Chapter 7 Acids Bases About Acids Sour taste

Chapter 7 Acids & Bases

About Acids… • • • Sour taste Neutralize base Dissolves active metal Litmus indicator = red Definitions – Arrhenius: form H+ in water – Bronsted-Lowry: donates a proton

About Bases… • • • Bitter taste Neutralize acid Feels slippery or “soapy” Litmus indicator = blue Definitions – Arrhenius: form OH- in water – Bronsted-Lowry: accepts a proton

Strong Acids & Bases • Come apart completely in water – Examples: Hydrochloric acid, sulfuric acid, nitric acid

Weak Acids & Bases • Come apart only partially in water – Examples: acetic acid, carbonic acid, hydrocyanic acid

Acids and Bases
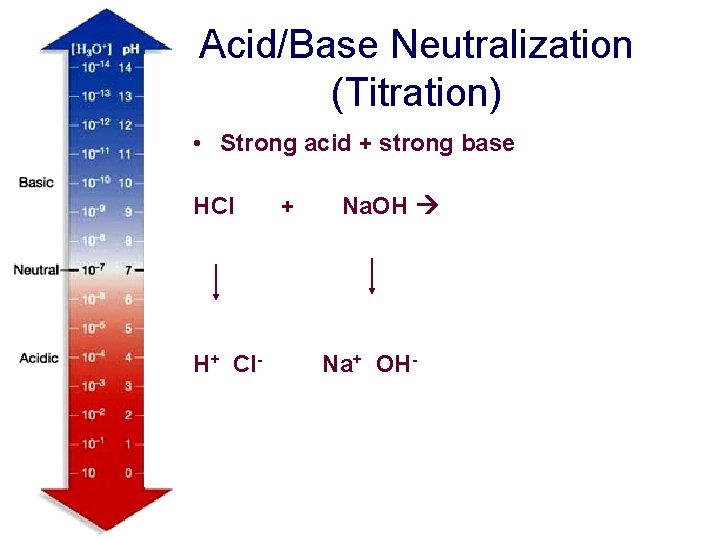
Acid/Base Neutralization (Titration) • Strong acid + strong base HCl + Na. OH Na. Cl H+ Cl- Na+ OH- H 2 O + Na. Cl
![p. H Calculations p. H = – log [H+] = 10 -p. H • p. H Calculations p. H = – log [H+] = 10 -p. H •](http://slidetodoc.com/presentation_image_h2/41ce78d37838497d1c515c46abbf90bc/image-9.jpg)
p. H Calculations p. H = – log [H+] = 10 -p. H • What is the p. H of a solution that has a hydrogen ion concentration of 8. 5 x 10 -4 M? • If a solution has a p. H of 9. 4, what is the hydrogen ion concentration?
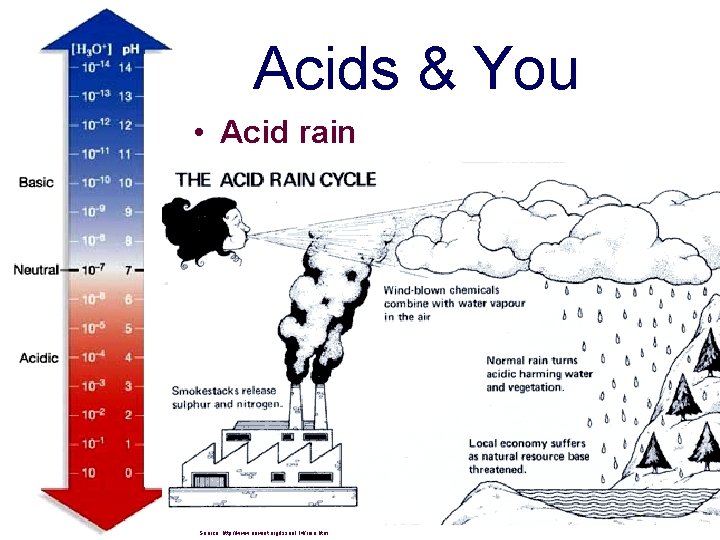
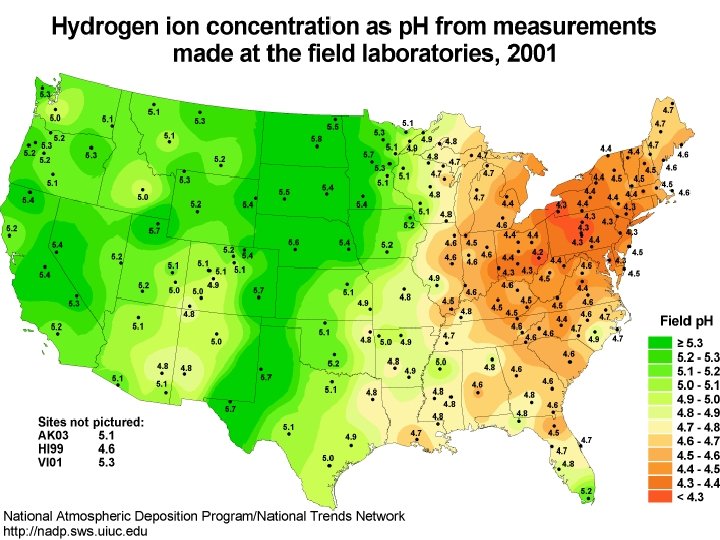
Acids & You • Acid rain Source: http: //www. newint. org/issue 114/rain. htm
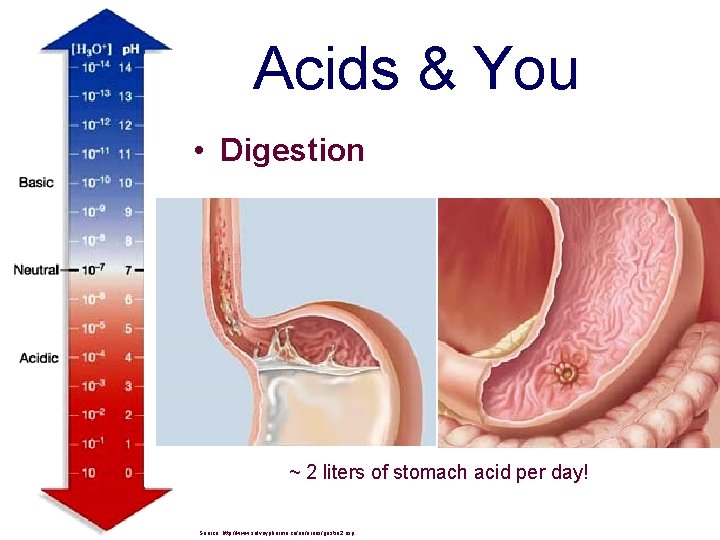
Acids & You • Digestion ~ 2 liters of stomach acid per day! Source: http: //www. solvaypharma. ca/en/areas/gastro 2. asp
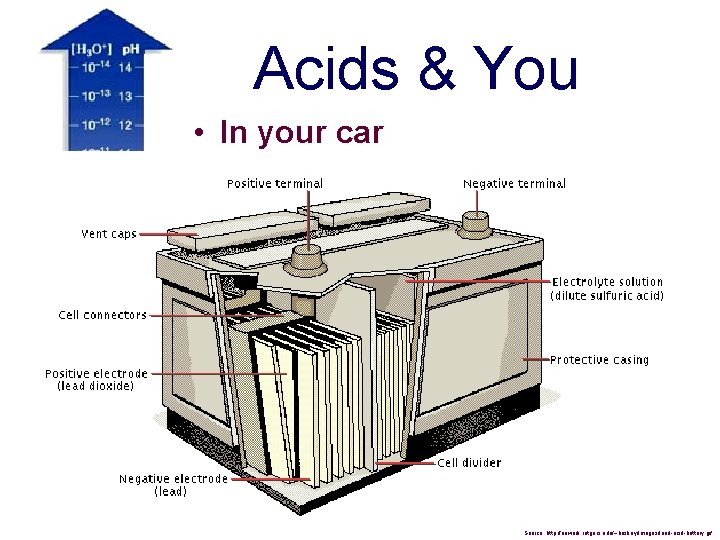
Acids & You • In your car Source: http: //newark. rutgers. edu/~huskey/images/lead-acid-battery. gif

Bases & You • Cleaning – Clarifying shampoos • Higher p. H than the hair itself makes the hair shaft swell and allows cleaning agents to penetrate more deeply – Detergents • Break up oil and grease – Drain cleaner • Very high p. H to eat through acidic hair & skin debris

Bases & You • Antacids – Acid-base neutralization – Calcium carbonate + stomach acid

Bases & You • Baking – Baking soda & baking powder react with acid in the batter to form bubbles of CO 2 Source: http: //www. joyofbaking. com/bakingsoda. html

Practice • Nitric acid (HNO 3) reacts with magnesium hydroxide in an acid-base neutralization reaction. – Write the balanced equation. 2 HNO 3 + Mg(OH)2 2 H 2 O + Mg(NO 3)2 – Name both products in the reaction. Water and magnesium nitrate

Practice • What is the molarity? – 50. 0 g of potassium bromate dissolved in enough water to make 1. 5 L of solution (2 decimal places) 0. 2 M • How many grams to weigh out? – Make 1. 8 L of a 0. 75 M solution of calcium perchlorate (2 decimal places) 322. 52 g

Practice • What is the percent concentration? (1 decimal place) – 45 m. L of ethanol dissolved in 1 L of water 4. 5% v/v – 85 g of Na. Cl dissolved in 315 g of water 21. 3% m/m

Practice • How many times more acidic is lemon juice (p. H 1. 5) than blood (p. H 7. 5)? 1, 000 or 106 times more acidic • Something that is 10, 000 times more basic than tomatoes (p. H 4. 2) would have a p. H of what? p. H of 8. 2
![Practice • What is the p. H? (2 decimal places) – [H+] = 1. Practice • What is the p. H? (2 decimal places) – [H+] = 1.](http://slidetodoc.com/presentation_image_h2/41ce78d37838497d1c515c46abbf90bc/image-21.jpg)
Practice • What is the p. H? (2 decimal places) – [H+] = 1. 87 x 10 -4 M 3. 73 • What is the hydrogen ion concentration? – p. H = 11. 45 3. 55 x 10 -12 M
- Slides: 21