Chapter 7 Dmitri Mendeleev arranged periodic table according

Chapter 7 Dmitri Mendeleev arranged periodic table according to atomic masses chemical periodicity- predicting a property based on an element’s position within the periodic table
-Henry Moseley later arranged according to increasing atomic # -elements in the same group have similar # of valence e- and similar chemical properties

valence orbitals- occupied orbitals which hold the electrons involved in bonding (outermost levels) -in transition metals the d e- are included in the valence electrons even though they are not in the outermost energy level core electrons- all other e- besides the valence e-

*Identify the valence and core e- for potassium, titanium and germanium K 1 valence e- and 18 core e. Ti 4 valence e- and 18 core e. Ge 4 valence e- and 28 core e-

Periodic Trends 1) Effective Nuclear Charge (Zeff) -the net positive charge of the nucleus experienced by an e- in a many-electron atom *Coulomb’s Law the strength of the interaction between two electrical charges depends on the magnitude of the charges and on the distance between them *force inc as Zeff inc and dec as distance inc

Zeff = Z – S Z = # of p+ S= screening constant (usually close to # of core e-) *Zeff < actual nuclear charge *Zeff includes the effect of the other e- in the atom -core e- partially screen valence e- from the attraction of the nucleus

**Zeff increases from left to right across a period -core e- stays the same, but p+ inc -added valence e- screen one another ineffectively

** Zeff going down a group changes far less than it does across a period ex- Li = 3 -2 = 1+ Na = 11 -10 = 1+ -inc slightly as you move down a group b/c the more diffuse core e- cloud is less able to screen the valence e- from the nuclear charge

2) Atomic Radii/Size nonbonding atomic radius/van der Waals radiustwice the radius of two nuclei bonding atomic radius/covalent radius- half the distance between two nuclei page 254 figure 7. 5

-increases as you move down a group *due to inc. in n (prin. quantum #) of outer e*as you inc. n outer e- are further from nucleus -decreases as you move across a period (left to right) *due to the inc. in Zeff which draws valence ecloser to the nucleus, causing smaller radius

-length of the atomic radius of elements will be given -page 255 figure 7. 6 *transition metals do not show a regular decrease moving across -given in angstroms 1Å = 10 -10 m

Problems: 1) Natural gas used in heating and cooking is odorless. Because natural gas leaks pose the danger of explosion or suffocation, various smelly substances are added to detect a leak. One example is mercaptan, CH 3 SH. Predict the lengths of C—S, C—H, and S—H bonds. C—S = 0. 77 + 1. 02 = 1. 79 Å C—H = 0. 77 + 0. 37 = 1. 14 Å S—H = 1. 02 + 0. 37 = 1. 39 Å

2) Predict which is longer: a) P—Br bond in PBr 3 b) As—Cℓ bond in As. Cℓ 3 a) P—Br = 1. 06 + 1. 14 = 2. 20 Å b) As—Cℓ = 1. 19 + 0. 99 = 2. 18 Å **P—Br

3) Arrange (as much as possible) the atoms P, S, As, and Se in order of increasing atomic size. S P Se As 4) Arrange Na, Be, and Mg in order of inc. atomic radius. Be Mg Na

3) Ionic Radius/Size -cations are smaller than their parent atoms *e- lost less e- repulsions -anions are larger than their parent atoms *e- gained more e- repulsions -for ions with the same charge, ionic radius inc. as you move down a group -dec. as you move across a period for ions with same # of e-

isoelectronic series- group of ions all containing the same # of eex: Na+, O 2 -, Mg 2+, F-, Aℓ 3+ -all have 10 e-ionic radius dec. with inc. atomic # b/c e- are more strongly attracted to nucleus **Put above in order of dec. ionic radius O 2 - F- Na 1+ Mg 2+ Aℓ 3+

Problems: 1) Arrange Mg 2+, Ca 2+ and Ca in order of decreasing radius. Ca Ca 2+ Mg 2+ *atoms larger than their cations, inc. as you move down 2) Which of the following atoms and ions is largest: S 2 - , S, O 2 -? S 2*anions larger than their atoms, inc. as you move down
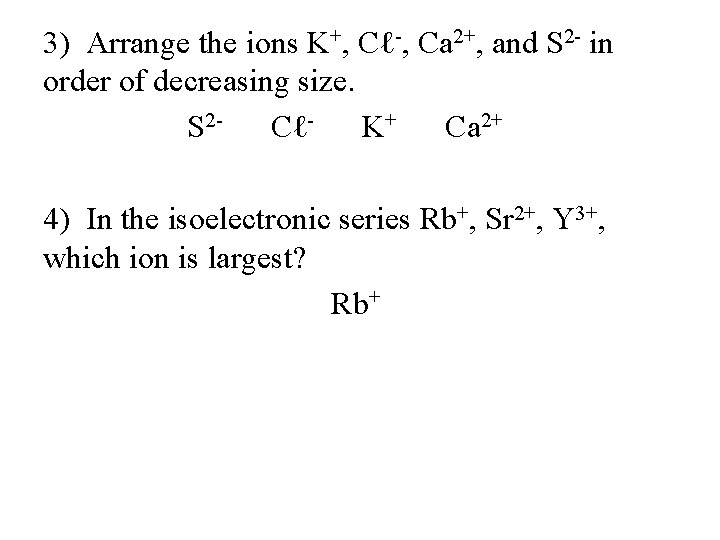
3) Arrange the ions K+, Cℓ-, Ca 2+, and S 2 - in order of decreasing size. S 2 CℓK+ Ca 2+ 4) In the isoelectronic series Rb+, Sr 2+, Y 3+, which ion is largest? Rb+

4) Ionization Energy -minimum energy needed to remove an electron from the ground state of the gaseous atom or ion (k. J/mol) first ionization energy (I 1)- energy needed to remove the first e- from a neutral atom second ionization energy (I 2)- energy needed to remove the second e-

*as each e- is removed the ion. energy increases *greatly increases when an inner e- is removed -due to the greater Zeff on inner e-page 259 Table 7. 2

Problems: 1) Of the following elements, which has the greatest I 2? Na Ca S Na= 1 val e. Ca= 2 val e. S= 6 val e. Na, b/c 2 nd will be a core e 2) Which has the greater I 3, O or Mg? O= 6 val e. Mg= 2 val e. Mg b/c 3 rd will be core e-

Trends in first ionization energy -inc. as you move across a period -dec. as you move down a group -tran. metals generally inc. left to right *alkali metals have the lowest *noble gases have the highest

*smaller atoms have higher I 1 -the energy depends on Zeff and distance of efrom nucleus -inc. Zeff or dec. the distance from the nucleus inc. attraction between e- and the nucleus and makes it harder to remove e-

Problems: 1) Arrange the atoms Ne, Na, P, Ar and K in order of increasing first ion. energy. K Na P Ar Ne 2) Which has the lowest first ion. energy, B, Aℓ, C, or Si? Aℓ

E- configs of ions *e- lost from highest occupied levels first ex: Li Li+ + eex: Fe 1+ + 1 e. Fe 1+ Fe 2+ +1 e-

5) Electron Affinity -energy change that occurs when an electron is added to a gaseous atom -in k. J/mol -measures the attraction of the atom for the added e-when e- is added energy is released, so negative values
*halogens have most negative e- affinities -due to only needing 1 e- to achieve octet *Group 2 A has + energy b/c added e- goes to p orbital which is higher in energy and unoccupied *Noble gases also + b/c added e- goes to next higher s -no definite trends to follow
- Slides: 27