Chapter 7 Electrochemistry 7 6 Reversible cell 7

Chapter 7 Electrochemistry § 7. 6 Reversible cell

7. 6. 1 Basic concepts of electrochemical apparatus 1) Electrochemical apparatus Electrolytic cell; Galvanic/voltaic cell Components: Electrodes; electrolytic solution Reaction: oxidation reaction: anode, anodic reaction reductive reaction: cathode, cathodic reactions.

2) Components of an electrode: 1. Current collector (first-type conductor) 2. Active materials: involves in electrochemical reaction 3. Electrolytic solution (second-type conductor). Question: Point out the current collector, active materials and electrolytic solution of the following electrode. 1) Zn(s)|Zn 2+(sln. ) 2) (Pt), H 2(g, p )|H+ (sln. )

3) Differences between chemical and electrochemical reactions e 2 Fe 3+ + Sn 2+ 2 Fe 2+ + Sn 4+ Sn 2+ Fe 3+ e Fe 3+ Sn 2+ e Fe 3+ Sn 2+ in bulk solution Interfacial reaction e e 2+ Fe 3+ Fe Fe 3+ e Fe 3+ Fe 2+ Fe 3+ cathode Sn 4+ Sn 2+ e Sn 2+ Sn 4+ anode at electrode / solution interface halfreactions: Sn 2+ Sn 4+ + 2 e 2 Fe 3+ + 2 e- 2 Fe 2+

4) Basic principle for cell design To harvest useful energy, the oxidizing and reducing agent has to be separated physically in two different compartments so as to make the electron passing through an external circuit.
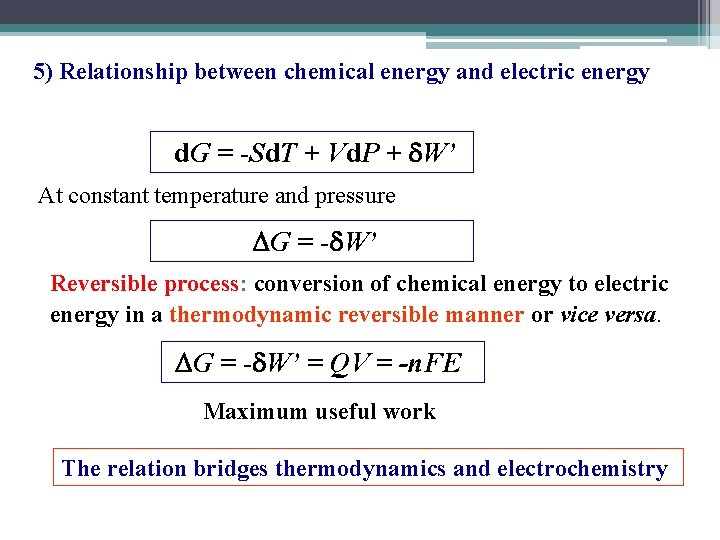
5) Relationship between chemical energy and electric energy d. G = -Sd. T + Vd. P + W’ At constant temperature and pressure G = - W’ Reversible process: conversion of chemical energy to electric energy in a thermodynamic reversible manner or vice versa. G = - W’ = QV = -n. FE Maximum useful work The relation bridges thermodynamics and electrochemistry

7. 6. 2. Reversibility of electrochemical cell Thermodynamic reversibility 1. Reversible reaction: The electrode reaction reverts when shift from charge to discharge. reversible electrode 2. Reversible process: I 0, no current flows.

7. 6. 3. Reversible electrodes 1) basic characteristics: 1) single electrode; Zn|Zn 2+; Zn|H+; 2) reversible reaction; Zn Zn 2+ + 2 e 3) the equilibrium can be easily attained and resumed. In order to acquire reversibility, all reactants and products of the electrode reaction must be present at the electrode. The stability of the electrode materials: According to the active series of metals, which kind of metal can form reversible electrode? K, Ca, Na, Mg, Al, Zn, Fe, Sn, Pb, (H), Cu, Hg, Ag, Pt, Au

2) Main kinds of reversible electrodes (1) The first-type electrode: metal – metal ion electrode A metal plate immersed in a solution containing the corresponding metal ions. Cu (s) Cu 2+ (m) Cu Cu 2+ Cu 2+ metal electrode; amalgam electrode; complex electrode; gas electrode.

amalgam electrode Zn(Hg)x Zn 2+(m 1): complex electrode Ag(s) Ag(CN)2(m 1): Cu Basic characteristics: 1) Two phases / One interface 2) Mass transport: metal cations only Cu 2+ Cu 2+

Gas electrode: Hydrogen electrode Pt(s) H 2(g, p ) H+(c) Three-phase electrode: H 2 gas H+ solution (liquid) Pt foil (solid) 1. 0 mol·dm-3 H+ solution

Acidic hydrogen electrode Basic hydrogen electrode Pt(s), H 2(g, p) H+(c) Pt(s), H 2(g, p) OH (c) 2 H+(c) + 2 e H 2(g, p) 2 H 2 O(l) + 2 e H 2(g, p)+2 OH (c) acidic oxygen electrode Basic oxygen electrode Pt(s), O 2(g, p) H+(c) Pt(s), O 2(g, p) OH (c) O 2(g, p) + 4 H+(c) + 4 e 2 H 2 O(l) O 2 (g, p)+ 2 H 2 O + 4 e 4 OH (c)

(2) The second-type electrode: metal – insoluble salt-anion electrode A metal plate coated with insoluble salt containing the metal, and immersed in a solution containing the anions of the salt. Ag Type II: Ag. Cl metal insoluble salt anion electrode Cl Cl Cl Ag(s) Ag. Cl(s) Cl

Important metal – insoluble salt-anion electrode Hg(l) Hg 2 Cl 2(s) Cl (c): Hg 2 Cl 2(s) + 2 e 2 Hg(l) + 2 Cl (c) There are three phases contacting with each other in the electrode. Pb(s) Pb. SO 4(s) SO 42 (c): in lead-acid battery Pb. SO 4(s) + 2 e Pb(s) + SO 42 (c)

(3) The third-type electrode: oxidation-reduction (redox) electrodes: immersion of an inert metal current collector (usually Pt) in a solution which contains two ions or molecules with the same composition but different states of oxidation. Type III: Pt oxidation-reduction electrodes Sn 2+ Sn 4+ Sn 2+ Pt(s) Sn 4+(c 1), Sn 2+(c 2) Sn 4+(c 1) + 2 e Sn 2+(c 2)
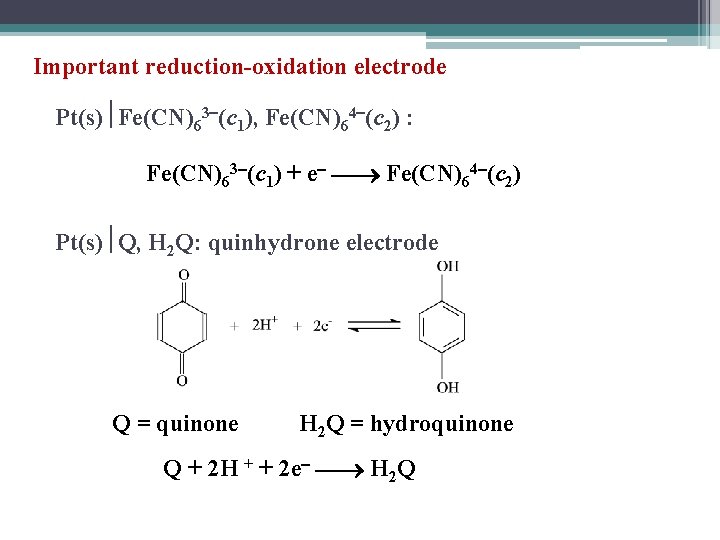
Important reduction-oxidation electrode Pt(s) Fe(CN)63 (c 1), Fe(CN)64 (c 2) : Fe(CN)63 (c 1) + e Fe(CN)64 (c 2) Pt(s) Q, H 2 Q: quinhydrone electrode Q = quinone H 2 Q = hydroquinone Q + 2 H + + 2 e H 2 Q

4) Membrane electrode: glass electrode The membrane potential can be developed by exchange of ions between glass membrane (thickness < 0. 1 mm) and solution. Reference:

7. 6. 4. Cell notations 1) conventional symbolism Zn(s)| Zn. SO 4(c 1) ||Cu. SO 4(c 2) |Cu(s) cell notation / cell diagram 1. The electrode on the left hand is negative, while that on the right hand positive; 2. Indicate the phase boundary usingle vertical bar “│”; 3. Indicate salt bridge using double vertical bar “||”; 4. Indicate state and concentration; 5. Indicate current collector if necessary.

(2) Steps for Reversible Cell Design 1. Separate the two half-reactions 2. Determine electrodes and electrolytes 3. Write out cell diagram 4. Check the cell reaction EXAMPLES: e. g. 1 Zn + Cu. SO 4 = Zn. SO 4 +Cu e. g. 2 Ag+(m) + Cl (m) = Ag. Cl(s) e. g. 3 H 2 O = H+ + OH-

Outside class reading Levine: pp. 417 14. 4 Galvanic cells: cell diagrams and IUPAC conventions Levine: pp. 423 14. 5 types of reversible electrodes metal-metal ion electrode amalgam electrode redox electrode metal-insoluble-salt electrode gas-electrode nonmetal electrode membrane electrode
- Slides: 20