Chapter 7 Membrane Structure and Function Cell Membrane
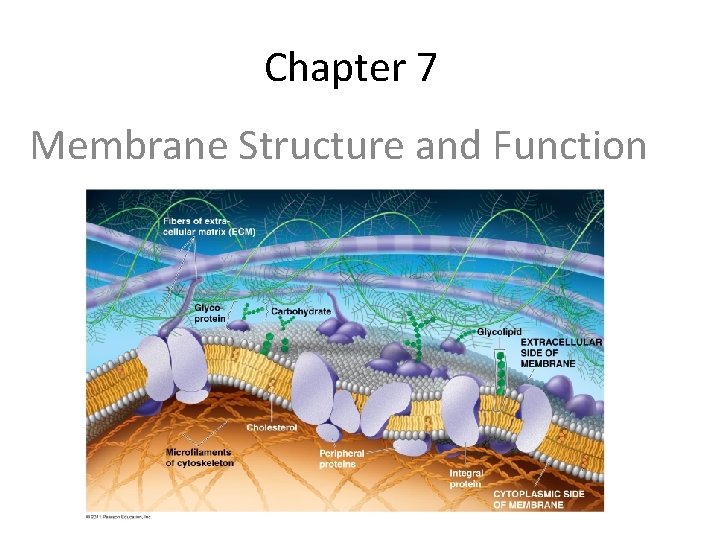
Chapter 7 Membrane Structure and Function

Cell Membrane Plasma membrane is selectively permeable – Allows some substances to cross more easily than others • hydrophobic (nonpolar) vs. hydrophilic (polar) – Cell membrane separates living cell from aqueous environment • Fluid Mosaic Model – Fluid: membrane held together by weak interactions – Mosaic: phospholipids, proteins, carbs
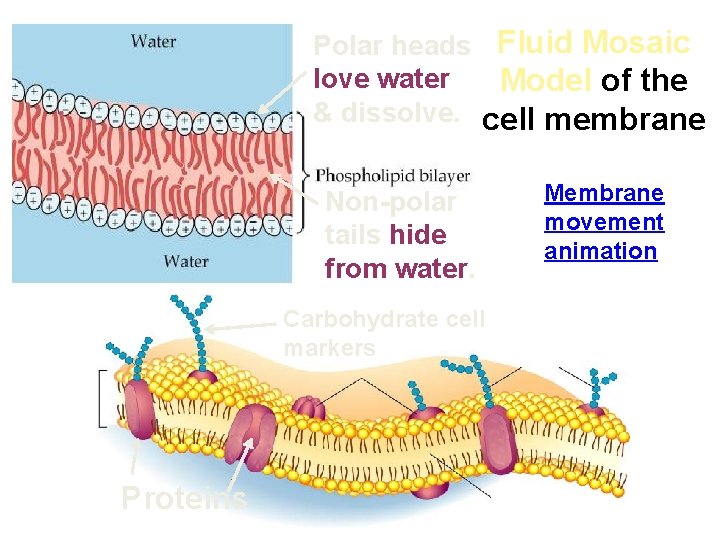
Polar heads Fluid Mosaic love water Model of the & dissolve. cell membrane Non-polar tails hide from water. Carbohydrate cell markers Proteins Membrane movement animation
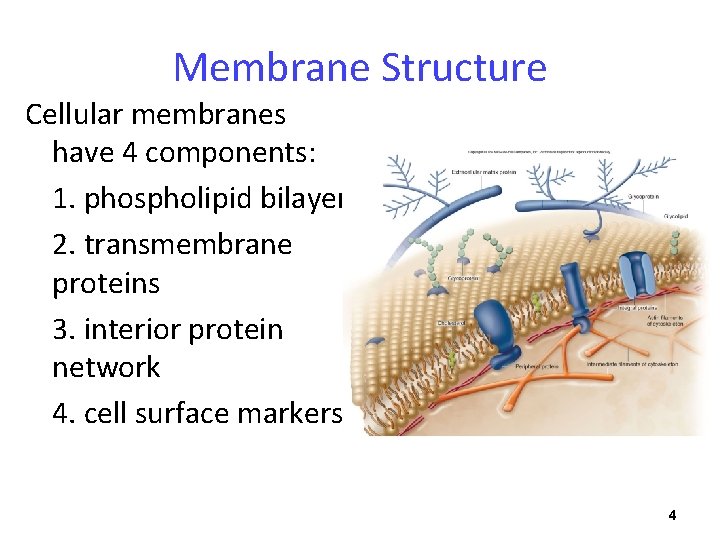
Membrane Structure Cellular membranes have 4 components: 1. phospholipid bilayer 2. transmembrane proteins 3. interior protein network 4. cell surface markers 4

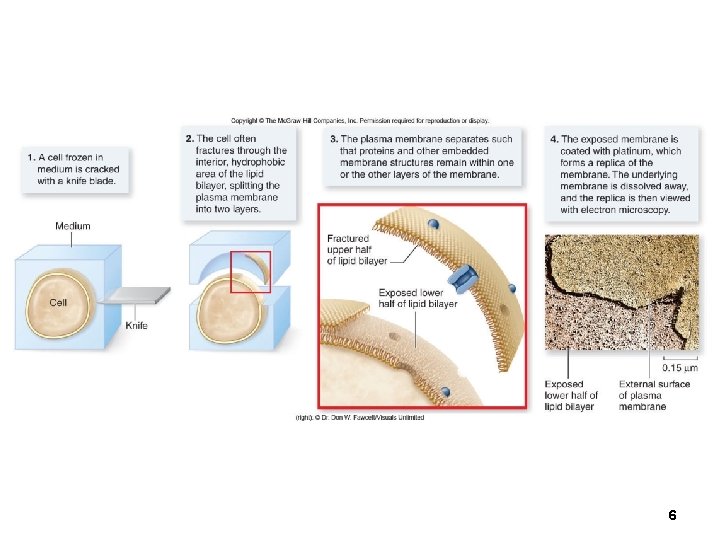
Membrane Structure Membrane structure is visible using an electron microscope. Transmission electron microscopes (TEM) can show the 2 layers of a membrane. Freeze-fracturing techniques separate the layers and reveal membrane proteins. 5
6
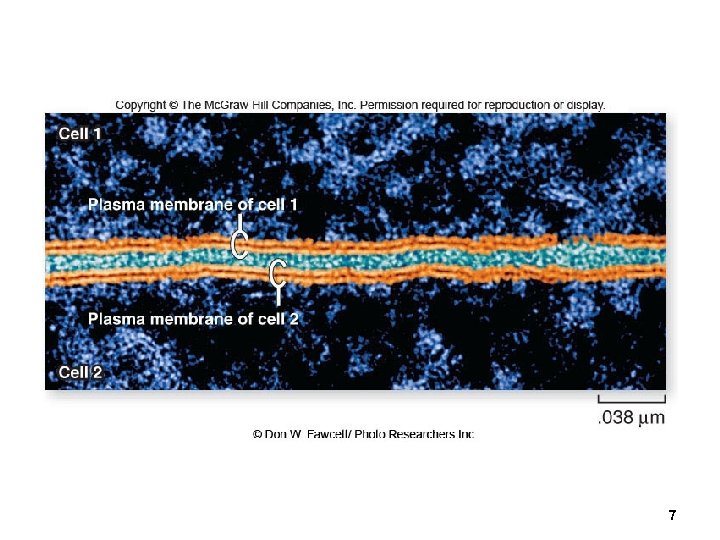
7
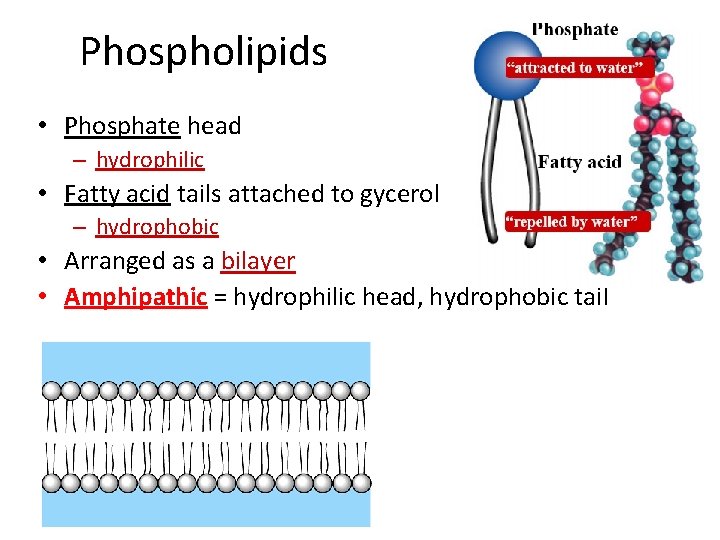
Phospholipids • Phosphate head – hydrophilic • Fatty acid tails attached to gycerol – hydrophobic • Arranged as a bilayer • Amphipathic = hydrophilic head, hydrophobic tail
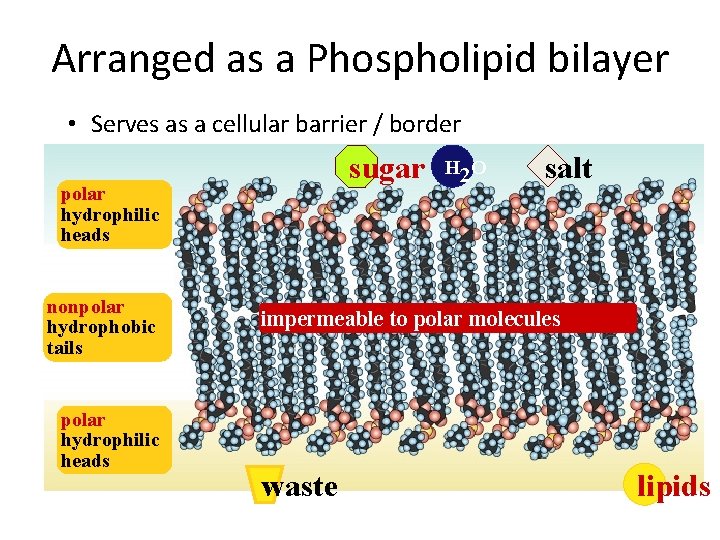
Arranged as a Phospholipid bilayer • Serves as a cellular barrier / border sugar polar hydrophilic heads nonpolar hydrophobic tails polar hydrophilic heads H 2 O salt impermeable to polar molecules waste lipids

Phospholipids Phospholipid bilayers are fluid -hydrogen bonding of water holds the 2 layers together -individual phospholipids and unanchored proteins can move through the membrane -saturated fatty acids make the membrane less fluid than unsaturated fatty acids -warm temperatures make the membrane more fluid than cold temperatures
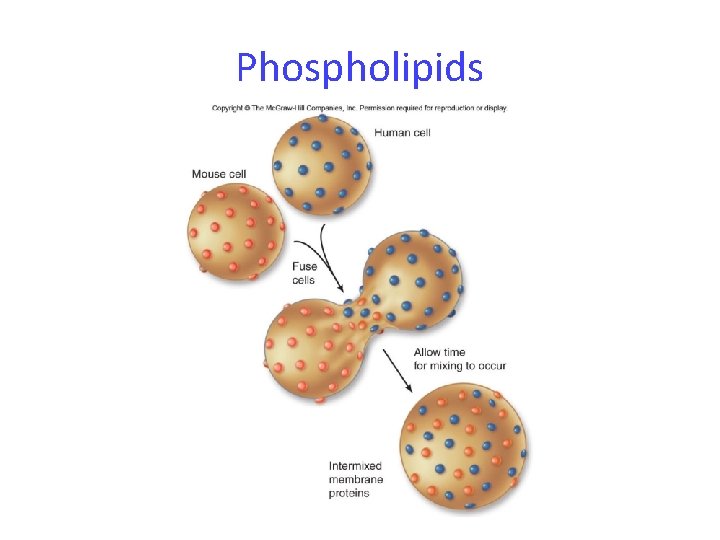
Phospholipids
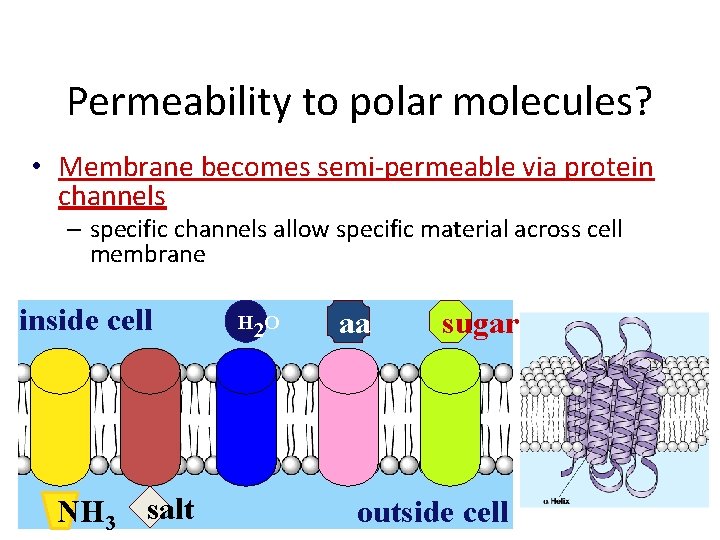
Permeability to polar molecules? • Membrane becomes semi-permeable via protein channels – specific channels allow specific material across cell membrane inside cell NH 3 salt H 2 O aa sugar outside cell
Why are proteins the perfect molecule to build structures in the cell membrane? 2007 -2008
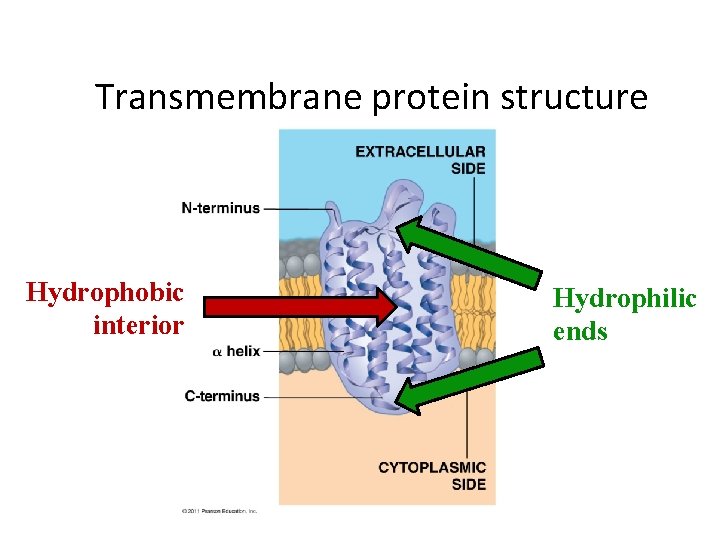
Transmembrane protein structure Hydrophobic interior Hydrophilic ends
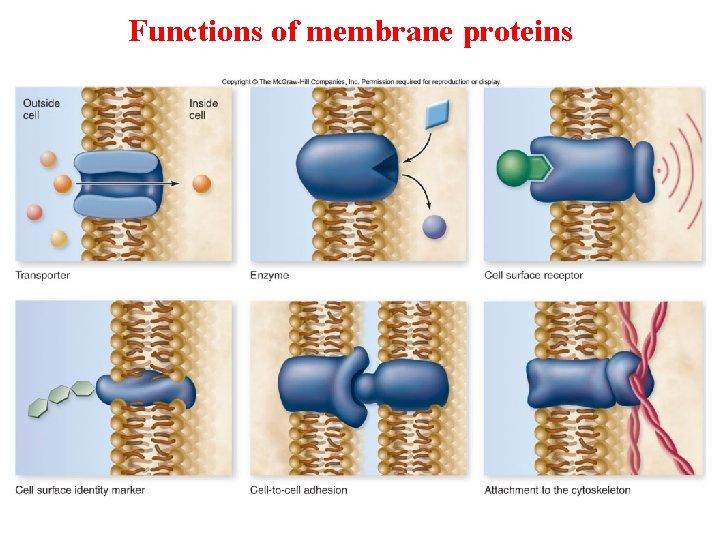
Functions of membrane proteins

Membrane Proteins Integral Proteins Peripheral Proteins • Span the length of membrane • Transmembrane with hydrophilic heads/tails and hydrophobic middles • Extracellular or cytoplasmic sides of membrane • NOT embedded, loosely bound • Held in place by the cytoskeleton or ECM • Provides stronger framework • antigens • ex: transport proteins – channels, permeases (pumps)
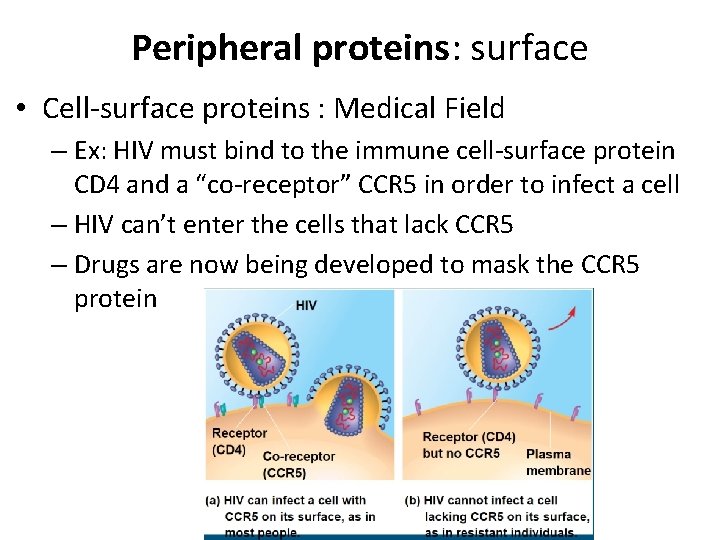
Peripheral proteins: surface • Cell-surface proteins : Medical Field – Ex: HIV must bind to the immune cell-surface protein CD 4 and a “co-receptor” CCR 5 in order to infect a cell – HIV can’t enter the cells that lack CCR 5 – Drugs are now being developed to mask the CCR 5 protein

Membrane fluidity • Low temps: phospholipids w/unsaturated tails (kinks prevent close packing) • Cholesterol resists changes by: – limit fluidity at high temps – hinder close packing at low temps
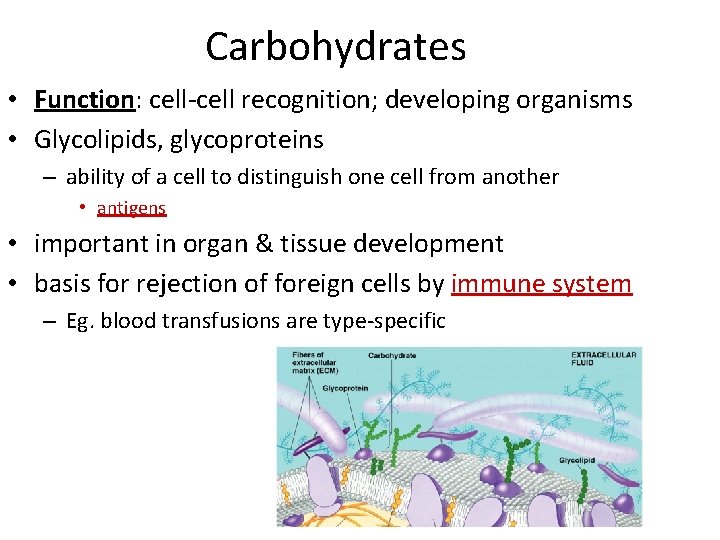
Carbohydrates • Function: cell-cell recognition; developing organisms • Glycolipids, glycoproteins – ability of a cell to distinguish one cell from another • antigens • important in organ & tissue development • basis for rejection of foreign cells by immune system – Eg. blood transfusions are type-specific

Selective Permeability • Small molecules (polar or nonpolar) cross easily (hydrocarbons, hydrophobic molecules, CO 2, O 2) • Hydrophobic core prevents passage of ions, large polar molecules

Evolution of Differences in Membranes • Variations in lipid composition of many species – adaptations to specific environmental conditions – arctic fish → high proportion of unsaturated tails – plants in winter → % unsaturated tails increases in autumn (winter wheat) – bacteria in hot springs – unusual lipids • Natural Selection

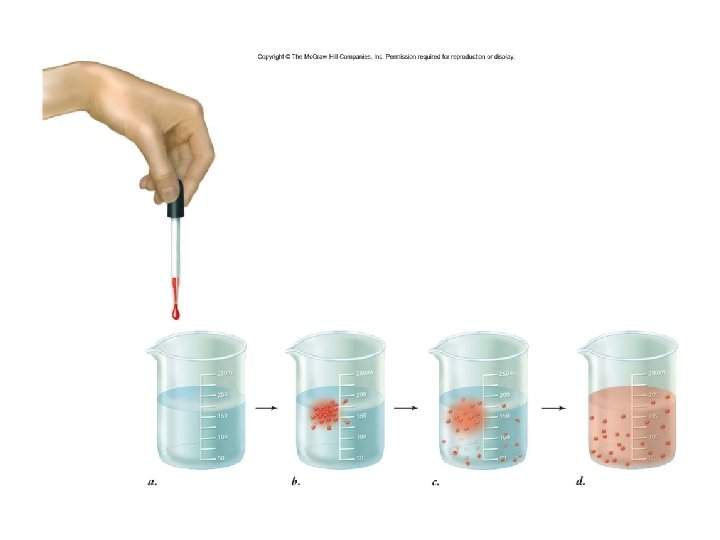
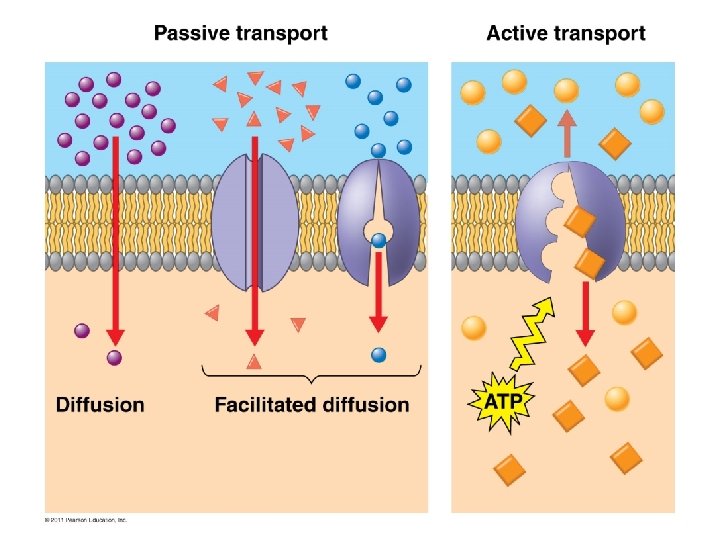
Passive Transport • NO ENERGY (ATP) needed! • Diffusion down concentration gradient – (high low concentration) – Eg. hydrocarbons, CO 2, H 2 O • Particles flow until equilibrium is reached – Dynamic equilibrium - Continuous particle movement w/no net change in concentration • Diffusion = particles moving across membrane • Osmosis = water moving across membrane
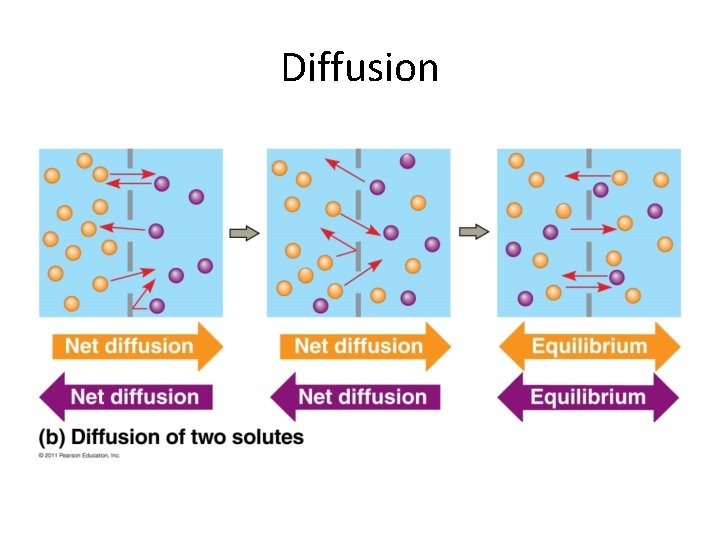
Diffusion

Facilitated Transport A type of passive transport Selective permeability: integral membrane proteins allow the cell to be selective about what passes through the membrane. Channel proteins have a polar interior allowing polar molecules to pass through. Carrier proteins bind to a specific molecule to facilitate its passage.

Passive Transport Channel proteins include: -ion channels allow the passage of ions (charged atoms or molecules) which are associated with water -gated channels are opened or closed in response to a stimulus -the stimulus may be chemical or electrical
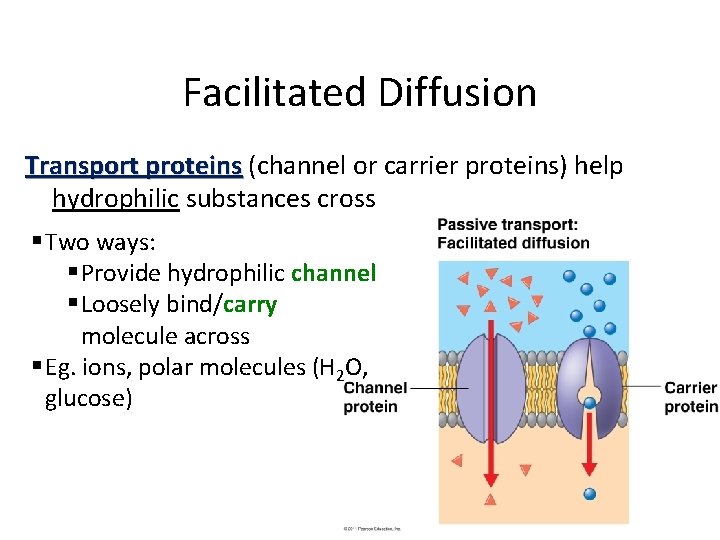
Facilitated Diffusion Transport proteins (channel or carrier proteins) help hydrophilic substances cross § Two ways: § Provide hydrophilic channel § Loosely bind/carry molecule across § Eg. ions, polar molecules (H 2 O, glucose)

Aquaporins 1991 | 2003 • Water moves rapidly into & out of cells – evidence that there water channels • protein channels allowing flow of water across cell membrane Peter Agre Roderick Mac. Kinnon John Hopkins Rockefeller
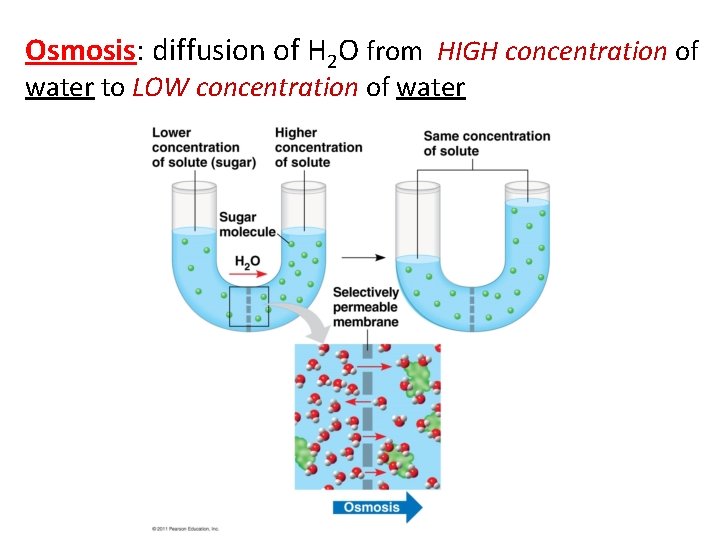
Osmosis: diffusion of H 2 O from HIGH concentration of water to LOW concentration of water
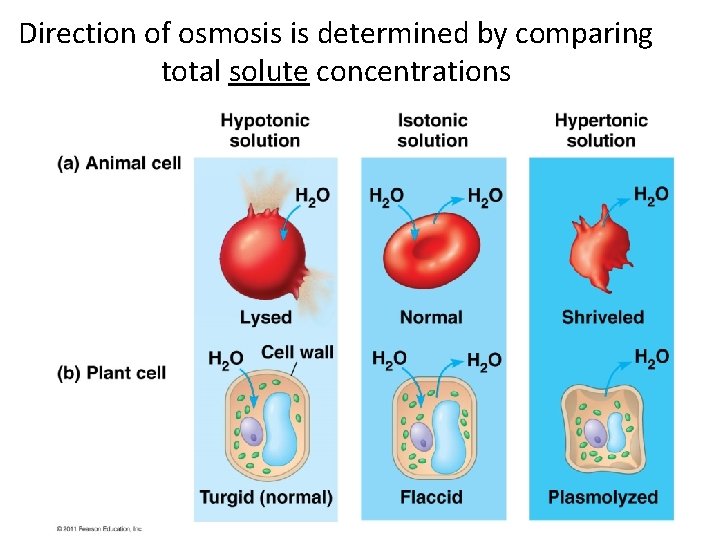
Direction of osmosis is determined by comparing total solute concentrations

Concentration of water • Hypertonic - more solute, less water • Hypotonic - less solute, more water • Isotonic - equal solute, equal water
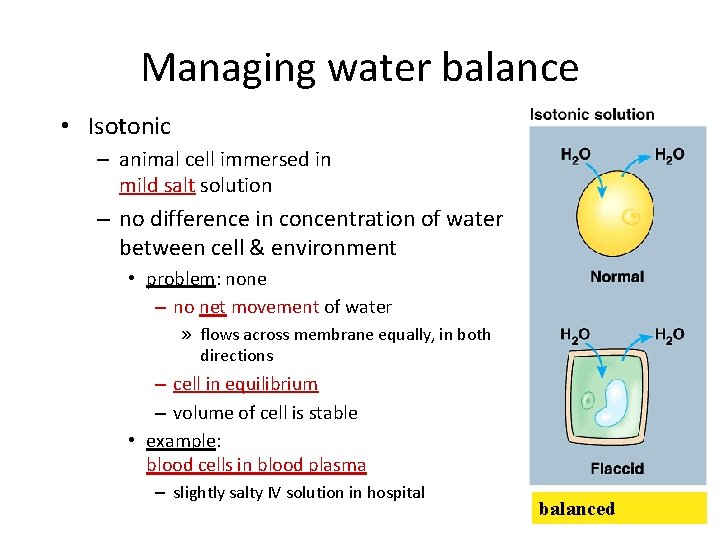
Managing water balance • Isotonic – animal cell immersed in mild salt solution – no difference in concentration of water between cell & environment • problem: none – no net movement of water » flows across membrane equally, in both directions – cell in equilibrium – volume of cell is stable • example: blood cells in blood plasma – slightly salty IV solution in hospital balanced
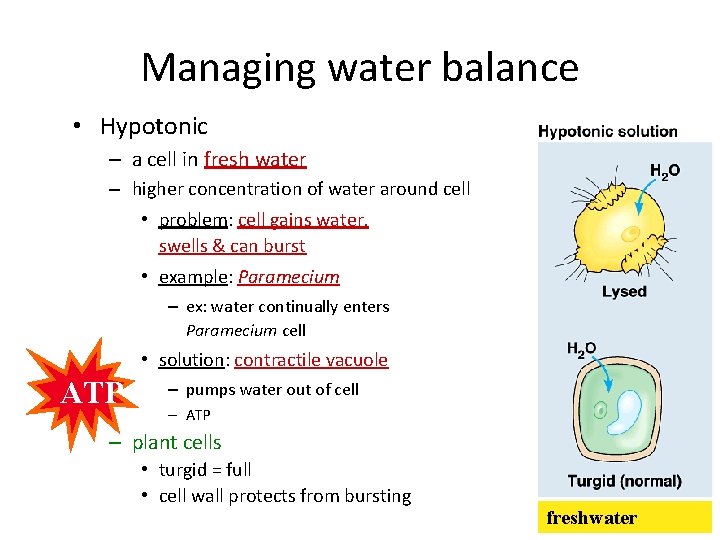
Managing water balance • Hypotonic – a cell in fresh water – higher concentration of water around cell • problem: cell gains water, swells & can burst • example: Paramecium – ex: water continually enters Paramecium cell • solution: contractile vacuole ATP – pumps water out of cell – ATP – plant cells • turgid = full • cell wall protects from bursting freshwater
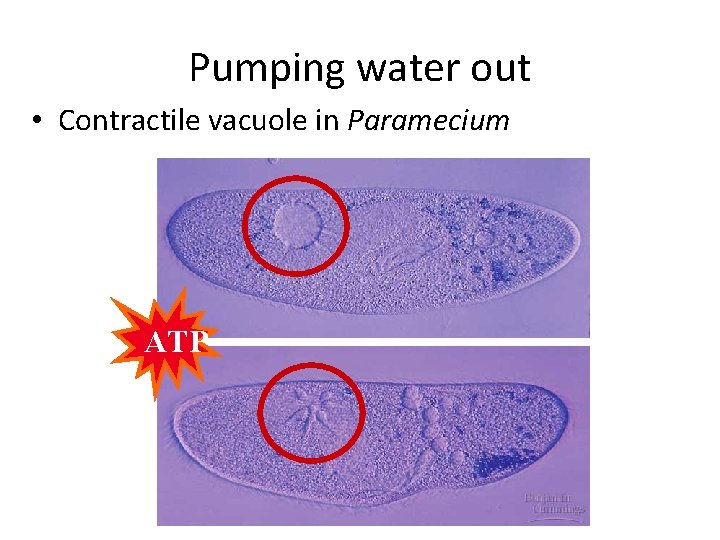
Pumping water out • Contractile vacuole in Paramecium ATP
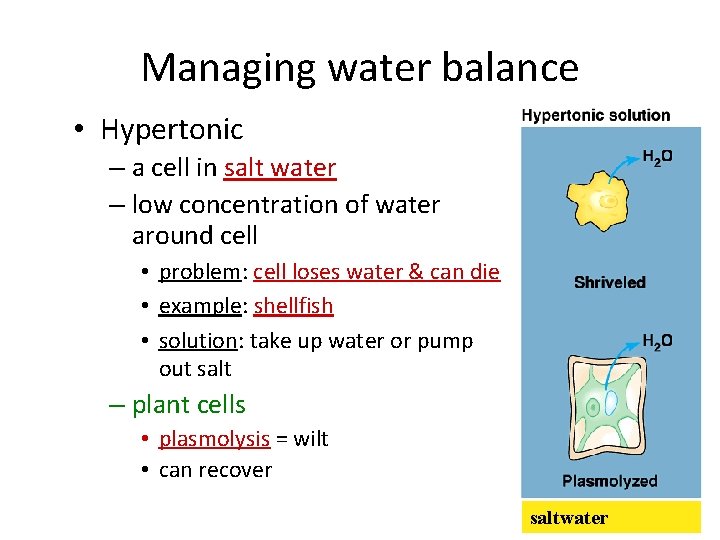
Managing water balance • Hypertonic – a cell in salt water – low concentration of water around cell • problem: cell loses water & can die • example: shellfish • solution: take up water or pump out salt – plant cells • plasmolysis = wilt • can recover saltwater
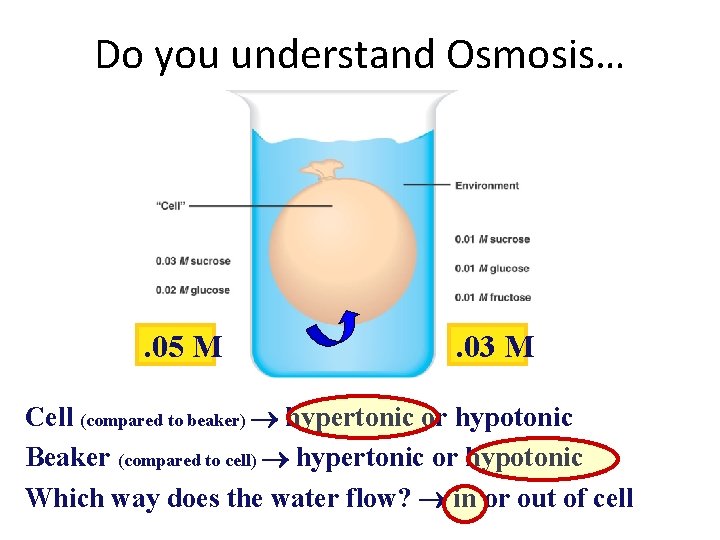
Do you understand Osmosis… . 05 M . 03 M Cell (compared to beaker) hypertonic or hypotonic Beaker (compared to cell) hypertonic or hypotonic Which way does the water flow? in or out of cell

Why is it important to know the concentration of a solution? 1. What happens if you give a person an IV of pure water? (hint our blood cells have about a 0. 9% Na. Cl solution) 2. What happens to a freshwater fish when placed in saltwater? 1. http: //www. bozemanscience. com/osmoregulation/
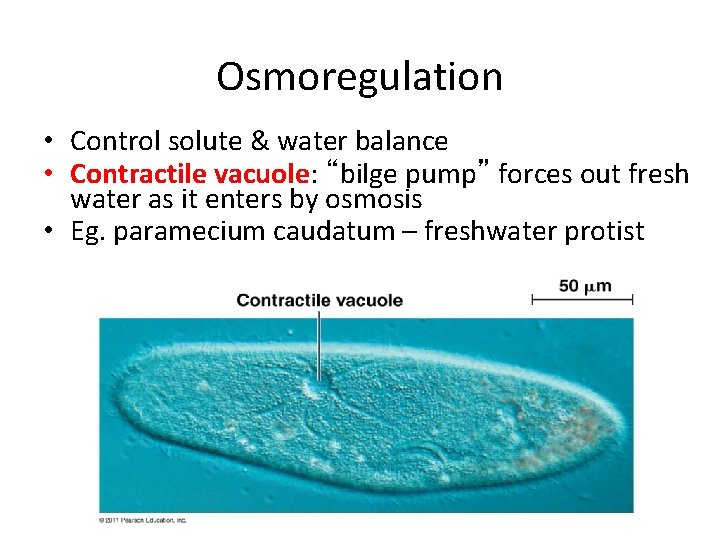
Osmoregulation • Control solute & water balance • Contractile vacuole: “bilge pump” forces out fresh water as it enters by osmosis • Eg. paramecium caudatum – freshwater protist
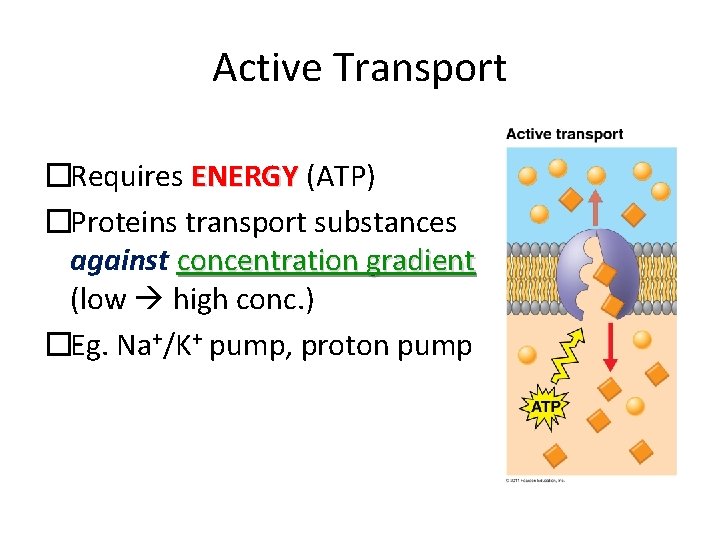
Active Transport �Requires ENERGY (ATP) �Proteins transport substances against concentration gradient (low high conc. ) �Eg. Na+/K+ pump, proton pump

Active Transport Carrier proteins used in active transport include: -uniporters – move one molecule at a time -symporters – move two molecules in the same direction -antiporters – move two molecules in opposite directions
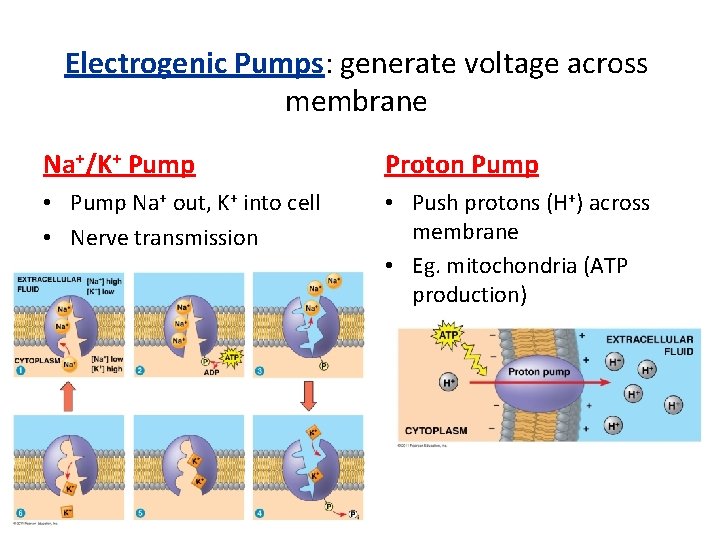
Electrogenic Pumps: generate voltage across membrane Na+/K+ Pump Proton Pump • Pump Na+ out, K+ into cell • Nerve transmission • Push protons (H+) across membrane • Eg. mitochondria (ATP production)
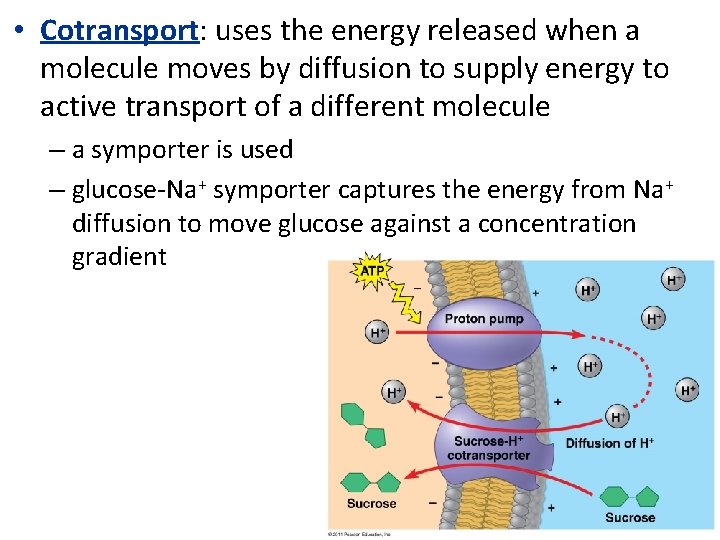
• Cotransport: uses the energy released when a molecule moves by diffusion to supply energy to active transport of a different molecule – a symporter is used – glucose-Na+ symporter captures the energy from Na+ diffusion to move glucose against a concentration gradient

Passive vs. Active Transport • Little or no Energy • High low concentrations • DOWN the concentration gradient • eg. diffusion, osmosis, facilitated diffusion (w/transport protein) • Requires Energy (ATP) • Low high concentrations • AGAINST the concentration gradient • eg. pumps, exo/endocytosis
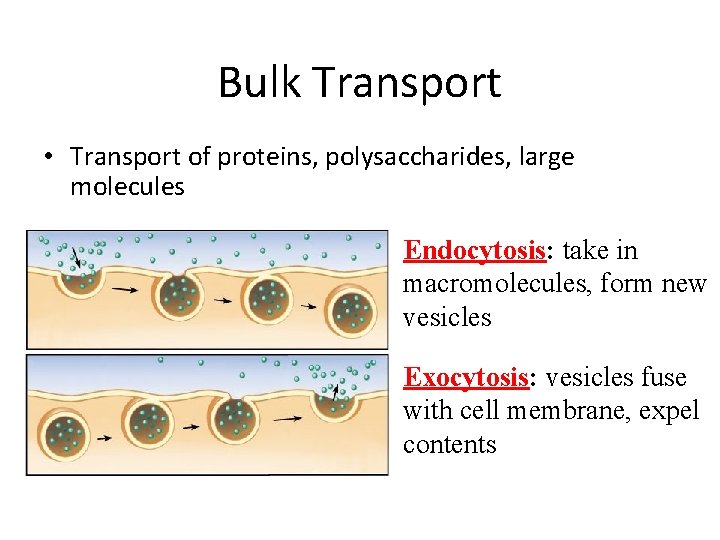
Bulk Transport • Transport of proteins, polysaccharides, large molecules Endocytosis: take in macromolecules, form new vesicles Exocytosis: vesicles fuse with cell membrane, expel contents

Bulk Transport Endocytosis occurs when the plasma membrane envelops food particles and liquids. 1. phagocytosis – the cell takes in particulate matter 2. pinocytosis – the cell takes in only fluid 3. receptor-mediated endocytosis – specific molecules are taken in after they bind to a receptor
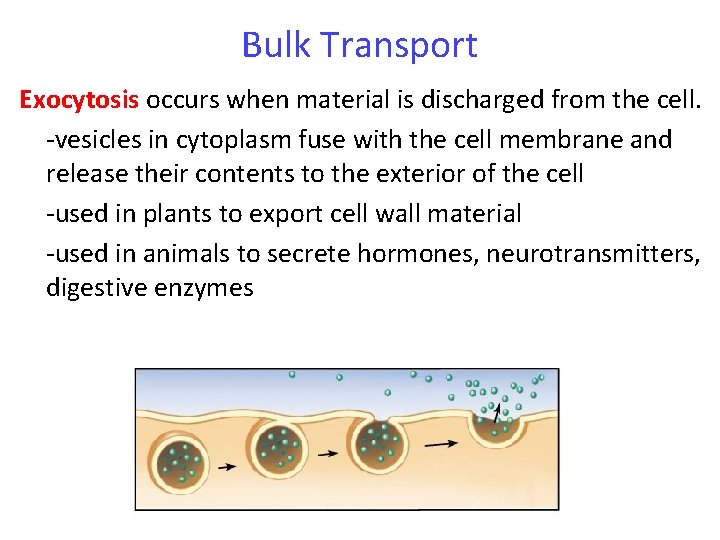
Bulk Transport Exocytosis occurs when material is discharged from the cell. -vesicles in cytoplasm fuse with the cell membrane and release their contents to the exterior of the cell -used in plants to export cell wall material -used in animals to secrete hormones, neurotransmitters, digestive enzymes
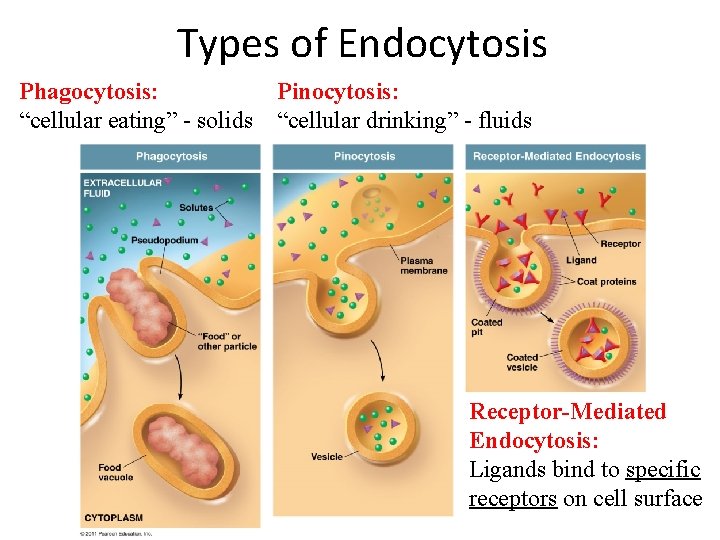
Types of Endocytosis Phagocytosis: “cellular eating” - solids Pinocytosis: “cellular drinking” - fluids Receptor-Mediated Endocytosis: Ligands bind to specific receptors on cell surface

Understanding Water Potential • Water potential = Ψ : psi (sounds like “sigh”) – measure of pressure: megapascal (MPa) or “bars” (1 MPa=10 Bars) • Ψ = 0 MPa for pure water in an open container at sea level and at room temperature – Note: 1 MPa is about 10 x atmospheric pressure at sea level

How Solutes and Pressure Affect Water Potential Equation: • Ψ = Ψ S + ΨP The solute potential “osmotic potential” (ΨS) of a solution is directly proportional to its molarity (pure water is 0) – Expresses as a negative (the more solute added, the more negative it becomes!!!) • Less “free” water • An increase in solute concentration has a negative effect on water potential • Pressure potential (ΨP) is the physical pressure on a solution (from cell wall) • Positive or negative – For example, solution withdrawn by a syringe is under negative pressure; it is under positive pressure when it is being expelled by the syringe – Water in cells usually under positive pressure – Turgor pressure is the push out on the cell wall • Air in a tire

Water Potential Water potential (ψ): H 2 O moves from high ψ low ψ potential Water potential equation: ψ = ψS + ψP • Water potential (ψ) = free energy of water • Solute potential (ψS) = solute concentration (osmotic potential) • Pressure potential (ψP) = physical pressure on solution; turgor pressure (plants) – Pure water: ψP = 0 MPa – Plant cells: ψP = 1 MPa
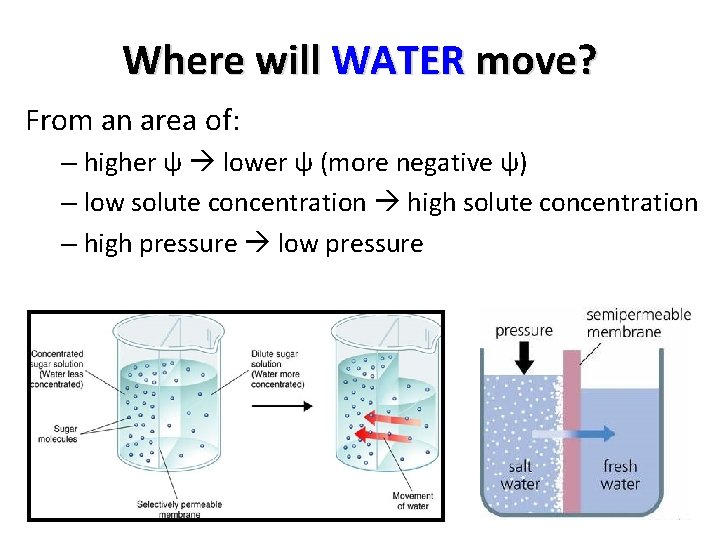
Where will WATER move? From an area of: – higher ψ lower ψ (more negative ψ) – low solute concentration high solute concentration – high pressure low pressure


Water potential (ψ ) = pressure potential (ψp ) + solute (osmotic) potential (ψs) Pressure potential ( Solute potential ( ψp): ψs): In a plant cell, pressure exerted by the rigid cell wall that limits further water uptake The effect of solute concentration. Pure water at atmospheric pressure has a solute potential of zero. As solute is added, the value for solute potential becomes more negative. This causes water potential to decrease also. *As solute is added, the water potential of a solution drops, and water will tend to move into the solution.
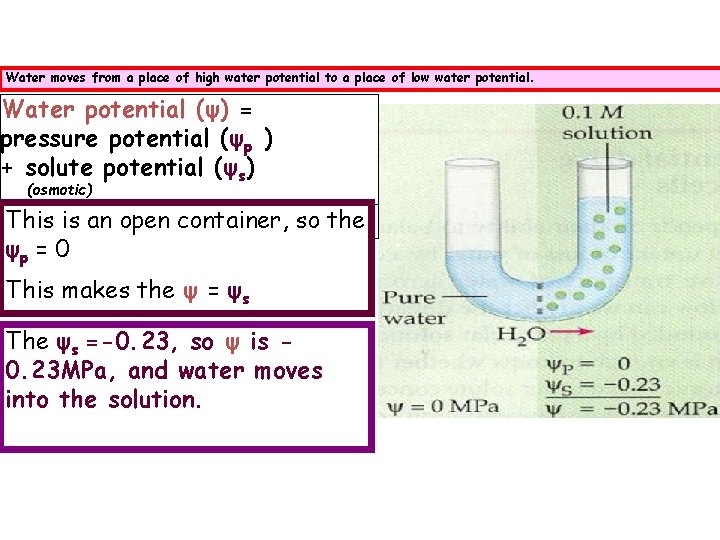
Water moves from a place of high water potential to a place of low water potential. Water potential (ψ) = pressure potential (ψp ) + solute potential (ψs) (osmotic) This is an open container, so the ψp = 0 This makes the ψ = ψs The ψs =-0. 23, so ψ is 0. 23 MPa, and water moves into the solution.
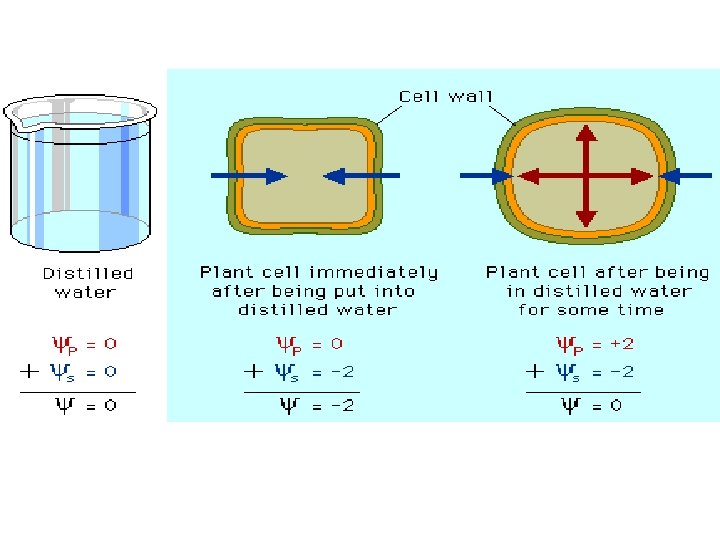
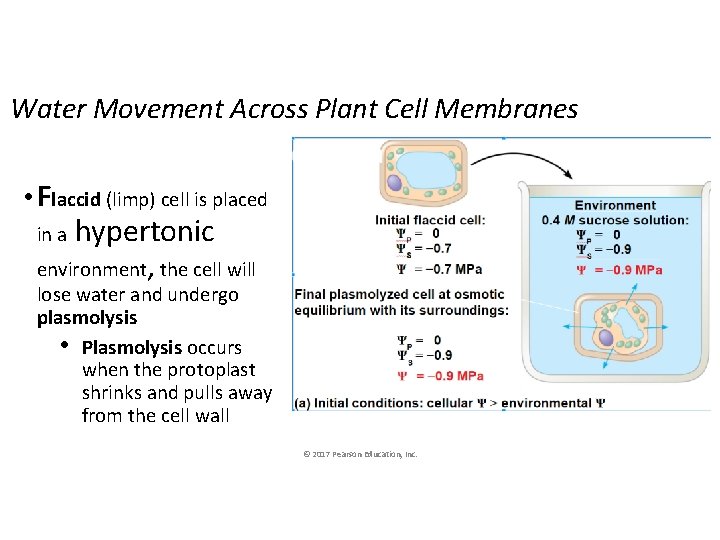
Water Movement Across Plant Cell Membranes • Flaccid (limp) cell is placed hypertonic environment, the cell will in a lose water and undergo plasmolysis • Plasmolysis occurs when the protoplast shrinks and pulls away from the cell wall © 2017 Pearson Education, Inc.
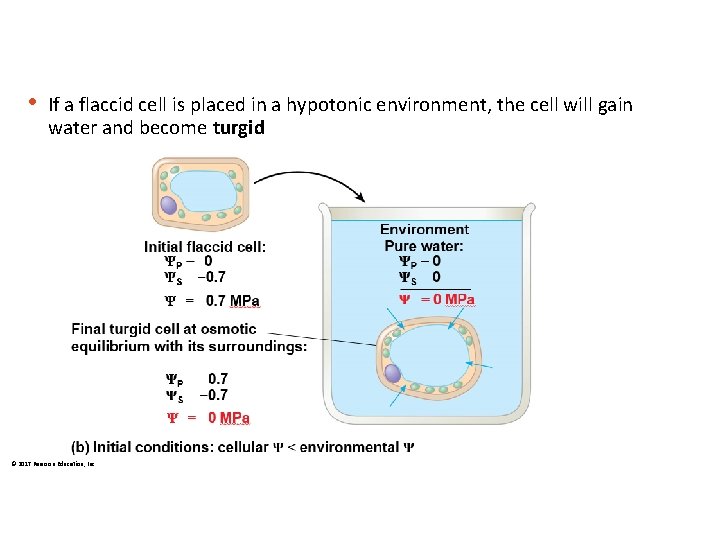
• If a flaccid cell is placed in a hypotonic environment, the cell will gain water and become turgid © 2017 Pearson Education, Inc.

Questions: If a plant cell immersed in distilled water has a ΨS of -0. 7 MPa and a Ψ of 0 MPa, what is the cell’s Ψp? If you put it in an open beaker of solution that has a Ψ of -0. 4 MPa what would be its Ψp at equilibrium? 0. 7 MPa 0. 3 MPa
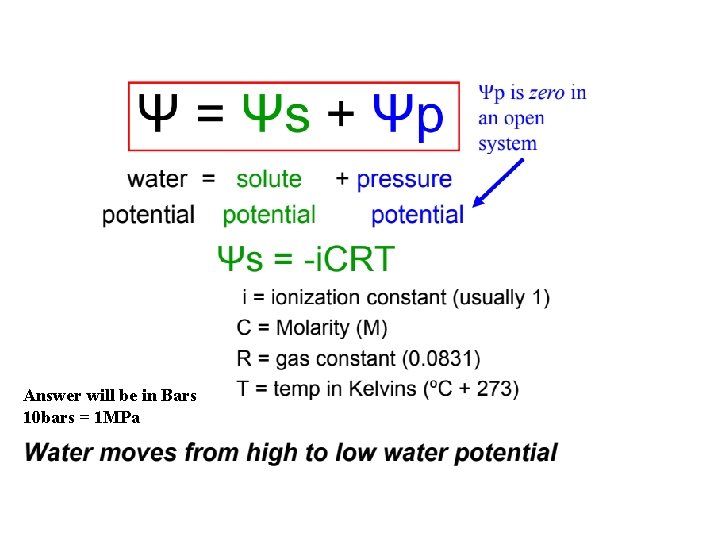
Answer will be in Bars 10 bars = 1 MPa

Calculating Solute Potential (ψS) ψS = -i. CRT • • i = ionization constant (# particles made in water) C = molar concentration R = pressure constant (0. 0831 liter bars/mole-K) T = temperature in K (273 + 0 C) • The addition of solute to water lowers the solute potential (more negative) and therefore decreases the water potential.

So what is the solute potential of a 0. 1 M solution of sucrose at 22 C? • Solute potential = -i. CRT • i (ionization constant) = 1 • R = 0. 0831 (from handbook) • T = temp K (273 + C of solution) Ωs = - (1) (0. 0831) (295) Ωs = - 2. 45 bars

• Calculate the water potential of a solution of 0. 15 M sucrose. The solution is at standard temperature (273 K) and pressure (. 0831 L bars/mol K). Ψ = Ψp + Ψs = 0 bars + [-i. CRT] = 0 bars + [-(1) (0. 15 mol/L) (0. 0831 L bars/mol K) (273 K) = 0 bars + -3. 40 bars Ψ = -3. 40 bars

1. Calculate the solute potential of a 0. 1 M Na. Cl solution at 25°C. 2. If the concentration of Na. Cl inside the plant cell is 0. 15 M, which way will the water diffuse if the cell is placed in the 0. 1 M Na. Cl solution?
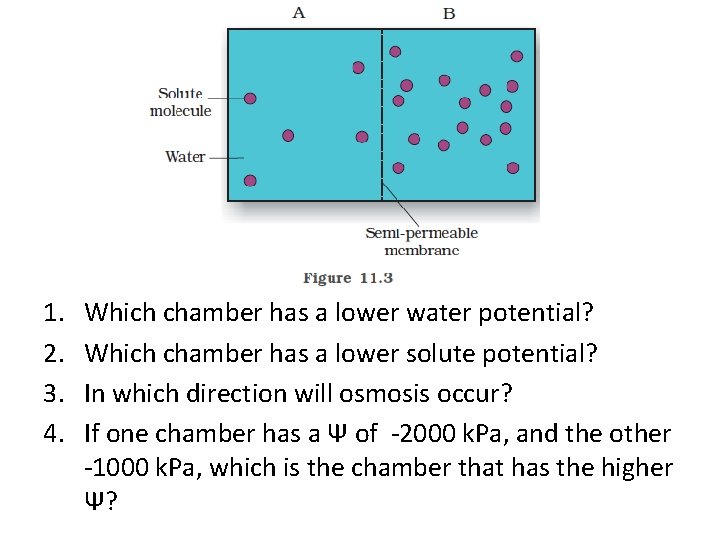
1. 2. 3. 4. Which chamber has a lower water potential? Which chamber has a lower solute potential? In which direction will osmosis occur? If one chamber has a Ψ of -2000 k. Pa, and the other -1000 k. Pa, which is the chamber that has the higher Ψ?

1. A solution in a beaker has Na. Cl dissolved in water with a solute potential of -0. 5 bars. A flaccid cell is placed in the above beaker with a solute potential of -0. 9 bars. p = 0 bars a) What is the pressure potential of the flaccid cell before it was placed in the beaker? p = 0 bars b) What is the water potential of the cell before it was placed in the beaker? w = p + s X = 0 bars + -0. 9 bars X = -0. 9 bars c) What is the water potential in the beaker containing the sodium chloride? w = p + s X = 0 bars + -0. 5 bars X = -0. 5 bars d) How will the water move? Water will enter the cell and leave the solution.

e) What is the pressure potential of the plant cell when it is in equilibrium with the Na. Cl solution outside? Equilibrium means w of cell = w of solution. wcell = pcell + scell -0. 9 bars = x + -0. 5 bars p of the cell = 0. 4 bars f) What is the cells final water potential when it is in equilibrium? Same as the solutions w which = -0. 5 bars g) Is the cell now turgid/flaccid/plasmolysed? turgid h) Is the cell hypotonic or hypertonic with respect to the outside? hypertonic i) If it is hypo/hyper (choose one) tonic – this means that its water potential is higher/lower (choose one) than the outside. Hypertonic means that the water potential is lower than the outside.

A solution in a beaker has sucrose dissolved in water with a solute potential of -0. 9 bars. A flaccid cell is placed in the above beaker with a solute potential of -0. 3 bars. a) What is the pressure potential of the flaccid cell before it was placed in the beaker? p = 0 bars since the cell is flaccid. b) What is the water potential of the cell before it was placed in the beaker? w = p + s X = 0 bars + -0. 3 bars X = -0. 3 bars c) What is the water potential in the beaker containing the sucrose? w = p + s X = 0 bars + -0. 9 bars X = -0. 9 bars d) How will the water move? Water will move out of the cell since it’s water potential value is smaller.

e) What is the pressure potential of the plant cell when it is in equilibrium with the sucrose solution outside? Think carefully – does the plant cell wall change shape? p = 0 bars No f) Also, what is the cell’s final water potential when it is in equilibrium? -0. 9 bars g) Is the cell now turgid/flaccid/plasmolysed? plasmolyzed What is the cell’s solute potential when it is in equilibrium? -0. 9 bars h) Is the cell hypotonic or hypertonic with respect to the outside? Cell would be hypotonic i) If it is hypo/hyper (choose one) tonic – this means that its water potential is higher/lower (choose one) than the outside. Hypotonic means that its water potential value is higher than the outside.
- Slides: 71