Chapter 7 Periodic Properties Overview u Periodic Table

Chapter 7 Periodic Properties

Overview u Periodic Table u Electron Shells & Sizes of Atoms u Ionization Energy u Electron Affinities u Metals, Nonmetals & Metalloids and Trends

Periodic Table u 1869 -- Dmitri Mendeleev & Lothar Meyer ° published classification schemes of elements ° based on order of increasing elemental atomic weight · which follows atomic number ° some missing elements were “discovered” based upon prediction that they should be there ¨ Henry Moseley ° related energy of nuclear x-rays to atomic number

Sizes of Atoms u Electron Shells oquantum mechanical model predicts shells of electron density oas number of protons increases, inner shells are held more tightly, closer to nucleus u Sizes of Atoms oatom boundaries are difficult to assess odefine atom size as ½ the distance between nuclei of two bound atoms
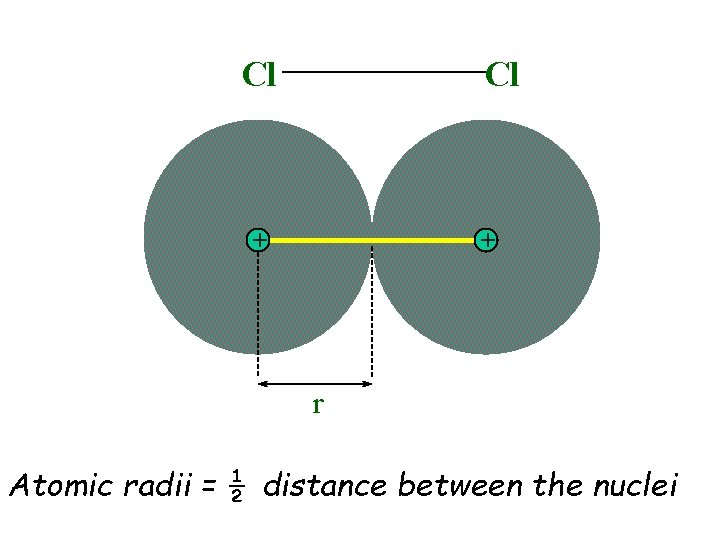
Cl Cl + + r Atomic radii = ½ distance between the nuclei

General Trends of Physical Properties u most trends are related to Zeff, effective nuclear charge odue to increase in distance from nucleus and shielding, Zeff decreases going down a group odue to ineffective shielding by electrons in the same shell, Zeff increases going across a row Zeff decreases increases

u Compare valence e- ‘s of Na and Cs owhich will be held more tightly by nucleus? • the e- that is closest to the nucleus owhich will be closest to the nucleus? • Na’s 3 s electron u Compare last electron of C and F owhich has more protons interacting? • ZF = 9 while ZC = 4 • do e- in the same subshell shield their fellow e- ‘s very well? – no • so which last e- will experience the greatest Zeff? –F

General Trends in Atomic Size u u radius increases on going down a group (Zeff decreases) radius tends to decrease on going left to right across a row (Zeff increases) size increases decreases

u We know that Zeff decreases going down a group, how does that affect size? o. As Zeff (on a valence e- ) decreases, it is less tightly held, hence can expand somewhat u We know that Zeff increases going across a row, how does that affect size? o. As Zeff (on a last e-) increases, it is more tightly held, hence contraction occurs and atom is smaller

u Compare valence e- ‘s of Na and Cs owhich experiences the greatest Zeff ? • Na’s 3 s electron (bec. it is closest to nucleus) owhich atom will be smaller? • Na < Cs u Compare last electron of C and F owhich has more protons interacting? • ZF = 9 while ZC = 4 • do e- in the same subshell shield their fellow e- ‘s very well? – no • so which last e- will experience the greatest Zeff? –F • which atom will experience greatest contraction? – F will be more contracted, hence smaller
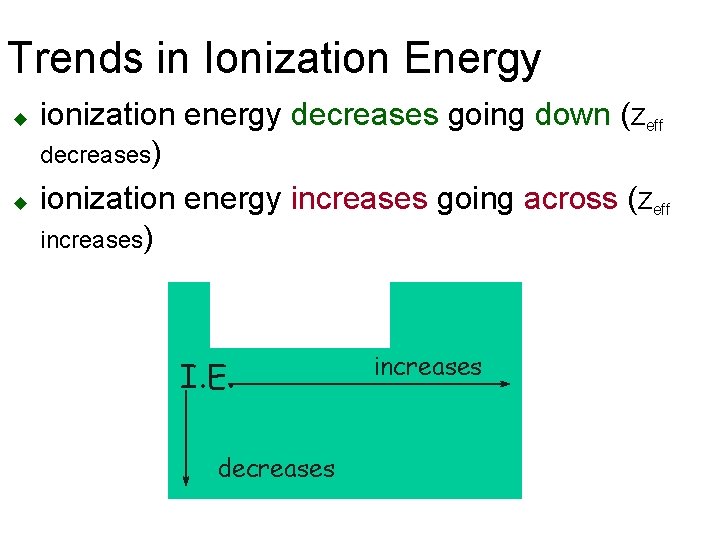
Trends in Ionization Energy u u ionization energy decreases going down (Zeff decreases) ionization energy increases going across (Zeff increases) I. E. decreases increases

u Ionization Energy -- endothermic process ofirst ionization energy, I 1 -- to remove first electron • M ® M+ + 1 e - osecond ionization energy, I 2 -- to remove second electron • M+ ® M 2+ + 1 e - othird ionization energy, I 3 -- to remove third electron • M 2+ ® M 3+ + 1 e u I 1 < I 2 < I 3

u Note: removal of inner core electrons is very energy expensive Element I 1 I 2 I 3 Na 496 4560 Mg 738 1450 7730 Al 578 1820 2750 I 4 11, 600

u The more tightly an e- is held, the more energy it takes to remove it oin other words, greater Zeff = greater I. E. u Compare valence e- ‘s of Na and Cs owhich has greater Zeff? • Na owhich has greater I 1? • Na (Cs’s 6 s electron is further away, less tightly held and easier to remove than Na’s 3 s electron) u Compare last e- of C and F owhich has greater Zeff? • F owhich has greater I 1? • F (more tightly held, more energy to remove)

u u u transition metals and f-block metals show small variation in ionization energies across rows representative elements show a larger range of values for I 1 irregularities in ionization energies occur due to shielding o. I 1 for Al < I 1 for Mg -- removal of p versus s electron o. I 1 for O < I 1 for N -- removal of p electron to produce stable configuration

Electron Affinities u energy change associated with adding an electron to gaseous atom omeasures attraction of atom for the added electron • Cl(g) + 1 e - ® Cl - DE = -349 k. J/mol oexothermic process u most electron affinities are exothermic osome exceptions & irregularities • nobel gases • Be & Mg • Group III elements

Metals u general trends and properties oto the left on periodic table oability to lose electrons (low ionization energies) -- what kind of ions? ohave a metallic or shiny luster, ductile oform crystalline solids ohave good thermal and electrical conductivity otransition metals vary in charge • most have +2 in addition to other charges (+1 to +5) oform ionic compounds with non-metals • especially with oxides or halides

ometal oxides are called basic oxides • react with water to form basic hydroxides • Na 2 O(s) + H 2 O(l) ® 2 Na. OH(aq) • react with acid to form water and salt

Nonmetals u general trends and properties oto the right on periodic table oability to gain electrons -- what kind of ions? onon-conductors and thermal insulators ohave lower melting points – are molecular solids rather than ionic solids oseven exist as diatomic molecules – H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2

oform molecular compounds with other nonmetals such as oxides, halides & hydrides ononmetal oxides are called acidic oxides • • u react with water to form acids CO 2(g) + H 2 O(l) ® H 2 CO 3(aq) react with bases to form salt and water CO 2(g) + 2 Na. OH(aq) ® Na 2 CO 3(aq) + H 2 O(l) Metalloids ohave a mixture of properties some metallic, some not

Group Trends for Active Metals u Group 1 A -- Alkali Metals overy reactive, only found in nature in oxidized form (compounds) ometals produced by electrolysis of molten compounds oreactivity increases down the group oreact rapidly with water 2 K(s) + 2 H 2 O(l) ® 2 KOH(aq) + H 2(g) oreact with acid: 2 K(s) + 2 HCl(aq) ® 2 KCl(aq) + H 2(g)

oreact with hydrogen to form hydrides • 2 K(s) + H 2(g) ® 2 KH(s) oreact with oxygen in different ways • 4 Li(s) + O 2(g) ® 2 Li 2 O(s) containing O 2 • this is the more common reaction oother alkali metals form peroxides, O 22 • 2 Na(s) + O 2(g) ® Na 2 O 2(s) o. K, Rb, Cs form superoxides, O 2 - • K(s) + O 2(g) ® KO 2(s) • peroxides and superoxides are not as common omost alkali metal compounds are colorless

u Group 2 A -- Alkaline Earth Metals oreactive – less than alkali metals (ionization energies higher) oreactivity increases down the group oheavier metals react with water to form bases • Ca(s) + 2 H 2 O(l) ® Ca(OH)2(aq) + H 2(g) • lighter metals react similarly with hot water or steam oreact with acids: • Mg(s) + 2 HCl(aq) ® Mg. Cl 2(aq) + H 2(g) oreact with O to produce oxides • 2 Mg(s) + O 2(g) ® 2 Mg. O(s)

Trends for Nonmetals u Hydrogen ononmetal, diatomic, colorless gas ohigh ionization energy (no shielding) otypically forms molecular compounds with other nonmetals • these reactions are fairly exothermic • 2 H 2(g) + O 2(g) ® 2 H 2 O(l) DHo = -571. 7 k. J/mol oforms hydrides (H-) with metals • 2 Na(s) + H 2(g) ® 2 Na. H(s)

u Group 6 A: Oxygen Group odensity increases, metallic character increases down group ooxygen is diatomic, & the only gas in the group oallotrope (different form) is ozone, O 3 • 3 O 2(g) ® 2 O 3(g) DHo = +284. 6 k. J/mol • is ozone more or less stable than O 2? less oions are in different forms • O 2 -, oxide • O 22 -, peroxide • O 2 -, superoxide

u Group 7 A: Halogens oincreased nonmetallic character olighter elements are diatomic gases & more reactive than heavier elements ohave very exothermic electron affinities and gain electron easily o. F has highest electron affinity • 2 Na(s) + F 2(g) ® 2 Na. F(s) DHo = -1147 k. J • 2 H 2 O(l) + 2 F 2(g) ® 4 HF(aq) DHo = -758. 7 k. J o. Cl reacts somewhat more slowly • Cl 2(g) + H 2 O(l) ® HCl(aq) + HOCl(aq)

u Group 8 A: Noble Gases oall are monotomic, quite stable and unreactive o. Xe was the first noble gas compound identified • Xe. F 6 o. Kr is less reactive & has one known compound • Kr. F 2 ono compounds of He, Ne or Ar are known • however, there have been implications of a possible Ar compound
- Slides: 27