Chapter 7 Chemical Formulas Chemical Compounds Ch 7










































- Slides: 44

Chapter 7 Chemical Formulas & Chemical Compounds Ch 7: Problem Set: pg 231: 2, 4 pg 251: 1, 3, 4, 6, 17, 23, 24, 33, 36, 37, 42, 50

7. 1 Chemical Names and Formulas n Naming monatomic ions n n End in -ide Look up the charges on the periodic table n F vs F-1 n n n F Fluorine F-1 Fluoride S vs S-2 n n S Sulfur S-2 Sulfide

Writing Formulas for Ionic Compounds n Cations – positive ions (metal) n n Anions – negative ions n n metal nonmetal Charge on compound = 0

Writing Formulas for Ionic Compounds n Binary ionic compounds n Rules n n if the charges on the ions are the same, drop ‘em if the charges are different, criss-cross n n Same charges – +1 Cl-1 n Na Na. Cl +2 O-2 n Mg Mg. O Different charges+1 S-2 - Na S n Na 2 +2 Cl-1 - Mg. Cl n Mg 2 Sodium Chloride Magnesium Oxide Sodium Sulfide Magnesium Chloride

Writing Formulas for Ionic Compounds n Ternary Ionic compounds n n Metal + (Polyatomic ion) When naming, do not use the ending –ide n Sodium Nitrate n n Sodium Carbonate n n Na+1 NO 3 -1 Na(NO 3) Na+1 CO 3 -2 Na 2(CO 3) Aluminum Nitrate n Al+3 NO 3 -1 Al(NO 3)3

Writing Formulas for Ionic Compounds n Aluminum Phosphate n n Al+3 PO 4 -3 Aluminum Bihypophosphite Al+3 HPO 2 -2 n Al(PO 4) Al 2 (HPO 2)3 Calcium Phosphite n Ca+2 PO 3 -3 Ca 3 (PO 3)2

Writing Names for Ionic Compounds n n n Front name – positive (cation – metal) Back name – negative (anion – nonmetal) Binary ionic compounds – composed of only 2 types of elements ( M + NM) – end in -ide n n Na. Cl Mg. Cl 2 Al 2 O 3 Na. H Sodium Chloride Magnesium Chloride Aluminum Oxide Sodium Hydride

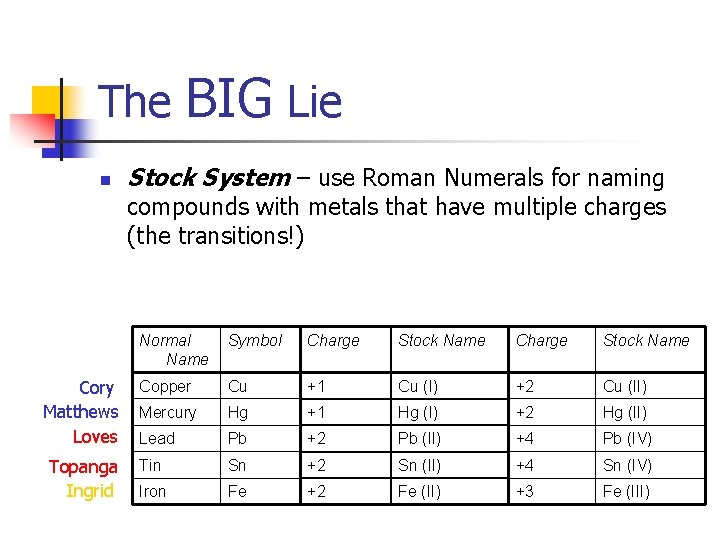
The n BIG Lie Stock System – use Roman Numerals for naming compounds with metals that have multiple charges (the transitions!) Cory Matthews Loves Topanga Ingrid Normal Name Symbol Charge Stock Name Copper Cu +1 Cu (I) +2 Cu (II) Mercury Hg +1 Hg (I) +2 Hg (II) Lead Pb +2 Pb (II) +4 Pb (IV) Tin Sn +2 Sn (II) +4 Sn (IV) Iron Fe +2 Fe (II) +3 Fe (III)

BIG Lie The n More exceptions to the Lie! n Ag is always = +1 charge n n DON’T write Ag I Zn always = +2 charge n DON’T write Zn II

Practice! n Sn 2 N 2 n n Ag. OH n n Lead (II) Carbonate Zn(OH)2 n n Silver Hydroxide Pb. CO 3 n n Tin (II) Nitride Zinc Hydroxide Fe 2(SO 4)3 n Iron (III) Sulfate

More Practice!! n Cu(HSO 2)2 n n Cu. SO 2 n n Copper (II) Bihyposulfite Copper (II) Hyposulfite Cu. HSO 2 n Copper (I) Bihyposulfite

Practice! n Li. Cl. O 3 n n Li. Cl. O 2 n n Calcium Carbonate Ca(HCO 2)2 n n Lithium Chlorite Ca. CO 3 n n Lithium Chlorate Calcium Bicarbonite Fe(NO 3)3 n Iron (III) Nitrate

Writing Names for Molecular Compounds n Molecular Compounds – covalent compounds n n 2 nonmetals To name, we use prefixes # Prefix 1 mono 6 hexa 2 di 7 hepta 3 tri 8 octa 4 tetra 9 nona 5 penta 10 deca don’t use the prefix Mono on the first atom

Writing Names for Molecular Compounds n CO Carbon Monoxide n n CO 2 Carbon Dioxide n n PCl 3 n n CBr 4 n n Phosphorous trichloride Carbon tetrabromide N 2 O 5 n n Dinitrogen pentoxide SF 6 n Sulfur hexafluoride

Writing Formulas for Molecular Compounds n n The prefixes = the subscripts. Do NOTlook at the charges. n Sulfur Dioxide SO 2 n Disulfur Trioxide S 2 O 3 n Dinitrogen pentoxide N 2 O 5

Naming Acids n n Acid - when a solution yields H+ ions in solution 2 types n Binary n n H and one other type of atom ternary (sometimes called oxy) n acids that have H with a polyatomic ion

Naming Binary Acids n Rules n Hydro__(beginning of name)__ic acid n Ex. HCl n n HBr n n H 2 S n n Hydrobromic acid HF n n Hydrochloric acid Hydrofluoric acid Hydrosulfuric acid H 3 P n Hydrophosphoric acid

Writing Formulas from Names for Acids n Do the criss-cross n Ex. Hydronitric acid n n H 3 N Hydroiodic acid n n H+1 N-3 H+1 I-1 HI Hydrosulfuric acid n H+1 S-2 H 2 S

Naming Ternary Acids n n H + polyatomic Rules n n n Do NOT start with hydro. If the ending of polyatomic is –ate If the ending of polyatomic is –ite n n Ate/ite Example: n ic/ous H 2 SO 4 + -2 – sulfate n H and SO 4 n sulfuric acid -ic + acid -ous + acid

Naming Ternary Acids n n n H 2 SO 3 HCl. O 4 HCl. O 3 HCl. O 2 HCl. O

Formulas for Ternary Acids n Use the criss-cross method n n Nitric acid Phosphorous acid

7. 2 Oxidation Numbers n Since electrons are shared, there is no definite charge - we assign the more electronegative element the “apparent” negative charge - this is known as the oxidation # n n oxidation numbers can also be positive. oxidation # - a number assigned to an atom to show the distribution of electrons

Oxidation Numbers n Rules n n n Free elements = 0 n Ex. Mg = O Ions = charges F = -1 S = -2 Oxygen (O) = -2 n except in peroxides (H 2 O 2) O = -1 H = +1 n except in metal hydrides (Mg. H, Na. H) H = -1

Oxidation Numbers n …. Rules n More electronegative atom gets a (-) charge n Ox #’s add up to 0 in compounds n Ox #’s = the charge in polyatomic ions

Oxidation # Practice n Fe. O (Iron II Oxide) O = -2 Fe = ? -2 + x = 0 x= 2 n Fe 2 O 3 (Iron III oxide) O = -2 Fe = ? 3(-2) + 2 x = 0 x= 3

Oxidation # Practice n H 2 SO 4 (Hydrogen Sulfate or Sulfuric Acid) O = -2 H = +1 S = SO 4 -2 X + 4(-2) = -2 X=6

Oxidation # Practice n H 2 SO 3 (Hydrogen sulfite or sulfurous acid) n n n H = +1 S = +4 O = -2 SO 3 -2 X + 3(-2) = -2 X=4

Oxidation # Practice n H 2 Cr 2 O 7 (Hydrogen Dichromate or Dichromic Acid n n NO 3 -1 (Nitrate) n n n H = +1 Cr = +6 O = -2 N = +4 O = -2 Mg. H 2 (Magnesium hydride) n n Mg = +2 H = -1

7. 3 Using Chemical Formulas n Step 1 – be able to calculate molar mass (aka – formula mass, molecular weight, atomic mass, gram formula weight, etc. ) n Add atomic weights from the periodic table n n round to the nearest 10 th place Examples n CH 4 12. 0 + 4(1. 0) = 16. 0 g/mol n Mg. SO 4· 7 H 2 O 24. 3 + 32. 1 + 4(16. 0) + 14(1. 0) + 7(16. 0) = 246. 4 g/mol

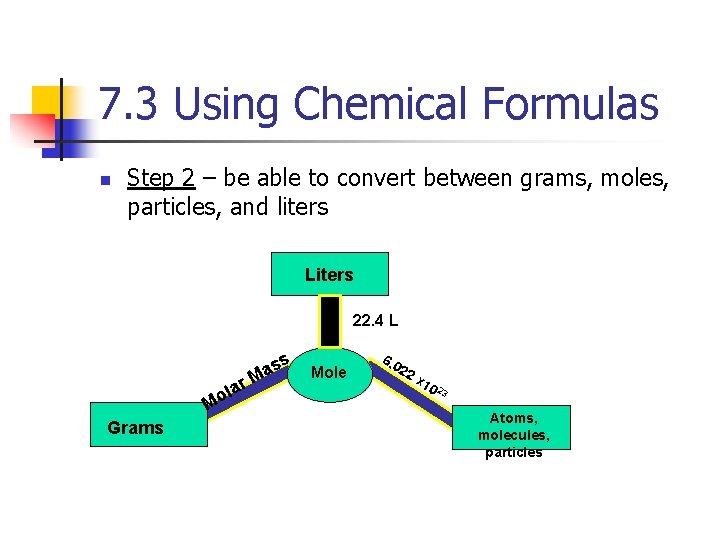
7. 3 Using Chemical Formulas n Step 2 – be able to convert between grams, moles, particles, and liters Liters 22. 4 L s as M lar Mo Grams Mole 6. 0 22 x 1 02 3 Atoms, molecules, particles

Using Chemical Formulas n Convert 32. 0 g of CH 4 to moles, liters, molecules, total atoms, atoms of H n n n Moles Liters Molecules Atoms H

Conversions n Moles CH 4 n n 32. 0 g 1 1 mole 2. 00 moles 16. 0 g CH 4 Liters n 32. 0 g 1 1 mole 16. 0 g 22. 4 L 1 mole 44. 8 Liters

Conversions n Molecules n n Atoms n n 32. 0 g 1 mole 6. 022 x 1023 molecules 1. 20 x 1024 molecules 1 16. 0 g CH 4 1 mole 32. 0 g 1 1 mole 16. 0 g 6. 022 x 1023 atoms 1 mole 5 1 mole 16. 0 g 6. 022 x 1023 atoms 1 mole 4 6. 00 x 1024 molecules Atoms H n 32. 0 g 1 4. 80 x 1024 molecules

Percent Composition n n Percentage Composition - every compound has a certain percentage of each type of atom (we measure it by mass) Formula n % composition = mass element mass compound X 100 =

Practice - % Composition n Calculate % composition if a compound contains 24 g of Carbon and 64 g of Oxygen n % composition = mass element mass compound n n n X 100 = Total mass compound = 24 + 64 = 88 g % Composition C = 24 x 100 = 27 % C 88 % Composition O = 64 x 100 = 73% O 88

Practice - % Composition n What is the % composition of Ba(OH)2?

Molecular Mass of Ba(OH)2 Ba= 1 x 137= O= 2 x 16= H=2 x 1= 137 g 32 g 2 g =171 g Ba: 137/171 = O: 32/171= H: 2/171 = X 100 = 80. 1% X 100 = 18. 7% X 100 = 1. 2%

Empirical Formula- the lowest whole number ratio of atoms in a compound (simplest formula) Three basic steps to the problems: n 1. Divide (%’s or grams) by the gram atomic mass n 2. Divide the resulting #’s by the smallest of those numbers n 3. Multiply by 2 or 3 only if a whole number ratio isn’t the result of step 2 n Ex 1: Calculate the empirical formula if there is 52. 17 % C, 13. 04% H, and 34. 78 % O. n C 52. 17/12. 0 = 4. 3475/2. 17375 = 2 n H 13. 04/1. 0 = 13. 04/2. 17375 = 5. 998849914 6 n O 34. 78/16. 0 = 2. 17375 /2. 17375 = 1 Thus, the empirical formula = C 2 H 6 O

n n n n Ex 2 Calculate the empirical formula if there is 26. 56 % K, 35. 41 % Cr, and 38. 03 % O. K 26. 56/39. 1 =. 679283887/. 679283887 = 1 Cr 35. 41/52. 0 =. 680961538 /. 679283887 = 1. 002469 1 O 38. 03/16. 0 = 2. 376875 /. 679283887 = 3. 499089 The formula is K 1 Cr 1 O 3. 5 (cannot have half of an atom) So, multiply each element by 2 The empirical formula = K 2 Cr 2 O 7

n n Ex 3: Find the empirical formula if a sample contains 5. 6 g N and 12. 8 g O. N 5. 6/14. 0 =. 4 . 4/. 4 = 1 O 12. 8/16. 0 =. 8 . 8/. 4 = 2 The empirical formula = NO 2

Finding Molecular Formulas: • The same steps as empirical formula with one additional step - use the gram formula mass of the empirical formula and its relationship to the gram formula mass of the molecular formula to find what number to multiply the empirical formula by to find the molecular formula (sounds more complicated than it is).

Ex 4 - Find the molecular formula of a compound (mw = 144. 0 g) with 66. 67 % C, 11. 11 % H, and 22. 22 % O C 66. 67/12. 0 = 5. 5558333 /1. 38875 = 4. 000600 4 H 11. 11/1. 0 = 11. 11 11. 11/1. 38875 = 8 O 22. 22/16. 0 = 1. 38875 /1. 38875 = 1 The empirical formula = C 4 H 8 O Check the empirical formula’s mass: C 4 (12. 0) = 48. 0 H 8 (1. 0) = 8. 0 O 1 (16. 0) = 16. 0 72. 0 (see next slide)

EX 4: continued The compound needs to have a mass of 144. 0 grams. C 4 H 8 O only has a mass of 72. 0 grams. By dividing 144. 0 by 72. 0, it is found that C 4 H 8 O is off by a factor of 2. So, multiply each element in C 4 H 8 O by 2. The molecular formula now equals C 8 H 16 O 2

EX 5: 19. 80% C, 2. 50% H, 66. 10% O, 11. 6% N and MW=242. 0 (C=12. 0, O=16. 0, H=1. 0 and N=14. 0)