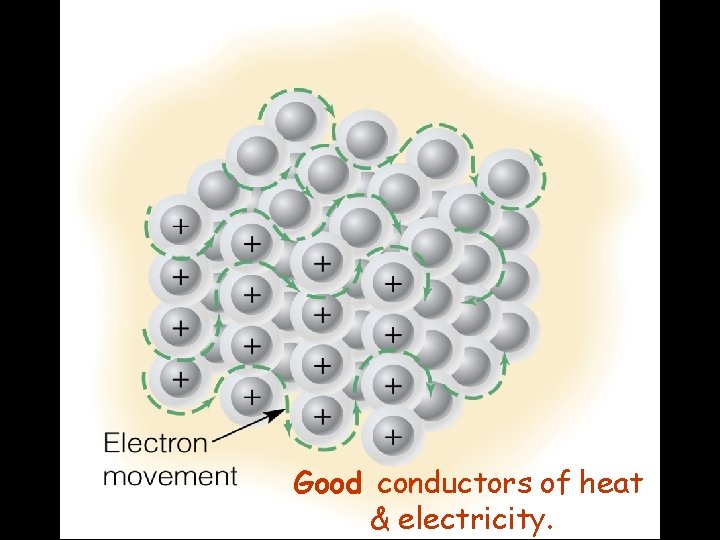
Conductors Metallic Solids: Good conductors of electricity Why? • Free-moving valence electrons Good conductors of heat Why? • Free-moving valence electrons

Good conductors of heat & electricity.

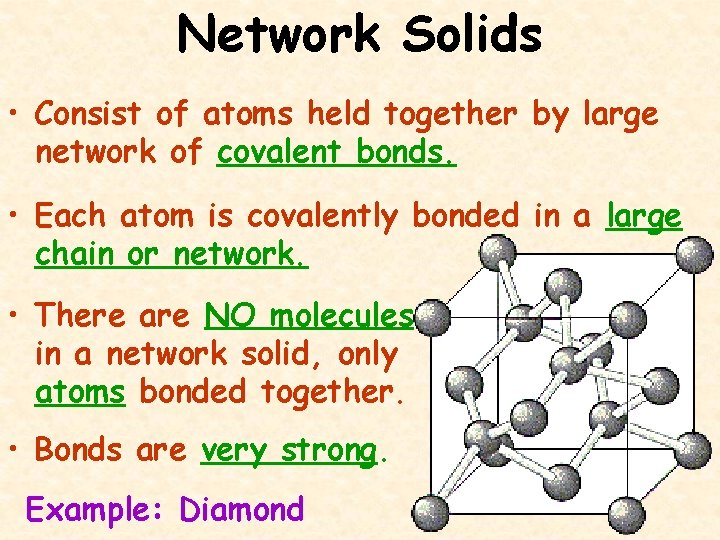
Network Solids • Consist of atoms held together by large network of covalent bonds. • Each atom is covalently bonded in a large chain or network. • There are NO molecules in a network solid, only atoms bonded together. • Bonds are very strong. Example: Diamond

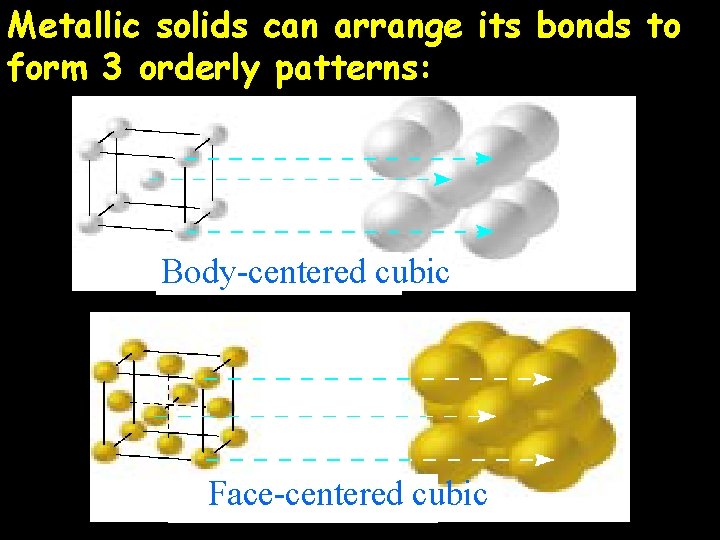
Metallic solids can arrange its bonds to form 3 orderly patterns: Body-centered cubic Face-centered cubic

Hexagonal close-packed Why are metallic solids the simplest crystalline solid?